Heidersbach R. Metallurgy and Corrosion Control in Oil and Gas Production
Подождите немного. Документ загружается.


CORROSION CONTROL 169
− 850 mV CSE Criterion Over the years, many author-
ities came to the conclusion that the − 850 mV CSE
potential advocated by R. Kuhn and his colleagues in
Louisiana was the easiest and most reliable way to
determine if cathodic protection had been achieved.
This idea was incorporated into the fi rst international
standard on cathodic protection, NACE RP0169 (since
changed to SP0169).
102
Mr. Kuhn ’ s arguments were
based on leak records that showed that if cathodically
protected structures were kept at − 850 mv or more com-
pared to CSE, then leaks due to corrosion were substan-
tially eliminated.
76
−
79,103,104
This approach was reinforced by Peabody, who pub-
lished a practical galvanic series of metals in soil in 1967
(Table 6.26 ).
103
This showed that carbon steel ( “ mild
steel ” in Peabody ’ s terminology) would have a native,
or unprotected, potential of somewhere between − 0.2
and − 0.8 V CSE. Thus, Kuhn ’ s recommended potential
of − 0.85 V ( − 850 mV) is at least a 50 mV shift in the
cathodic direction, and usually much more. Peabody
and Parker were the two standard references on cathodic
protection of pipelines in 1969 when NACE RP169 was
fi rst published, and both books advocated the − 850 mV
criterion, although they do discuss other criteria for
determining if cathodic protection has been achieved.
103,104
Many authorities pointed out, and still do, that it is
unnecessary to have steel at − 850 mV CSE in order to
achieve cathodic protection.
105
While this has always
been the case, most owner operators choose to use the
− 850 mV criterion in NACE SP0169 and similar stan-
dards, because it is easy to measure and to train inspec-
tors on how to perform the necessary measurements.
Electricity is generally cheaper than trained labor, which
is necessary to inspect according to the other, more
complicated, criteria.
In cases where microbially infl uenced corrosion
(MIC) is suspected or at elevated temperatures, the
protection potential is considered to be − 950 mV
CSE.
79,85,102
Little controversy has appeared over the idea of a
similar criterion for locations where MIC is suspected.
The change of potential to − 950 mV due to temperature
is not controversial and can be understood by anyone
who considers the Nernst equation, developed long
before cathodic protection was common, that clearly
explains why electrode potentials for any reaction will
be affected by temperature. This is in contrast to the
continuing controversy over the necessity to use an
“ instant off ’ ” or similar IR - compensation technique to
identify the “ true ” potential of a structure.
All of the above discussion has related to buried
structures, primarily pipelines. Other reference elec-
trodes are used in different applications. The corre-
sponding voltage for silver - silver chloride electrodes,
which are used in seawater, is − 805 mV, although this is
usually rounded to − 800 mV.
100
− 100 mV Shift Criterion Advocates of the − 100 mV
shift criterion point out that − 850 mV CSE is not neces-
sary to achieve cathodic protection (an acceptable
reduction in corrosion activity) in many, perhaps most,
cases.
105,106
They also claim that in some circumstances − 850 mV
CSE, however determined, may not produce protection.
This latter claim is very controversial and, except in the
cases of elevated temperature, parallel zinc anodes, or
microbial activity, it has not been unequivocally
documented.
The − 100 mV shift criterion assumes that unshifted
potentials can be determined. This is impractical for
galvanic anode systems. It also assumes that the
unshifted (or native) potential of the structure does not
change with time. Areas with changing groundwater
levels due to seasonal wet and dry seasons are one
example of where native (unprotected) potentials are
likely to change.
Turning off ICCP systems to determine the native
potential requires up to 48 h for the potential to decay
to the unshifted potential.
The diffi culties and limitations discussed above have
led most operators to prefer to use the − 850 criteria for
determining cathodic protection.
E Log i Criterion There are structures where it is
inconvenient or impossible to place reference elec-
trodes along the structure being protected. Well casings
are an excellent example of this situation. While the top
of the well casing is available for electrical connections,
the bottom of the casing is inaccessible. The E log i
criterion (Tafel curve method) is used in these situations
to measure the current necessary to provide cathodic
protection.
TABLE 6.26 Practical Galvanic Series
Metal
Volts
(CSE)
Noble or
cathodic
Copper, brass, bronze
− 0.2
Mill scale on steel
− 0.2
Mild steel (rusted)
− 0.2 to − 0.5
Mild steel (clean and shiny)
− 0.2 to − 0.8
Active or
Anodic
Zinc
− 1.1
Magnesium
− 1.75
Note : Condensed from Table 2.2 in Peabody, Control of Pipeline
Corrosion .
103
c06.indd 169c06.indd 169 10/29/2010 10:35:57 AM10/29/2010 10:35:57 AM
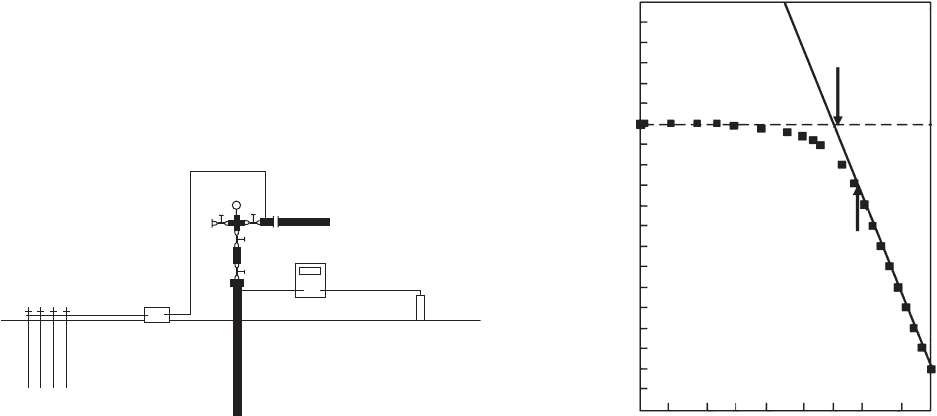
170 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
Figure 6.74 Test setup for E - log i testing to determine the
necessary cathodic protection current for a well casing.
112
Electrical isolation
Voltmeter
Rectifier
+–
Test ground
bed
Well casing
Reference electrode
Figure 6.75 Plot of E log i data for a well casing.
Log current →
I
CP
I
corr
E
corr
Potential →
Figures 6.74 and 6.75 show how this is done. A
cathodic current source is connected to the casing and
current is applied. The potential at the casing head is
measured before the current is applied and the change
in potential is monitored as additional cathodic current
is applied to the casing.
At the corrosion potential no current is being sup-
plied from the external power supply, and the corrosion
potential, E
corr
, is due to the natural oxidation and reduc-
tion reactions on the structure. The total oxidation and
reduction currents are unknown. As current is supplied
from an external power source, the potential versus log
of applied current plot begins to curve downward when
the applied current is similar in magnitude to the natural
current. Increased applied current leads to a situation
where virtually all of the cathodic current is coming
from the power supply. The plot then becomes linear or
straight. The applied current where the potential - log
current plot becomes “ straight ” or linear is assumed to
be the current necessary to provide adequate cathodic
protection. At one time, it was suggested that the “ linear ”
portion of the E log i plot should extend over one
decade (or order of magnitude) of current. In recent
years, a two - decade (100 - fold change in current) linear
region has been recommended.
107 – 109
The current requirement from the E log i method is
considered conservative, and leak records seem to
confi rm that idea.
110 – 112
The E log i or Tafel extrapolation method can also
be used to determine the corrosion current. This is dis-
cussed in Chapter 7 , Inspection, Monitoring, and Testing.
Inspection and Monitoring
The continued operation of cathodic protection systems
requires monitoring to insure that the system is working
adequately. Third - party damage, coating degradation
leading to increased current demands, changes in envi-
ronment, and ageing of cathodic protection components
can all cause systems to degrade. There are many inspec-
tion and monitoring methods used in conjunction with
cathodic protection. This section only discusses some of
the more important methods. More complete discus-
sions are available.
78,106 – 118
Inspection and monitoring is
required on at least an annual basis for most pipeline
systems in the United States, and more frequent inter-
vals are sometimes necessary. Rectifi er operations must
be monitored every 2.5 months in the United States.
Potential Surveys The most common means of inspect-
ing a cathodically protected structure is by means of a
potential survey. In any potential survey, it is necessary
to measure the potential of the structure in question
relative to a standard potential. The most commonly
used reference electrode is the saturated copper - copper
sulfate electrode (CSE), which is used onshore and in
freshwater applications. This is shown in Figure 6.76 .
Silver - silver chloride electrodes are used in marine
applications, and the conversion from one standard to
the other is fairly simple. The − 850 mV CSE standard
theoretically becomes − 805 mV with the silver - silver
chloride electrode, but it is usually rounded to − 800 mV.
Zinc is sometimes used as a robust reference anode
for permanently mounted test stations on offshore
structures.
Reference electrodes can degrade and must be main-
tained.
119
It is common for inspectors to carry three
electrodes with them. Two are used and checked against
each other. If they do not produce the same result, they
are then checked against a “ less weathered ” electrode
c06.indd 170c06.indd 170 10/29/2010 10:35:57 AM10/29/2010 10:35:57 AM
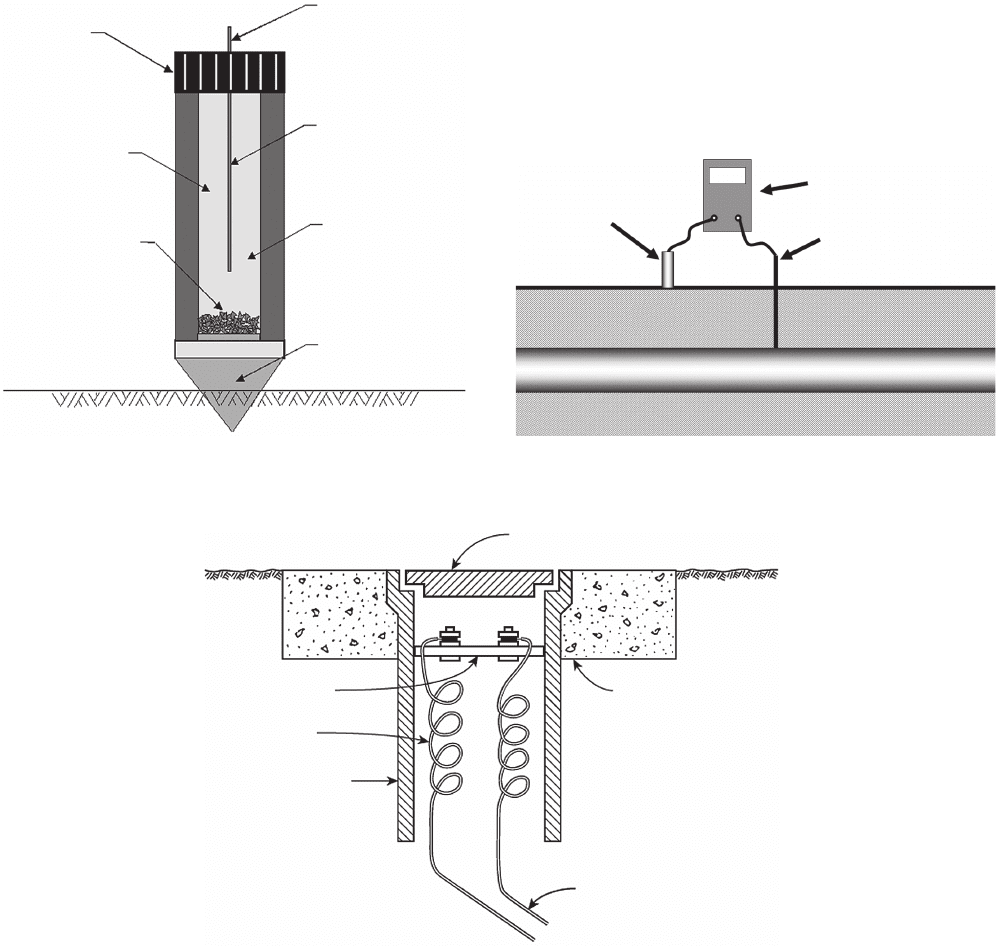
CORROSION CONTROL 171
Figure 6.76 Saturated copper - copper sulfate electrode.
Removable
Cap
Transparent
Window
Undissolved
Crystals
Copper Rod
Connection
to Test Lead
Saturated
Copper
Sulfate
Solution
Porous
Ceramic Plug
Figure 6.77 Measurement of pipe - to - soil potential.
Reference
Electrode
High-Impedance
Voltmeter
Connection to
Structure
Soil
0.907
+–
Pipeline
Figure 6.78 Typical at - grade test station.
114
Removable cover
Terminal
block
Lead
block
Housing
To pipeline
Street paving
or 18″ × 18″ × 6″
concrete block
in the hopes that one or the other electrode will still be
in calibration.
In order to measure the potential of a structure, it
must be connected through a high - impedance voltmeter
to a reference cell in direct electrical contact with the
same electrolyte. This is shown in Figure 6.77 .
At one time, it was common for pipeline surveyors
to make electrical connections with the buried pipeline
by driving a pointed rod into the soil over the pipeline.
This caused unnecessary coating damage. It is now more
common to locate test points along the right of way.
These test points are electrical connections to the pipe-
line and allow the surveyor to make electrical connec-
tions to the pipeline without damaging the coating.
A secondary advantage of using test points is that they
are permanent locations and insure that connections
on subsequent surveys will be made at the same loca-
tion. A typical fl ush - mounted test point is shown in
Figure 6.78 .
Test stations of this type are available from most
cathodic protection equipment suppliers. The at - grade
design has the advantage of being less likely to suffer
vandalism or other third - party damage. It is, however,
hard to fi nd and is subject to being covered over by soil
erosion. Above - ground designs are also available. The
minimum spacing for test stations is at the midpoint
between anode locations, the most likely location for
c06.indd 171c06.indd 171 10/29/2010 10:35:57 AM10/29/2010 10:35:57 AM
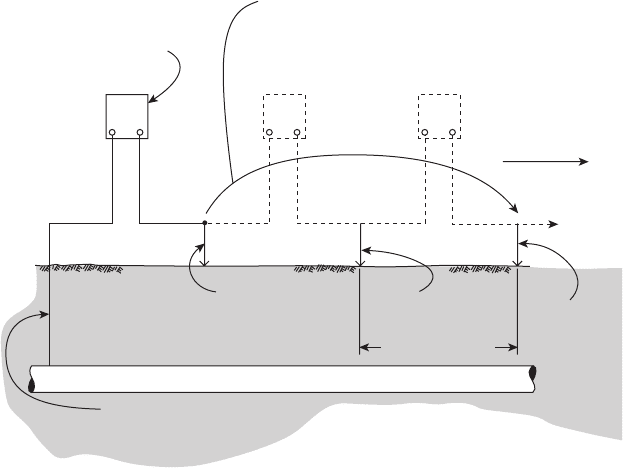
172 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
the pipeline potential to be unprotected. These test sta-
tions are often required by regulatory agencies, and
their locations are recorded on maps of the cathodic
protection system. It is possible to instrument these test
points and relay the readings to remote locations.
Test points on cross - country transmission pipeline
are typically located at intervals up to several kilome-
ters (miles) and at cased road crossings, wherever they
cross another utility, and at buried insulated joints.
Pipelines run for long distances, and the most common
surveys are over - the - line close - interval potential surveys
(CIPS) where the surveyors follow the right of way and
make measurements at predetermined intervals. This is
shown in Figure 6.79 . The intervals between readings
can vary but are typically in the hundreds of meters
(yards) for many cross - country pipelines. These surveys
supplement the information obtained from readings at
the test points, which usually are spaced much farther
apart.
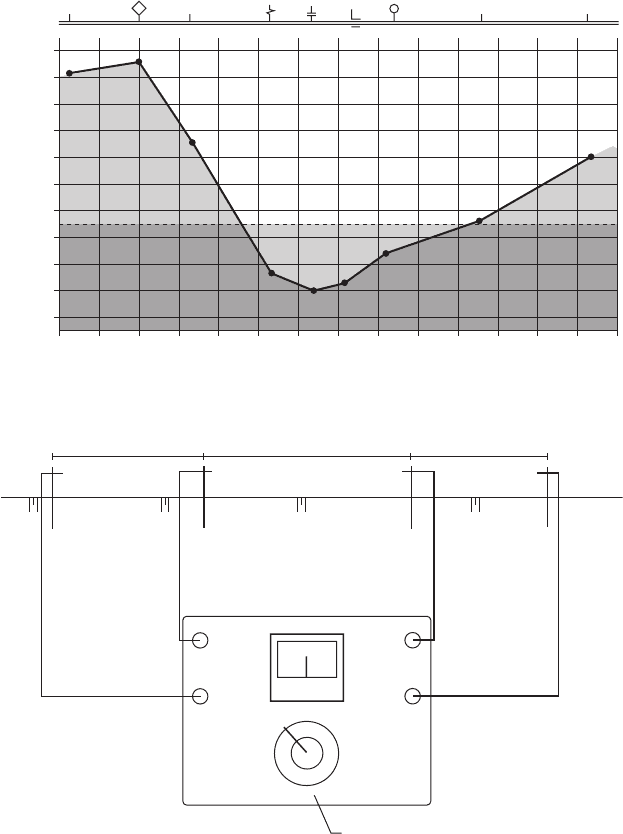
A typical pipe - to - soil potential profi le is shown in
Figure 6.80 . Virtually all cathodic potentials are nega-
tive, and it is common to plot the larger negative volt-
ages higher on the vertical axis. As long as the potential
is more negative than − 850 mV CSE, most authorities
will consider the pipeline to be protected. The problem
areas identifi ed by this survey are shown near the center
of Figure 6.79 where the negative voltages are less than
− 850 mV. The problem could be caused by a discon-
nected magnesium anode or by other factors. Once the
unsatisfactory potential survey results are available, it is
usually necessary to inspect the pipeline in these loca-
tions in greater detail to determine the source of the
problem.
Resistivity Surveys Soil resistivity is commonly mea-
sured when planning ICCP ground - bed locations or
when determining the corrosivity of pipeline rights of
way. The most accurate method is the in situ Wenner
four - pin method, which has been the industry standard
for over 50 years. The Wenner method has the advan-
tage of measuring the average soil resistivity at a depth
determined by the pin spacings. Thus, it can measure,
without disturbing the soil, the resistivity near the
surface, frequently high due to drying between rainfalls,
and at the depth of the proposed structure.
The setup for this measurement is shown in Figure
6.81 . Four electrical contact pins are placed in the soil
surface. An AC electrical current is applied between
the outer pins to produce current fl ow through the soil.
The voltage measured between the inner pins is used
to calculate resistivity. The four pins are arranged in a
straight line, and the distance between pins is adjusted
to refl ect the depth of the soil of interest. Once
the measurements have been made, it is easy to
calculate the average resistivity of the soil at the depth
equal to the pin spacing. Adjusting the pin spacing
allows determination of changes in resistivity with
depth.
113,115,116
Figure 6.79 Over - the - line potential survey setup.
118
Electrodes are
placed on surface
directly above pipe
High resistance
voltmeter
First reading
Second reading
Survey
direction
Rear electrode “leap-frogged”
to forward position for each
subsequent reading
Subsequent
voltmeter
locations
V
V
V
+– +– +–
Pipeline
Etc.
Test point wire or other
metallic contact with
pipeline
CuSO
4
electrode “A”
position 1
CuSO
4
electrode “B”
position 2
CuSO
4
electrode “A”
position 3
Known spacing
c06.indd 172c06.indd 172 10/29/2010 10:35:57 AM10/29/2010 10:35:57 AM
CORROSION CONTROL 173
The soil resistivity is calculated from the following
formula:
118,122
ρπ= 2AR
(Eq. 6.9)
where:
ρ = soil resistivity (ohm - centimeters)
A = distance between pins
R = resistance measured with the ohmmeter
Most commercial instruments for measuring resistivity
automatically calculate the resistivity based on pin
spacing and measured resistance.
Changes in resistivity are often indications of changes
in moisture levels. Low - lying riparian areas, often with
more vegetation, are typical examples of where the
resistivity would be lower and expected corrosion rates
would increase. Some locations have widely varying soil
resistivities depending on the time of the year.
It is sometimes desirable to measure the maximum
conductivity (reciprocal of resistivity) by placing the soil
from the appropriate depth into a soil box with four - pin
connections. The four - pin method is then used to deter-
mine the conductivity of the wetted soil. This method
cannot determine the effects of soil compaction on con-
ductivity and is not as reliable as the in situ four - pin
measurements described above.
Figure 6.80 Protective potential profi le indicating a lack of protection near the center
of the plot.
116
9
–1.5
–1.4
–1.3
–1.2
–1.1
–1.0
–0.9
–0.8
–0.7
–0.6
–0.5
10
PIPE-TO-SOIL POTENTIAL-V (CSE)
11
0.1
Ω
CD-TP 4
CD-TP 5
CD-TP6-F
CD-TP7-I
CD-TP 8
CD-TP 9
CD-TP 10
CD-MG 3
CD-R 2
12 13 14 15 16 17 18 19 20 21 22 23
Figure 6.81 Wenner four - pin soil resistivity measurement.
113
C1
PIN
C1
PIN
P1
PIN
P2
PIN
C2
aaa
P1
C2
Soil Resistance Meter
P2
c06.indd 173c06.indd 173 10/29/2010 10:35:57 AM10/29/2010 10:35:57 AM

174 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
TABLE 6.27 Recommended Minimum Cathodic
Protection Design Current Densities for Different Soils
117
Soil resistivity ohm.m
Design Current Density
mA/m
2
(bare steel)
< 10
20
10 – 100 10
100 – 1000 5
> 1000
1
Note : A 2.5% increase for each degree C rise above 30 ° C.
TABLE 6.28 Recommended Minimum Cathodic
Protection Design Current Densities for Different Soils
117
Coating Design Current Density mA/m
2
None 20
Tape wrap 1.25
Coal tar epoxy 0.75
FBE 0.1
Polyethylene 0.1
Single - probe conductivity measurement is also pos-
sible. This is less work but is less accurate; only the
conductivity at the depth of the local probe is deter-
mined at the precise location where the probe is placed.
Cathodic Protection Design Procedures
Most of the following discussion will emphasize design
of cathodic protection systems for pipelines. The
Handbook of Cathodic Protection contains complete
chapters on cathodic protection of ships (applicable to
designs for spar platforms and fl oating production,
storage, and offl oading [FPSO] structures or vessels ),
marine structures, well casings, water tanks and boilers,
and process equipment.
117
The fi rst step in any cathodic protection design is to
determine the total electrical current demand. For exist-
ing structures, this can be done by measuring the current
necessary to produce the desired potential shift. This is
done by connecting the structure to a temporary DC
power source and varying the current until the neces-
sary polarization, determined by either the E log i
method or by simple measurement of the potential at
remote locations from the temporary anodes. The choice
of method depends on whether or not the remote loca-
tion is accessible for potential measurement. In either
case, it is necessary to wait until changes in the applied
current have produced steady - state potentials before
increasing the current to the next level. This can take
minutes to hours depending on the size of the structure
involved.
New pipeline cathodic protection design is often
based on the current expected to be necessary after 20
years of service. The coating degradation is not expected
to worsen after that period of time. Designs provide
more current than is necessary during the early life of
the system and are intended to last indefi nitely. For this
reason, it is common to overdesign the system, because
it is easier, and is supposedly less expensive, to install a
somewhat larger than necessary system at the beginning
than it is to retrofi t at some later date. Most authorities
recommend current densities based on expected bare
metal exposed area. Sometimes the assumption is that
1% of the possible surface area will be exposed, but this
is seen as very conservative for some of the newer pipe-
line coating systems. Tables 6.27 and 6.28 show guide-
lines from several different oil companies ’ published
recommendations for the minimum current densities
necessary for buried pipeline cathodic protection.
117
The
effectiveness of high - quality coatings is obvious from
the reduced current demands shown for FBE and
polyethylene.
Once the current requirements have been identifi ed,
the design procedure then must consider a number of
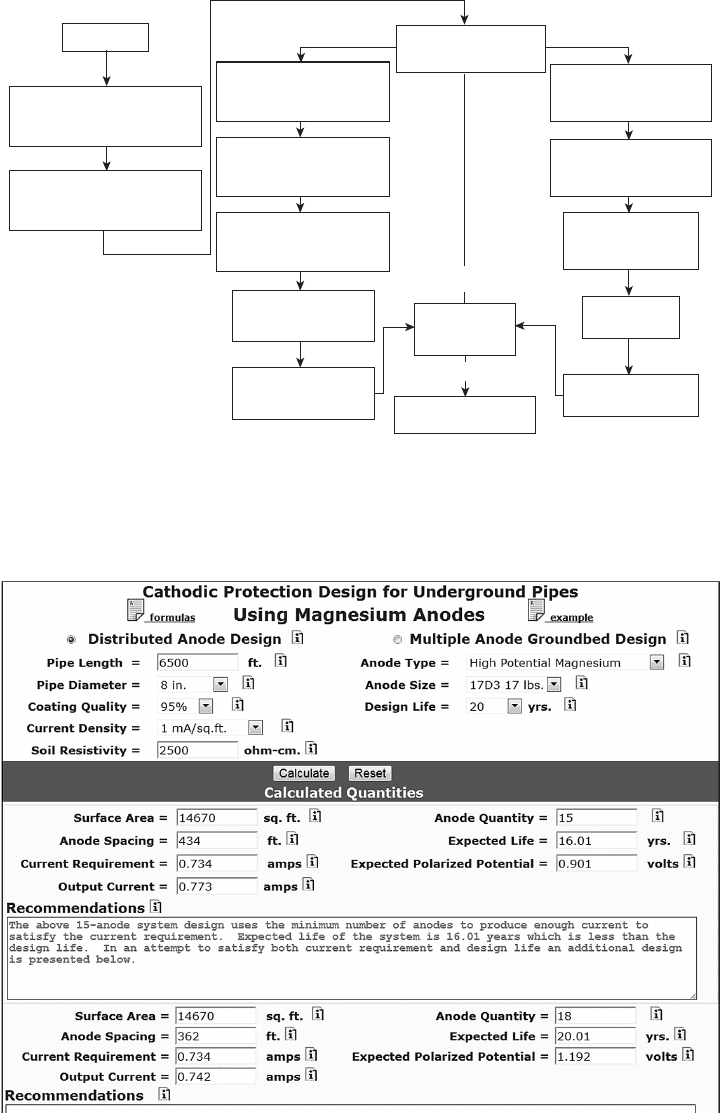
alternatives based on the choice of anode type. Figure
6.82 is one of a number of recommended design proce-
dures that are available.
The procedures for calculating all of the above design
steps have been standardized for many years. Many of
the necessary formulas, for example, for calculating
ground - bed resistance, are based on work done in the
1930s. While the calculations can be done by hand (as
they were originally), it is more common to do them
using computer software. Most of this software is based
on spreadsheets, and many cathodic protection contrac-
tors have developed their own in - house software for this
purpose. In recent years, several websites have become
available that do these calculations. Figures 6.83 and
6.84 are examples of what is available. As the use of
computers increases, this kind of user - friendly software
will become more common.
For many years, the 1960s ’ book, Control of Pipeline
Corrosion by A. W. Peabody, was considered to be one
of the premier reference materials on cathodic protec-
tion.
103
The book was updated in 2001, and the compact
disc that accompanies the updated book has spreadsheet -
based software included. Figure 6.85 is one example of
the screens used by the compact disc that accompanies
this widely used handbook.
The compact disc also contains sample problems
showing the following calculations:
•
Determining protective current requirements
•
Anode resistance to earth
•
Conventional ground - bed design
c06.indd 174c06.indd 174 10/29/2010 10:35:58 AM10/29/2010 10:35:58 AM
CORROSION CONTROL 175
Figure 6.82 Cathodic Protection Design Procedure. Adapted from NACE International CP
Technology Course Slide5/140.
Sample Cathodic Protection Design Procedure
Start
Evaluate Pipeline
and
Environmental Factors
Determine Current
Requirements to Achieve
Desired Criterion
Calculate Resistance
of Anode or
Groundbed
Calculate Number
and Spacing of
Anodes or Groundbeds
Calculate
System Life
Choose Anode Type,
Size, Weight, and
Arrangement
Galvanic
Galvanic or
Impressed Current?
Is Design
Acceptable?
No
Yes
Design Complete
Impressed
Current
Calculate Resistance
of Anode or
Groundbed
Choose
Power Supply,
Type, and Rating
Calculate
System Life
Estimate Installed
Cost of System
Choose Anode
Size, Weight, and
Arrangement
Estimate Installed
Cost of System
Figure 6.83 Online screen for calculating galvanic anode life. Image courtesy of Mesa
Products, Inc., http://www.mesaproducts.com and http://www.cpdesigncenter.com/public/cp_
calculators/currentrequirement.htm , May 2, 2010.
c06.indd 175c06.indd 175 10/29/2010 10:35:58 AM10/29/2010 10:35:58 AM

176 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
Figure 6.84 Online screen for calculating total current requirement for cathodic protection
of pipelines or storage tanks. Image courtesy of Mesa Products, Inc., http://www.mesaproducts.
com and http://www.cpdesigncenter.com/public/cp_calculators/currentrequirement.htm , May
2, 2010.
Figure 6.85 Screen from CD accompanying Peabody ’ s Control of Pipeline Corrosion,
2nd edition.
118
NACE
Companion to the Peabody Book
October 26, 2000
Revision 1.1M
Dwight’s Equation for Single Vertical Anode Resistance to Earth - millimeters
Dwight’s Equation for Single Vertical Anode Resistance to Earth—meters
25.6 ohms
25.6 ohms
10,000 ohm-cm
10,000 ohm-cm
2.13 m
0.203 m
203 mm
2134 mm
r
r =
= Soil resistivity in ohm-cm
L
= Rod length in mm
d
L =
d =
= Rod diameter in mm
R
V
R
V
=
L
1.59 r
⎧
⎩
⎧
⎩
ln
d
8L
–1
= Resistance of vertical rod in ohms
r = Resistivity of backfill material (or earth) in ohm-cm
L
= Length of anode in meters
d
= Diameter of anode in meters
R
V
= Resistance of one vertical anode to earth in ohms
R
V
R
V
=
r =
L =
d =
R
V
=
=
L
0.00159 r
⎧
⎩
⎧
⎩
ln
d
8L
–1
•
Deep anode bed design
•
Cathode resistance to earth
•
Total DC circuit resistance
•
Current attenuation
•
System life of galvanic anode systems
They allow the user to input the data in metric or U.S.
conventional units.
Temperature Effects on Cathodic Protection Chemical
reactions associated with corrosion are highly tempera-
ture dependent. Many design guidelines contain advice
c06.indd 176c06.indd 176 10/29/2010 10:35:58 AM10/29/2010 10:35:58 AM
CORROSION CONTROL 177
on increasing current density for above - ambient tem-
peratures. The consumption rate of anodes depends on
temperature, and this must be considered in cathodic
protection design and replacement scheduling. The
increased consumption rates of anodes can be mini-
mized by using remote anode locations in cooler envi-
ronments, but this leaves some designs more prone to
mechanical damage due to soil movement and other
causes.
Computer - Aided Cathodic Protection Design
Computer - aided cathodic protection designs for off-
shore structures have been tried by several organiza-
tions in recent years. These computer - aided designs are
of two types:
82,119,120
•
Personal computers used to make the types of cal-
culations (such as the wetted surface area calcula-
tions discussed above) that have commonly been
used for cathodic protection design. The computer
is a time - saver in these calculations and allows a
greater number of alternatives to be considered,
but the actual design methodology is not changed.
•
The use of numerical techniques, such as fi nite
element, fi nite difference, or boundary integral, to
model the potential current distribution around a
structure. Initial efforts to use these techniques
found limited acceptance because of the time delay
caused by communications diffi culties between the
operator, the cathodic protection designer, and the
computer expert.
The increased memory capabilities of personal comput-
ers now allow design engineers to make calculations
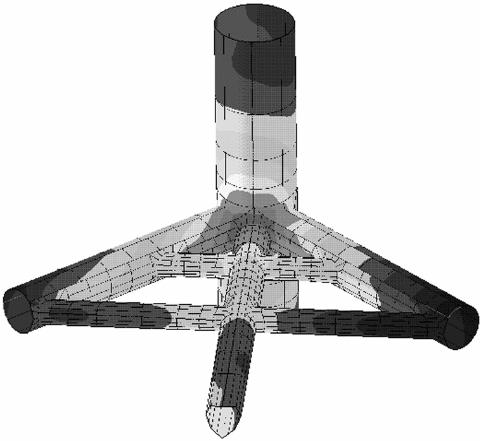
once requiring mainframe computers. Figure 6.86 shows
a sample plot of the cathodic protection on an offshore
platform node. The various color arrangements or shad-
ings allow quick assessment of areas that might be inad-
equately protected.
82,119,120
Comparisons of plots for
different anode arrangements allow the designer to
quickly determine which anode locations are the most
effective and where inspection points should be located
to determine if adequate cathodic protection is being
achieved at high - stressed node welds and other critical
locations.
Additional Topics Related to Cathodic Protection
Some of the problems associated with cathodic protec-
tion include stray current corrosion, hydrogen embrit-
tlement and stress corrosion cracking of high - strength
(or high - hardness) steel, and cathodic disbondment of
coatings. In addition to these well - documented prob-
lems, many authorities have questioned the standards
used over recent decades for determining if a structure
is cathodically protected.
“ Instant - Off ” Potentials In recent years, the biggest
controversy in cathodic protection has been over the
idea of measuring “ instant - off ” potentials to determine
if a structure is protected from corrosion. There are
many publications pro and con on this subject, and the
ideas behind “ instant - off ” potentials are the subject of
continuing debates.
The fi rst advocates of “ instant - off ” potentials cited
the need for accounting for IR drops between the struc-
ture and the electrolyte. This was based on the mistaken
assumption that the − 850 mV CSE potential was the
“ equilibrium potential ” for carbon steel in soil. This is
not the case because:
•
T h e − 850 mV criterion came from leak records and
measurements of “ current - on potentials ” on cath-
odically protected pipelines, primarily on the Gulf
Coast of the United States. R. Kuhn from Louisiana
was the most prominent early advocate of this idea,
and he based his reasoning on leak records that
showed that structures held at potentials at least as
negative as − 850 mV CSE had much lower leak
records than unprotected steel. This standard was
considered acceptable for most situations, although
a − 950 mV criterion was generally recommended
when microbial activity was likely.
76,77,79,85
Figure 6.86 Computerized model of the cathodically pro-
tected region around a node on an offshore platform.
126,127
Reprinted with permission of ASM International. All rights
reserved, http://www.asminternational.org .
c06.indd 177c06.indd 177 10/29/2010 10:35:58 AM10/29/2010 10:35:58 AM
178 METALLURGY AND CORROSION CONTROL IN OIL AND GAS PRODUCTION
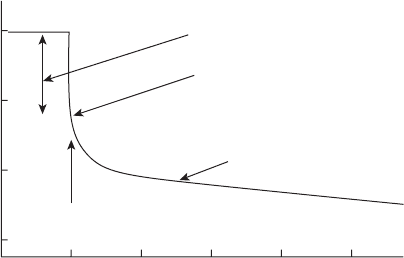
Figure 6.87 Idealized instant - off potential plot.
IR Error
–1200
–1000
–800
CP On
CP Off
Switch Off
Time (milliseconds)
0 100 500 1000 1500 2000
–600
“Instant Off” Potential
Depolarization
Pipe / Soil Potential (V SCE)
•
T h e − 850 mV criterion is at a lower (smaller) nega-
tive number than the equilibrium potential. This
was shown in Figure 6.64 where the equilibrium
potential is shown at − 950 mV in an acid environ-
ment. The equilibrium potential will vary with pH
in accordance with the Nernst equation discussed
in earlier chapters, but the equilibrium potential
will always be at a greater negative potential than
the − 850 CSE protection potential used for cathodic
protection. As stated previously, cathodic protec-
tion does not eliminate oxidation or corrosion on
a protected structure, but it has been shown to sig-
nifi cantly reduce corrosion.
In the decades after the NACE RP 0169 standard was
adopted, many authors discussed errors in potential
measurement, but these errors were considered to be
insignifi cant in most cases.
79
The IR drops that were originally considered to be
insignifi cant for bitumastically coated pipelines pro-
tected with galvanic anodes were questioned for
impressed current systems and for measurements
directly over galvanic anodes.
108
This led to the develop-
ment of NACE RP 0169 (92) which mandated that IR
drops must be compensated for using an “ instant - off ”
criterion. Diffi culties in defi ning how this “ instant off ”
should be done, and questions on whether or not this
“ instant - off ” potential is necessary, continue as of this
writing. Since the requirement to determine an “ instant -
off ” negative potential means that the negative “ current -
on ” potential will be greater than the negative
“ instant - off ” potential, the requirement is conservative.
It is, nonetheless, questioned by many and is unpopular
with fi eld personnel.
Figure 6.87 illustrates the concept of the “ instant - off ”
method of determining IR errors in cathodic protection.
When the cathodic protection current is applied, the
structure assumes a potential which is intended to
reduce corrosion. In order to measure the potential, a
voltmeter is connected to the system and the current - on
potential is measured. The measured potential includes
the potential of the structure and any current - resistance
(IR) drops in the circuit, for example, the reference
electrode - electrolyte IR drop and the structure -
electrolyte IR drop . By turning off the current, the
voltage supposedly “ instantaneously ” drops to the
potential of the structure in the electrolyte. The poten-
tial then decays to the unprotected potential, a process
that can take anywhere from minutes to days. Proponents
of the “ instant - off ” method suggest that the “ instant -
off ” potential must satisfy whatever criterion, either
− 850 mV CSE or − 100 mV potential shift, is used for the
system. The IR drop can be as large as 1 V in some
instances.
Proponents cite electrochemistry textbooks which
identify several IR drops in an electrochemical circuit
and the supposed failures of the “ current - on ” criterion
that had been in use for several decades. Opponents of
this concept argue that measuring the potential with the
current applied has worked for decades. The original
“ current - on ” criterion was developed for onshore pipe-
lines along the Gulf Coast of the United States, a region
where soils generally have low resistance. The IR drop
of concern is more likely to be of concern with high -
resistivity environments. The length of time (from
microseconds to several seconds) of the “ instant decay ”
depends, at least in part, on the environment and must
be empirically determined.
108,121,122
Stray Current Corrosion Caused by Cathodic Protection
Systems The potential fi eld around a cathodically pro-
tected structure can cause stray current corrosion on
nearby structures. This is a signifi cant problem with
ICCP systems that share rights of way with nearby
structures/utility systems. Galvanic anodes, with their
much lower driving potentials, are unlikely to cause this
problem.
Figure 6.88 shows two pipelines crossing each other.
The cathodic protection system is causing corrosion
where current from the cathodic protection system
leaves the unprotected line.
Stray or interference current is detected by turning
the cathodic protection system on and off and monitor-
ing the potential of the unprotected line. If the potential
of the unprotected line varies with the cycle of turning
the cathodic protection system on and off, then stray
current corrosion will occur.
Stray current corrosion can usually be handled by
bonding a short section of the unprotected line to the
protected line and using current from the cathodic pro-
tection system to protect both structures. “ Hot spot ”
c06.indd 178c06.indd 178 10/29/2010 10:35:59 AM10/29/2010 10:35:59 AM
