Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

At high concentrations, ammonia seems to affect the central nervous system of fish,
resulting in hyperventilation, convulsions, and death. At chronic levels of exposure, the
toxicity seems to involve histological and reproductive effects. The LC
50
falls in the range
0.49 to 4.6 mg of nonionized ammonia per lite r for a variety of aquatic animals. These
include Daphnia, cray fish, and the fish bluegills, red shiners, channel catfish, and large-
mouth bass. One group that seems more sensitive is the salmonids, which include pink
salmon (0.08 to 0.1 mg/L) and rainbow trout (0.2 to 1.1 mg/L). Chronic effects include
reduced uptake of food and reduced growth at levels as low as 0.002 to 0.15 mg/L NH
3
.
Ammonia is oxidized by bacteria such as Nitrosomonas to nitrite (NO
2
), which is
then converted to nitrate (NO
3
) by, among others, Nitrobacter. The first step is usually
rate limiting. Thus, nitrite does not normally accumulate to appreciable levels in aquatic
systems. Nonionized ammonia inhibits Nitrobacter at 0.1 to 1.0 mg NH
3
/L and inhibits
Nitrosomonas at 10 to 150 mg/L. Thus, in between these two levels, the second step is
inhibited and not the first, and nitrite can accumulate. Thus, lowering the pH can cause
nitrite accumulation by its effect on ammonia. Furthermore, nitrite is in equilibrium with
nitrous acid (HNO
2
), with a K
a
of about 3.39 at 10
C. Nitrous acid inhibits Nitrobacter
and Nitrosomonas at concentrations starting at about 0.22 to 2.8 mg/L. This inhibition also
favors nitrite formation. Some organic chemicals have similar effects. Concentrations of
nitrite up to 73 mg NO
2
-N /L have been found in lakes and streams.
Nitrite is toxic to fish and mammals by forming methemoglobin, limiting the oxygen-
carrying capacity of the blood (see Section ) LC
50
for fish has been measur ed at 0.1 to
0.4 mg NO
2
-N=L for rainbow trout, 1.6 mg NO
2
-N/L for mosquitofish, 7.5 to 13
mg NO
2
-N /L for catfish, and greater than 67 mg NO
2
-N/L for the mottled sculpin.
Toxicity to fish decreases above pH 6.4, apparently due to decreasing HNO
2
levels.
Nitrate toxicity is very low. One of the most sensit ive aquatic species is the guppy
(Poecilia reticulata), with a 96-hour LC
50
of 180 to 200 mg NO
3
-N/L. Other species
range from 420 to 2000 mg NO
3
-N/L. Some of this toxicity may be related to a salinity
effect rather than a specific toxicity of nitrate.
In mammals, high concentrations of nitrites can react with some amines under the acid
conditions of the stomach to form N-nitroso compounds (nitrosamines and nitrosa-
mides). Many of these are highly carcinogenic. Nitrate is used to cure meats and to pre-
serve other foods and is found in water contaminated by agricultural fertilizers or
domestic wastewater. It is also found naturally in some vegetables such as turnip greens,
beets, and celery. Nitrate in food or water can be converted to nitrite by microorganisms in
the mouth and stomach. Nitrites and nitrates also cause ‘‘aging’’ of coronary arteries. N-
nitroso compounds can also be formed by cooking or drying foods over open flames when
the NO
x
compounds formed in combustion react with amines in the food.
R
1
NR
2
N
O
nitrosamine
Suspended solids may come from inert materials such as soil particles or from pollution
sources such as sewage. In the case of sewage, other harmful effects, such as oxygen limi-
tation, predominate over the physical effect of the solids. Forest streams typically have very
low suspended solids: less than 20 to 50 mg/L. Some rivers, such as the Mississippi River in
Louisiana, can reach almost 300 mg/L. The higher levels can be abrasive for aquatic
plants, and can coat the bottom, reducing survival of benthic organisms and fish eggs.
828 TOXICITY OF SPECIFIC SUBSTANCES
Fish production is thought to be reduced at levels from 25 to 80 mg/L. From 80 to
400 mg/L, fish production is likely to be affected severely. Lethal effects in fish have been
measured at suspend ed solids levels ranging from 40,000 to 160,000 mg/L. However, a
single value cannot be determined because it will depend on shape and size distribution of
the particles. Presumably the lethality is due to clogging or abrasion of the gil ls.
21.7 TOXICITY TO MICROBES
Bacteria tend to be less sensitive than eukaryotic organisms to toxic substances. This may
be due to the relative simplicity of their metabolic processes and the lower degree of spe-
cialization. For example, organophosphate pesticides act on acetylcholinesterase in the
nervous system, which has no counterpart among bacteria. Thus, bacteria are fairly insen-
sitive to these substances. General metabolic inhibitors, however, can affect bacteria in a
manner similar to that of higher organisms. Bacterial membranes, cell walls, and slime layers
offer greater resistance to many toxins than animal structures such as gills, lungs, and intest-
inal surfaces. Furthermore, bacteria can become dormant when conditions are not favorable
for growth or survival. Cyst formation can be another barrier to a toxicant entering the cell.
Besides reduced sensitivity, three factors relating to microbial ecology serve to miti-
gate the impact of toxins on microbial communities:
1. Bacteria grow rapidly and can quickly regain their numbers if only part of the
population is eliminated.
2. Bacterial groups have redundant capabilities. That is, although bacteria may be
metabolically specialized individually, many different types share similar speciali-
zation. Consequently, when one type is poisoned, there will often be others ready to
take their place. There are some exceptions, such as the nitrifying bacteria. These
grow slowly, and are replaced slowly, yet are important to the ecosystem. Thus,
toxicity assessments should consider possible effects on this group.
3. Bacteria are ubiquitous in natural systems. Even in laboratory or industrial systems
it is very difficult to sterilize an open system chemically and at the same time to
isolate it from external resupply. In the natural environment, it is all but impossible.
Nevertheless, inhibition of bacteria has been measured by various means, including plate
counts, biochemical measurements, respirometry, and biomass growth rates. Table 21.12
TABLE 21.12 Effect of Herbicides on Microbial Respiration
Erwinia carotovora Pseudomonas fluorescens Bacillus sp.
Conc. % Conc. % Conc. %
Herbicide (mg/L) Inhibition (mg/L) Inhibition (mg/L) Inhibition
Diquat 25 40.8 50 0 25 100
Loxynil 25 71.0 50 0 10 29.0
Paraquat 50 25.5 25 43.7 5 100
PCP 10 29.6 25 65.8 5 100
Picloram 50 0 50 28.8 50 0
Nitralin 50 0 25 22.0 50 0
Source: Rand and Petrucelli (1985).
TOX ICITY TO MICROBES
829
shows the reduction in respiration measured with several herbicides. It is interesting
that Bacillus spp. were completely inhibited by diquat at levels that had no effect on
P. fluorescens respiration. Note that inhibition of respiration does not necessarily mean
that the organisms were killed.
Toxic effects on bacteria can be studied by determination of effects on specific func-
tions, such as saprophytic activity, methanogenesis, nitrification, or other biogeochemi cal
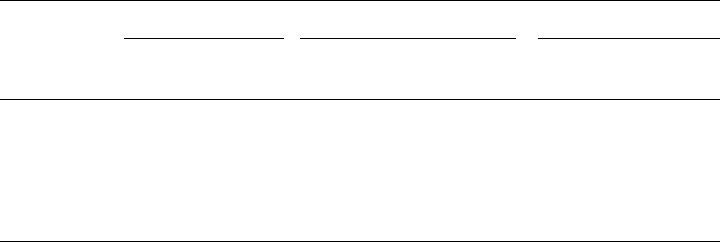
cycle activities. Figure 21.3 shows an example of this. The amount of inhibi tion was
observed to vary with incubation time. For example, methylparathion’s inhibition
decreases up to 9 days, then increases again. This may be due to the degradation of the
pesticide, followed by an accumulation of an even more toxic by-product, aminophenol.
Nitrogen fixation can be studied by measuring the conversion of acetylene to ethylene
in the absence of nitrogen. This is because the enzyme nitrogenase catalyzes both reac-
tions. Stimulation is often found with pesticides, as well as inhibition. For example,
malathion causes an initial period of inhibition, followed by more than double the activity
after 8 days. Other combinations of pesticides and species produced up to five times the
activity.
Soil fungi and actinomycetes were found to bioaccumulate organochlorine pesticides.
Bioconcentration factors ranged from 10 for lindane to 59,000 for chlordane. Bacteria are
also involved in biotransformation reactions that can enhance or reduce toxicity of sub-
stances to other organisms. One of the most important is the conversion of inorganic mer-
cury to methylmercury. Other examples include the conversion of crude oil components to
genotoxic agents, the conversion of P
S groups in organophosphate pesticides to the
more active P
O groups, and the formation of nitrosamines from secondary amines
and nitrites.
0
10
20
30
40
50
60
70
80
579111315
t (da
y
s)
Percent activity
Fonofos
Guthion
Parathion
Methyl Parathion
Control
Figure 21.3 Effect of thiophosphorus pesticides on ammonia oxidation by estuarine nitrifiers.
(From Rand and Petrucelli, 1985; original source: Jones and Hood, 1980, Can. J. Microbiol., Vol.
26, pp. 1296–1299.)
830
TOX ICITY OF SPECIFIC SUBSTANCES
21.8 IONIZING RADIATION
An important physical cause of toxic effects is a variety of forms of radiation. Ionizing
radiation possesses enough energy to strip an electron from an atom. This can result in
the formation of damaging free radicals or directly damage bonds in biochemical sub-
stances. The most sensitive system in living things is the DNA, since damage to a single
molecule can transform a cell to malignancy. It is not necessary for a radioactive emission
to damage a DNA molecule directly. The most abundant molecule in living things is
water. Water can form free radicals when irradiated, and these in turn can produce
toxic effects, including genotoxicity.
Ionizing radiation may be electromagnetic (g-rays, x-rays, ultraviolet rays) o r particu-
late. The major particulate forms of radiation are a and b, but they may include neutrons,
other subatomic particles, or larger particles; such as various atomic nuclei found in cos-
mic rays. Of primary interest here the a, b, and g emissions from the decay of radioactive
atoms, or radionuclides. Each particle of radiation possesses energy, commonly mea-
sured in millions of electron volts (MeV). A change to SI units is under way that will
replace the MeV with the joule: 1MeV ¼ 1:6 10
13
J:
The energy of a particle relates to the amount of damage a particle can do to a tissue.
As radiation passes through material such as tissue, it is absorbed, depositing its energy in
the material. Much of this energy goes into stripping electrons from atoms. This requires
an average of 33.85 eV per electron. This ability to eject electrons confers a capacity to
disrupt chemical bonds with potentially damaging results.
Gamma (g) radiation is electromagnetic radiation (photons) of very high energy,
usually less than 1 MeV. They are produced by radioactive decay of unstable atomic
nuclei. X-rays are somewhat less energetic, up to 0.25 MeV for radiation from medical
x-ray machines. Ultraviolet (UV) are even less energetic, ranging from about 4.13 to 155
eV. X-ray and UV photons are produced by excitation of an atom’s electrons. Alpha (a)
particles consist of two protons and two neutrons (helium nuclei). They have energies that
range from 3 to 9 MeV. Beta (b) particles are high-energy electrons, with energies ranging
up to 3.5 MeV. Alpha, beta, and gamma particles are all emitted by spontaneous reactions
in the nuclei of unstable (radioactive) isotopes.
Although alpha particles are high in energy, they have the least penetrating power, due
to their mass and charge. They are strongly absorbed and deposit their energy rapidly,
producing a large amount of damage. For example , alpha particles with 5 MeV pene-
trate about 40 mm of soft tissue or about 5 cm in air. Thus, they do not penetrate the
skin. However, if they are inhaled or inge sted, they do not need to pe netrate very far
to reach cell nuclei, potentially causing gen etic damage. Beta particles are charged,
but have little mass. They penetrate about 3 m in a ir, but they are stopped by thin sheets
of many solids. Thus, alpha and be ta radiation is a problem mostly if their emitters
are ingested . Gamma rays are the most penetrating. Their range in air is about 4 m,
and th ey will penetrate several centimeters of lead. Co nsequently, g rays can cause
their effects o n living things eit her when emitters are ingested or by external radiation
sources.
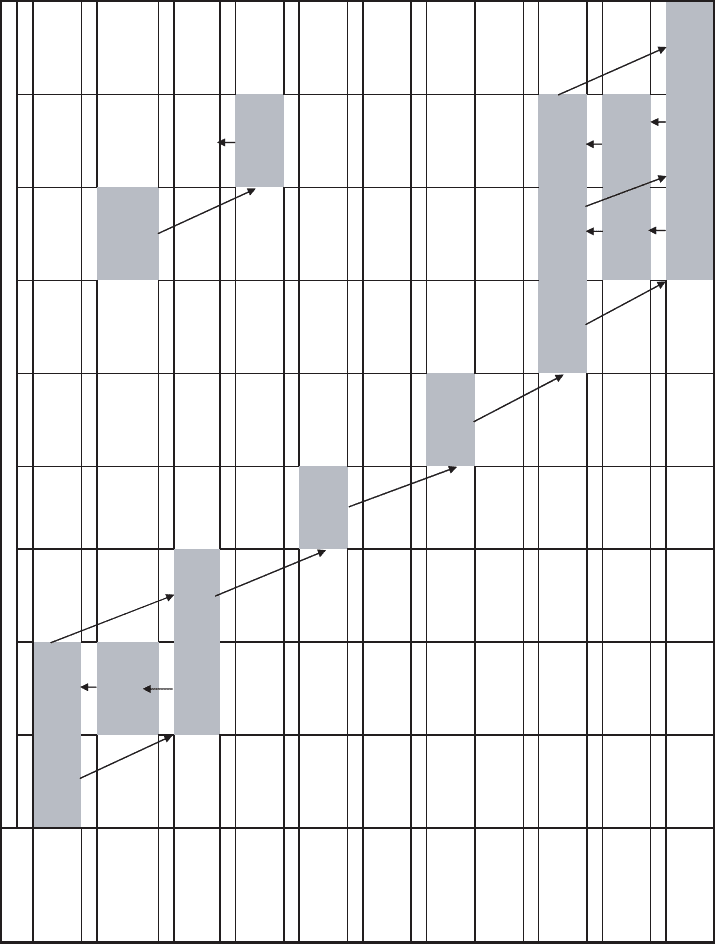
Many of the environmentally important radionuclides are products of the sequence of
nuclear decay reactions that start with
238
U. Each beta emission increases the atomic num-
ber without significantly changing the atomic mass by converting a neutron to a proton.
An alpha emission reduces the atomic number by two and the atomic mass by four.
Figure 21.4 shows the members of the series, with their half-lives and the energy carr ied
IONIZING RADIATION 831
238
234
230
226
222
218
214
210
206
92 - (U)
Uranium
U-238
4.5e9 yr
4.2 MeV
U-234
2.5e5 yr
4.7-4.8 MeV
91 - (Pa)
Protactinium
Pa
Pa-234m
1.2 months
2.3 MeV
Alpha Decay
(helium nucleus
ejected)
90 - (Th)
Thorium
Th
Th-234
24 days
0.2
,
0.1 MeV
Th-230
8.0e4 yr
4.6-4.7 MeV
89 - (Ac)
Actinium
Ac
Beta Decay
(electron
e
j
ected)
88 - (Ra)
Radium
Ra
Ra-226
1600 yr
4.8 MeV
87 - (Fr)
Francium
Fr
86 - (Rn)
Radon
Rn
Rn-222
3.82 days
5.5 MeV
85 - (At)
Astatine
At
84 - (Po)
Polonium
Po
Po-218
3.05 min
6.0 MeV
Po-214
1.6e-4 sec
7.7 MeV
Po-210
138 days
5.3 MeV
83 - (Bi)
Bismuth
Bi
Bi-214
19.7 min
0.4-3.3 MeV
Bi-210
5.0 days
1.2 MeV
82 - (Pb)
Lead
Pb
Pb-214
26.8 min
0.7, 1.0 MeV
Pb-210
22 yr
<0.1 MeV
Pb-206
Stable
MASS NUMBER
ATOMIC NUMBER
Figure 21.4
Uranium-238 decay series.
832

by their alpha and beta particles. Some of these reactions may also produce g radiation,
usually at lower energies.
Table 21.13 summarizes the emissions from some other important radionuclides. Note
that the smaller radionuclides tend not to emit alpha radi ation.
Each type of decay is characterized by its half-life, the type of particle(s) emitted, and
the energy carried by the particles. The half-life defines the rate of disintegrations. The
half-life is related to the decay rate coefficient, k, by equation (18.25). The rate coefficient
can then be used in equation (18.23) to compute the rate of decay.
Example 21.1 One gram of
226
Ra has a half-life of 1600 years. Thus, k ¼ 0:693=
1600 years ¼ 4:33 10
4
yr
1
or 1:374 10
11
s
1
. The number of atoms in 1 g of
radium is
1g
226 g=mol
ð6:022 10
23
Þ¼2:665 10
21
atoms
TABLE 21.13 Some Important Radionuclides and Energies of Dominant Radiation Particle
a
Avg. Particle Nutrient Relative Food
Dominant Energy Analogs and Chain
Particle (MeV) Critical Transport
Isotope Half-Life Emission (% intensity) Tissue (BAF) Source
3
H 12.26 yr b 0.01861 H, whole body High (1) Nuclear weapons,
nuclear power
14
C 5730 yr b 0.156 C NA Natural, nuclear
power
40
K1:28 10
9
yr b 1.35 (1.35%) K, whole body High (1) Natural
32
P 14.3 days b 1.71 P NA Natural
85
Kr 10.76 yr b 0.67 NA NA Nuclear weapons,
nuclear power
87
Rb 4:8 10
11
yr b 0.274 NA NA Natural
90
Sr 28.1 yr b 0.546 Ca, bones High (1) Nuclear weapons
106
Ru 367 days b 0.0392 NA NA Nuclear weapons
129
I1: 6 10
7
yr b 0.189 NA NA Nuclear power
131
I 8.07 days b 0.582 I, thyroid High (10) Nuclear weapons,
power, medical
134
Cs 2.05 yr b 0.499 NA NA Nuclear power
135
Cs 3 10
6
yr b 0.210 NA NA Nuclear weapon
tests
137
Cs 30.23 yr b 0.551 K, whole body High (3.0) Nuclear power
222
Rn 3.82 days a NA None, lungs, Negligible Natural
S, Se?
238
U3:5 10
9
yr a 4.2 GI tract, Low–moderate Natural,
kidney, lung (<1.0) contaminant in
depleted
uranium
projectiles
239
Pu 24,400 yr a 5.149 None, bone, Very low Nuclear weapons
lung (<0.01)
a
Energy of dominant radiation particle given, or weighted average for type of particles. NA, not available.
Source: Based on Connell and Miller (1984); Weast (1979); Newman (1998).
IONIZING RADIATION
833
Then the rate of decay can be computed as
r ¼ kc ¼ð1:374 10
11
s
1
Þð2:665 10
21
atomsÞ¼3:66 10
10
atoms=s
That is, 1 g of radium disintegrates at the rate of 3:66 10
10
atoms per second.
21.8.1 Dosimetry
In measuring radiation, it is necessary to discriminate among emission, exposure, and
dose. Emission is the rate at which particles are produced or energy is released. One dis-
integration per second is termed a becquerel (Bq). The curie (Ci) is defined as
1Ci¼ 3:70 10
10
Bq. Note that this is approximately the number of becquerels emitted
by 1 g of radium. Historically, the Curie was defined in terms of radium decay, although
now it is given the fixed value. Also, note that 1 curie emitted by different radionuclides
can have different amounts of energy. Thus, the number of curies by itself does not indi-
cate the amount of potential harm that might be cause d by a radionuclide. Environmental
concentrations of radionuclides are often expressed in picocuries: 1 pCi ¼ 10
12
Ci ¼
0:037 Bq. For example, radon in drinking water is regulated in terms of pCi/L. The
U.S. EPA has proposed a maximum contaminant level for radon in drinking water of
300 pCi/L and has an action level in ambient air of 4 pCi/L.
Exposure is defined only for electromagnetic radiation such as g -rays. The SI unit is
based on the number of charges of one sign produced by complete absorption in air. The
unit is coulombs per kilogram of air. The older unit of exposure is the roentgen (R):
1 roentgen ¼ 2:58 10
4
C=kg
Exposure can occur due to external sources, such as g-, b-, or x-ray emissions from
outside the body. Alternatively, they may be from internal sources, such as any radionu-
clide that is ingested or absorbed and decays within the body.
The dose refers to the amount of energy actually absorbed. The SI unit is the gray
(Gy), which is 1 J/kg. The older unit is the rad. The conversion is 100 rad ¼ 1 Gy.
Because of the previously mentioned average ionization energy, exposure can be con-
verted into dosage in either system of units assuming 100% absorption of the energy:
1C=kg ¼ 33:85 Gy
1 roentgen ¼ 0:87 radðin airÞ
For tissues this conversion is approximate and may vary with type of tissue and g
energy. Exposure and dose have frequently been confused with each other, partly because
in the units of roentgens and rads they are numerically similar. Exposure refers only to
ionization that would occur in air due to electromagnetic radiation such as g- or x-rays.
It is the radiation that would be measured by an external dosimeter, such as a film badge
clipped to the shirt of a worker. However, it only indirectly indicates how much radiation
has been absorbed by the worker. Dose applies to energy absorbed by any material and
due to any type of radiation.
The differing ability of various emissions to damage living tissue is accounted for by
what is called the relative biological effectiveness (RBE) in biology. It is defined as the
ratio of the toxicological effect of the radiation in question to the effect produced by g
834 TOXICITY OF SPECIFIC SUBSTANCES

radiation of the same dose. The value depends not only on the radiation but on the type of
endpoint (e.g., cancer, cell necrosis). Because the RBE is difficult to determine for human
health effects, the relative rate at which energy is deposited in water by the emission, com-
pared to g radiation, is used in place of RBE. This ratio is called the radiation weighting
factor (W
r
; formerly the quality factor). Thus; g radiation has a W
r
value of 1. The W
r
of
a radiation is about 20. The W
r
for b is also about 1. Th e product of dose and W
r
gives the
dose equivalent (H), which is measured in sieverts (Sv); Gy W
r
¼ Sv. (In the older
measurement units, rad W
r
¼rem. Then 100 rem ¼ 1 Sv.) An interpretation of dose
equivalent is that it is the dose of g radiation that would produce the same amount of tis-
sue damage as the radiation under consideration.
Dose equivalent, however, still does not provide enough information to estimate the
risk of cancer. The reason is that all parts of the body rarely receive the same exposure,
and in the case of ingested radionuclides, they often concentrate in various organs or tis-
sues with varying sensitivities. For example, dent al x-rays are concentrated on the head
and neck area, and ingested radioactive iodine tends to concentrate in the thyroid gland.
To account for factors such as these, dose equivalent is multiplied by a tissue weighting
factor, w
t
, which is the ratio of the cancer risk to the organ per sievert divided by the
cancer risk per sievert to the whole body. The product of dose equivalent and w
t
is called
the effective dose equivalent (H
E
). The w
t
for the gonads is 0.20; for bone marrow, colon,
lung, and stomach it is 0.12; for the bladder, breast, liver, esophagus, and thyroid it is
0.05; for the skin and bone surface it is 0.01; for the rest of the body it is 0.05. The effec-
tive dose for parts of the body are computed separately because the dose received by the
different parts are rarely the same. The sum of the effective dose equivalents for all the
parts of the body is used to compute overall effective dose equivalent. This value is then
used to compute a person’s risk of cancer. Table 21.14 summarizes the units used in radia-
tion dosimetry.
The dosage values described so far are in units of energy deposited per unit mass of
tissue. However, the dosage may decrease with time because of the elimination of the
radionuclide by radioactive decay, by biological elimination, or by both. The decrease
TABLE 21.14 Units and Conversion Factors Used in Radiation Health Measurements
a
Fundamental Units Traditional Units SI Units
Disintegration Rate
1 disintegration/s 1 becquerel (Bq) 27.0 picocuries (pCi)
3:70 10
10
Bq 1 curie (Ci)
Exposure (g- or X-Rays): Number of Charges of One Sign Produced by Complete Absorption in Air
2:58 10
4
C/kg 1 roentgen (R) 2:58 10
4
C/kg
3876 R 1 C/kg
Dose: Amount of Energy Actually Absorbed
1 J/kg 100 rad 1 gray (Gy)
Dose Equivalent: Dose RBE or Dose W
r
: Capability to Damage Living Tissue
100 rem 1 sievert (Sv)
Effective Dose Equivalent: Dose Equivalent Tissue Weighting Factor
100 rem 1 sievert (Sv)
a
Units on the same row are equal to each other.
IONIZING RADIATION
835

is a first-order process, with an effective rate constant, k
E
, equal to the sum of the rate
constants for radioactive decay, k
R
, and for biological elimination, k
B
. The initial rate at
which energy is being deposited in a tissue is D
0
. Then the amount that will be deposited
in the first t time units will be
D ¼
D
0
k
E
½1 expð k
E
tÞ ð21:3Þ
The dose that is deposited over a lifetime (assumed to be 50 years for adults and
70 years for exposure to children) is called the committed effective dose (CED).
For an infinite time period, or for practical purposes when the lifetime is four or
more times the effective half-life (1=k
E
), the committed effective dose is equal to the
total dose:
D ¼
D
0
k
E
ð21:4Þ
Internal nuclides with long effective half-lives do not change appreciably in concentra-
tion in the body over a person’s lifetime . In such cases it is appropriate to expressed the
dosage as a rate of energy delivered per mass of tissue per unit time, D
0
.
Example 21.2
131
Iisab emitter with a half-life of 8 days and a 180-day biological half-
life in the thyroid. Thus, k
R
¼ 0:693=8:07 days ¼ 0:0859day
1
and k
B
¼ 0:693=
180 days ¼ 0 :00385 day
1
. Then k
E
¼ 0:0897 and the effective half-life is 7.72 days.
About 60% of the iodine in the body concentrates in the thyroid gland. If a person receives
3 mCi of
131
I, what will be the effective dose rate to the thyroid? What will be the effec-
tive dose delivered over the first 15 days, and what will be the total dose?
Answer From Table 21.13,
131
I produces 0.582 MeV per disintegration; 60% of the
total dose results in ð0:6Þð3ÞmCi ¼ 1:8 mCi dose to the thyroid. Thus, the dose rate will
be
ð1:8 mCiÞ
3:7 10
4
Bq
mCi
0:582 MeV
Bq s
1:6 10
13
J
MeV
86; 400 s
day
¼ 5:3610
4
J=day
This is also the dose equivalent, H, since the radiation weighting factor can be taken as 1.0
for b emiss ions. Also, since we are dealing with a known dose to an organ rather than a
whole-body dose, a tissue weighting factor is not needed and the dosage we are computing
is the effective dose equivalent, H
E
. Assume that the mass of the thyroid is 25 g. Since the
penetration depth of b emissions is much smaller than the thyroid, one can assume that all
the energy is deposited in that organ. Thus, the initial dose rate, D
0
, will be
D
0
¼
5:36 10
4
J=day
0:025 kg
¼ 0:0214 Gy=day ¼ 0:0214 Sv=day
The dose delivered over the first 15 days will be
D ¼
0:0214 Sv=day
0:0897 day
1
½1 expð 0 :0897 day
1
Þð15 dayÞ ¼ 0:177 Sv
836 TOXICITY OF SPECIFIC SUBSTANCES

Over the long term, the total dose will be
D ¼
0:0214 Sv=day
0:0897 day
1
¼ 0:239 Sv
Dosimetry calculations can easily become complicated by additional factors not con-
sidered here. The distribution of radionuclides can be described by pharmacokinetic mod-
els to more accurately describe their spatial and temporal distributions in the b ody.
Conversion of exposure to absorbed dose can be complicated by external and internal
shielding, which can be difficult to quantify. Often, it is necessary to take into account
not only a particular radionuclide, but its daughters as well. Radon is one example
where this is critical (see below). In such cases the daughters can have greatly different
emission characteristics and pharmacokine tics. Once the radionuclide loading to a parti-
cular tissue is determined, it may be necessary to consider secondary forms of radiation as
the energy interacts with the matter (e.g., neutrons or positrons). It may be necessary to
examine radiation to tissues adjacent to the organ containing the radionuclide. An exam-
ple of the latter is that some of the radioactive isotopes of strontium may deposit at the
inner surface of hard bone tissue, where their emissions can irradiate the sensitive bone
marrow tissue. Many radioisotopes produce more than one type of emission. For example,
some 8% of the emission from
131
Iisg radiation. In the case of g-ray dosimetry from
internal emitters, the spatial distribution of the nuclides affects the absorbed dose. This
can be taken into account either by detailed integration or by use of predetermined
geometry factors.
21.8.2 Radiation Exposure and Risks
The major sources of radionuclides in the environment are natural sources, fallout from
nuclear detonations in the atmosphere, and sources associated with nuclear power. Natural
sources include a number of minerals, such as
238
U and its derivatives (Figure 21.4),
40
K
and the gas tritium (
3
H), and
14
C, which is continually produced by cosmic ray bombard-
ment of the atmosphere. One of the more important by-products of
238
U is radon ,
222
Rn.
The natural backgr ound radiation to which the public is exposed is about 3 mSv per year,
as shown in Table 21.15.
Inhalation of radon is the largest source of natural exposure. Outdoor radiation in the
United States has been measured at an average of 7 Bq/m
3
(0.19 pCi/L); indoor levels tend
TABLE 21.15 Estimated Total Effective Dose
from Several Forms of Natural Radiation for the U.S.
and Canadian Populations
Source Dose Rate (mSv/yr)
Cosmic 0.27
Cosmogenic 0.01
Terrestrial 0.28
Inhaled 2.0
In body 0.40
Total 3.0
Source: Harley (1996).
IONIZING RADIATION
837
