Givan A.L. Flow Cytometry. First Principles
Подождите немного. Документ загружается.

trol of an exogenous inducer) and into the nematode Caenorhabditis
elegans (under the control of a cell-lineage-related promoter). Subse-
quent work in this ®eld has elaborated a series of GFP-derived ¯uo-
rochromes of di¨erent colors (BFP, CFP, and YFP). They have been
used as reporters to detect induction of di¨erent proteins or, alter-
natively, have been engineered as fusion proteins so that, without
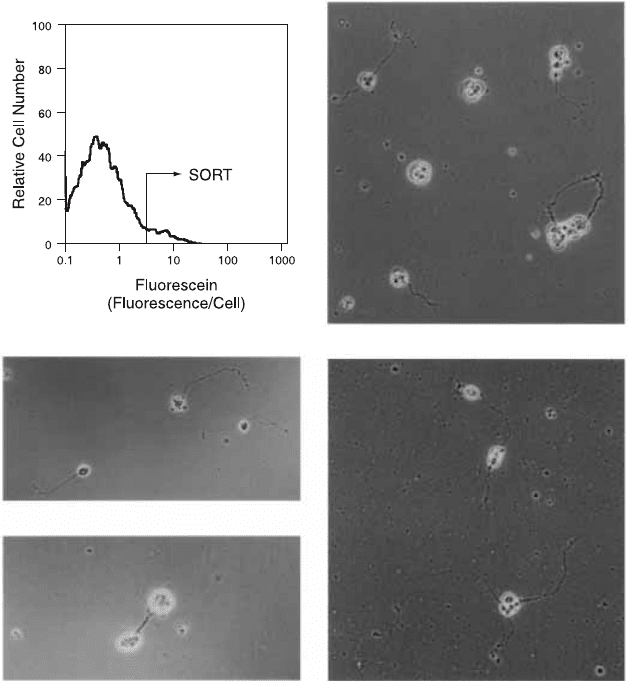
Fig. 11.10. When lacZ has been transfected into Drosophila embryos in association
with a neuronal-cell-speci®c promoter, the cells that ¯uoresce brightly in the presence
of ¯uorogenic b-galactosidase substrate will, when sorted, develop neuronal pro-
cesses in culture. From Krasnow et al. (1991). Science 251:81±85. Copyright AAAS.
Flow Cytometry210
staining, the presence of these proteins can be assayed by ¯ow cy-
tometry and their cellular location visualized by microscopy. Flow
sorting has had an important role to play in the development of
stable GFP-fusion protein transfected cell lines (Fig. 9.4 shows an
example of sorting for GFP ¯uorescence).
MICROBIOLOGY
Although microorganisms would seem to be ideal candidates for ¯ow
analysis, ¯ow cytometry has been slow to make its presence felt in the
®eld of microbiology. To a great extent, this is attributable to limi-
tations of the instrumentation; ¯ow chambers and sheath ¯uid and
electronics designed for eukaryotic cells have often not worked de-
pendably well with the smaller members of our universe. Without
the help of auto¯uorescent pigments that aid oceanographers in the
identi®cation of phytoplankton, microbiologists have di½culty in re-
solving low-intensity scatter and ¯uorescence signals from debris and
instrument noise. A considerable amount of work has aimed at
overcoming some of these instrumental di½culties; many cytometers,
if well-aligned, perform acceptably with small bacterial cells.
Much of the work on microorganisms in ¯ow systems has con-
cerned yeast, algae, and protozoa; although smaller than mammalian
cells, these eukaryotes are considerably larger than most bacteria.
DNA, RNA, protein, and light scatter measurements have been
made on these organisms, and the feasibility of cell cycle analysis has
been demonstrated. Bacteria, however, present more acute di½culties.
The diameter of bacterial cells is perhaps 1 mm (compared with 10 mm
for mammalian blood cells), and therefore the surface area to be
stained (and resulting ¯uorescence intensity) is 10
2
less than that of a
mammalian cell. The DNA content of the E. coli genome is about
10
ÿ3
times that of a diploid human cell. Hence, bright dyes and sen-
sitive instrumentation are required for studies of bacteria. Neverthe-
less, reasonable DNA histograms of bacteria can be obtained by ¯ow
cytometry. Methods are being developed to investigate cell cycle
kinetics, the e¨ects of antibiotics, and the detection and identi®cation
of bacteria for clinical investigations.
However, a technique developed at the Massachusetts Institute of
Research Frontiers 211
Technology has approached the problem with the ``if you can't beat
them, join them'' philosophy of allowing bacteria to masquerade as
larger particles. The technique involves the creation of ``salad oil''
emulsions of drops of agar within bacterial suspensions in bu¨er
solution. By adjusting the size of the drops and the concentration of
the bacteria, it is possible to arrange conditions so that, on average,
each drop of gel contains one bacterial cell. The microdrop then be-
comes a minicontainer for the bacterial cell, allowing the di¨usion of
stain, nutrient, antibiotics, and so forth, but containing the bacterial
cell and all its progeny.
The usefulness of this technique has been shown by its ability to
detect the division of these bacterial cells. By staining the cells within
the droplets in some way (e.g., for DNA or protein content), the
original culture will form a single ¯ow histogram peak representing
gel microdroplets, each ¯uorescing with an intensity related to the
DNA or protein content of its entrapped single bacterial cell. After
one replication cycle in which all the bacteria are replicating, each
droplet will then contain two cells and have twice the original ¯uo-
rescence intensity. Alternatively, if only some of the bacteria are
replicating, a small population of gel droplets with twice the ¯uores-
cence intensity will appear. The droplets containing replicating cells
will then progress to 4-fold, 8-fold, and 16-fold intensity as replica-
tion continues (Fig. 11.11). The technique can provide a sensitive
method for studying small particles as well as a very rapid assay for
the replication of a small proportion of bacterial cells in the presence
of antibiotics, growth factors, or varied growth conditions (Fig.
11.12).
Although this gel microdroplet method is still new (even after 10
years) and its potential applications relatively untested, it has been
described here because it can teach us certain general lessons. It
serves to remind us that cytometry, despite its name, does not neces-
sarily involve the ¯ow analysis of cells; particles of many sorts will do
just as well. It is also of interest as a method that has, in fact, in-
stitutionalized the formation of clumped cells that most workers try
so hard to avoid. In addition, it has provided us with a way to make
a small cell into a larger (¯ow-friendly) particle. Finally, it has given
us inspiration by exemplifying the way in which lateral thinking can
extend the impact of ¯ow cytometry in new directions.
Flow Cytometry212
MOLECULAR BIOLOGY
Flow cytometry has been applied in many creative ways to the
science of molecular biology. Flow sorting, based on Hoechst 33258
and chromomycin A3 ¯uorescence, turns out to be one of the best
ways available for obtaining relatively pure preparations of each type
of chromosome. Even those chromosomes that are not distinguish-
able by their ¯uorescence (e.g., 9-12) can usually be sorted from
hamster±human hybrid cell lines. These preparations of reasonably
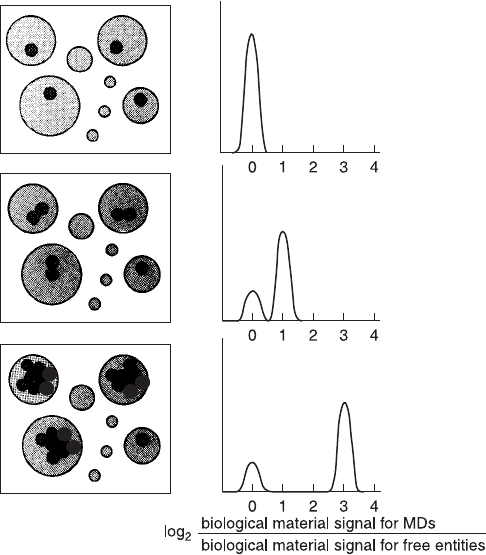
Fig. 11.11. Illustration of the use of gel microdroplets for sensing growth at the level
of one cell growing into a two-cell microcolony. By staining the cells within micro-
droplets with, for example, a DNA-speci®c ¯uorochrome, a small subpopulation of
cells dividing more or less rapidly than most could be detected in a ¯ow histogram of
microdroplet ¯uorescence. From Weaver (1990).
Research Frontiers 213
pure ¯ow-sorted chromosomes have been the starting material for
obtaining chromosome-speci®c DNA libraries. The Human Genome
Project has made extensive use of chromosomes obtained with the
sorting cytometers at Los Alamos and Livermore. The bottleneck in
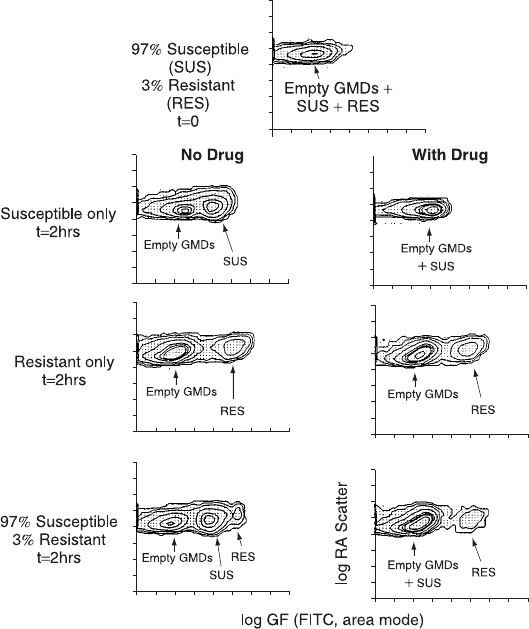
Fig. 11.12. The use of gel microdroplets and ¯ow cytometry to assay drug sensitivity
of bacterial cells. The ®gure shows side scatter and green ¯uorescence contour plots
of gel microdroplets (GMDs) containing E. coli cells that have been stained with
¯uorescein isothiocyanate for total protein. The microdroplets have been analyzed in
the ¯ow cytometer either at time 0 or 2 h after incubation in control medium (left
plots) or medium containing penicillin (right plots). A model system was created by
mixing two strains of bacteria (susceptible or resistant to penicillin). The data show
that a small subpopulation of resistant cells could be detected within 2 h because of
its rapid growth in comparison to susceptible cells. From Weaver et al. (1991).
Flow Cytometry214

this technique is the time it takes for sorting. In a conventional sorter,
if the ¯ow rate is limited to about 1000 particles per second, then
human chromosomes of a particular type could theoretically be
sorted at a rate of about 40 per second (1000/23). Thus it would take
about 7 h to obtain 10
6
chromosomes of a given type under optimal
conditions. The high-speed drop sorters at Los Alamos and Liver-
more were developed with the demands of the Human Genome
Project in mind. The technique for optical (zapper inactivation) sort-
ing is the next step in this evolution (Table 11.1).
Even small numbers (20,000 or so) of sorted chromosomes can be
used quite neatly to aid in the mapping of genes to chromosomes.
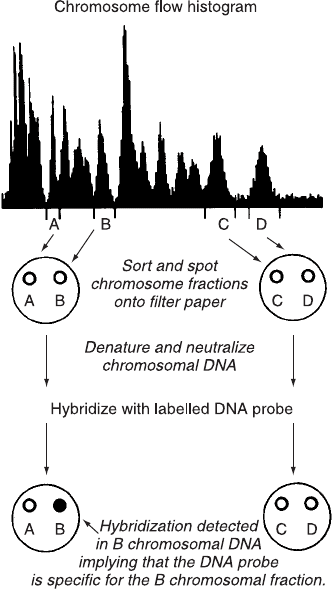
Chromosomes of each type are simply sorted (two at a time: one type
to the left, the other to the right) onto a nitrocellulose ®lter. The
DNA is then denatured on the ®lter where it can be hybridized to a
radioactive gene probe. Autoradiography of the ®lter will then reveal
whether the probe has hybridized to the DNA from any given chro-
mosome (Fig. 11.13). In this way, the sorting of small numbers of
each of the chromosomes onto ®lters allows the mapping of any
available gene probe to its chromosome.
Much like chromosomes, DNA fragments generated by restriction
enzyme digestion of native DNA, when stained with DNA-speci®c
TABLE 11.1. Chromosome Sorting
Application Chromosomes required
Polymerase chain reaction chromosome paints 3:0 10
2
Viral cloning (17 kb inserts) 1.6 10
6
Cosmid cloning (37 kb inserts) 6:4 10
6
Yeast arti®cial chromosome cloning 6:4 10
7
Sorter
Chromosomes
sorted per
24 h day
Estimated sorting time
for YAC cloning
(6:4 10
7
chromosomes)
Conventional 3 10
6
21 days
High speed 1.5 10
7
4 days
Optical zapper 7:5 10
7
0.8 days
After Roslaniec MC et al. (1997). Hum. Cell 10: 3±10.
Research Frontiers 215
¯uorochromes, ¯uoresce more or less brightly according to their
length. Flow data acquisition could, in theory, provide histograms
indicating the relative proportions of di¨erent-sized fragments in a
digest. Implementation of this application has not been trivial be-
cause, compared with chromosomes, fragments of DNA are much
smaller and ¯uoresce much less brightly. Rising to this challenge,
scientists have developed ultrasensitive ¯ow cytometric systems by
using high-e½ciency optics and light detection, bright ¯uorochromes,
and very slow sheath velocities of approximately 2±4 cm per second
(compare this with the usual cytometers with velocities of 10 m [1000
cm] per second). With slow ¯ow rates, DNA fragments spend longer
Fig. 11.13. DNA blot analysis using spots from chromosomes sorted directly onto
®lter paper on the basis of their Hoechst 33258 ¯uorescence. From Van Dilla et al.
(1990).
Flow Cytometry216
time in the laser beam, thereby emitting more ¯uorescence per frag-
ment and increasing instrument sensitivity for detection of short
fragments with low intensities. As an alternative to pulsed-®eld gel
electrophoresis, the ¯ow methodology records similar information
about the distribution of fragment sizes, but with better resolution,
greater accuracy, and higher sensitivity. In addition the ¯ow method
requires less DNA and less time. In one proof-of-principle applica-
tion of this technology, when DNA from bacteria has been broken
into fragments by digestion with restriction enzymes, the size distri-
bution of the fragments is typical of a given strain of bacteria.
Therefore the ¯ow histogram that is derived from sending the frag-
ments through a ¯ow cytometer is a signature of the particular bac-
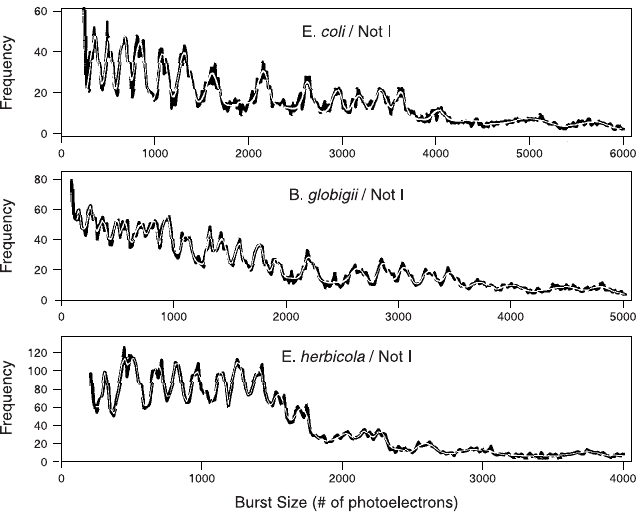
terial strain and can be used for identi®cation (Fig. 11.14).
Fig. 11.14. Flow cytometric signatures distinguishing three di¨erent strains of bac-
teria according to the size distributions of DNA fragments generated by restriction
enzyme digestion. From Kim et al. (1999).
Research Frontiers 217
Workers with James Jett at Los Alamos have been developing
cytometers that are able to detect the ¯uorescence from single mole-
cules and have been applying this capability to the problem of DNA
sequencing. The proposed technique involves the synthesis of a com-
plementary strand of DNA with ¯uorescently tagged precursors (each
base of a di¨erent color). The labeled (¯uorescent) duplex DNA
molecule is then attached to a microsphere and the microsphere sus-
pended in a ¯ow stream where it can be sequentially cleaved with an
exonuclease. The cleaved ¯uorescent bases from the molecule would
then be identi®ed by their color as they pass through the laser beam,
and the sequence in which the colors appear will re¯ect the base se-
quence in the DNA. It is projected that between 100 and 1000 bases
per second could be sequenced, although there are still obstacles to be
surmounted before this technique becomes a practical reality.
MULTIPLEX CYTOMETRY FOR SOLUBLE ANALYTES
With a continuation of the lateral thinking that has marked devel-
opments in ¯ow cytometry for many years, it is now possible to use a
simple ¯ow cytometer to analyze multiple soluble analytes in a single
test tube. Sets of beads can be manufactured with distinctive ratios
of two di¨erent ¯uorochromes (for example, red and orange). By
precisely controlling the ratios of these two dyes, bead sets can be
obtained with 100 di¨erent speci®cations (according to their red/
orange balance). When run on a ¯ow cytometer, the beads cluster
into 100 separate regions on a two-color (red vs. orange) dot plot
(Fig. 11.15). In a multiplexed version of an ELISA assay, each bead
type can be linked to a di¨erent capture molecule; the capture mole-
cule will bind a soluble target analyte to the bead (compare this with
the solid-phase capture molecules on an ELISA plate). For example,
capture molecules on the beads might be a range of allergens (to bind
antibodies from the serum for allergy pro®ling) or nucleotide probes
for single-nucleotide polymorphisms (SNPs) or antibodies to a range
of cytokines.
After preparation (with beads of each red/orange ratio having
been linked to capture molecules for speci®c targets), the beads can
all be combined in a single tube. For the assay, a test solution is
added to the mixed bead suspension. After incubation, centrifugation,
Flow Cytometry218
and washing, ¯uorescein-conjugated detection reagents are added to
the incubation mixture. Much like the top layer in an ELISA assay,
the detection reagents will bind to the target analytes on the beads.
After running the tube of beads through the ¯ow cytometer, the ®nal
read out comes from gating on each one of the 100 types of beads in
turn and then (knowing the particular capture antibody on that type
of bead) reading the green ¯uorescence intensity on that red/orange
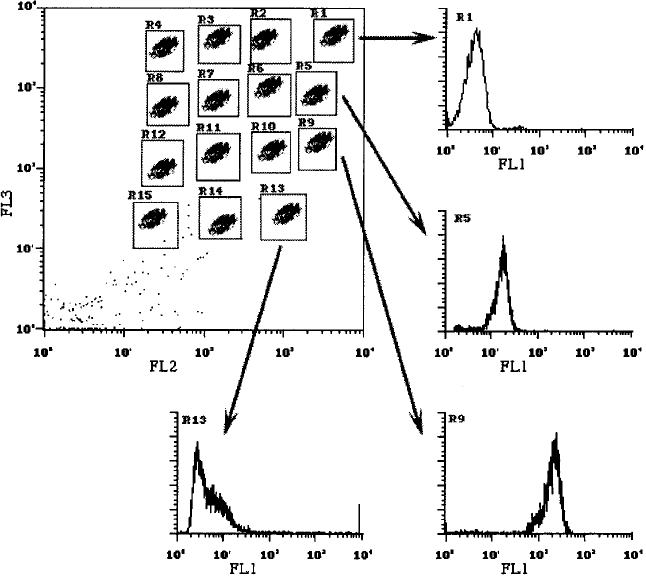
Fig. 11.15. A multiplex bead array that can be used to capture multiple soluble an-
alytes. In the dot plot, 15 types of beads are distinguished by their red/orange ratios;
they capture 15 di¨erent cytokines. For example, the beads in Region 1 capture
interleukin (IL)-9; in Region 5, IL-2; in Region 9, IL-5; and in Region 13, MCP-1.
The green ¯uorescence histograms from each region show, by their intensity, how
much cytokine has been captured by each type of bead. Standard curves can relate
the green ¯uorescence intensity to the concentration of cytokine in the sample. The
experimental format illustrated in the cartoon here is patterned directly from work
by RT Carson and DAA Vignali (1999).
Research Frontiers 219
