Chen C.J. Physics of Solar Energy
Подождите немного. Документ загружается.


i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 254 — #281
i
i
i
i
i
i
254 Energy Storage
final temperature T
2
is
Q = M
T
2
T
1
c
p
dT, (12.1)
where M is the mass, c
p
is the isobaric specific heat. In most applications, the density
and specific heat can be treated as a constant. Equation 12.1 can be simplified to
Q = Mc
p
(T
2
− T
1
) . (12.2)
The quantity of material required for the storage tank and the heat losses are
approximately proportional to the surface area of the tank. The storage capacity is
proportional to the volume of the tank. Larger tanks have a smaller surface area–
volume ratio and therefore are less expensive and have less heat losses per unit energy
stored.
An important issue in thermal energy storage is thermal conduction, or temper-
ature equalization in the medium. In liquids, heat conduction has two major paths:
conduction and convection. Temperature in a liquid medium can become equalized
much faster than in a solid. Therefore, liquid is preferred whenever applicable. Ta-
ble 12.1 shows some thermal properties of commonly used liquids for sensible heat
thermal energy storage.
12.1.1 Water
As shown in Table 12.1, water has the largest heat capacity both per unit volume and
per unit weight. And it is free. Therefore, it is logical to use water as the material for
sensible heat storage. A typical case is the hot-water tank used in most homes. The
tank is typically insulated by foam polyurethane, which has a thermal conductivity κ
= 0.02 W/mK and density ρ = 30 kg/m
3
.
Table 12.1: Thermal Properties of Some Commonly Used Materials
Materials Density Heat Product Temperature
ρ capacity c
p
ρc
p
range ΔT
10
3
kg/m
3
10
3
J/kg·K10
6
J/m
3
·K
◦
C
Water 1.00 4.19 4.19 0 to 100
Ethonal 0.78 2.46 1.92 -117 to 79
Glycerine 1.26 2.42 3.05 17 to 290
Canola Oil 0.91 1.80 1.64 -10 to 204
Synthetic Oil 0.91 1.80 1.64 -10 to 400
Source: American Institute of Physics Handbook,3rdEd.,
American Institute of Physics, New York, 1972.
i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 255 — #282
i
i
i
i
i
i
12.1 Sensible Heat Energy Storage 255
If the entire tank is filled with water, the volume is
V =
1
4
πD
2
L (12.3)
and the heat capacity is
C
p
= ρc
p
V. (12.4)
The total surface area of the tank is
A =
1
2
πD
2
+ πD L. (12.5)
The rate of heat loss is
dQ
dt
=
κA
τ
(T
w
− T
a
) , (12.6)
where T
w
−T
a
is the difference of water temperature T
w
and ambient temperature T
a
.
The rate of temperature loss is
dT
dt
=
2κ(D +2L)
τρc
p
DL
(T
w
− T
a
) . (12.7)
The rate of temperature drop through the tank skin is proportional to the total
surface area and inversely proportional to the volume. If the tank is too thin or too
flat, then the heat loss is high. Therefore, for a tank of fixed volume, there should be
an optimal ratio L/D to minimize the heat loss. Intuitively, the condition should be
L ≈ D. In Problem 12.1, one can show that the intuition is correct: The optimum
condition is L = D, and Eq. 12.7 becomes
dT
dt
=
6κ
τρc
p
D
(T
w
− T
a
) . (12.8)
It verifies another qualitative argument: The larger the dimension, the better the tank
could preserve temperature.
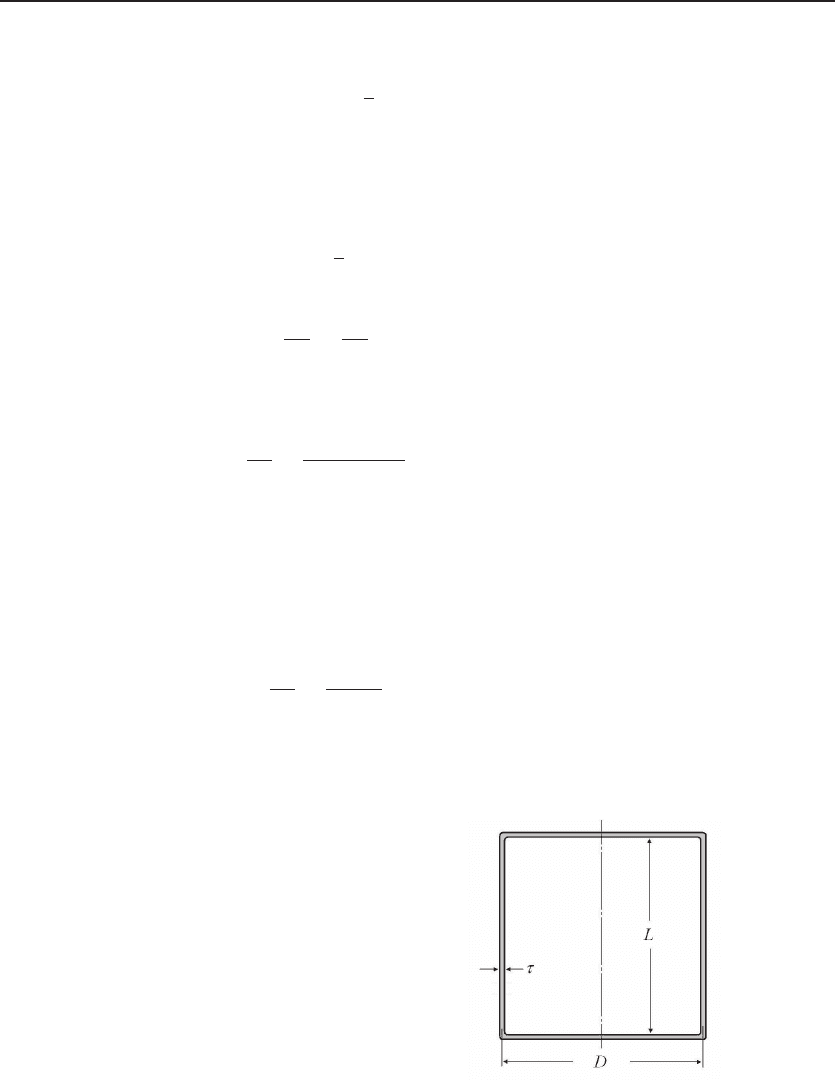
Figure 12.1 Water in an insulating tank.
Calculation of energy storage behavior of an in-
sulated water tank. The energy loss is propor-
tional to the total surface area, and the energy
content is proportional to the volume. The rate
of temperature drop is minimized when the diam-
eter D equals the length L. With a tank of lin-
ear dimension about 1 m with a 5-cm-thick foam
polyurethane insulation, it take 8 h for the water
temperature to drop by 1
◦
C.

i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 256 — #283
i
i
i
i
i
i
256 Energy Storage
Here is a numerical example. If D = L =1m,τ = 5 cm = 0.05 m, T
w
=80
◦
C, and
T
a
=20
◦
C, the rate of temperature drop is
dT
dt
=
6 × 0.02
0.05 × 4.19 × 10
6
× 1
× 60 = 3.4 ×10
−5 ◦
C/s=0.124
◦
C/h. (12.9)
The temperature takes 8 h to drop by 1
◦
C. Such an energy storage unit is extensively
used in hot-water systems.
12.1.2 Solid Sensible Heat Storage Materials
In contrast to water and other liquids, solid materials can provide a larger temperature
range and can be installed without a container. However, thermal conductivity be-
comes a significant parameter. Table 12.2 shows the thermal properties of typical solid
materials. For many items, such as soil and rock, the values are only approximate or
an average, because those materials vary widely. For example, the thermal parameters
of soil could vary by one order of magnitude depending on the water content.
As shown in Table 12.2, materials with high thermal conductivities usually have
low heat capacity. To use solid materials with high heat capacities, a long temperature
equalizing time is expected.
Table 12.2: Thermal Properties of Solid Materials
Material Density Heat Product Thermal
ρ Capacity c
p
ρc
p
Conductivity k
(10
3
kg/m
3
)(10
3
J/kg·K) (10
6
J/m
3
·K) (W/m·K)
Aluminum 2.7 0.89 2.42 204
Cast iIron 7.90 0.837 3.54 29.3
Copper 8.95 0.38 3.45 385
Earth (wet) 1.7 2.1 3.57 2.5
Earth (dry) 1.26 0.795 1.00 0.25
Limestone 2.5 0.91 2.27 1.3
Marble 2.6 0.80 2.08 2.07 - 2.94
Granite 3.0 0.79 2.37 3.5
Bricks 1.7 0.84 1.47 0.69
Concrete 2.24 1.13 1.41 0.9 - 1.3
Wood (oak) 0.48 2.0 0.96 0.16
Source: American Institute of Physics Handbook ,3rdEd.,
American Institute of Physics, New York, 1972; and Ref. [31].
i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 257 — #284
i
i
i
i
i
i
12.2 Phase Transition Thermal Storage 257
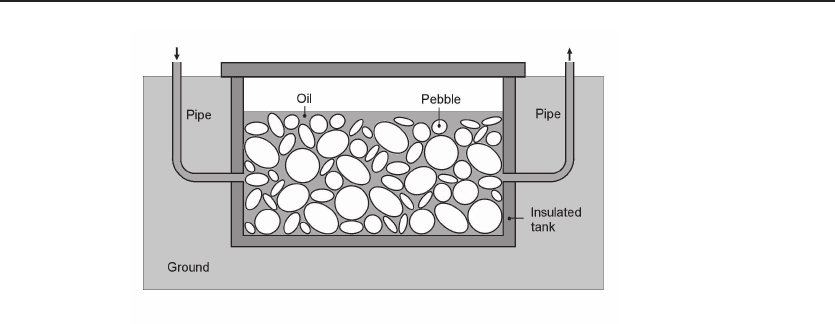
Figure 12.2 Rock-bed thermal energy storage system. Using a mixture of synthetic oil
and pebbles, a thermal energy storage system at high temperature (e.g. 400
◦
C) can be built with
reasonable cost. The heat conduction is mainly through convection of oil, and the pebbles provide
heat capacity.
12.1.3 Synthetic Oil in Packed Beds
Because the temperature range of water is limited, in order to store sensible heat
at higher temperature, for example, in solar power generation systems, synthetic oil
should be used. However, synthetic oil is expensive. A compromised solution is to use a
mixture of synthesized oil and inexpensive solid materials, such as pebbles. Figure 12.2
shows such a thermal energy storage system schematically. A thermal energy storage
system at high temperature (e.g. 400
◦
C) can be built with limited cost. The heat
conduction is mainly through convection of oil, and the pebbles provide heat capacity.
12.2 Phase Transition Thermal Storage
In sensible heat thermal energy storage systems, the process of charging or discharging
of energy is related to a change of temperature, and the temperature is related to the
amount of heat energy content. The storage density is limited by the heat capacity
of the material. Using phase-change materials (PCMs), a considerably higher thermal
energy storage density can be achieved that is able to absorb or release large quantities
of energy (“latent heat”) at a constant temperature by undergoing a change of phase.
Theoretically, three types of phase changes can be applied: solid–gas, liquid–gas and
solid–liquid. The first two phase changes are generally not employed for energy storage
in spite of their high latent heats, since gases occupy large volumes. Large changes in
volume make the system large, complex, and impractical. Solid–liquid transformations
involve only a small change in volume (often only a few percent) and are therefore
appropriate for phase-change energy storage.

i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 258 — #285
i
i
i
i
i
i
258 Energy Storage
Table 12.3: Commonly Used Phase-Change Materials.
Materials Transition Density Latent
Temperature ρ Heat h
◦
C10
3
kg/m
3
10
6
J/m
3
Water-ice 0 1.00 335
Paraffin wax 58 – 60 0.90 180 – 200
Animal fat 20 – 50 0.90 120 – 210
CaCl
2
(6→2)H
2
O 29 1.71 190.8
Na
2
SO
4
(10→0)H
2
O 32.4 1.46 251
Ba(OH)
2
(8→0)H
2
O 72 2.18 301
MgCl
2
(6→4)H
2
O 117 1.57 172
Source: Refs. [28] and [31].
In general, the heat absorbed or released during the phase transition is
ΔQ = H
2
− H
1
, (12.10)
where H is the enthalpy before and after the transition. The latent heat h, or specific
enthalpy, is defined by
H = hV, (12.11)
where V is the volume of the material.
Table 12.3 shows the thermal properties of several commonly used PCMs. A good
PCM should provide energy storage at the desired temperature, have a large latent
heat and a small volume change, and be non-flammable, noncorrosive, nontoxic, and
inexpensive.
In general, PCMs are more expensive than sensible heat systems. They undergo
solidification and therefore cannot generally be used as heat transfer media in a solar
collector or the load. A separate heat transport medium must be employed with a
heat exchanger in between. Many PCMs have poor thermal conductivity and therefore
require large amount of heat exchange liquid. Others are corrosive and require special
containers. These increase the system cost.
12.2.1 Water–Ice Systems
As shown in Table 12.3, the latent heat of the freezing of water or melting of ice is one
of the highest. The water-ice system has already used in industry to save energy in
air-conditioning systems. Figure 12.3 is a photo of a water–ice energy storage system,
named Ice Bear, designed and manufactured by Ice Energy, Inc. The system has a
large insulated tank filled with water and a lot of copper heat exchange coils. During
the night, the refrigerator uses inexpensive electricity and cool air to make ice from the
water. As we shown in Chapter 6, the lower the ambient temperature, the higher the

i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 259 — #286
i
i
i
i
i
i
12.2 Phase Transition Thermal Storage 259
Figure 12.3 Ice Bear energy storage system. An insulated tank is filled with water and many
copper heat exchange coils. During the night, the refrigerator uses inexpensive electricity and cool air
to make ice from the water. As shown in Chapter 6, the lower the ambient temperature, the higher
COP. Therefore, to make a well-defined mass of ice during the night, the electricity cost is much lower
than in the hot daytime. Courtesy of Ice Energy, Inc.
coefficient of performance (COP). Therefore, to make a well-defined mass of ice during
the night, the electricity cost is much lower than in the hot daytime. During the hot
daytime, the system uses the ice to cool the building. With this system, the efficiency
of energy storage can be better than 90%. The overall energy savings can be as high
as 30%.
Below is a numerical example of a water–ice system. Using the insulated container
in Fig. 12.1, the total latent heat is
Q =ΔH = H
2
− H
1
, (12.12)
Using Eq. 12.3,
ΔH =
1
4
πhD
2
L. (12.13)
For a tank of D = L = 1 m, the total enthalpy of phase transition is
ΔH = 335 × π × 4 ×10
6
J=2.63 × 10
8
J. (12.14)
Iftheambienttemperatureis20
◦
C, for such a tank, according to Eq. 12.6, the rate of
heat loss is
dQ
dt
=
3 × π × 0.02
2 × 0.05
× 20 = 1.89 W. (12.15)
If at the beginning the tank is full of ice, then it can keep the temperature at 0
◦
Cfor
2.63 × 10
8
/1.89 = 1.39 × 10
8
s, or 4.4 years. Therefore, using a moderate means, the
energy storage is efficient.
The freezing temperature of pure water is 0
◦
C. If the working temperature is other
than 0
◦
C, a mixture of water and other materials can do the job. An example is
solar-operated refrigerator (U.S. Patent 7,543,455). The freezer should work at around
-10
◦
C. Because the refrigerator is used for food and medicine, the additive must be
i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 260 — #287
i
i
i
i
i
i
260 Energy Storage
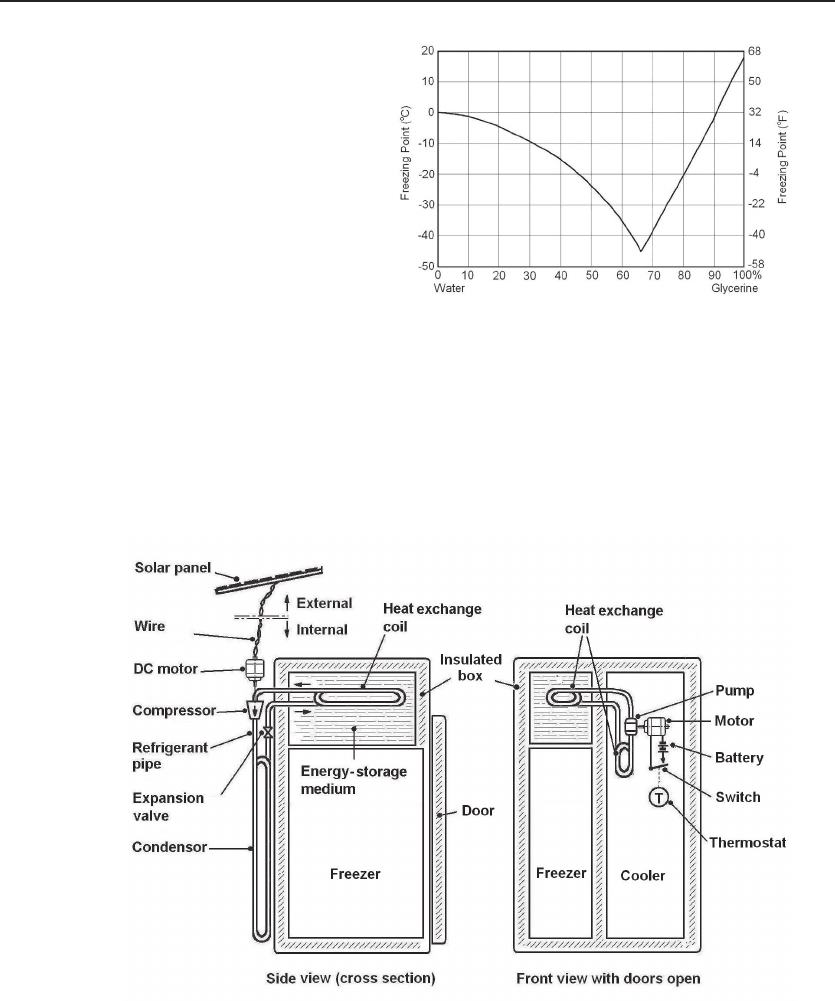
Figure 12.4 Freezing point
of water–glycerin system.
By mixing water and glycerin,
the freezing point can be low-
ered from 0 to -40
◦
C depend-
ing on the ratio. By mixing
with a few percent of alcohol,
the freeing point ca be lowered
further.
nontoxic. Glycerin and ethanol are good additives because both are ingredients of food
and medical substances. Figure 12.4 shows the freezing points of a mixture of water
and glycerin. By mixing with alcohol, the freezing temperature can be lowered further.
Glycerin is a byproduct of biodiesel production, and there is a surplus of raw glyc-
erin. The impurities in raw glycerin as byproducts of biodiesel production are water,
salt, fatty acids and alcohol, and there is no need to use high-purity glycerin. Therefore,
Figure 12.5 Solar-powered refrigerator. The design of a solar-powered refrigerator. The com-
position of the water–glycerin–alcohol system determines the working temperature of the freezer. The
temperature of the cooler is controlled by a thermostat to the brine circulation, which can be set by
the user. See U.S. Patent 7,543,455.

i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 261 — #288
i
i
i
i
i
i
12.2 Phase Transition Thermal Storage 261
economically it is advantageous. Figure 12.5 shows the design of a solar-powered refrig-
erator. The composition of the water–glycerin–alcohol system determines the working
temperature of the freezer. The temperature of the cooler is controlled by a thermostat
to the brine circulation, which can be set by the user.
12.2.2 Paraffin Wax and Other Organic Materials
Paraffin wax is a byproduct of petroleum refining. The melting point of paraffin wax
ranges from 50 to 90
◦
C. Currently, paraffin wax only has a few commercially valuable
applications, such are candles and floor wax. For such applications, only these with
melting temperature between 58 and 60
◦
are usable. But the supply is abundant.
The melting temperature of paraffin matches the range needed for space heating and
domestic hot water. It is also nontoxic and noncorrosive. One problem is its low
thermal conductivity. This can be mitigated with encapsulation; see Section 12.2.4.
Other organic materials have similar properties as paraffin wax. An example is
animal fat. Lard and chicken fat are considered harmful to human health because they
can increase blood triglyceride and cause obesity. In some sense they are wastes of the
food-processing industry. Animal fat is nontoxic and noncorrosive, thus it can be safely
utilized for energy storage in residential environments.
12.2.3 Salt Hydrates
Many inorganic salts crystallize with a well-defined number of water molecules to be-
come salt hydrates. Heating a salt hydrate can change its hydrate state. For example,
hydrated sodium sulfate (Glauber’s salt) undergoes the transition at 32.4
◦
C
Na
2
SO
4
10H
2
O+ΔQ −→ Na
2
SO
4
+ 10H
2
O. (12.16)
In general, the transition is
Salt mH
2
O+ΔQ −→ Salt nH
2
O+(m −n)H
2
O. (12.17)
Thus, at the melting point the hydrate crystals break up into anhydrous salt and water
or into a lower hydrate and water. The latent heat could be quite large, thus the
storage density could be very high. If the water released is sufficient, a water solution
of the (partially) dehydrated salt is formed.
These salt hydrates can be used in solar-operated space-heating or hot-water sys-
tems to provide uniform temperature over a longer period of time.
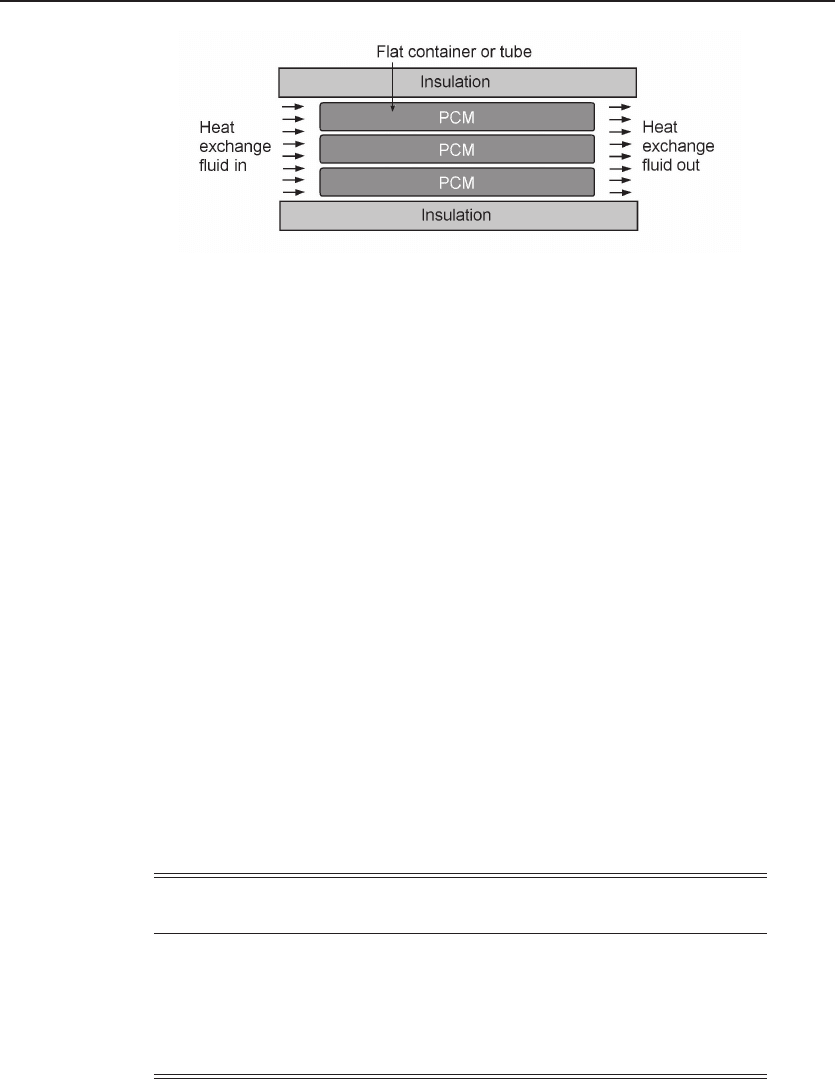
12.2.4 Encapsulation of PCM
To mitigate the problem of low thermal conductivity of PCMs, the material is often
encapsulated in various forms. Figure 12.6 shows an example of a PCM encapsulated
in flat or tubular parcels. A heat transfer fluid is required to make it operational.
i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 262 — #289
i
i
i
i
i
i
262 Energy Storage
Figure 12.6 Encapsulation of PCM. An example of PCM encapsulated in flat or tubular parcels,
to mitigate the low thermal conductivity of PCM.
12.3 Rechargeable Batteries
In the last decades, the technology of rechargeable batteries has observed a phenomenal
expansion. To date, the century-old lead–acid rechargeable battery has been constantly
improved and is still in widespread use. New types of batteries, especially lithium ion
rechargeable batteries, are experiencing an explosive growth, and will soon become the
dominating distributed storage device for electricity.
Table 12.4 gives the specifications of several types of rechargeable batteries. One of
the basic parameters is nominal voltage. Cells with higher nominal voltage are certainly
advantageous, because fewer cells are needed to construct the desired system. Energy
density and specific energy are also significant parameters. For static applications such
as street lights, a smaller specific energy is not a serious problem. For automobile
applications, energy density and specific energy are critical parameters. Lifetime is
also a critical parameter for automobile applications. Not shown here is the unit cost.
Currently, lead–acid batteries are still the least expensive and thus widely used.
Table 12.4: Comparison of Rechargeable Batteries
Type Voltage Energy Density Specific Energy Lifetime
(V) (Wh/liter) (Wh/kg) (cycles)
Lead–acid 2.1 70 30 300
NiMH 1.4 240 75 800
LiCoO
2
3.7 400 150 1000
LiMn
2
O
4
4.0 265 120 1000
LiFePO
4
3.3 220 100 3000
i
i
“ChenSolarEnergy” — 2011/5/17 — 17:56 — page 263 — #290
i
i
i
i
i
i
12.3 Rechargeable Batteries 263
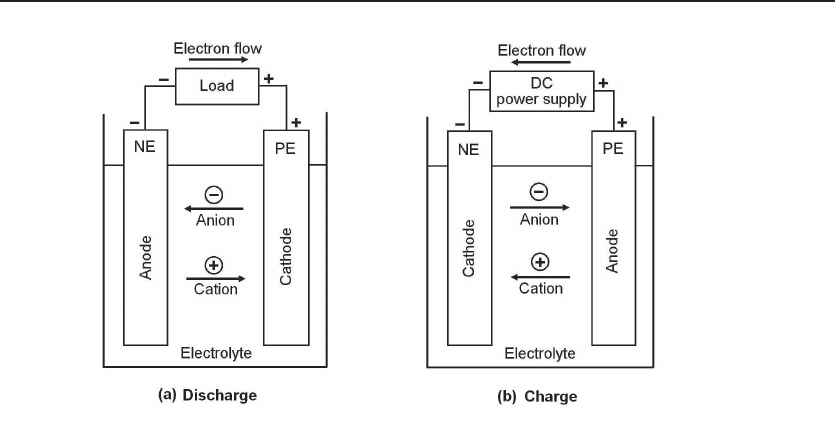
Figure 12.7 Electrochemistry of rechargeable batteries. (a) Discharging process. By con-
necting the cell to an external load, electrons flow from NE, the anode, to PE. The electric circuit is
completed in the electrolyte by the flow of anions and cations to the anode and cathode, respectively.
(b) Charging process. An external DC power supply forces electrons to flow into NE, then the circuit
is completed in the electrolyte by the flow of anions and cations.
12.3.1 Electrochemistry of Rechargeable Batteries
The basic structure and the charging–discharging processes of rechargeable batteries
are shown in Fig. 12.7. For reference, definitions are provided as below. For more
details, see, for example, Handbook of Batteries [51].
Cell The basic electrochemical unit converting electrochemical energy to electrical
energy.
Battery One or more electrochemical cells connected in series or parallel to provide
electrical power.
Primary cells or batteries One-time source of electricity, cannot be recharged after
usage. Are discarded after usage.
Secondary (rechargeable) cells or batteries Can be recharged electrically after
usage to their original condition.
Oxidation Loss of electron(s).
Reduction Gain of electron(s).
Redox Reduction and oxidation.
