Berg J.M., Tymoczko J.L., Stryer L. Biochemistry
Подождите немного. Документ загружается.

III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids 24.4. Amino Acids Are Precursors of Many Biomolecules
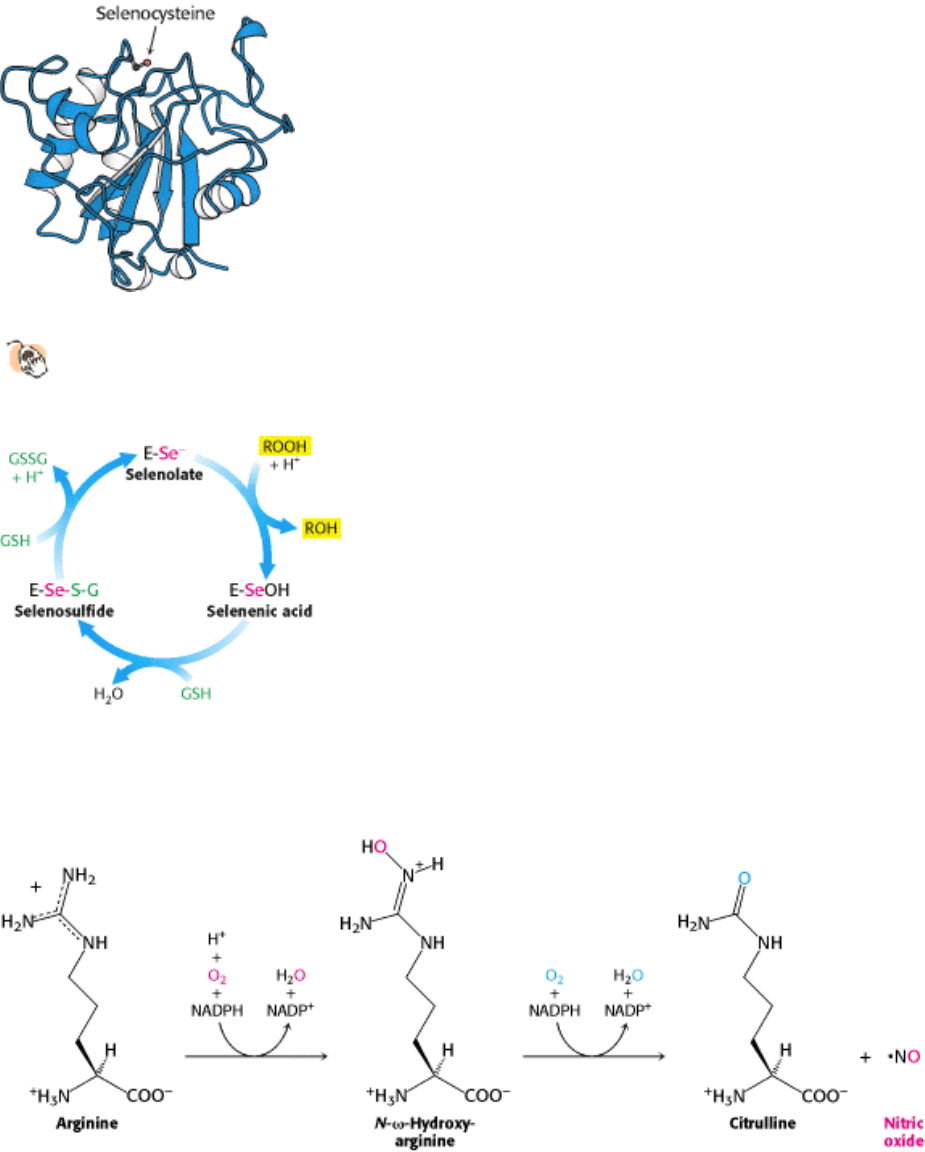
Figure 24.31. Structure of Glutathione Peroxidase.
This enzyme, which has a role in peroxide detoxification, contains
a selenocysteine residue in its active site.
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids 24.4. Amino Acids Are Precursors of Many Biomolecules
Figure 24.32. Catalytic Cycle of Glutathione Peroxidase. [After O. Epp, R. Ladenstein, and A. Wendel. Eur. J.
Biochem. 133(1983):51.]
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids 24.4. Amino Acids Are Precursors of Many Biomolecules
Figure 24.33. Formation of Nitric Oxide. NO is generated by the oxidation of arginine.
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids 24.4. Amino Acids Are Precursors of Many Biomolecules
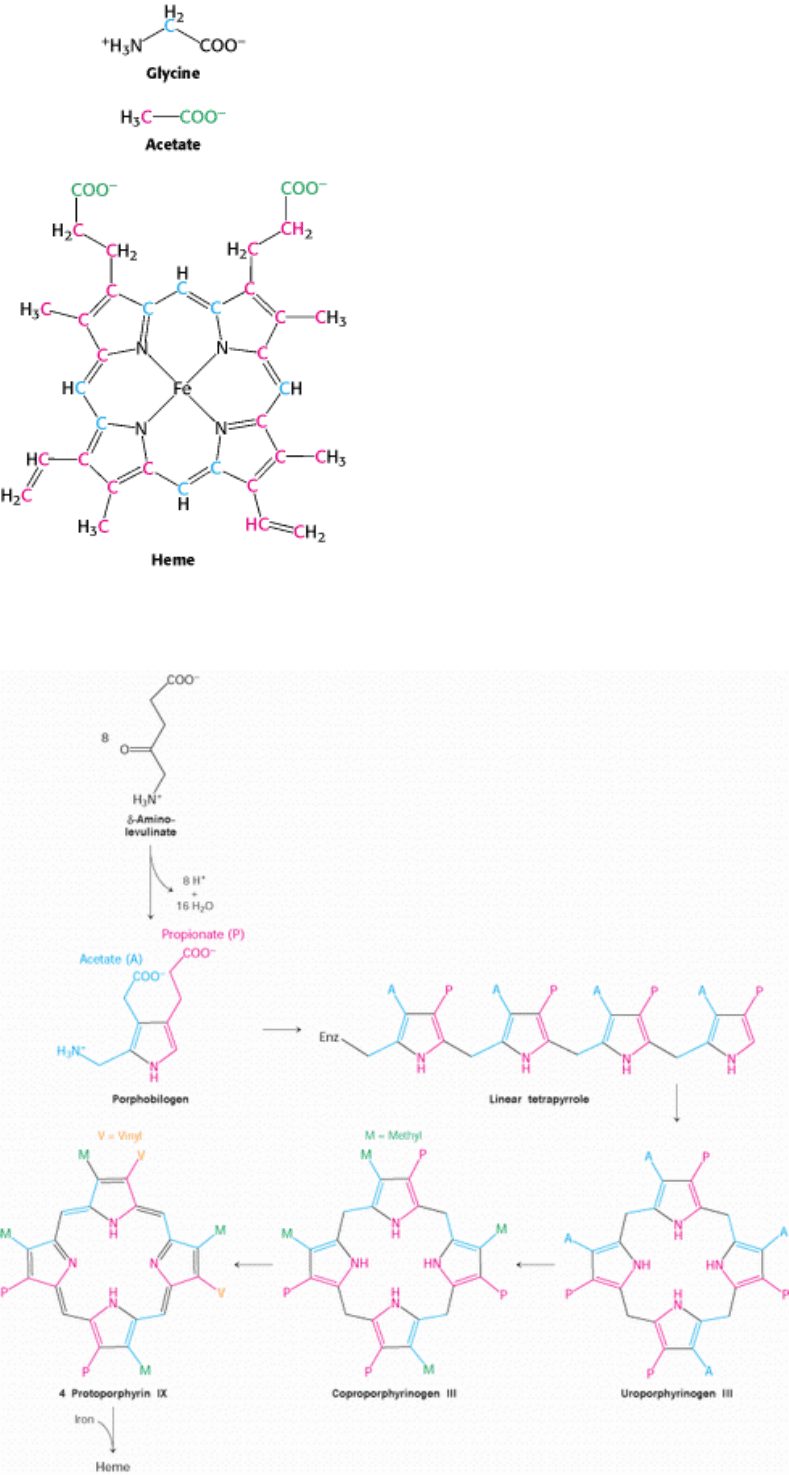
Figure 24.34. Heme Labeling. The origins of atoms in heme revealed by the results of isotopic labeling studies.
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids 24.4. Amino Acids Are Precursors of Many Biomolecules
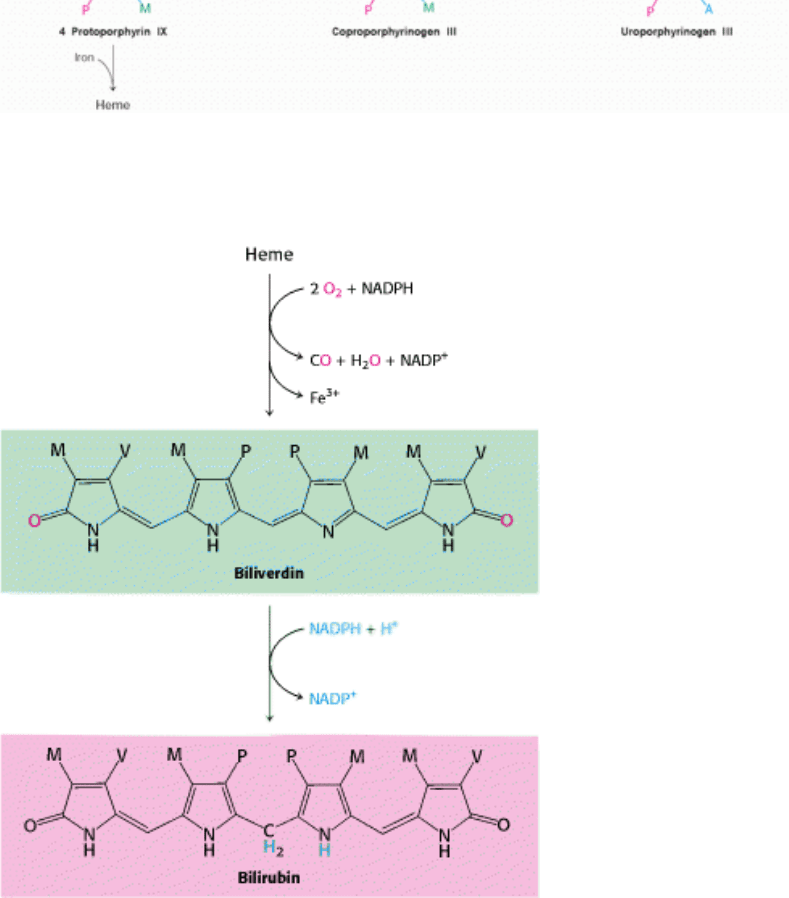
Figure 24.35. Heme Biosynthetic Pathway. The pathway for the formation of heme starts with eight molecules of δ-
aminolevulinate.
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids 24.4. Amino Acids Are Precursors of Many Biomolecules
Figure 24.36. Heme Degradation. The formation of the heme-degradation products biliverdin and bilirubin is
responsible for the color of bruises. Abbreviations: M, methyl; V, vinyl.
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids
Summary
Nitrogen Fixation: Microorganisms Use ATP and a Powerful Reductant to Reduce
Atmospheric Nitrogen to Ammonia
Microorganisms use ATP and reduced ferredoxin, a powerful reductant, to reduce N
2
to NH
3
. An iron-molybdenum
cluster in nitrogenase deftly catalyzes the fixation of N
2
, a very inert molecule. Higher organisms consume the fixed
nitrogen to synthesize amino acids, nucleotides, and other nitrogen-containing biomolecules. The major points of entry
of NH
4
+
into metabolism are glutamine or glutamate.
Amino Acids Are Made from Intermediates of the Citric Acid Cycle and Other Major
Pathways
Human beings can synthesize 11 of the basic set of 20 amino acids. These amino acids are called nonessential, in
contrast with the essential amino acids, which must be supplied in the diet. The pathways for the synthesis of
nonessential amino acids are quite simple. Glutamate dehydrogenase catalyzes the reductive amination of α-

ketoglutarate to glutamate. A transamination reaction takes place in the synthesis of most amino acids. At this step, the
chirality of the amino acid is established. Alanine and aspartate are synthesized by the transamination of pyruvate and
oxaloacetate, respectively. Glutamine is synthesized from NH
4
+
and glutamate, and asparagine is synthesized similarly.
Proline and arginine are derived from glutamate. Serine, formed from 3-phosphoglycerate, is the precursor of glycine
and cysteine. Tyrosine is synthesized by the hydroxylation of phenylalanine, an essential amino acid. The pathways for
the biosynthesis of essential amino acids are much more complex than those for the nonessential ones.
Tetrahydrofolate, a carrier of activated one-carbon units, plays an important role in the metabolism of amino acids and
nucleotides. This coenzyme carries one-carbon units at three oxidation states, which are interconvertible: most
reduced methyl; intermediate methylene; and most oxidized formyl, formimino, and methenyl. The major donor
of activated methyl groups is S-adenosylmethionine, which is synthesized by the transfer of an adenosyl group from ATP
to the sulfur atom of methionine. S-Adenosylhomocysteine is formed when the activated methyl group is transferred to
an acceptor. It is hydrolyzed to adenosine and homocysteine, the latter of which is then methylated to methionine to
complete the activated methyl cycle.
Amino Acid Biosynthesis Is Regulated by Feedback Inhibition
Most of the pathways of amino acid biosynthesis are regulated by feedback inhibition, in which the committed step is
allosterically inhibited by the final product. Branched pathways require extensive interaction among the branches that
includes both negative and positive regulation. The regulation of glutamine synthetase from E. coli is a striking
demonstration of cumulative feedback inhibition and of control by a cascade of reversible covalent modifications.
Amino Acids Are Precursors of Many Biomolecules
Amino acids are precursors of a variety of biomolecules. Glutathione (γ-Glu-Cys-Gly) serves as a sulfhydryl buffer and
detoxifying agent. Glutathione peroxidase, a selenoenzyme, catalyzes the reduction of hydrogen peroxide and organic
peroxides by glutathione. Nitric oxide, a short-lived messenger, is formed from arginine. Porphyrins are synthesized
from glycine and succinyl CoA, which condense to give δ-aminolevulinate. Two molecules of this intermediate become
linked to form porphobilinogen. Four molecules of porphobilinogen combine to form a linear tetrapyrrole, which
cyclizes to uroporphyrinogen III. Oxidation and side-chain modifications lead to the synthesis of protoporphyrin IX,
which acquires an iron atom to form heme.
Key Terms
nitrogen fixation
nitrogenase complex
essential amino acids
nonessential amino acids
pyridoxal phosphate
tetrahydrofolate
S-adenosylmethionine (SAM)
activated methyl cycle
substrate channeling

committed step
enzyme multiplicity
cumulative feedback inhibition
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids
Problems
1.
From sugar to amino acid. Write a balanced equation for the synthesis of alanine from glucose.
See answer
2.
From air to blood. What are the intermediates in the flow of nitrogen from N
2
to heme?
See answer
3.
One-carbon transfers. Which derivative of folate is a reactant in the conversion of
(a) glycine into serine?
(b) homocysteine into methionine?
See answer
4.
Telltale tag. In the reaction catalyzed by glutamine synthetase, an oxygen atom is transferred from the side chain of
glutamate to orthophosphate, as shown by the results of
18
O-labeling studies. Account for this finding.
See answer
5.
Therapeutic glycine. Isovaleric acidemia is an inherited disorder of leucine metabolism caused by a deficiency of
isovaleryl CoA dehydrogenase. Many infants having this disease die in the first month of life. The administration of
large amounts of glycine sometimes leads to marked clinical improvement. Propose a mechanism for the therapeutic
action of glycine.
See answer
6.
Deprived algae. Blue-green algae (cyanobacteria) form heterocysts when deprived of ammonia and nitrate. In this
form, the algae lack nuclei and are attached to adjacent vegetative cells. Heterocysts have photosystem I activity but
are entirely devoid of photosystem II activity. What is their role?
See answer
7.
Cysteine and cystine. Most cytosolic proteins lack disulfide bonds, whereas extracellular proteins usually contain
them. Why?
See answer
8.
To and fro. The synthesis of δ -aminolevulinate takes place in the mitochondrial matrix, whereas the formation of
porphobilinogen takes place in the cytosol. Propose a reason for the mitochondrial location of the first step in heme
synthesis.
See answer
9.
Direct synthesis. Which of the 20 amino acids can be synthesized directly from a common metabolic intermediate by
a transamination reaction?
See answer
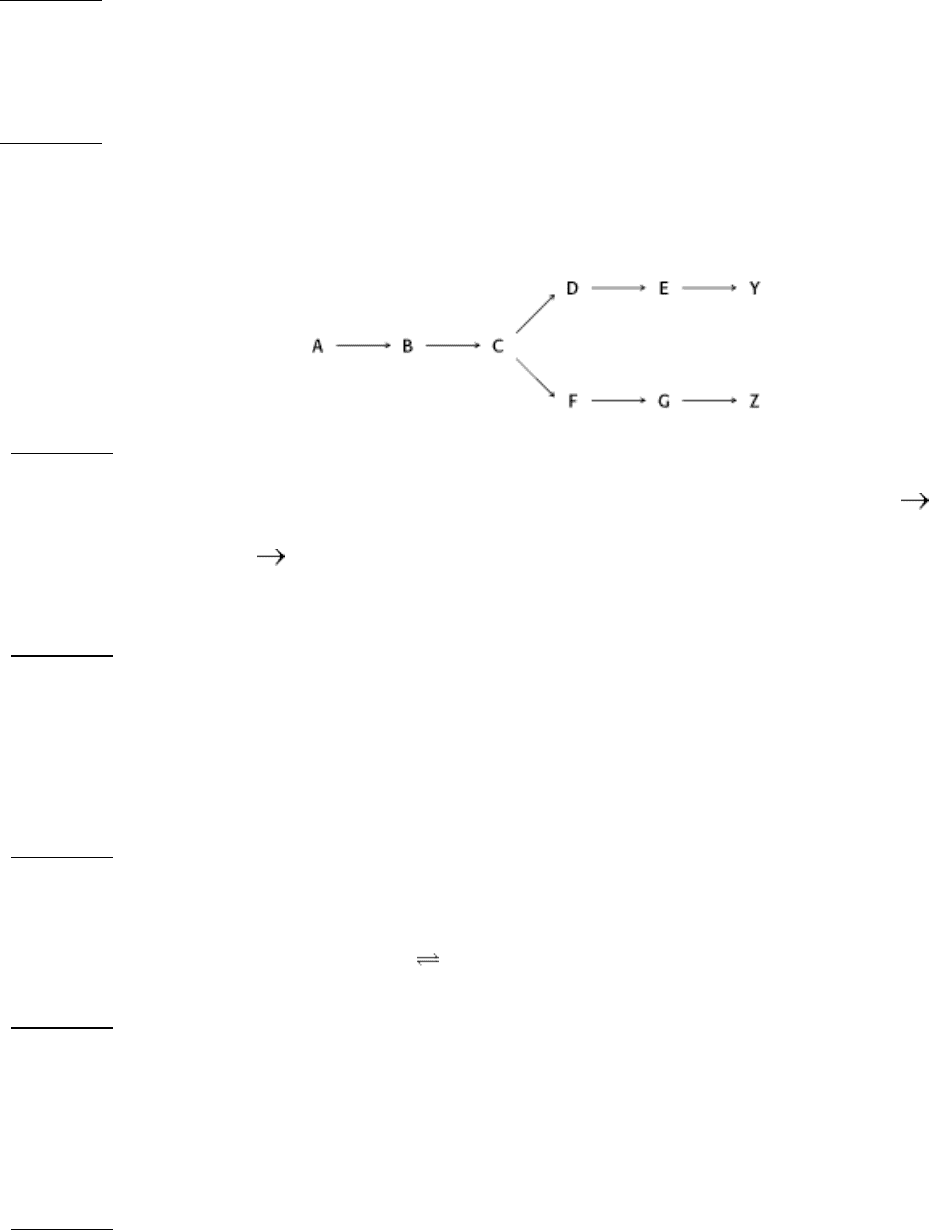
10.
Lines of communication. For the following example of a branched pathway, propose a feedback inhibition scheme
that would result in the production of equal amounts of Y and Z.
See answer
11.
Cumulative feedback inhibition. Consider the branched pathway above. The first common step (A B) is partly
inhibited by both of the final products, each acting independently of the other. Suppose that a high level of Y alone
decreased the rate of the A
B step from 100 to 60 s
-1
and that a high level of Z alone decreased the rate from
100 to 40 s
-1
. What would the rate be in the presence of high levels of both Y and Z?
See answer
Mechanism Problems
12.
Ethylene formation. Propose a mechanism for the conversion of S-adenosylmethionine into 1-aminocyclopropane-1-
carboxylate (ACC) by ACC synthase, a PLP enzyme. What is the other product?
See answer
13.
Mirror-image serine. Brain tissue contains substantial amounts of d-serine which is generated from l-serine by
serine racemase, a PLP enzyme. Propose a mechanism for the interconversion of l- and d-serine. What is the
equilibrium constant for the reaction l-serine d-serine?
See answer
Chapter Integration Problems
14.
Connections. How might increased synthesis of aspartate and glutamate affect energy production in a cell? How
would the cell respond to such an effect?
See answer
15.
Protection required. Suppose that a mutation in bacteria resulted in diminished activity of methionine
adenosyltransferase, the enzyme responsible for the synthesis of SAM from methionine and ATP. Predict how this
might affect the stability of the mutated bacteria's DNA.
See answer
Chapter Integration and Data Interpretation Problem
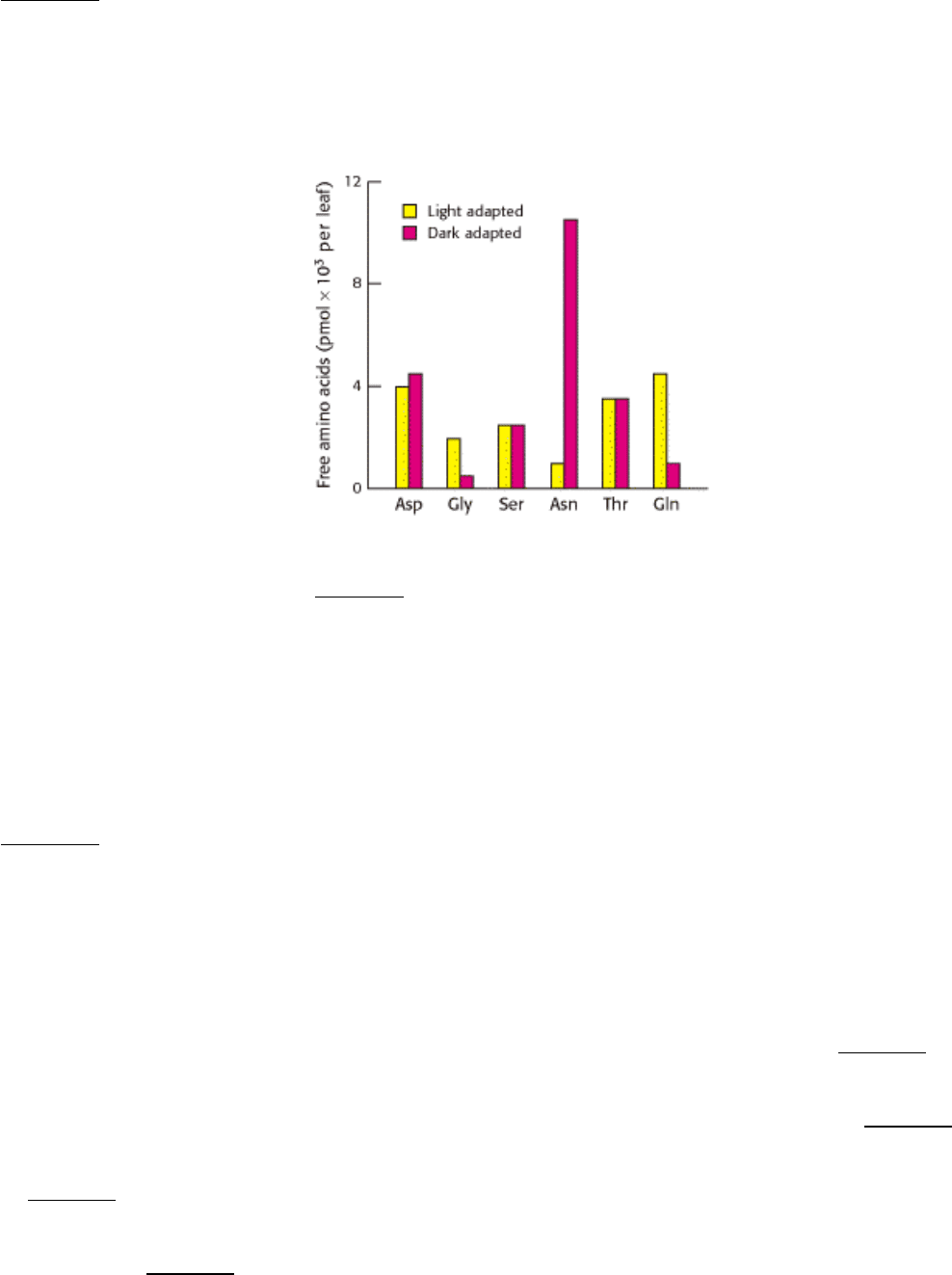
16.
Light effects. The adjoining graph shows the concentration of several free amino acids in light- and dark-adapted
plants.
[After B. B. Buchanan, W. Gruissem, and R. L. Jones, Biochemistry and Molecular Biology of Plants. (American
Society of Plant Physiology, 2000), Figure 8.3, p. 363.]
(a) Of the amino acids shown, which are most affected by light-dark adaptation?
(b) Suggest a plausible biochemical explanation for the difference observed.
(c) White asparagus, a culinary delicacy, is the result of growing asparagus plants in the dark. What chemical might
you think enhances the taste of white asparagus?
See answer
III. Synthesizing the Molecules of Life 24. The Biosynthesis of Amino Acids
Selected Readings
Where to start
J. Kim and D.C. Rees. 1989. Nitrogenase and biological nitrogen fixation Biochemistry 33: 389-397. (PubMed)
P. Christen, R. Jaussi, N. Juretic, P.K. Mehta, T.I. Hale, and M. Ziak. 1990. Evolutionary and biosynthetic aspects of
aspartate aminotransferase isoenzymes and other aminotransferases Ann. N. Y. Acad. Sci. 585: 331-338. (PubMed)
G. Schneider, H. Kack, and Y. Lindqvist. 2000. The manifold of vitamin B6 dependent enzymes Structure Fold Des. 8:
R1-R6. (PubMed)
S.G. Rhee, P.B. Chock, and E.R. Stadtman. 1989. Regulation of Escherichia coli glutamine synthetase Adv. Enzymol.
Mol. Biol. 62: 37-92. (PubMed)
D. Shemin. 1989. An illustration of the use of isotopes: The biosynthesis of porphyrins Bioessays 10: 30-35. (PubMed)
Books
Bender, D. A., 1985. Amino Acid Metabolism (2d ed.). Wiley.
Jordan, P. M. (Ed.), 1991. Biosynthesis of Tetrapyrroles . Elsevier.
Scriver, C. R., Beaudet, A. L., Sly, W. S., and Valle, D. (Eds.), 1995. The Metabolic Basis of Inherited Disease (7th ed.).
McGraw-Hill
Meister, A., 1965. Biochemistry of the Amino Acids (vols. 1 and 2, 2d ed.). Academic Press.
Blakley, R. L., and Benkovic, S. J., 1989. Folates and Pterins (vol. 2). Wiley.
Walsh, C., 1979. Enzymatic Reaction Mechanisms . W. H. Freeman and Company.
Nitrogen fixation
C.M. Halbleib and P.W. Ludden. 2000. Regulation of biological nitrogen fixation J. Nutr. 130: 1081-1084. (PubMed)
S.M. Mayer, D.M. Lawson, C.A. Gormal, S.M. Roe, and B.E. Smith. 1999. New insights into structure-function
relationships in nitrogenase: A 1.6 Å resolution X-ray crystallographic study of Klebsiella pneumoniae MoFe-protein J.
Mol. Biol. 292: 871-891. (PubMed)
J.W. Peters, K. Fisher, and D.R. Dean. 1995. Nitrogenase structure and function: A biochemical-genetic perspective
Annu. Rev. Microbiol. 49: 335-366. (PubMed)
G.J. Leigh. 1995. The mechanism of dinitrogen reduction by molybdenum nitrogenases Eur. J. Biochem. 229: 14-20.
(PubMed)
M.K. Chan, J. Kim, and D.C. Rees. 1993. The nitrogenase FeMo-cofactor and P-cluster pair: 2.2 Å resolution studies
Science 260: 792-794. (PubMed)
M.M. Georgiadis, H. Komiya, P. Chakrabarti, D. Woo, J.J. Kornuc, and D.C. Rees. 1992. Crystallographic structure of
the nitrogenase iron protein from Azotobacter vinelandii Science 257: 1653-1659. (PubMed)
Regulation of amino acid biosynthesis
D. Eisenberg, H.S. Gill, G.M. Pfluegl, and S.H. Rotstein. 2000. Structure-function relationships of glutamine synthetases
Biochim. Biophys. Acta 1477: 122-145. (PubMed)
D.L. Purich. 1998. Advances in the enzymology of glutamine synthesis Adv. Enzymol. Relat. Areas Mol. Biol. 72: 9-42.
(PubMed)
M.M. Yamashita, R.J. Almassy, C.A. Janson, D. Cascio, and D. Eisenberg. 1989. Refined atomic model of glutamine
synthetase at 3.5 Å resolution J. Biol. Chem. 264: 17681-17690. (PubMed)
D.J. Schuller, G.A. Grant, and L.J. Banaszak. 1995. The allosteric ligand site in the V
max
-type cooperative enzyme
phosphoglycerate dehydrogenase Nat. Struct. Biol. 2: 69-76. (PubMed)
S.G. Rhee, R. Park, P.B. Chock, and E.R. Stadtman. 1978. Allosteric regulation of monocyclic interconvertible enzyme
cascade systems: Use of Escherichia coli glutamine synthetase as an experimental model Proc. Natl. Acad. Sci. USA 75:
3138-3142. (PubMed)
P.M. Wessel, E. Graciet, R. Douce, and R. Dumas. 2000. Evidence for two distinct effector-binding sites in threonine
deaminase by site-directed mutagenesis, kinetic, and binding experiments Biochemistry 39: 15136-15143. (PubMed)
Y. Xu, P.D. Carr, T. Huber, S.G. Vasudevan, and D.L. Ollis. 2001. The structure of the PII-ATP complex Eur. J.
Biochem. 268: 2028- 2037. (PubMed)
Aromatic amino acid biosynthesis
P. Pan, E. Woehl, and M.F. Dunn. 1997. Protein architecture, dynamics and allostery in tryptophan synthase channeling
Trends Biochem. Sci. 22: 22-27. (PubMed)
A. Sachpatzidis, C. Dealwis, J.B. Lubetsky, P.H. Liang, K.S. Anderson, and E. Lolis. 1999. Crystallographic studies of
phosphonate-based alpha-reaction transition-state analogues complexed to tryptophan synthase Biochemistry 38: 12665-
12674. (PubMed)
M. Weyand and I. Schlichting. 1999. Crystal structure of wild-type tryptophan synthase complexed with the natural
substrate indole-3-glycerol phosphate Biochemistry 38: 16469-16480. (PubMed)
I.P. Crawford. 1989. Evolution of a biosynthetic pathway: The tryptophan paradigm Annu. Rev. Microbiol. 43: 567-600.
(PubMed)
E.P. Carpenter, A.R. Hawkins, J.W. Frost, and K.A. Brown. 1998. Structure of dehydroquinate synthase reveals an
active site capable of multistep catalysis Nature 394: 299-302. (PubMed)
I. Schlichting, X.J. Yang, E.W. Miles, A.Y. Kim, and K.S. Anderson. 1994. Structural and kinetic analysis of a channel-
impaired mutant of tryptophan synthase J. Biol. Chem. 269: 26591-26593. (PubMed)
Glutathione
R. Edwards, D.P. Dixon, and V. Walbot. 2000. Plant glutathione S-transferases: Enzymes with multiple functions in
sickness and in health Trends Plant Sci. 5: 193-198. (PubMed)
S.C. Lu. 2000. Regulation of glutathione synthesis Curr. Top. Cell Regul. 36: 95-116. (PubMed)
J.B. Schulz, J. Lindenau, J. Seyfried, and J. Dichgans. 2000. Glutathione, oxidative stress and neurodegeneration Eur. J.
Biochem. 267: 4904-4911. (PubMed)
S.C. Lu. 1999. Regulation of hepatic glutathione synthesis: Current concepts and controversies FASEB J. 13: 1169-1183.
(PubMed)
A.E. Salinas and M.G. Wong. 1991. Glutathione S-transferases: A review Curr. Med. Chem. 6: 279-309. (PubMed)
Ethylene and nitric oxide
J. Haendeler, A.M. Zeiher, and S. Dimmeler. 1999. Nitric oxide and apoptosis Vitam. Horm. 57: 49-77. (PubMed)
G. Capitani, E. Hohenester, L. Feng, P. Storici, J.F. Kirsch, and J.N. Jansonius. 1999. Structure of 1-aminocyclopropane-
1-carboxylate synthase, a key enzyme in the biosynthesis of the plant hormone ethylene J. Mol. Biol. 294: 745-756.
(PubMed)
A.J. Hobbs, A. Higgs, and S. Moncada. 1999. Inhibition of nitric oxide synthase as a potential therapeutic target Annu.
Rev. Pharmacol. Toxicol. 39: 191-220. (PubMed)
D.J. Stuehr. 1999. Mammalian nitric oxide synthases Biochim. Biophys. Acta 1411: 217-230. (PubMed)
C. Chang and J.A. Shockey. 1999. The ethylene-response pathway: Signal perception to gene regulation Curr. Opin.
Plant Biol. 2: 352-358. (PubMed)

P.R. Johnson and J.R. Ecker. 1998. The ethylene gas signal transduction pathway: A molecular perspective Annu. Rev.
Genet. 32: 227-254. (PubMed)
A. Theologis. 1992. One rotten apple spoils the whole bushel: The role of ethylene in fruit ripening Cell 70: 181-184.
(PubMed)
Biosynthesis of porphyrins
F.J. Leeper. 1989. The biosynthesis of porphyrins, chlorophylls, and vitamin B
12
Nat. Prod. Rep. 6: 171-199. (PubMed)
R.J. Porra and H.-U. Meisch. 1984. The biosynthesis of chlorophyll Trends Biochem. Sci. 9: 99-104.
III. Synthesizing the Molecules of Life
25. Nucleotide Biosynthesis
An ample supply of nucleotides is essential for many life processes. First, nucleotides are the activated precursors of
nucleic acids. As such, they are necessary for the replication of the genome and the transcription of the genetic
information into RNA. Second, an adenine nucleotide, ATP, is the universal currency of energy. A guanine nucleotide,
GTP, also serves as an energy source for a more select group of biological processes. Third, nucleotide derivatives such
as UDP-glucose participate in biosynthetic processes such as the formation of glycogen. Fourth, nucleotides are
essential components of signal-transduction pathways. Cyclic nucleotides such as cyclic AMP and cyclic GMP are
second messengers that transmit signals both within and between cells. ATP acts as the donor of phosphoryl groups
transferred by protein kinases.
In this chapter, we continue along the path begun in Chapter 24, which described the incorporation of nitrogen into
amino acids from inorganic sources such as nitrogen gas. The amino acids glycine and aspartate are the scaffolds on
which the ring systems present in nucleotides are assembled. Furthermore, aspartate and the side chain of glutamine
serve as sources of NH
2
groups in the formation of nucleotides.
Nucleotide biosynthetic pathways are tremendously important as intervention points for therapeutic agents. Many of the
most widely used drugs in the treatment of cancer block steps in nucleotide biosynthesis, particularly steps in the
synthesis of DNA precursors.
25.0.1. Overview of Nucleotide Biosynthesis and Nomenclature
The pathways for the biosynthesis of nucleotides fall into two classes: de novo pathways and salvage pathways (Figure
25.1). In de novo (from scratch) pathways, the nucleotide bases are assembled from simpler compounds. The framework
for a pyrimidine base is assembled first and then attached to ribose. In contrast, the framework for a purine base is
synthesized piece by piece directly onto a ribose-based structure. These pathways comprise a small number of
elementary reactions that are repeated with variation to generate different nucleotides, as might be expected for pathways
that appeared very early in evolution. In salvage pathways, preformed bases are recovered and reconnected to a ribose
unit.
Both de novo and salvage pathways lead to the synthesis of ribonucleotides. However, DNA is built from
deoxyribonucleotides. Consistent with the notion that RNA preceded DNA in the course of evolution, all
deoxyribonucleotides are synthesized from the corresponding ribonucleotides. The deoxyribose sugar is generated by the
reduction of ribose within a fully formed nucleotide. Furthermore, the methyl group that distinguishes the thymine of
DNA from the uracil of RNA is added at the last step in the pathway.
The nomenclature of nucleotides and their constituent units was presented earlier (Section 5.1.2). Recall that a
nucleoside consists of a purine or pyrimidine base linked to a sugar and that a nucleotide is a phosphate ester of a
nucleoside. The names of the major bases of RNA and DNA, and of their nucleoside and nucleotide derivatives, are
