Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

21.2.1 Toxic Effects
Tables 21.4 and 21.5 contains toxicity data on some pesticides. Most insecticides act by
interfering with the nervous system. For example, DDT binds to lipoproteins in axon
membrane, holding a gate open for sodium. Organochlorines in general stimulate the ner-
vous system, causing irritability, tremor, and convulsions.
OP and carbamate inhibit acetylcholinesterase. In the Central nervous system (CNS)
they cause tremor and convulsion. In the autonom ic nervous system they cause diarrhea,
involuntary urination, and bronchoconstriction. At the neuromuscular junction they cause
muscle contraction, weaknes s, and loss of reflexes, followed by paralysis. Chronic expo-
sure causes neuropathy (destruction of nerve fibers). Motor effects predominate, includ-
ing muscle wasting, difficulty with movement, and inability to locate one’s joints in space,
making walking difficult. Exposure to OPs were observed in both human and animal tests
to result in myopathy (damage to muscle tissue), including necrosis of skeletal muscles.
This is thoug ht to be caused by the high levels of acetylcholine resulting from inhibition
Dichlorodiphenyltrichloroethane (DDT)
Chlordane
Lindane (a fungicide)
Dieldrin
2,4-dichlorophenoxyacetic acid
(2,4-D)
C
HLORINATED INSECTICIDES
Cl
H
C
CCl Cl
Cl
Cl
CCl
2
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
CCl
2
Cl
Cl
Cl
Cl
CH
2
O
CHLORINATED HERBICIDES
OCH
2
COOH
Cl
Cl
OCH
2
COOH
Cl
Cl
Cl
2,4,5-trichlorophenoxyacetic acid
(2,4,5-T)
Figure 21.1 Structures of some organochlorine pesticides.
808
TOX ICITY OF SPECIFIC SUBSTANCES
Parathion
Malathion
Carbar
y
l (Sevin) Atrazine (a triazine used as a herbicide)
Glyphosate − a herbicide
O
RGANOPHOSPHATE PESTICIDES
SP
O
O
ONO
2
C
2
H
5
C
2
H
5
SP
O
O
SCH
CH
3
CH
3
H
2
C
C
C
O
O
C
2
H
5
C
2
H
5
O
O
OHCCH
2
NCH
2
PO
OH H
OOH
NITROGEN-BASED PESTICIDES
CARBAMATE INSECTICIDES
N
N
N
Cl
NHCH
2
NH CH
CH
3
CH
3
H
3
C
OCNHCH
3
O
Figure 21.2 Structures of some unchlorinated pesticides.
TABLE 21.4 Mammalian Toxicity of Some Insecticides
a
NOEL (mg/kg)
LD
50
ADI
Pesticide (mg/kg) Rat Dog Human (mg/kg)
Diazinon 108 0.1 0.02 0.02 0.002
Dichlorvos 80 NA NA 0.033 0.004
Malathion 1375 5.0 0.2 NA 0.02
Parathion 13 NA NA 0.05 0.005
Trichlorfon 630 2.5 1.2 NA 0.005
Aldicarb 0.8 0.125 0.25 0.005
Carbaryl 850 10 NA 0.06 0.01
DDT 113 0.05 NA NA 0.01
Aldrin/dieldrin 40 0.025 0.025 NA 0.001
Chlordane 335 0.25 0.075 NA 0.001
Endrin 18 0.05 0.02 NA 0.0002
Heptachlor 100 0.25 0.0625 NA 0.0005
Lindane 88 1.25 1.6 NA 0.01
Methoxychlor 6000 10 NA NA 0.1
a
NA: Not available.
Source: Lu (1991).
PESTICIDES
809

of acetylcholinesterase. Symptoms include muscle tenderness. Adverse psychological dis-
turbances have been identified which lasted from six months to a year. These included
depression, nightmares, and emotional instability.
There is positive interaction between many OPs. One of the strongest is a 100-fold
increase in the combined toxici ty of malathion and tri-o-cresyl phosphate (TOCP).
Some pesticide formulations include potentiators to increase their effect, although this
TABLE 21.5 Acute Aquatic Toxicity of Example Pesticides
a
Photosyn. % Daphnia
Pesticide Red.
b
48-h EC
50
Amphipod Stonefly Naiad Fish Tadpole
Organochlorine
Aldrin 85 28 9800 1.3 NA 150
Chlordane 94 29 26 15 69 NA
DDT 77 0.36 1.0 7.0 34 800–1000
Dieldrin 85 250 460 0.5 16 100–150
Endrin 46 20 3.0 0.25 1.3 120–180
Heptachlor 94 42 29 1.1 56 440
Lindane 28 460 48 4.5 56 2700–4400
Methoxychlor 81 0.78 0.8 1.4 35 330
Toxaphene 42 15 26 2.3 5.1 140–500
Organophosphorus
Diazinon 7 0.9 200 25 NA NA
Dibrom 56 0.35 110 8.0 NA 1700
Dichlorvos NA 0.066 0.5 0.1 NA NA
Malathion 7 1.8 1.8 10 12,500 200–420
Methyl parathion NA 0.14 NA NA 7,500 NA
Parathion NA 0.6 0.6 5.4 1,600 1000
Phosdrin NA 0.16 0.16 5.0 NA NA
Herbicides
Dinitrocresol NA 0.014
c
NA 0.32 NA NA
Diquat NA NA NA NA 91
d
NA
2,4-D acid NA 1.4 NA 15 NA NA
2,4-D butoxyethanol NA >100
c
0.44 1.6 2.1
d
NA
ester
Naphtha NA 3.7 0.84 2.8 NA NA
Paraquat NA 3.7 11 >100 400
d
28
Picloram NA NA 27 48 26.5
d
NA
Silvex NA 2 NA 0.34 83
e
10
Trifluralin NA 0.24 2.2 3.0 8.4
d
0.1
e
2,4,5-T NA NA NA NA 0.5
d
NA
a
The fish are freshwater fathead minnow for OC and OP pesticides, and bluegill for the herbicides; 96-h LC50
except where noted. Concentrations in mg/L for organochlorine and organophosphorus pesticides and mg/L
for herbicides. NA, not available.
b
Reduction of estuarine phytoplankton photosynthetic productivity during 4 h exposed to a concentration
of 1.0 mg/L.
c
26-h EC
50
.
d
48-h LC
50
.
e
24-h LC
50
.
Source: Connell and Miller (1984).
810 TOXICITY OF SPECIFIC SUBSTANCES
increases mammalian toxicity. Antagonistic effects occur as well. Substituted urea herbi-
cides reduce the toxicity of parathion, probably by inducing MFO detoxification.
OP and carbamates are not carcinogens, in general, except some that are chlorinated, or
some like carbaryl, which can be nitrosated to a carcinogenic form. All organochlorines
cause hepatoma in mice. DDT is controversial because it does not cause cancer in rats,
hamsters, and a few other animals. Only for hexachlorocyclohexane is there epidemiolo-
gical evidence of cancer in humans. The fumigants ethylene dibromide (EDB) and 1,2-
dibromo-3-chloropropane (DBCP) cause stomach cancer in rats and mice and have been
restricted. The herbicide amitrole (aminotriazole) and the ethylenebisdithiocarbamates
(such as maneb) produce thyroid tumors. Carbaryl (sevin) is a teratogen in dogs but
not in other animals. The dithiocarbamate fungicides, captan, paraquat, and others, are
reportedly teratogenic. The herbicide 2,4,5-T is known to cause teratological effects in
rats.
Carbaryl produced effects attributed to decr eased renal resorption in human experi-
ments. Paraquat causes edema, hemorrhage, and fibrosis in the lungs when inhaled. Pyr-
ethrum is associated with hypersensitivity, contact dermatitis, and asthma. OCs are
hepatotoxic and induce microsomal monooxygenases. Some pesticides from all the
major groups have an effect on the immune system. These include malathion, methylpar-
athion, carbaryl, DDT, paraquat, and diquat.
Herbicides act on plants by affecting either growth or photosynthesis. The phenoxya-
cids simulate natural auxins (plant growth hormones). Urea herbicides inhibit photosys-
tem II, preventing ATP and NADPH formation. Diquat and paraquat inhibit photosynthesis.
Some are selective for either monocots (grassy plants) or dicots (broadleaf).
The manufacture of the pesticide 2,4,5-T produces 2,3,7,8-tetrachlorodibenzo-
p-dioxin (TCDD) as a trace impurity. This is one of the most toxic substances known.
The manufacture and use of 2,4,5-T and related herbicides has resulted in some of the
most notorious environmental contamination incidents known, which involve TCDD
(see Section 21.4).
Dinitrophenols are used for weed control. They uncouple oxidative phosphorylation in
respiration, causing metabolic activity to increase out of control. Effects of acute exposure
in humans include the sensation of heat, and rapid breathing and heart rate. Chronic symp-
toms include anxiety, sweating, thirst, and fatigue. Paraquat causes lung, kidney, and liver
damage and can cause pulmonary fibrosis even from routes other than inhalation.
Fate and Transport: Dieldrin depuration half-life ranges from 148 days for the guppy
Lebistes reticulatus to 12.2 days for tubificid worms (Tubifex sp.). Depuration half-lives
are correlated with lipophilicity. Plants cannot excrete toxins, but instead, segregate them
in vacuoles within the cells. Table 21.6 summarizes some of the environmental fate and
transport properties for a number of pesticides.
21.2.2 Ecosystem Effects
Biomagnification is a problem mostly with organochlorine pesticides. The classic case is
DDT and its metabolite DDD. Figure 18.9 illustrates two examples of organochlorine bio-
magnification.
By their very nature, pesticides can produce pronounced effects on populations and
ecosystems. These include many that were dramatically publicized by Rachel Carson
in her book Silent Spring, including fish and bird kills, development of insect resistance,
and actually causing an increase in some pests by destroying their predators. In the latter
PESTICIDES 811

case, the target species is sometimes the beneficiary of a pesticide application, if its pre-
dators and competitors are more strongly affected. An example of this counterproductive
situation is the application of pesticide to control boll weevils and leafworms infesting
cotton farms in Central America. From 1950 to 1955 the number of pesticide applications
had to increase from few or none to 8 to 10 per season. By 1960, 28 applications per sea-
son were needed. Over the same period, the number of pest species increased from the
original two in 1950, to five in 1955, and then eight in 1960. Many similar examples exist.
Many pesticides, such as insecticides, fungicides, and fumigants, target soil organisms
and have their greatest environmental effect on nontarget organisms in the soil. Suscep-
tible invertebrates include insects, earthworms, slugs, and gastropods. Some of these
organisms contribute to topsoil formation. Insecticides applied to natural systems, such
as for the control of forest pests, can reduce the species diversity within the invertebrates.
Among the terrestrial vertebrates, avian species are especially susceptible to the insec-
ticides. LD
50
values are often less than 100 mg/kg. However, the subleth al effects are of
greatest concern. In particular, bird reproduction is very sensitive to organochlorine insec-
ticides. The best known effect is the thinning of eggshells due to DDT, its metabolite
DDE, and dieldrin, among others. Th ese prevent the embryos from surviving to hatch.
However, many other reproductive effects are also found, including direct embryo toxicity
TABLE 21.6 Fate and Transport Properties of Some Pesticides
Aqueous Log
10
BCF Typical
Pesticide Solubility (mg/L) Log
10
K
OW
(Fish) (L/kg) Environmental Half-Life
Organochlorines 1–12 years
2,3,7,8-TCDD 0.0002 6.15 4.73
DDT 0.0012 5.98 4.78
Aldrin 0.01
Chlordane 0.056 5.15 4.05 >100 days
Heptachlor 0.056
Dieldrin 0.18 5.48 3.76
Lindane 7.0 4.82 2.51 >100 days
Organophosphates 72–180 days
Chlorpyrophos 0.3 4.99 2.65
Parathion 24
Diazinon 40
Malathion 145 <30 days
Dimethoate 2,500
Carbamates 20–365 days
Carbaryl 40
Carbofuran 700
Pyrethrin <1day
Herbicides
Simazine 5.0 30–100 days
Atrazine 34 30–100 days
2,4,5-T 280 0.6 1.63 <30 days
2,4-D 890 1.57 1.30 <30 days
Glyphosate 12,000 30–100 days
Diquat (70%)
Dalapon (80%)
Source: Rand and Petrucelli (1985); LaGrega et al. (2001); Rao et al. (2004); Ferruzzi and Gan (2004).
812 TOXICITY OF SPECIFIC SUBSTANCES
and aberrant parental behavior such as destruction of eggs. The greatest concern with
these effects is with birds of prey, because the organochlorine pesticides are biomagnified.
Species that have experienced serious population declines include the bald eagle (Halia-
cetus leucophalus) and the osprey (Pondion haliatetus).
DDT has not been found to seriously affect wild mammals. However, other organo-
chlorine pesticides, especially the cyclodienes such as heptachlor, dieldrin, and endrin ,
have resulted in mortalities. Although the cholinesterase inhibitors are very toxic to
mammals, no significant effects have been observed in the wild. Development of resis-
tance has been observed in a variety of aquatic invertebrates.
Aquatic ecosystems have been affected both by direct application of pesticides, such as
for control of mosquitoes, and by runoff from agricultural, forest spraying, and so on.
Arthropods such as crustraceans are very susceptible to insecticides. Heavy killings of
crabs and shrimp have occurred in marshes sprayed with organochlorines. The microcrus-
taceans are an important component of the food chain and are also affected heavily.
Mollusks and annelids, on the other hand, are relatively tolerant. Fish tend to be very sus-
ceptible to the organochlorines, especially the cyclodienes. Some warm-water, rapidly
reproducing speci es of fish in heavily sprayed areas of Mississippi have become resistant
to levels of some of the organochlorines 100 times higher than that which is normally
toxic.
Chronic toxicity is repor ted as the MATC in Section 19.7. Application factors (LC
50
/
MATC) for chronic toxicity vary greatly. For Kepone the application factor for sheeps-
head minnows ranges from 0.001 to 0.002. However, for endrin the AP is 0.35 to 0.91.
In other words, Kepone causes chronic effects at concentrations as much as 1000 times
lower than the LC
50
, whereas the chron ic effects of endrin do not appear until the con-
centration reaches one-third of the LC
50
.
Pesticides interact with each other, often positively. The toxicity of pesticides to aqua-
tic organisms als o interacts with the abiotic factors temperature and pH, as described in
Section 17.3. Both positive and negative effects of temperature have been observed. End-
rin was found to be several hundred times as toxic to carp ( Cyprinus carpio )at27
Casit
was at 7
C. DDT and methoxychlor, on the other hand, were more toxic at lower tempera-
tures. Hardness does not seem to produce an effect independent of pH, and many of the
pH effects are associated with ionizable pesticides or those that react to form other pro-
ducts at certain pH ranges.
21.3 HYDROCARBONS, SOLVENTS, PAHs, AND SIMILAR
COMPOUNDS
Here we consider other anthropogenic organic chemicals and their by-products. Solvents
include many types of liquid hydrocarbons, which are used as carriers for other materials,
such as pigments in paints, or for rinsing oil and grease from manufactured items. Some,
such as benzene, may also be components of fuel or raw materials for synthesis of other
compounds. Some of the larger molecules we consider include the polynuclear aromatic
hydrocarbons (PAHs), phthalate esters, and surfactants. Chlorinated organics are consid-
ered separately in the next section.
Many hydrocarbons are biotransformed by cytochrome P450 enzymes. Thus, they can
interact with other toxins either positively by increasing cytochrome activity (as benzene
does) or negatively by competing for the enzyme (as toluene does with benzene).
HYDROCARBONS, SOLVENTS, PAHs, AND SIMILAR COMPOUN DS 813
The solvents are hydrocarbons up to five or six carbons in size. They have appreciable
vapor pressures, which may result in significant inhalation exposure. Solvents are also
absorbed significantly through the skin in occupational settings. The larger compounds
are more likely to be adsorbed to particles. Exposure can result from inhalati on or inges-
tion of the particles. Some of them enter the food chain, where they can be bioaccumu-
lated and ingested.
The major toxic effects common to these compounds are CNS depression, including
narcosis, and irritati on. These are of special concern with acute exposure to the solvents.
However, these types of compounds can cause other effects, ranging from liver or kidney
damage to carcinogenesis. CNS depression is essentially the action of a general anaes-
thetic. The potency of organics to produce CNS depression increases with the chain length
of the compound. Halogenation, addition of an alcohol group, or unsaturation (removal of
hydrogen to form a double carbon–carbon bond) increase potency as well.
One mechanism of irritation by hydrocarbons and surfactants is by the extraction of
fats from the skin, lungs, eyes, or other cell membranes contacted. Addition of functional
groups to an organic molecule tends to increase irritant properties. Amines and acidic
groups make the compound corrosive, and alcohol, aldehyde, and ketone groups can pre-
cipitate and denature proteins associated with the membr anes.
The aliphatic (saturated) hydrocarbons have relatively less toxicity than others do in
this group. Ingestion of more than 1 mL/kg can produce systemic effects. For lower
amounts, aspiration to the lungs is the principal concern. Chronic exposure, such as to
hexane or heptane, produces neuropathy, probably due to metabolism to alcoholic and
ketone forms. Olefinic (unsaturated) forms are stronger CNS depressants. Interestingly,
the presence of doubl e bonds eliminates the neurotoxicity of hexane and heptane. The
cyclic hydrocarbons, such as cyclohexane, are similar to the open-chain forms, except
that they are higher in irritancy, and do not seem to produce chronic effects.
Alcohols, including glycols, are much stronger CNS depressants than aliphatics are and
slightly mor e irritating. As carbon chain length increases, irritation decreases but lipophi-
licity increases, as does systemic toxicity. Methanol is less inebriating than ethanol but
has the unusual property of destroying the optic nerve. Fifteen milliliters can cause blind-
ness. As with ethanol, it is metabolized by a zero-order rate mechanism, but at one-
seventh the rate. Ethanol acts as an irritant by dehydrating protoplasm. An initial stimu-
lant effect is caused by depression of control mechanisms in the brain. Pain sensitivity is
greatly reduced. Cutaneous (skin) blood vessels become dilated. The resulting increased
heat loss can be dangerous in cold weather. It increases gastric secretion, which can aggra-
vate stomach ulcers. It causes fat accumulation and cirrhosis in the liver. The latter can be
fatal itself or can cause progression to cancer. Ethanol increases urine flow through a
mechanism involving pituitary and adrenal hormones. The resulting water loss, along
with the acetaldehyde by-product, may be a cause of the headache in a hangover. On
the other hand, there is evidence that consumption of small amou nts of alcoholic bev-
erages with meals may have some benefits for the cardiovascular system.
Isopropyl alcohol is less toxic than n-propanol, but both these and butanol are more
toxic than ethanol. Allyl alcohol is highly irritating. It can be absorbed through the
skin, resulting in deep pain and possible burns of the eye. Glycols are compounds that
have two hydroxyls on adjacent carbons. They are less toxic than the monohydroxy alco-
hols. Ethylene glycol can be fatal to humans with a single dose of 100 mL. It is biotrans-
formed to oxalic acid, which blocks kidney function. Ethanol can inhibit this
transformation, making it an antidote.
814 TOXICITY OF SPECIFIC SUBSTANCES

Aldehydes tend to be more irritating than they are CNS depressants. One unique toxi-
city of the aldehydes, especially formaldehyde, is sensitization. That is, it can increase a
person’s response to other chemicals. Formaldehyde is a common industrial chemical
used in plastics and resins. The LD
50
in humans for formalin (37 to 50% formaldehyde
solution) is about 45 g, although deaths have been reported at as low as 30 g. Ingestion can
produce headaches, GI tract corrosion, pulmonary edema, fatty liver, kidney necrosis,
unconsciousness, and vascular collapse. Formaldehyde has been associated with muta-
genicity and carcinogenicity in laboratory tests, but a steep dose–response relationship,
the lack of epidemiological evidence for carc inogenicity in humans, plus the following
facts, suggest the presence of a carcinogenicity threshold. Formaldehyde is a metabolic
by-product, normally present at several ppm in tissues. Thus, it appears that carcinogeni-
city is associated with exposures high enough to cause irritation and tissue injury. Acet-
aldehyde is less irritating and toxic. Acrolein is an unsaturated propionaldehyde. The
double bond greatly increases its reactivity as well as its irritant and toxic effect.
Ketones produce fewer occupational health problems, possibly because their irritant
properties serve as a warning before other effects occur. Acetone causes skin irritation
only after repeated lengthy contact. Eye irritation occurs with unacclimated persons at
air concentrations of 500 ppmv, and repeated exposure can produce tolerance up to
2500 ppmv. Carboxylic acids are mostly irritants. Acidity, and irritation, decreases with
chain length and increases with halogenation. Esters are stronger anesthetics than alco-
hols. The lower-carbon esters are stronger irritants than alcohols and can cause lacrima-
tion (eye tearing). Phosphate esters are used as plasticizers and can cause CNS damage.
Ethers are effective anesthetics and slightly irritat ing.
Amines have many uses, including as disinfectants in consumer products. They include
some of the most toxic common industrial chemicals. Most simple amines have pK
a
values between 10.5 and 11.0, making them ionized at physiological pH and corrosive
to tissues.
R
H
H
NR
1
R
2
H
NR
1
R
2
R
3
primary
amine
secondary
amine
tertiary
amine
An important property of amines is that they are easily absorbed by all routes of expo-
sure, including the skin. Thus, the dermal toxicity is often similar to the ingested toxicity
(Table 21.7). Systemic effects include pulmonary edema and hemorrhage, liver and
TABLE 21.7 Toxicity of Some Amines in Animal Tests (mg/kg)
Amine Oral LD
50
Skin LD
50
Methylamine 0.02 0.04
Ethylamine 0.4 0.4
Propylamine 0.4 0.4
Butylamine 0.5 0.5
Hexylamine 0.7 0.5
Source: Williams and Burson (1985).
HYDROCARBONS, SOLVENTS, PAHs, AND SIMILAR COMPOUN DS
815
kidney necrosis, and coronary degeneration. They react to form methemoglobin in the
blood. Repeated exposure can produce strong allergic reactions. Aromatic amines or
diphenylamine comp ounds are carcinogens, such as benzidine, 2-naphthylamine, or 4-
aminodiphenyl, which cause ‘‘aniline tumors’’ in dye industry workers. Any alkyl
amine (such as those that can be produced in digestion of food) can react with nitrites
in food and acid in the stomach to form nitrosamines, which can potentially cause liver
cancer.
Analine itself is an important industrial compound that is also used in household pro-
ducts such as polishes, paints, and inks. It is considered very toxic and is easily absorbed
through the skin. In the blood, aniline oxidize s Fe(II) to Fe(III), turning the blood to a
brown-black color and eliminating its ability to transport oxy gen.
Nitro compounds are mild irritants. Unsaturated compounds are easily absorbed der-
mally. Nitrated aromatics produce a variety of effects, including methemoglobinemia.
Some produce bladder tumors. Trinitrotoluene and dinitrobenzene are easily absorbed
by all routes of exposure. Besides the other toxic effects, they can uncouple oxidative
phosphorylation and cause liver damage. Nitriles (compounds with structure RC
N)
are easily absorbed, and many slowly degrade to cyanide. Acetonitrile is considered
moderately to very toxic. Acrylonitrile is classified as highly toxic and is a suspected
human carcinogen.
H
2
C
CHC
N
acrylonitrile
Single-ring aromatics include benzene and its derivatives. Aromatics tend to be more
toxic than aliphatics with a similar number of carbons. CNS depression caused by aro-
matics differs qualitatively from that produced by aliphatics. Coma induced by aliphatics,
such as by gasoline, exhibit inhibited reflexes. On the other hand, aromatics produce
hyperactive reflexes, sometimes with convulsions.
Benzene is reportedly fatal at about 10 to 15 mL for an adult, and possibly as low as 2
to 5 mL. Inhalation at 250 ppmv produces headache, nausea, vertigo, and drowsiness;
3000 ppmv in air is irritating to the eyes and respirato ry tract. Acute symptoms appear
after 30 to 60 minutes at 7500 ppmv; 20,000 ppmv for 5 to 10 minutes has been reported
to be fatal. Acute symptoms are respiratory inflammation, pulmonary edema and hemor-
rhage, renal congestion, and cerebral edema. Chronic dermal exposure can produce
damage resembling first- or secon d-degree burns. Blood disorders progress through
three stages: First is a reversible reduction in blood clotting and a mild anemia. Continued
exposure produces increased leukocyte production. Finally, a more severe anemia and
hemorrhage occurs. Less com monly, benzene also causes a cancer, leukemia. It is a
class A carcinogen. Chomosome damage has also been observed in humans. Toluene is
a stronger CNS depressant than benzene but is not known to cause permanent blood dis-
orders. The xylenes are even strong er CNS depressants than toluene.
Phenols are cytotoxic, acting as a local anesthetic and CNS depressant. They are easily
absorbed but produce strong irritation and burns. Phenol easily penetrates the skin, where
it causes deep burns. Burns from ingestion can lead to extreme shock and death. Phenol
can also cause necrosis systemically in the liver and kidneys as well as the heart and urin-
ary tract. Death from systemic poisoning is usually from respiratory depression as a result
of its CNS depression activity. Ingestion of as little as 1 or 2 g reportedly can be fatal. The
816 TOXICITY OF SPECIFIC SUBSTANCES
dihydroxyphenols catechol, resorcinol and hydroquinone, and cresol, behave similarly to
phenol, except that they are more toxic.
OH
OH
HO
OH
H
3
C
benzene
phenol
hydroquinone
p-cresol
Polynuclear aromatic hydrocarbons (PAHs, also referred to as PNAs or polycyclic aro-
matic hydrocarbons) are fused benzene rings and their derivatives. Many are prevalent in
coal and crude oil. They also are significant produc ts of partial combustion; that is, they
are found in soot and smoke particles, and in pyrolytically produced tars and oils. The
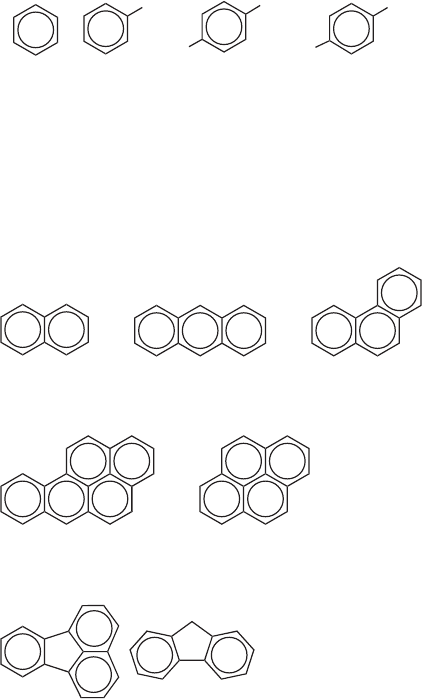
simplest PAH is naphthalene.
naphthalene
anthracene phenanthrene
pyrene
benzo[a]pyrene
fluorenefluoranthene
PAHs are strongly absorbed to soil and sediment particles in the environment and can
even be transported long d istanc es by the wind. Aqueous solubility decreases with molar
mass and is lower for linear ring arrangements than more condensed forms (compare
anthracene and phenanthrene in Table 21.8). In aquatic environments, PAHs tend to
be most concentrated in sediments, least in the water column, and intermediate in organisms.
Some bacteria and fungi can mineralize some PAHs, but others can only cometabolize
them. Fungi and some animals utilize the cytochrome P450 (MFO) system to hydroxylate
PAHs to diols. Certain intermediates of this conversion in mammals are thought to be
responsible for carcinogenicity in this group. Injection of PAHs in fish can induce
MFO activity severalfold within several days.
PAHs cause acute toxic effects in humans only at high doses that are not common
in environmental or occupational settings. Napthalene inhalation cause s headache,
confusion, nausea, and profuse perspiration in humans. For a variety of marine and
HYDROCARBONS, SOLVENTS, PAHs, AND SIMILAR COMPOUN DS 817
