Versteeg H., Malalasekra W. An Introduction to Computational Fluid Dynamics: The Finite Volume Method
Подождите немного. Документ загружается.


12.15 THE SIMPLE CHEMICAL REACTING SYSTEM 367
ρ
=
R
u
T (12.86)
where (MW )
k
is the molecular weight of species k and R
u
is the universal gas
constant (8.314 kJ/kmol.K).
The flow field is in turn affected by changes in temperature and density,
so in addition to the species and enthalpy equations we must solve all the
flow equations. The resultant set of transport equations can be very large.
Models that consider many intermediate reactions require vast computing
resources, so simple models that incorporate only a few reactions are often
preferred in CFD-based combustion procedures. The simplest known pro-
cedure is the simple chemical reacting system (SCRS; see Spalding, 1979),
which is described below in some detail. Other approaches of modelling
turbulent combustion, such as the eddy break-up model and the laminar
flamelet model, are discussed later.
If we are concerned with the global nature of the combustion process
and with final major species concentrations only, the detailed kinetics is
unimportant and a global one-step, infinitely fast, chemical reaction can be
assumed where oxidant combines with fuel in stoichiometric proportions to
form products:
1 kg of fuel + s kg of oxidant → (1 + s) kg of products (12.87)
For methane combustion the equation becomes
CH
4
+ 2O
2
→ CO
2
+ 2H
2
O
1 mol of CH
4
2 mol of O
2
1 mol of CO
2
2 mol of H
2
O
1 kg of CH
4
+ kg of O
2
→ 1 + kg of products
The stoichiometric oxygen/fuel ratio by mass s is equal to 64/16 = 4 for
methane combustion. However, equation (12.87) also shows that the rate of
consumption of fuel during stoichiometric combustion is 1/s times the rate
of consumption of oxygen, i.e.
M
fu
= M
ox
In the SCRS infinitely fast chemical reactions are assumed and the inter-
mediate reactions are ignored. The transport equations for fuel and oxygen
mass fraction are written as
+ div(
ρ
Y
fu
u) = div(Γ
fu
grad Y
fu
) + M
fu
(12.88)
+ div(
ρ
Y
ox
u) = div(Γ
ox
grad Y
ox
) + M
ox
(12.89)
where Γ
fu
=
ρ
D
fu
and Γ
ox
=
ρ
D
ox
.
∂
(
ρ
Y
ox
)
∂
t
∂
(
ρ
Y
fu
)
∂
t
1
s
D
E
F
64
16
A
B
C
64
16
Y
k
(MW )
k
all species
∑
k
p
The simple
chemical reacting
system (SCRS)
12.15
ANIN_C12.qxd 29/12/2006 04:44PM Page 367
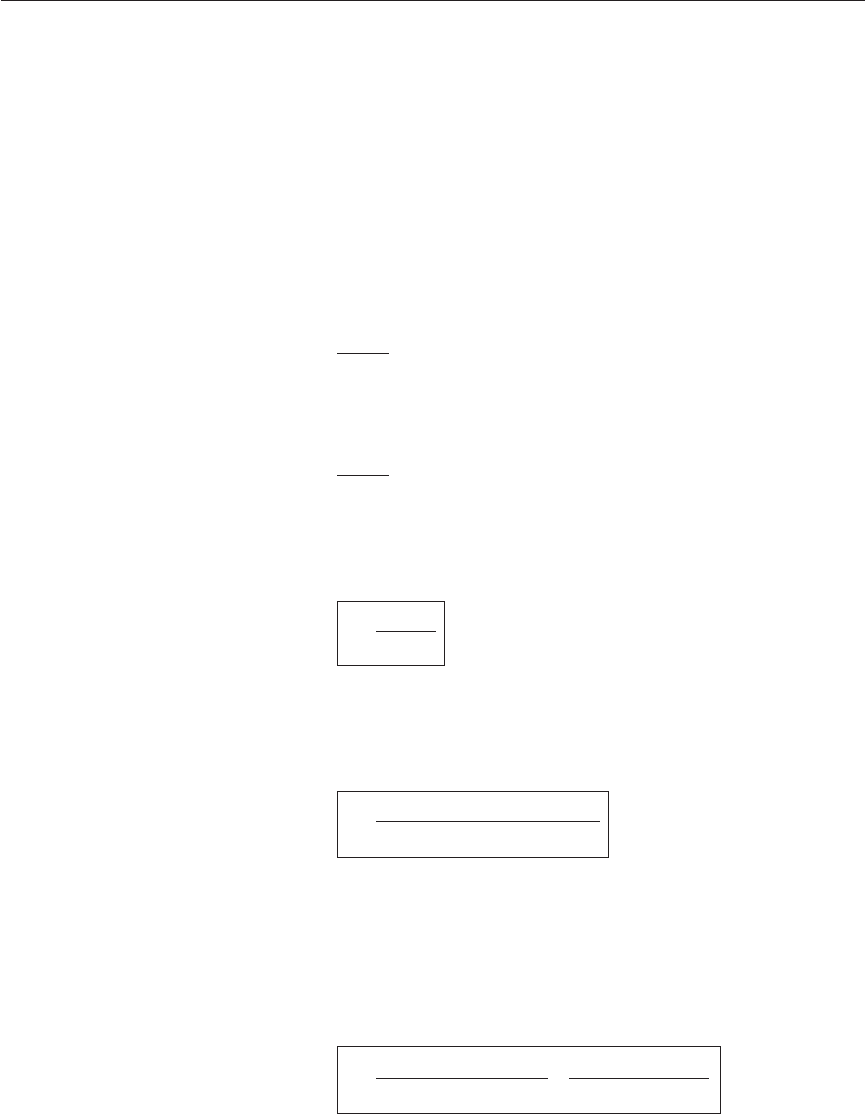
368 CHAPTER 12 CFD MODELLING OF COMBUSTION
The oxidant stream will have nitrogen which remains unaffected during
simple combustion; the mass fraction of inert species (Y
in
) remains the same
before and after combustion. Local values of Y
in
are determined by mixing
only, since the inert species does not take part in combustion (unless forma-
tion of NO is considered). Since the mass fraction of products Y
pr
= 1 − (Y
fu
+ Y
ox
+ Y
in
) it is unnecessary to solve a separate equation for Y
pr
.
It is possible to reduce the number of transport equations even further by
introducing a variable defined as follows:
φ
= sY
fu
− Y
ox
(12.90)
Application of the single diffusion coefficient assumption, Γ
fu
=Γ
ox
=
ρ
D =Γ
φ
,
allows us to subtract equation (12.89) from s times equation (12.88) and com-
bine the result into a single transport equation for
φ
:
+ div(
ρφ
u) = div(Γ
φ
grad
φ
) + (sM
fu
− M
ox
) (12.91)
From the one-step reaction assumption (12.87), we have M
fu
= (1/s)M
ox
,
giving (sM
fu
− M
ox
) = 0, and equation (12.91) reduces to
+ div(
ρφ
u) = div(Γ
φ
grad
φ
) (12.92)
Hence,
φ
is a passive scalar; it obeys the scalar transport equation without
source terms. A non-dimensional variable
ξ
called the mixture fraction
may be defined in terms of
φ
as follows:
ξ
= (12.93)
where suffix 0 denotes the oxidant stream and 1 denotes the fuel stream. The
local value of
ξ
equals 0 if the mixture at a point contains only oxidant and
equals 1 if it contains only fuel.
Equation (12.93) may be written in expanded form as
ξ
= (12.94)
If the fuel stream has fuel only we have
[Y
fu
]
1
= 1[Y
ox
]
1
= 0 (12.95)
and if the oxidant stream contains no fuel we have
[Y
fu
]
0
= 0[Y
ox
]
0
= 1 (12.96)
In such conditions equation (12.94) may be simplified as follows:
ξ
== (12.97)
In a stoichiometric mixture neither fuel nor oxygen is present in the prod-
ucts, and the stoichiometric mixture fraction
ξ
st
may be defined as
sY
fu
− Y
ox
+ Y
ox,0
sY
fu,1
+ Y
ox,0
[sY
fu
− Y
ox
] − [−Y
ox
]
0
[sY
fu
]
1
− [−Y
ox
]
0
[sY
fu
− Y
ox
] − [sY
fu
− Y
ox
]
0
[sY
fu
− Y
ox
]
1
− [sY
fu
− Y
ox
]
0
φ
−
φ
0
φ
1
−
φ
0
∂
(
ρφ
)
∂
t
∂
(
ρφ
)
∂
t
ANIN_C12.qxd 29/12/2006 04:44PM Page 368
12.15 THE SIMPLE CHEMICAL REACTING SYSTEM 369
ξ
st
= (12.98)
Fast chemistry implies that, at a certain location with a lean mixture, there
is an excess of oxidant and no fuel is present in the products. Hence Y
fu
= 0
if Y
ox
> 0, so
if
ξ
<
ξ
st
then
ξ
= (12.99)
Conversely, in a rich mixture there is a local excess of fuel in the mixture
and there is no oxidant in the products. Hence Y
ox
= 0 if Y
fu
> 0, so
if
ξ
>
ξ
st
then
ξ
= (12.100)
The above formulae show that the mass fractions of the fuel Y
fu
and oxygen
Y
ox
are linearly related to the mixture fraction
ξ
. This is illustrated graph-
ically in Figure 12.2.
sY
fu
+ Y
ox,0
sY
fu,1
+ Y
ox,0
−Y
ox
+ Y
ox,0
sY
fu,1
+ Y
ox,0
Y
ox,0
sY
fu,1
+ Y
ox,0
Figure 12.2 Mixing and
fast reaction between fuel
and oxidant streams
(SCRC relationships)
By equation (12.93)
ξ
is linearly related to
φ
so the mixture fraction is
also a passive scalar and obeys the transport equation
+ div(
ρξ
u) = div(Γ
ξ
grad
ξ
) (12.101)
Written in suffix notation the transport equation for the mixture fraction is
(
ρξ
) + (
ρ
u
i
ξ
) =Γ
ξ
(12.102)
To obtain the distribution of
ξ
we solve equation (12.101) subject to suitable
boundary conditions, e.g. mixture fractions of fuel and oxidant inlet streams
are known, zero normal flux of
ξ
across solid walls and zero-gradient out-
flow conditions. Given the resulting mixture fraction we can rearrange
D
E
F
∂ξ
∂
x
i
A
B
C
∂
∂
x
i
∂
∂
x
i
∂
∂
t
∂
(
ρξ
)
∂
t
ANIN_C12.qxd 29/12/2006 04:44PM Page 369
370 CHAPTER 12 CFD MODELLING OF COMBUSTION
equations (12.98)–(12.100) to give values for oxygen and fuel mass fractions
after combustion:
ξ
st
≤
ξ
< 1: Y
ox
= 0 Y
fu
= Y
fu,1
(12.103)
0 <
ξ
<
ξ
st
: Y
fu
= 0 Y
ox
= Y
ox,0
(12.104)
The reactants may be accompanied by inert species such as N
2
, which do not
take part in the reaction. The mass fraction of inert species in the mixture
varies linearly with
ξ
, as illustrated in Figure 12.2. Simple geometry gives the
total mass fraction of the inert species Y
in
after combustion at any value of
ξ
as
Y
in
= Y
in,0
(1 −
ξ
) + Y
in,1
ξ
(12.105)
The mass fraction of products (Y
pr
) of combustion may be obtained from
Y
pr
= 1 − (Y
fu
+ Y
ox
+ Y
in
) (12.106)
The above equations, (12.101) and (12.103)–(12.106), represent the SCRS
model.
When the reaction products contain two or more species, the ratio of the
mass fraction of each component to the total product mass fraction is known
from the equation for the chemical reaction and can be used to deduce the
mass fraction of different product components. For example, consider the
burning of methane with O
2
:
CH
4
+ 2O
2
⎯→ CO
2
+ 2H
2
O
1 mol of CH
4
2 mol of O
2
1 mol of CO
2
2 mol of H
2
O
16 kg 64 kg 44 kg 36 kg
Ratio of CO
2
in products by mass (r
CO
2
) = 44/80
Ratio of H
2
O in products by mass (r
H
2
O
) = 36/80
If the product mass fraction from equation (12.106) is Y
pr
then the CO
2
mass
fraction in the products is Y
pr
r
CO
2
and the H
2
O mass fraction is Y
pr
r
H
2
O
.
The SCRS model has made the following simplifications: (i) single-step
reaction between fuel and oxidant, and (ii) one reactant which is locally in
excess causes all the other reactant to be consumed stoichiometrically to
form reaction products. These assumptions fix algebraic relationships between
the mixture fraction
ξ
and all the mass fractions Y
fu
, Y
ox
, Y
in
and Y
pr
. As a
consequence of the additional assumption that the mass diffusion coefficients
of all species are equal, it is only necessary to solve one partial differential
equation for
ξ
to calculate combusting flows rather than individual partial
differential equations for the mass fraction of each species. An example which
uses this approach for combustion calculations is presented below.
The SCRS model can be readily applied to calculate temperatures and
species distribution in laminar diffusion flames. For example, consider the
axisymmetric laminar non-premixed diffusion flame geometry shown in
ξ
st
−
ξ
ξ
st
ξ
−
ξ
st
1 −
ξ
st
Modelling of a
laminar diffusion
flame --- an example
12.16
ANIN_C12.qxd 29/12/2006 04:44PM Page 370
12.16 MODELLING OF A LAMINAR DIFFUSION FLAME 371
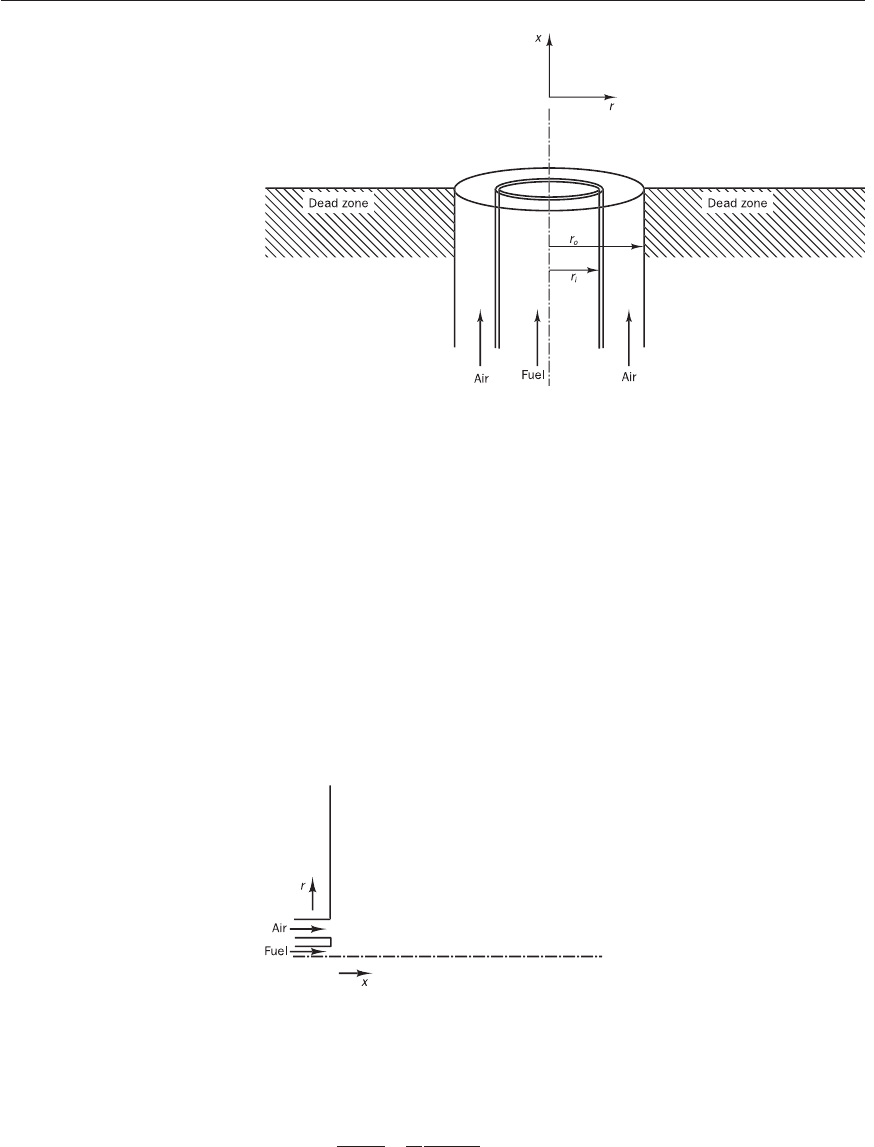
Figure 12.3 Schematic of the
problem considered
Figure 12.4 A part of the
computational geometry
Figure 12.3. The geometry considered here is an experimental configuration
which has been used by a number of studies documented in the combustion
literature (Bennett and Smooke, 1998; Smooke et al., 1990; Smooke, 1991).
For illustrative purposes we use our own operating conditions to formulate
the problem in this example. The radius of the fuel jet (r
i
) is 0.2 cm and the
radius (r
o
) of the co-flowing air jet is 2.5 cm. The thickness of the wall
between fuel and air streams is 0.05 cm. Let us consider burning of a pure
methane jet with co-flowing air. Both fuel and air velocities are taken as
0.2 m/s (20 cm/s) and enter at a temperature of 25°C (298 K). We would
like to calculate the resulting flame temperature and major species distribu-
tion field for this flame.
For calculation convenience we turn the actual geometry shown in Fig-
ure 12.3 through 90°. The axisymmetry of the problem allows us to adopt
a cylindrical x,r coordinate system to formulate the problem as shown in
Figure 12.4. Here we use u to represent the velocity component in the (axial)
x-direction and v to represent the velocity component in the (radial) r-direction.
The governing equations for steady laminar flow in cylindrical coordinates
in expanded form are as follows (see section 2.3 and section 12.14 above).
Continuity
+=0 (12.107)
∂
(
ρ
rv)
∂
r
1
r
∂
(
ρ
u)
∂
x
ANIN_C12.qxd 29/12/2006 04:44PM Page 371

Momentum equations
u-momentum equation:
(
ρ
ruu) + (
ρ
ruv) = (r
τ
xx
) + (r
τ
rx
) − r (12.108)
v-momentum equation
(
ρ
ruv) + (
ρ
rvv) = (r
τ
rx
) + (r
τ
rr
) − r (12.109)
The shear stress terms are
τ
xx
=
µ
2 − (div u) (12.110)
τ
rr
=
µ
2 − (div u) (12.111)
τ
rx
=
µ
2 + (12.112)
div u =+ (rv) (12.113)
The momentum equations can be rearranged in the usual form as follows:
u-momentum equation:
(
ρ
ruu) + (
ρ
ruv) = r
µ
+ r
µ
− r + S
u
(12.114)
v-momentum equation:
(
ρ
ruv) + (
ρ
rvv) = r
µ
+ r
µ
− r + S
v
(12.115)
where S
u
and S
v
contain the additional terms arising from the shear stress terms.
Enthalpy equation
(r
ρ
uh) + (r
ρ
vh) = rD
h
+ rD
h
+ S
rad
(12.116)
where D
h
is the diffusion coefficient for enthalpy (i.e. D
h
≡
α
= k/
ρ
C
p
) and
S
rad
is the radiation source (or sink) term.
Combustion model
Here we use the SCRC model described in section 12.15. The assumption of
fast chemistry gives the stoichiometric equation
CH
4
+ 2(O
2
+ 3.76N
2
) ⎯→ CO
2
+ 2H
2
O + 2 × 3.76N
2
(12.117)
D
E
F
∂
h
∂
r
A
B
C
∂
∂
r
D
E
F
∂
h
∂
x
A
B
C
∂
∂
x
∂
∂
r
∂
∂
x
∂
p
∂
r
D
E
F
∂
v
∂
r
A
B
C
∂
∂
r
D
E
F
∂
v
∂
x
A
B
C
∂
∂
x
∂
∂
r
∂
∂
x
∂
p
∂
x
D
E
F
∂
u
∂
r
A
B
C
∂
∂
r
D
E
F
∂
u
∂
x
A
B
C
∂
∂
x
∂
∂
r
∂
∂
x
∂
∂
r
1
r
∂
u
∂
x
J
K
L
∂
v
∂
x
∂
u
∂
r
G
H
I
J
K
L
2
3
∂
v
∂
r
G
H
I
J
K
L
2
3
∂
u
∂
x
G
H
I
∂
p
∂
r
∂
∂
r
∂
∂
x
∂
∂
r
∂
∂
x
∂
p
∂
x
∂
∂
r
∂
∂
x
∂
∂
r
∂
∂
x
372 CHAPTER 12 CFD MODELLING OF COMBUSTION
ANIN_C12.qxd 29/12/2006 04:44PM Page 372

12.16 MODELLING OF A LAMINAR DIFFUSION FLAME 373
The stoichiometric oxygen/fuel ratio by mass is
s = 2 × (MW )
O
2
/(MW )
CH
4
= 2 × 32/16 = 4
The mass fraction of fuel in the fuel stream is
Y
fu,1
= 1.0
The mass fraction of oxygen in the air stream is
Y
ox,0
= 0.233 (see section 12.5)
The mass fraction of inert (N
2
) in the air stream is
Y
in,0
= 0.767
We solve the following equation for the mixture fraction:
(r
ρ
u
ξ
) + (r
ρ
v
ξ
) = rΓ
ξ
+ rΓ
ξ
(12.118)
The mass fractions for the fuel and oxidant streams are as follows.
Since the fuel stream has fuel only we have
[Y
fu
]
1
= 1[Y
ox
]
1
= 0 (12.119)
The oxidant stream contains no fuel but oxygen and nitrogen (inert), so
we have
[Y
fu
]
0
= 0[Y
ox
]
0
= 0.233 [Y
in
]
0
= 0.767 (12.120)
The stoichiometric mixture fraction from equation (12.98) is
ξ
st
==0.055 (12.121)
If heat loss by radiation is considered in the calculation then solution of
the enthalpy equation is required. If radiation is considered to be negligible
then it can be seen that from equation (12.116) when S
rad
= 0 the transport
equation for enthalpy becomes another conserved scalar equation like the
mixture fraction equation. Therefore the enthalpy and mixture fraction are
both scalars and linearly related. The relationship can be further illustrated
as below.
Taking the reference temperature as zero, enthalpy is defined as
h = Y
fu
∆h
fuel
+ c
p
T (12.122)
Enthalpy of the fuel stream where
ξ
= 1 is
h
fu,in
= Y
fu,in
×∆h
fuel
+ Y
fu
c
p
× T
fu,in
where ∆h
fuel
is the enthalpy or formation of fuel. Enthalpy of the air stream
where
ξ
= 0 is
h
air,in
= c
p
× T
air,in
If we define a non-dimensional enthalpy as
h* = (12.123)
we can see that when
ξ
= 0, Y
fu
= 0, h* = 0 and when
ξ
= 1, Y
fu
= 1, h* = 1. If
we make the usual simplifying assumptions – single diffusion coefficient, unity
h − h
air,in
h
fu,in
− h
air,in
0.233
4 × 1 + 0.233
D
E
F
∂ξ
∂
r
A
B
C
∂
∂
r
D
E
F
∂ξ
∂
x
A
B
C
∂
∂
x
∂
∂
r
∂
∂
x
ANIN_C12.qxd 29/12/2006 04:44PM Page 373

Lewis number, negligible pressure work and radiation source – for enthalpy
transport, the governing transport equations for the enthalpy and mixture
fraction are the same (both variables are passive scalars). The resulting spatial
distributions of non-dimensional enthalpy h* and mixture fraction will be
the same (it is easy to verify that the boundary conditions for both variables
are also identical in this problem). Therefore, we do not need to solve a sep-
arate transport equation for enthalpy, but can calculate this variable from
ξ
= h* = (12.124)
Once the enthalpy value and mass fraction of fuel Y
fu
are known from SCRC
relationships, the temperature is obtained from
T = (12.125)
The boundary conditions for the problem are: at the inlet, u
in
= 0.2 m/s
for both fuel and air streams;
ξ
fu
= 1.0 for the fuel stream; and
ξ
air
= 0.0
for the air stream. Zero-velocity and zero-mixture-fraction-gradient wall
boundary conditions are used at all solid walls and a constant pressure
boundary condition is imposed on open boundaries with pressure set to
ambient. At the symmetry axis all gradients are set to zero.
Solution of the fluid flow equations and the mixture fraction equation
gives the distribution of mixture fraction which defines the flame structure
and species distribution. Equations (12.103) and (12.104) are used to obtain
species mass fractions. Equations (12.124) and (12.125) give the enthalpy and
temperature fields. Using pressure and temperature the density field is
obtained from (12.86). Because of the coupled nature of the equations, the
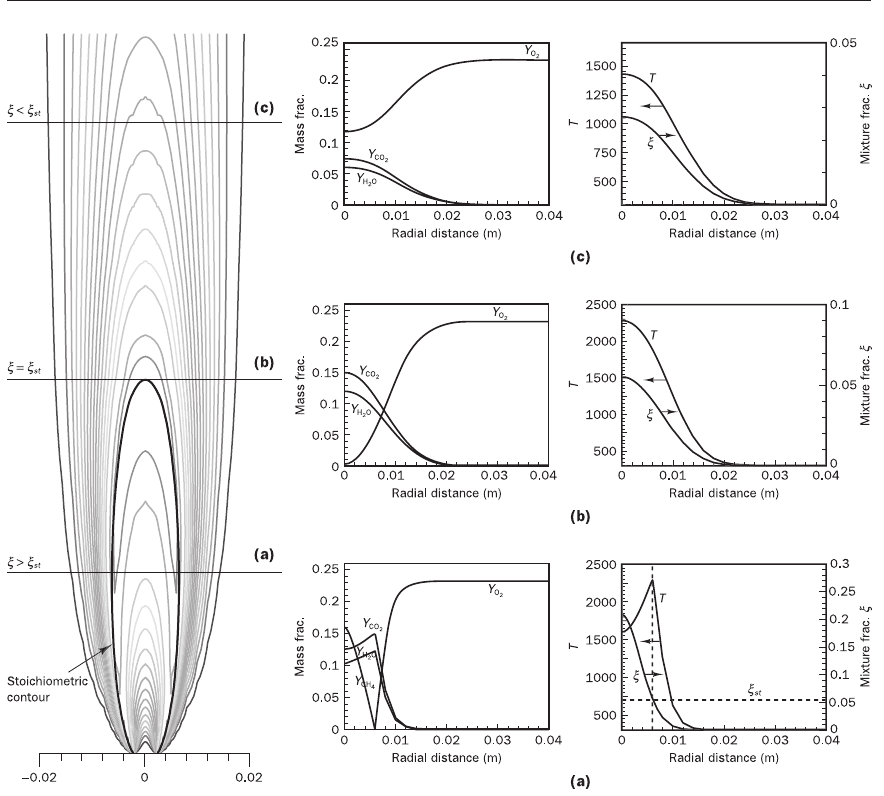
entire solution process is iterative. Figure 12.5 shows some typical results
obtained from this simulation. To highlight the consequences of the fast
chemistry assumption of the SCRS model we show radial profiles of tem-
perature and species mass fraction at three different points along the axis as
well as the temperature contours in Figure 12.5. The stoichiometric contour
corresponds with the contour of maximum temperature and defines the
flame. In the region inside the stoichiometric contour
ξ
>
ξ
st
, so by condition
(12.103) fuel exists here without oxidant. On the other hand, outside the
stoichiometric contour
ξ
<
ξ
st
, so by (12.104) no fuel can exist. At the axial
location (a) where
ξ
>
ξ
st
the radial profiles show no oxygen near the axis, and
fuel which gets completely consumed at the
ξ
st
contour. At axial location (b)
where centreline
ξ
=
ξ
st
neither fuel nor oxygen is present and the tempera-
ture is at a maximum. At axial location (c) where centreline
ξ
<
ξ
st
no fuel
exists and the temperature is lower.
Calculations with finite rate chemistry
To include finite rate and detailed chemistry in this combustion calculation
one has to consider a detailed mechanism and solve many species transport
equations of the form
(r
ρ
uY
k
) + (r
ρ
vY
k
) = r
ρ
D
k
+ r
ρ
D
k
+ rM
k
(12.126)
where M
k
is the rate of generation of species k, which is determined from
chemical kinetic expressions such as (12.69)–(12.71). Numerical solutions
D
E
F
∂
Y
k
∂
r
A
B
C
∂
∂
r
D
E
F
∂
Y
k
∂
x
A
B
C
∂
∂
x
∂
∂
x
∂
∂
x
h − Y
fu
(∆h
fu
)
O
p
h − h
air,in
h
fu,in
− h
air,in
374 CHAPTER 12 CFD MODELLING OF COMBUSTION
ANIN_C12.qxd 29/12/2006 04:44PM Page 374
12.16 MODELLING OF A LAMINAR DIFFUSION FLAME 375
including detailed and finite rate chemistry result in some variations to the
curves shown in Figure 12.5. The main difference is an overlap of fuel and
oxygen profiles and small curvature around the stoichiometric mixture frac-
tion showing a corresponding drop in temperature: see Warnatz et al. (2001).
Comprehensive combustion calculations for this geometry (for different
operating conditions) including detailed chemistry can be found in Smooke
et al. (1990), Bennett and Smooke (1998), Smooke and Bennett (2001).
The method presented above illustrates how CFD can be used for laminar
non-premixed combustion calculations. Unfortunately, the fast chemistry
assumption does not give adequate details of minor species, since it is neces-
sary to include more detailed chemistry and finite rate kinetics for a more
comprehensive account of combustion. Prediction of pollutants, such as
NO
x
discussed later in this chapter, inevitably requires inclusion of transport
equations for the most important minor species.
Figure 12.5 Calculated flame structure of a laminar diffusion flame
ANIN_C12.qxd 29/12/2006 04:44PM Page 375

The CFD calculation of turbulent non-premixed combustion is not as
straightforward as laminar calculations, even with the fast chemistry assump-
tion. In Chapter 3 we showed that Reynolds averaging and modelling of
the resulting averages of fluctuating product terms made it possible to
predict incompressible turbulent flow fields. Equations governing turbulent
non-premixed combustion also require averaging and modelling. The first
problem that needs to be addressed is the fact that strong and highly localised
heat generation in combusting flows causes the density to vary as a function
of position in combusting flows. There will also be density fluctuations if the
flow is turbulent. The Reynolds decomposition of a general flow variable is
as follows:
φ
= 2 +
φ
′
For the variables in a reacting flow this yields
u
i
= R
i
+ u′
i
p = Q + p ′
ρ
= 4 +
ρ
′
h = P + h′
T = E + T ′
Y
k
= F
k
+ Y′
k
It is easy to demonstrate that the presence of density fluctuations gives
rise to additional terms when Reynolds averaging is used. For example,
consider the instantaneous continuity equation in suffix notation:
+=0 (12.127)
After substituting for u and
ρ
the Reynolds-averaged equation is
++ =0 (12.128)
Compare this with the Reynolds-averaged equation for a constant density
flow:
+ (
ρ
R
i
) = 0 (12.129)
The additional term
∂
()/
∂
x
i
in equation (12.128) arises from correla-
tions between the velocity and density fluctuations in a reacting flow and
has to be modelled. Many more terms of this type appear in the Reynolds-
averaged momentum, scalar and species transport equations.
To reduce the number of separate terms requiring modelling in reacting
flows with variable density, we use a density-weighted averaging procedure
known as Favre averaging (Favre, 1969; Jones and Whitelaw, 1982).
In Favre averaging the density-weighted mean velocity is defined as
follows:
U = (12.130)
ρ
u
4
ρ
′u′
i
∂
∂
x
i
∂ρ
∂
t
∂
(
ρ
′u′
i
)
∂
x
i
∂
(4R
i
)
∂
x
i
∂
4
∂
t
∂
(
ρ
u
i
)
∂
x
i
∂ρ
∂
t
376 CHAPTER 12 CFD MODELLING OF COMBUSTION
CFD calculation
of turbulent non-
premixed combustion
12.17
ANIN_C12.qxd 29/12/2006 04:44PM Page 376
