Versteeg H., Malalasekra W. An Introduction to Computational Fluid Dynamics: The Finite Volume Method
Подождите немного. Документ загружается.


12.11 REACTION RATE 357
For example, consider the simple reaction
2H
2
+ O
2
→ 2H
2
O (12.57)
For this reaction the total number of species involved is N = 3; the species
involved are M
1
= H
2
, M
2
= O
2
and M
3
= H
2
O.
Considering the left hand side of equation (12.57) we have
Stochiometric coefficient
ν
′
1
for H
2
= 2
Stochiometric coefficient
ν
′
2
for O
2
= 1
Stochiometric coefficient
ν
′
3
for H
2
O = 0, no H
2
O on the left hand
side of the equation
Considering the right hand side of equation (12.57) we have
Stochiometric coefficient
ν
″
1
for H
2
= 0, no H
2
on the right hand
side of the equation
Stochiometric coefficient
ν
″
2
for O
2
= 0, no O
2
on the right hand
side of the equation
Stochiometric coefficient
ν
″
3
for H
2
O = 2
The mass conservation (12.56) can be verified for this equation as
Left hand side (2 × 2 + 1 × 32) = (2 × 18) Right hand side
The law of mass action (see Kuo, 2005; Turns, 2000), which has been
confirmed by experimental observations, states that the rate of consumption
of a chemical species in a reaction is proportional to the (mathematical) prod-
uct of the concentration of the reacting species, each concentration being
raised to the powers corresponding to the stoichiometric coefficients of the
reactants.
The reaction rate RR (units kmol/m
3
.s) for the reaction (12.55) is given
by (see Kuo, 2005)
RR ==k
f
(C
M
k
)
v ′
kj
(12.58)
Since species M
k
appear on both sides of equation (12.55), the net progress
rate of reaction j is given by the production of the species M
k
minus the
destruction of the species M
k
:
L
kj
==(
ν
″
k
−
ν
′
k
)k
f
(C
M
k
)
v ′
kj
(12.59)
Here C denotes the molar concentration of the species in units kmol/m
3
(or gmol/cm
3
in CGS units) and the proportionality constant k
f
is called the
specific reaction rate constant. It should be noted here that the subscript k
used to identify species is different from this k. The reaction rate constant k
f
is independent of the concentration and is usually expressed as
k
f
= AT
α
exp − (12.60)
This is known as the Arrhenius law. In this expression A is called the
pre-exponential constant. Parameter
α
is a temperature exponent and E
a
is
the activation energy. These constants are specific to a given reaction. R
u
is the universal molar gas constant (= 8.314 kJ/kmol.K) and T is absolute
D
E
F
E
a
R
u
T
A
B
C
N
∏
k=1
dC
M
k
dt
N
∏
k=1
d(C
product
)
dt
ANIN_C12.qxd 29/12/2006 04:44PM Page 357
358 CHAPTER 12 CFD MODELLING OF COMBUSTION
temperature. A note about units is required here. Consider the simple
forward-only reaction
aA + bB → cC + dD (12.61)
The progress rates of reactant consumption and product formation for this
reaction can be evaluated using equation (12.59) as
L
A
==−ak
f
(C
A
)
a
(C
B
)
b
(12.62)
L
B
==−bk
f
(C
A
)
a
(C
B
)
b
(12.63)
L
C
==ck
f
(C
A
)
a
(C
B
)
b
(12.64)
L
D
==dk
f
(C
A
)
a
(C
B
)
b
(12.65)
Note that equation (12.61) is a forward-only reaction. When a backward
reaction is also present similar expressions can be written using a backward
reaction rate constant k
b
. The units of C
A
and C
B
are kmol/m
3
. The units
of the progression rate are kmol/m
3
.s. Because of the involvement of
exponents in equations (12.59) it can be seen that the units of k
f
, which is
given by equation (12.60), depend on the stoichiometry of the reaction. For
the reaction (12.61), the sum of the coefficients a and b is called the reaction
order n = a + b. E
a
has units kJ/kmol. RT has the same units. The exponen-
tial term −E
a
/RT does not contribute to units. The factor T
α
has units
depending on the value of
α
(see Turns, 2000). Therefore for this particular
reaction the constant A has units (m
3
/kmol)
n−1
/(K
−
α
s). The units of k
f
are
(m
3
/kmol)
n−1
/s. In most cases
α
is zero, in which case the units required for
k
f
are the same as those for A (see e.g. Henson and Salimian, 1984).
For a simple reversible reaction of the form
aA + bB bcg cC + dD (12.66)
the progress rate of consumption of the reactant species A in equation
(12.66) is
L
A
==−a[k
f
(C
A
)
a
(C
B
)
b
− k
b
(C
C
)
c
(C
D
)
d
] (12.67)
Similarly for a large scheme of reactions of the form
ν
′
kj
M
k
bcg
ν
″
kj
M
k
for j = 1, 2,..., m (12.68)
the most general form of the reaction progress rate for species k by reaction
j is given by
L
kj
=
j
= (
ν
″
k
−
ν
′
k
) . k
f
(C
M
k
)
ν
′
kj
− k
b
(C
M
k
)
ν
″
kj
(12.69)
J
K
L
N
∏
k=1
N
∏
k=1
G
H
I
dC
M
k
dt
N
∑
k=1
N
∑
k=1
dC
A
dt
dC
D
dt
dC
C
dt
dC
B
dt
dC
A
dt
ANIN_C12.qxd 29/12/2006 04:44PM Page 358

12.11 REACTION RATE 359
where k
f
is the forward reaction rate constant and k
b
is the backward reaction
rate constant for the reaction j. This kind of compact notation is very useful
in handling large systems of kinetic schemes containing many equations
(more than 100 or so) and is used in common kinetic solution algorithms
such as CHEMKIN (Kee et al., 1996).
In complex chemical schemes involving m equations the total rate of
production rate L
k
of a certain species k is the sum of individual rates of each
equation producing the species k, i.e.
L
k
= L
kj
(12.70)
where L
k
is the total reaction rate for the species k and L
kj
is the reaction
rate for species k for the specific reaction j.
These reaction rates are used in the transport equations for each of the
species in the solution of combustion problems. When reaction rate expres-
sions are written for each species participating in the mechanism this yields
a system of first-order differential equations describing the progress of a
chemical system with time. Given the initial conditions (initial concentration
of the reacting species and temperature), additional equations for conservation
of mass, momentum and energy can be written, and the system of differential
equations can be numerically integrated to obtain a solution. In the solution
of such systems, when one or more variables change rapidly in comparison
with other variables, the resulting system is said to be stiff. Chemical kinetic
solution packages use special routines to deal with such stiff systems (see Kee
et al., 1996; Radhakrishnan and Pratt, 1988).
The reaction rates are used as source terms (see later) in transport equa-
tions for species mass fractions, which have the units kg/m
3
.s. To convert to
required units we multiply L
kj
, which is in kmol/m
3
.s, by the molecular
weight (MW). After converting to mass rate units the usual reaction rate
source term used in species transport equations may be written as
M
k
= (MW )
k
L
k
(12.71)
where L
k
is obtained from (12.70) and (12.69) using details of kinetic data
(values of constants A,
α
, E
a
etc.) from Baulch et al. (1994), Turns (2000),
Smith et al. (2003), Kuo (2005), Gardiner (1984) and many other publica-
tions cited later. In some of the early combustion literature tabulated kinetic
data have been tabulated in CGS units. Care must be given in using these
datasets, and appropriate SI unit conversion is required when incorporating
reaction rates.
A further point to note is the link between forward and backward reaction
rate constants. When the forward reaction rate constant (k
f
) is known, the
backward rate constant (k
b
) for the jth reaction can be calculated through the
equilibrium constant introduced in section 12.8:
(12.72)
kkC
X
X
KRTp
fj bj M
i
i
Pu
v
j
kj kj
k
N
i
i
ii
( ) ( /)
()
=
∑
==
∑∑
−
−
=
∏
∏
νν
ν
ν
ν
″′
″
′
′″
1
products
reactants
m
∑
j =1
ANIN_C12.qxd 29/12/2006 04:44PM Page 359
360 CHAPTER 12 CFD MODELLING OF COMBUSTION
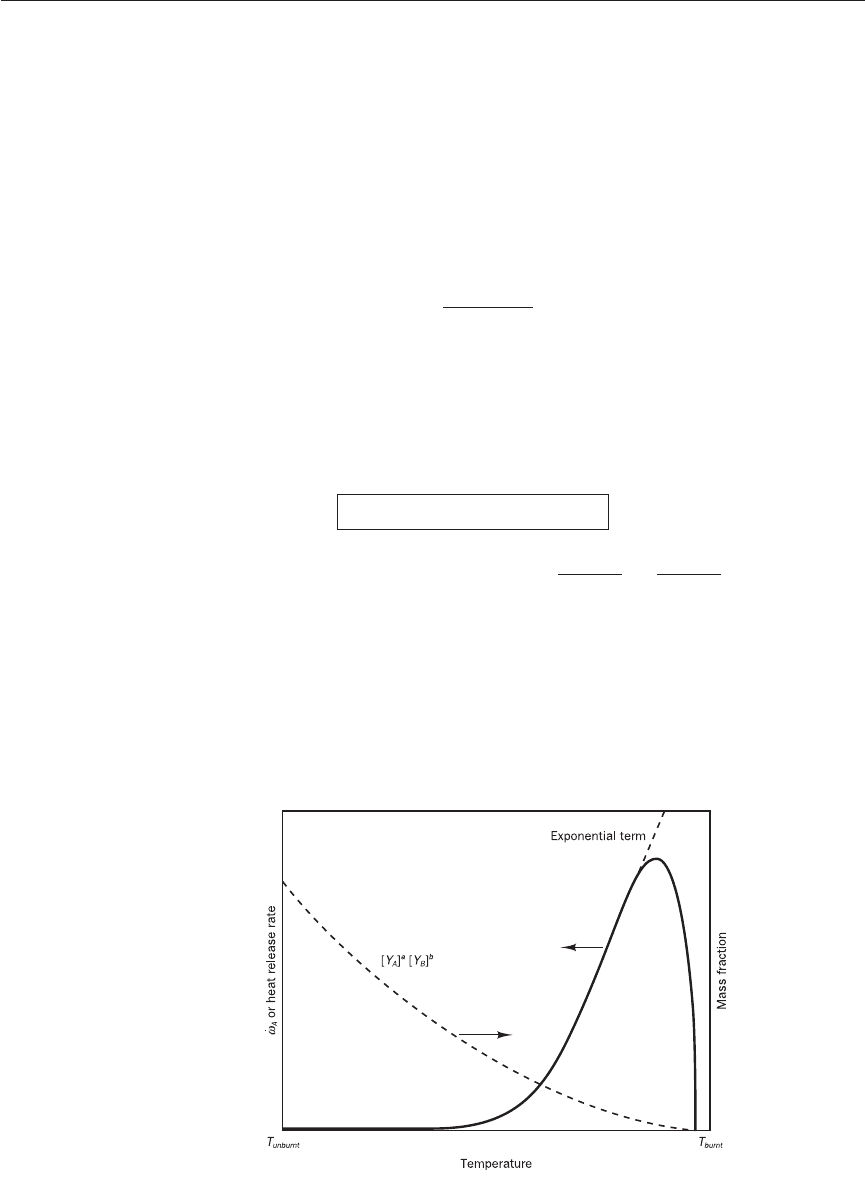
Figure 12.1 Schematic
representation of the reaction
rate expression for a single-
step reaction
For example, one of the reactions known as the Zel’dovich mechanism for
thermal NO formation (discussed later) is
NO + O → N + O
2
The rate coefficient (from Turns, 2000) for the forward reaction is
k
f
= 3.80 × 10
9
T
1.0
exp(−20820/T) cm
3
/gmol.s
At 2300 K, the value is k
f
= 1.029 × 10
9
cm
3
/gmol.s. For this reaction
(∑
ν
′
i
−∑
ν
″
i
) = 0. The K
P
-value for the above reaction at 2300 K is
K
P
= 1.94 × 10
−4
. Using equation (12.72) the backward reaction rate
coefficient is obtained as
k
b
= k
f
/K
P
==5.278 × 10
12
cm
3
/(gmol s)
Further details of chemical kinetics, the fundamental development of reac-
tion rate expressions, reaction kinetic data and how they are used in packages
like CHEMKIN are available in Turns (2000) and Kuo (2005).
To illustrate the behaviour of the reaction rate expression for a simple
reaction of the form (12.61) the rate of consumption of species A can be
written as
M
A
= A′[Y
A
]
a
[Y
B
]
b
exp(−E/R
u
T) (12.73)
where A′=(MW )
A
a AT
α
ab
It should be noted that constant A′ contains density and molecular weight
terms, which arise from the conversion from molar concentrations (12.69) to
mass fractions (as used in CFD computations). According to this expression,
it can be seen that the rate increases exponentially with temperature. As
the reaction progresses, the mass fractions of reactants decrease as they get
consumed. Therefore the reaction rate decreases as species A and B are con-
sumed. The typical behaviour of the rate expression is shown in Figure 12.1.
D
E
F
ρ
(MW )
B
A
B
C
D
E
F
ρ
(MW )
A
A
B
C
1.024 × 10
9
1.94 × 10
− 4
ANIN_C12.qxd 29/12/2006 04:44PM Page 360

12.13 REDUCED MECHANISMS 361
The reaction rate increases almost exponentially with temperature (T) at
first because of the dominant role of the term exp(−E/RT). As T increases
towards the final temperature T
burnt
, the reaction rate drops in spite of the
exponential term because [Y
A
]
a
[Y
B
]
b
decreases rapidly as reactants A and B
species are consumed in the reaction. This demonstrates that the availability
of reactants (concentrations) and temperature both play major roles in deter-
mining the reaction rate.
There are many types of reaction: reactions of various orders (first-order,
second-order etc.), consecutive reactions, competitive reactions, opposing
reactions, chain reactions, chain branching reactions etc. For oxidation of
various fuels detailed reaction mechanisms and appropriate rate constants
for those reactions are available in the literature. For example, see Gardiner
(1984), Drake and Blint (1988), Dryer (1991), Smooke (1991), Peters (1993),
Turns (2000), Seshadri and Williams (1994), Warnatz et al. (2001), the
GRI 3.0 mechanism (Smith et al., 2003 and references therein), and also
the San Diego mechanism (2003, http://maemail.ucsd.edu/combustion/
cermech/).
Detailed mechanisms for methanol and the CO/H
2
/O
2
system are
available in Dryer (1991). This reference also gives one-step, two-step and
four-step mechanisms for a variety of hydrocarbon fuels. Peters (1993) is
a valuable source book which has contributions from many other authors
and contains details of multi-step and reduced mechanisms for a range
of fuels. A detailed mechanism for methane combustion, NO formation,
single-step and multi-step reaction mechanisms for common hydrocarbons
are also available in Turns (2000). A mechanism containing 46 steps for
methane combustion has been reported in Smooke et al. (1986). A very well-
known and widely used mechanism for methane combustion is GRI 2.11
(Bowman et al., 1996). The most recent version of this mechanism is GRI 3.0
(Smith et al., 2003), which includes a detailed mechanism for NO formation.
However, it has been observed in some test cases that the previous version
GRI 2.11 appears to give better NO predictions (at the time of writing this
text) than the latest version GRI 3.0 (see Kim and Huh, 2002). Further
details of detailed mechanisms can be found in Seshadri and Williams
(1994), Warnatz et al. (2001), and also at the Lawrence Livermore Laboratory
mechanisms link (http://www-cms.llnl.gov/combustion/combustion2.html).
It should be noted that rate constants of various mechanisms have been
derived on the basis of computational estimations and comparisons with
measurable combustion properties such as flame speeds, temperatures and
species mass fractions obtained in controlled experiments. For large systems
of chemical equations dedicated computer programs are required to solve
chemical kinetic problems. CHEMKIN (Kee et al., 1996), for example, is a
widely used software package for such problems, and many commercial CFD
codes allow the incorporation of its information relating to chemical reactions.
The computational cost of chemical kinetics evaluations and the associated
species transport equations is substantial, and grows rapidly as the reaction
mechanisms become more elaborate and detailed. Consequently, efforts have
Detailed
mechanisms
12.12
Reduced
mechanisms
12.13
ANIN_C12.qxd 29/12/2006 04:44PM Page 361

been made to develop more practical reaction schemes involving fewer reac-
tions to represent combustion of basic fuels. These simplified schemes are
called reduced mechanisms and consist of a few key equations to predict
major and important minor species (see e.g. Dryer, 1991; Seshadri and
Williams, 1994). For example, the detailed mechanism for H
2
oxidation given
in Conaire et al. (2004) consists of 19 reactions. Massias et al. (1999) give a
five-step reduced mechanism for H
2
combustion including NO formation
(Table 12.1).
362 CHAPTER 12 CFD MODELLING OF COMBUSTION
Table 12.1
1H
2
bcg 2H
2H
2
+ O bcg H
2
O
3 4H + O
2
bcg 2H
2
O
4N
2
+ O
2
bcg 2NO
5N
2
+ O bcg N
2
O
The detailed chemical–kinetic mechanism for methane combustion
shown in Seshadri and Williams (1994) consists of 39 elementary reactions
involving 17 chemical species including N
2
. A five-step reduced mechanism
by Hewson and Bollig (1996) for CH
4
combustion is shown in Table 12.2.
The same reference gives a 52-step, 13-species reaction detailed mechanism
for nitrogen chemistry (species NO, NH
3
, HCN etc.) and a reduced six-step
reduced mechanism for nitrogen chemistry.
Table 12.2
1CH
4
+ 2H + H
2
O bcg 4H
2
+ CO
2H
2
O + CO bcg H
2
+ CO
2
32Hbcg H
2
4O
2
+ 3H
2
bcg 2H
2
O + 2H
5H
2
+ 2CO bcg C
2
H
2
+ O
2
Rate constants and further data for the reduced mechanisms shown
in Tables 12.1 and 12.2 can be obtained from the above cited references.
Extensive studies have been made of most practically important combustion
reactions, resulting in a vast body of publications with proposals for detailed
and reduced mechanisms. Some detailed mechanisms involve many equa-
tions – the above-mentioned GRI 3.0 mechanism for natural gas combus-
tion including NO
x
chemistry, for example, consists of 325 reactions and
53 species. The accuracy of reduced mechanisms has been compared with
detailed mechanisms for many practically important combustion reactions
(see e.g. Barlow et al., 2001; Massias et al., 1999). The choice of a reduced
mechanism is always a trade-off between computational cost and accuracy
required. The user should carefully select a suitable mechanism depend-
ing on the particular application and available resources. When results are
interpreted it should also be noted that some reduced mechanisms have
been tuned for specific temperature ranges and specific types of combustion
processes.
ANIN_C12.qxd 29/12/2006 04:44PM Page 362

12.14 GOVERNING EQUATIONS FOR COMBUSTING FLOWS 363
So far we have discussed thermodynamics, chemical equilibrium and chem-
ical kinetics without any reference to flow conditions. In many combustion
situations fluid flow is an integral part of the combustion process. In non-
premixed combustion situations such as furnaces, fuel and air streams are
mixed by fluid flow and turbulence, and the resulting combustion tempera-
tures, species concentrations and distribution of species are very much con-
trolled by fluid flow. Similarly, in premixed combustion, e.g. a spark ignition
IC engine, the geometry, fluid flow and turbulence created by the induction
and compression processes prior to ignition play an important role in the
combustion characteristics of the engine. We now present the transport
equations governing gaseous fuel combustion. We quote all equations in
compact suffix notation, introduced first in Chapter 3.
The governing equations of continuity and momentum developed in
Chapter 2 can be used in unchanged form.
Continuity
The continuity equation is
+ (
ρ
u
i
) = 0 (12.74)
It should be noted that density in combusting flows is a variable, and depends
on pressure, temperature and species concentration.
Momentum equations
As in other flows the velocity field is governed by momentum equations
(
ρ
u
i
) + (
ρ
u
i
u
j
) =− + +F
i
(12.75)
where
τ
ij
is the viscous stress tensor and F
i
is the body force (which includes
gravity):
τ
ij
=
µ
+−
δ
ij
(12.76)
The first transport equation that is specific to reacting flows is the equation
for the conservation of mass of a species k. It can be easily developed from
the general transport equation (2.39) by setting
φ
= Y
k
. In suffix notation the
statement is as follows.
Transport equations for species (k)
(
ρ
Y
k
) + (
ρ
u
i
Y
k
) =
ρ
D
k
+ M
k
(12.77)
Rate of Net rate of Net rate of Net rate of
change of
+
decrease of mass
=
increase of mass
+
increase of mass
mass of of species k due of species k due of species k due
species k to convection to diffusion to sources
D
E
F
∂
Y
k
∂
x
i
A
B
C
∂
∂
x
i
∂
∂
x
i
∂
∂
t
D
E
F
∂
u
k
∂
x
k
2
3
∂
u
j
∂
x
i
∂
u
i
∂
x
j
A
B
C
∂τ
ij
∂
x
i
∂
p
∂
x
i
∂
∂
x
i
∂
∂
t
∂
∂
x
i
∂ρ
∂
t
Governing
equations for
combusting flows
12.14
ANIN_C12.qxd 29/12/2006 04:44PM Page 363
364 CHAPTER 12 CFD MODELLING OF COMBUSTION
In equation (12.77) D
k
is the species diffusion coefficient (units m
2
/s). The
volumetric rate of generation (or destruction) of a species due to chemical
reactions appears as the source (or sink) term M
k
in each of their transport
equations. For kinetically controlled combustion this term takes the form of
(12.71) or (12.73). A wide variety of different models are available for phys-
ically controlled combustion. The most important ones will be discussed in
the remaining sections of this chapter.
It is common practice to assume a single diffusion coefficient for all
species. This simplifies equation (12.77) to
(
ρ
Y
k
) + (
ρ
u
i
Y
k
) =
ρ
D + M
k
(12.78)
Whilst the single D assumption is not always accurate (and may in fact be
quite inaccurate) it is very attractive, since it enables far-reaching simplifica-
tion of combustion calculations.
Energy equation
In combusting flows the temperature depends on the thermodynamic state
and the composition of the mixture. Some combustion models do not require
a transport equation for enthalpy: for example, in the laminar flamelet model
the temperature is obtained from the laminar flamelet library curves. Other
combustion submodels, however, require the solution of the transport equa-
tion for enthalpy. Chemical energy is released as heat during combustion,
and the resulting enthalpy is obtained by solving its transport equation:
(
ρ
h) + (
ρ
u
i
h) =+
µ
− h
k
++S
rad
(12.79)
Net rate of Net rate of
Net rate of Net rate of
Rate of
Net rate of increase of increase of
increase of increase of
change of
decrease of enthalpy due enthalpy due to
enthalpy due enthalpy due
enthalpy
+
enthalpy due
=
to diffusion
+
mass diffusion
+
to pressure
+
to radiative
to convection along gradients along gradients of
work heat transfer
of enthalpy species concentration
In equation (12.79) the source term of the transport equation for enthalpy
S
rad
is the radiation loss or gain. Viscous energy dissipation is normally
assumed to be negligible in low Mach number combusting flows. Here h is
the mixture enthalpy per unit mass and h
k
is the specific enthalpy of species
k, and the summation is carried out over all N species considered in the
chosen reaction mechanism. The mixture Prandtl number is
σ
h
, and Sc
k
is
the species Schmidt number, Sc
k
≡
µ
/
ρ
D
k
.
The Prandtl number (
σ
h
) is defined as
σ
h
==
Rate of momentum transport
Rate of energy transport
c
p
µ
k
∂
p
∂
t
J
K
L
∂
Y
k
∂
x
i
N
∑
k =1
D
E
F
1
σ
h
1
Sc
k
A
B
C
∂
h
∂
x
i
µ
σ
h
G
H
I
∂
∂
x
i
∂
∂
x
i
∂
∂
t
D
E
F
∂
Y
k
∂
x
i
A
B
C
∂
∂
x
i
∂
∂
x
i
∂
∂
t
ANIN_C12.qxd 29/12/2006 04:44PM Page 364
12.14 GOVERNING EQUATIONS FOR COMBUSTING FLOWS 365
The Lewis number is defined as
Le
k
≡=
The Schmidt number is defined as
Sc
k
==
= Le
σ
h
If a single diffusion coefficient is used, i.e. D
k
= D for k = 1, 2,..., N, as in
equation (12.78), equation (12.79) can be written as
(
ρ
h) + (
ρ
u
i
h) =+− 1 h
k
++S
rad
(12.80)
This can be rewritten as
(
ρ
h) + (
ρ
u
i
h) =+− 1 h
k
++S
rad
(12.81)
Therefore for the case where the Lewis number is unity the enthalpy equa-
tion simplifies to
(
ρ
h) + (
ρ
u
i
h) =++S
rad
(12.82)
For low-speed flows
∂
p/
∂
t can be neglected, so under the assumptions of
low-speed flow, single diffusion coefficient and unity Lewis number, the
enthalpy equation has exactly the same form as the general transport equa-
tion (2.39). Furthermore, if the radiation source term S
rad
is also small, the
enthalpy is a conserved or passive scalar.
It should be noted that the thermal conductivity of individual species is a
function of temperature and can be evaluated using a polynomial expression
in the form k = a + bT + cT
2
+ dT
3
, where coefficients are available in Reid
et al. (1987), for example. The binary diffusion coefficient D
ij
between two
species depends on the concentration of components, temperature and pres-
sure. Key equations which can be used to calculate D
ij
and then a single
diffusion coefficient D
k
(diffusion coefficient of a single species to the rest
of mixture) are also available in the above reference. D
k
also depends on
individual species concentrations, temperature and pressure. The reader is
also referred to Paul and Warnatz (1998), who describe transport models
for species properties. In combustion modelling the required diffusion co-
efficients and other transport properties are calculated (in detailed chemistry
∂
p
∂
t
J
K
L
∂
h
∂
x
i
µ
σ
h
G
H
I
∂
∂
x
i
∂
∂
x
i
∂
∂
t
∂
p
∂
t
J
K
L
∂
Y
k
∂
x
i
N
∑
k =1
D
E
F
1
Le
A
B
C
µ
σ
h
∂
h
∂
x
i
µ
σ
h
G
H
I
∂
∂
x
i
∂
∂
x
i
∂
∂
t
∂
p
∂
t
J
K
L
∂
Y
k
∂
x
i
N
∑
k=1
D
E
F
σ
h
Sc
A
B
C
µ
σ
h
∂
h
∂
x
i
µ
σ
h
G
H
I
∂
∂
x
i
∂
∂
x
i
∂
∂
t
Rate of momentum transport
Rate of mass transport
µ
ρ
D
k
Rate of energy transport
Rate of mass transport
k
ρ
c
p
D
k
ANIN_C12.qxd 29/12/2006 04:44PM Page 365

models) using packages such as CHEMKIN (Kee et al., 1996). Just to quote
some rough values, thermal conductivities of H
2
, H
2
O and CO
2
at 1000 K are
0.4 W/mK, 0.097 W/mK and 0.068 W/mK respectively. Approximate
values of binary diffusion coefficient for mixtures with N
2
at 1000 K reported
in Paul and Warnatz (1998) are: for H–N
2
, 10 cm
2
/s; for OH–N
2
, 2 cm
2
/s;
and for O
2
–N
2
, 1.5 cm
2
/s. Lewis numbers for species in an H
2
/air mixture
in a one-dimensional laminar flame calculation evaluated at the maximum
species mass fraction location are shown in Table 12.3. Apart from species H
and H
2
, other species have Lewis numbers close to 1.0. These values are
presented for information only, and the actual values should be evaluated
case by case depending on local conditions. The reader is also referred to
Clarke (2002) for further details on the calculation of Lewis numbers for
various fuel/air mixtures.
366 CHAPTER 12 CFD MODELLING OF COMBUSTION
Table 12.3 Lewis numbers for species evaluated in H
2
/air combustion
N
2
O
2
HOHOH
2
H
2
OH
2
O
2
0.920 1.052 0.200 0.726 0.700 0.175 0.995 1.005
Other relationships
The total of mass fractions of fuel, oxidant and inert species is equal to 1, so
Y
k
= 1 (12.83)
where k represents species.
The temperature can be calculated from the enthalpy by means of
T = (12.84)
where ∆h
fu
is the enthalpy of combustion.
The average value of the specific heat O
p
is defined as follows:
O
p
= c
p
dT (12.85)
Here
c
p
≡ Y
k
c
p,k
and c
p,k
is the specific heat of species k. Polynomial fits for temperature-
dependent c
p,k
for various species are available in various texts: see for
example Cengel and Boles (2002).
The local density of the mixture is dependent on pressure, reactant and
product concentrations and on the mixture temperature. Its value can be
calculated from the following equation of state:
all species
∑
k
T
T
0
1
(T − T
ref
)
fuel species
h −
∑
Y
k
∆h
fk
k
O
p
all species
∑
k
ANIN_C12.qxd 29/12/2006 04:44PM Page 366
