Klipp E., Herwig R., Kowald A., Wierling C., Lehrach H. Systems Biology in Practice: Concepts, Implementation and Application
Подождите немного. Документ загружается.


2.1
The Origin of Life
The earliest development on earth began 4
1
/
2
billion years ago. Massive volcanism re-
leased water (H
2
O), methane (CH
4
), ammonia (NH
3
), sulfur hydrogen (H
2
S), and
molecular hydrogen (H
2
), which formed a reducing atmosphere and the early ocean.
By loss of hydrogen into space and gas reactions, an atmosphere consisting of nitro-
gen (N
2
), carbon monoxide (CO), carbon dioxide (CO
2
), and water (H
2
O) was
formed. The impact of huge amounts of energy (e. g., sunlight with a high portion of
ultraviolet radiation and electric discharges) onto the reducing atmosphere along
with the catalytic effect of solid-state surfaces resulted in an enrichment of simple or-
ganic molecules such as amino acids, purines, pyrimidines, and monosaccharides in
the early ocean. This is called the prebiotic broth hypothesis and is based on the ex-
periments of Miller and Urey (1959). Another possibility is that the first forms of life
formed in the deep sea and utilized the energy of hydrothermal vents, well protected
from damaging UV radiation and the unstable environment of the surface (Wächters-
häuser 1988). Once simple organic molecules were formed in significant amounts,
they presumably assembled spontaneously into macromolecules such as proteins
and nucleic acids. Through the formation of molecular aggregates from these col-
loidally dissolved macromolecules, the development of simple compartmented reac-
tion pathways for the utilization of energy sources was possible. Besides this, en-
zymes appeared that permitted specific reactions to take place in ordered sequences
at moderate temperatures, and informational systems necessary for directed synth-
esis and reproduction were developed. The appearance of the first primitive cells –
the last common ancestors of all past and recent organisms – was the end of abiotic
(chemical) evolution and the beginning of biotic (biological) evolution. Later, these
first primitive cells evolved into the first prokaryotic cells (prokaryotes). About 3.5 bil-
lion years ago, the reducing atmosphere was very slowly enriched by oxygen (O
2
)
due to the rise of photosynthesis that resulted in an oxidative atmosphere (1.4 billion
years ago: 0.2% O
2
; 0.4 billion years ago: 2% O
2
; today: about 21% O
2
).
Prokaryotes (eubacteria and archaebacteria) are mostly characterized by their size
and simplistic structure compared to the more evolved eukaryotes. Table 2.1 sum-
marizes several differences between these groups. The evolutionary origin of the eu-
karyotic cells is explained by the formation of a nucleus and several compartments,
and by the inclusion of prokaryotic cells which is described by the endosymbiont hy-
pothesis. This hypothesis states that cellular organelles, such as mitochondria and
chloroplasts, are descendants of specialized cells (e.g., specialized for energy utiliza-
tion) that have been engulfed by the early eukaryotes.
Prokaryotes and these early eukaryotes are single-celled organisms. Later during
evolution, single-celled eukaryotes evolved further into multicellular organisms.
Their cells are mostly genetically identical but differentiate into several specialized
cell types during development. (Genetically identical cells are usually called cell
clones. The term clone is used in molecular biology mostly to denote colonies of
identical cells in culture, such as bacterial or yeast cells on a petri dish.) Most of
these organisms reproduce sexually.
20
2 Biology in a Nutshell

The developmental process that takes place by sexual reproduction starts with a
fertilized egg (zygote) that divides several times (the cell division underlying this pro-
cess is discussed in more detail in Section 2.5 and in Section 7.2). For instance, in
the frog Xenopus laevis – which is a vertebrate and belongs to the amphibians – the
development starts with the zygote and passes through several developmental
phases, i. e., morula (64 cells), blastula (10,000 cells), gastrula (30,000 cells), and
neurula (80,000 cells), before forming the tadpole (with a million cells 110 hours
after fertilization) that develops into the adult frog later on. This process is geneti-
cally determined and several phases are similar among species that are closely re-
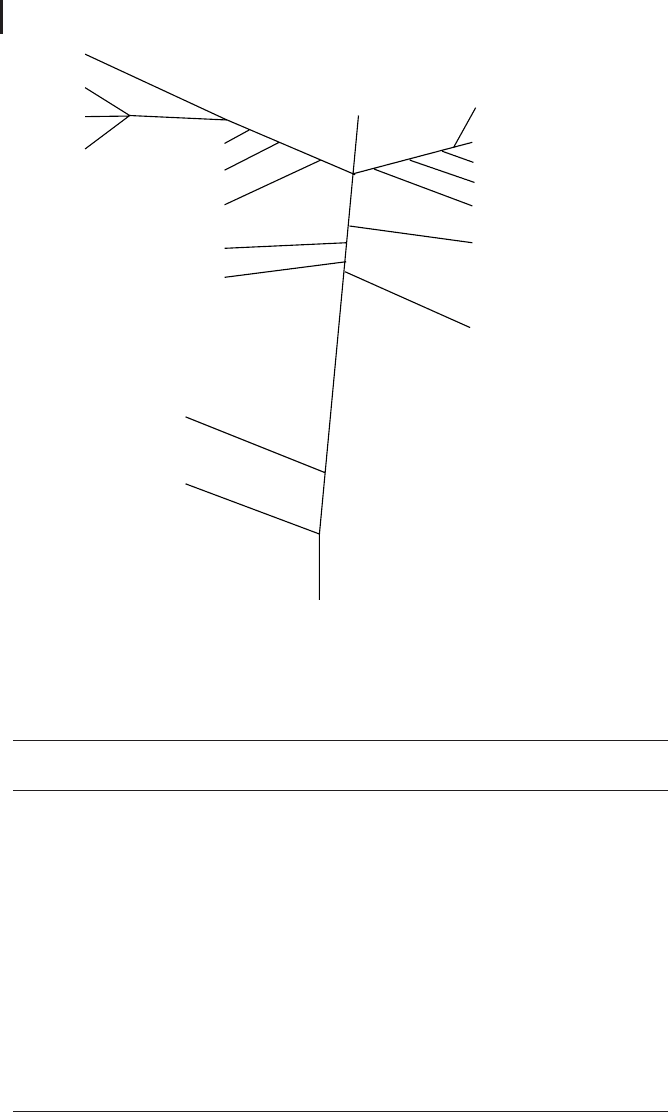
lated to each other due to their identical evolutionary origin. Figure 2.1 shows a sim-
plified tree of life that illustrates major evolutionary relations.
While most places with moderate aerobic conditions were populated by eukar-
yotes, the prokaryotic archaebacteria in particular have specialized to survive under
extreme conditions (e.g., the thermophile bacteria, which propagate at temperatures
of 85–105 8C in the black smokers of the deep sea, or the halobacteria, which live un-
der high salt concentrations).
Along with organisms that have their own metabolism, parasitic viruses and vir-
oids that utilize cells for reproduction have developed. Viruses consist of a very small
genome surrounded by a protein envelope (capsid); viroids are single-stranded circu-
lar RNAs. Due to the absence of a metabolism and a cellular structure, these para-
sites are usually not regarded as living organisms.
The phenotypical diversity of organisms observed is also displayed in the structure
of their hereditary information: the size of this genomic information can vary, as can
its organization into different elements, i.e., plasmids and chromosomes. Table 2.2
summarizes some data acquired from commonly investigated organisms.
21
2.1 The Origin of Life
Tab. 2.1 Some important differences between prokaryotic and eukaryotic cells.
Prokaryotes Eukaryotes
Size Mostly about 1–10 mm in length. Mostly about 10–100 mm in length.
Nucleus Nucleus is missing. The chromosomal
region is called nucleolus.
Nucleus is separated from the
cytoplasm by the nuclear envelope.
Intracellular
organization
Normally, no membrane-separated
compartments and no supportive intra-
cellular skeletal framework are present
in the cells’ interior.
Distinct compartments are present,
e.g., nucleus, cytosol with a cyto-
skeleton, mitochondria, ER, Golgi
complex, lysosomes, plastids
(chloroplasts, leucoplasts).
Gene structure No introns; some polycistronic genes. Introns and exons.
Cell division Simple cell division. Mitosis or meiosis.
Ribosome Consists of a large 50S subunit and
a small 30S subunit.
Consists of a large 60S subunit and
a small 40S subunit.
Reproduction Parasexual recombination. Sexual recombination.
Organization Mostly single cellular. Mostly multicellular, and with cell
differentiation.
22
2 Biology in a Nutshell
Eubacteria
Archaebacteria
Fungi
Flagellates
Ciliates
Animals
Plants
Algae
Mosses
Ferns
Conifers
Flowering plants
Slime molds
Cnidarians
Flatworms
Roundworms
Mollusks
Annelids
Arthropods
Chordates
Amoebae
Fig. 2.1 The tree of life shows phylogenetic relations between
some major groups of organism.
Tab. 2.2 Genome sizes of different organisms from the prokaryotic and eukaryotic kingdom. Infor-
mation about further organisms can be found, e. g., at http://www.cbs.dtu.dk/services/GenomeAtlas.
Organism Number of chromosomes
(haploid genome)
Genome size (base pairs; genes)
Mycoplasma genitalium
(prokaryote)
1 circular chromosome 580710
3
bp; 480 genes
Escherichia coli (prokaryote) 1 circular chromosome 4.6710
6
bp; 4,290 genes
Saccharomyces cerevisiae
(budding yeast; eukaryote)
16 chromosomes 12.5710
6
bp; 6,186 genes
Arabidopsis thaliana
(flowering plant; eukaryote)
5 chromosomes 100710
6
bp; ~25,000 genes
Drosophila melanogaster
(fruit fly, eukaryote)
4 chromosomes 180710
6
bp; ~14,000 genes
Mus musculus
(mouse, eukaryote)
20 chromosomes 2.5710
9
bp; ~30,000 genes
Homo sapiens
(human, eukaryote)
23 chromosomes 2.9710
9
; ~30,000 genes

2.2
Molecular Biology of the Cell
Cellular structures and processes result from a complex interaction network of biolo-
gical molecules. The properties of these molecules determine possible interactions.
Although many of these molecules are highly complex, most fall into one of the fol-
lowing four classes or contain substructures that belong to one of these classes: car-
bohydrates, lipids, proteins, and nucleic acids. Along with these four classes, water is
highly important for all living systems. Molecules are held together by and interact
through chemical bonds and forces of different types: ionic, covalent and hydrogen
bonds, nonpolar associations, and van der Waals forces. The following sections will
provide a foundation for the understanding of molecular structures, functions, and
interactions by giving a brief introduction to chemical bonds and forces, to the most
important classes of biological molecules, and to complex macromolecular struc-
tures formed by these molecules.
2.2.1
Chemical Bonds and Forces Important in Biological Molecules
The atom model introduced by Rutherford and significantly extended by Bohr de-
scribes the atom as a positively charged nucleus surrounded by one or more shells (or,
more exactly, energy levels) that are filled with electrons. Most significant for the che-
mical properties of an atom is the number of electrons in its outermost shell. Atoms
tend to fill up their outermost shell to obtain a stable state. The innermost or first shell
is filled up by two electrons. The second and further shells are filled up by 2n
2
elec-
trons, where n depicts the number of the shell. However, due to reasons of energetic
stability, the outermost shell will not contain more than eight electrons. For example,
helium, with two electrons in its single shell, or atoms such as neon or argon, with
eight electrons in their outermost shells, are essentially chemically inert. Atoms with
a number of electrons near to these numbers tend to lose or gain electrons to attain
these stable states. For example, sodium (one electron in its outer shell) and chlorine
(seven electrons in its outer shell) can both achieve such a stable state by transferring
one electron from sodium to chlorine, thus forming the ions Na
+
and Cl
–
. The force
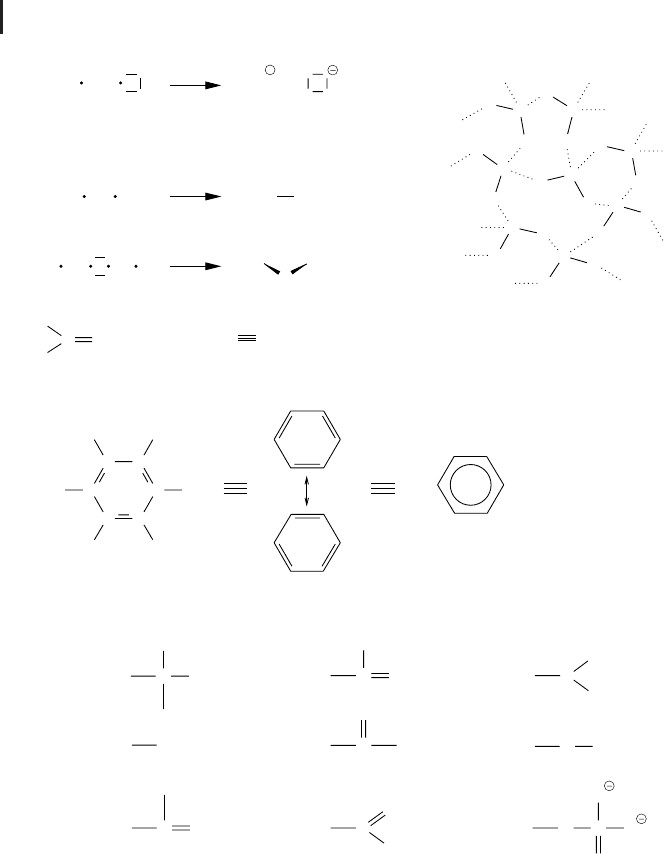
holding together the oppositely charged ions in solid state is called the ionic or electro-
static bond (Fig. 2.2a). If the number of electrons in the outer shell differs by more
than one, atoms tend to share electrons by forming a so-called covalent bond (Fig.
2.2b). Atoms held together by covalent bonds are called molecules. If the shared elec-
tron pair is equally distributed between the two atoms, this bond is called nonpolar
(e.g., the hydrogen molecule). If one atom has a higher attraction to the shared elec-
tron pair, it becomes partially negatively charged. Then the other atom in this polar as-
sociation becomes partially positively charged, as is the case with the water molecule
(H
2
O), where the oxygen attracts the shared electron pairs stronger than the hydrogen
atoms do. Thus –OH and –NH groups usually form polar regions in which the hydro-
gen is partially positively charged. A measurement for the affinity of an atom to attract
electrons in a covalent bond is given by its electronegativity, which was introduced by
23
2.2 Molecular Biology of the Cell
24
2 Biology in a Nutshell
NN
HH HH
HOH
O
H
H
CO
CC
C
CC
CH
HH
H
HH
Na Cl
SH
N
H
H
O
O
P
O
O
O
H
H
O
H
H
O
H
H
O
H
H
O
H
H
O
H
H
O
H
H
O
H
H
O
C
C
H
O
C
O
OH
OHC
OC
OH
Na Cl
+
++
A Ionic bond
D Functional groups
B Covalent bonds
+
+
C
Nonpolar electron sharing
Polar electron sharing
Delocalized electrons
+
Amino
Phosphate
Alcohol
Carbonyl
Aldehyde
Carboxyl
SulfhydrylHydroxyl
Ketone
Hydrogen bonds
Double bond Triple bond
Fig. 2.2 Chemical bonds and functional organic
groups. Single electrons in the outer shell are vi-
sualized by a dot; electron pairs are replaced by a
dash. Shared electron pairs are represented by a
dash between two atoms. (a) Single charged Na
+
and Cl
–
ions are formed by the transition of the
single outermost electron of sodium to chlorine.
(b) In a covalent bond, electrons are shared be-
tween two atoms. If the shared electron pair is at-
tracted more strongly by one of the participating
atoms than by the other, this bond is called a po-
lar bond. Depending on the molecule structure,
double and triple bonds can occur as well. Some-
times, binding electron pairs might also be de-lo-
calized among several atoms, as is the case in
benzol. (c) Unequal electron sharing causes the
formation of hydrogen bonds (shown by dotted
lines) as found in water. (d) The skeleton of or-
ganic molecules essentially consists of carbon
atoms bound to each other or to hydrogen. Some
of these carbons are bound to or are part of func-
tional groups with special chemical characteris-
tics. Hence, these influence the reactivities and
physicochemical properties of the molecule.

Linus Pauling. In addition to single covalent bonds, double and triple bonds also exist.
These kinds of bonds are more exactly described by the quantum-mechanical atom
model, in which the electron shells of an atom can be described by one of several dif-
ferently shaped orbitals that represent the areas where the electrons are located with
highest probability (electron clouds). A covalent bond is then described by molecule
orbitals, which are derived from atom orbitals. Furthermore, if single and double
bonds are altered in a single molecule or a double bond is in the direct vicinity of an
atom with a free electron pair, then one electron pair of the double bond and the free
electron pair can delocalize across the participating atoms, e.g., the three electron
pairs in benzol (Fig. 2.2b) or the double bond between C and O and the free electron
pair of N in a peptide bond (Fig. 2.6 a). Such electrons are called de-localized p-elec-
trons. For a more detailed description, please consult books about general and anor-
ganic chemistry or introductory books about biochemistry.
Hydrogen atoms with a positive partial charge that are bound to oxygen or nitro-
gen (as in H
2
OorNH
3
) are able to interact with free electron pairs of atoms with a
negative partial charge. These attractions are called hydrogen bonds and are rela-
tively weak compared to solid-state ionic bonds or covalent bonds. To break a hydro-
gen bond, only about 4 kJ mol
–1
is required. Therefore, hydrogen bonds separate
readily at elevated temperatures, which is often the reason that proteins such as
enzymes lose their function during heating. Likewise, the hydrogen bonds that hold
together the double strands of nucleic acids (see Section 2.2.3) can be separated at
high temperatures. This fact is utilized for several molecular biological methods,
e.g., polymerase chain reaction (PCR) and radioactive labeling of DNA (deoxyribonu-
cleic acid) fragments. Hydrogen bonds also explain why water is liquid at room tem-
perature and boils at 100 8C. Small alcohols, such as methanol or ethanol, are fully
soluble in water due to their hydroxyl group, which interacts with the hydrogen
bonds of water, whereas larger alcohols, such as hexanol or heptanol, are weakly so-
luble or insoluble in water due to their longer unpolar carbohydrate tail. As we have
seen, polarized functional groups can interact with water, which is why they often
are called hydrophilic (or lipophobic), while nonpolar molecules or molecule parts
are called hydrophobic (or lipophilic).
Also critical to structures and interactions of biological molecules are the van der
Waals forces. The electron clouds surrounding atoms that are held together by cova-
lent bonds are responsible for these forces. Momentary inequalities in the distribu-
tion of electrons in any covalent bond, due to chance, can make one end of the cova-
lent bond more negative or positive than the other for a short moment, which results
in rapid fluctuations in the charge of the electron cloud. These fluctuations can set
up opposite fluctuations in nearby covalent bonds, thus establishing a weak attrac-
tive force. This attractive force is stronger the closer the electron clouds are, but if
the outermost electron orbitals begin to overlap, the negatively charged electrons
strongly repel each other. Thus, van der Waals forces can be either attractive or repul-
sive. Their binding affinity is, at 0.4 kJ mol
–1
in water, even lower than that of hydro-
gen bonds. The optimal distance for maximum van der Waals forces of an atom is
called its van der Waals contact radius. The van der Waals repulsions have an impor-
tant influence on the possible conformations of a molecule.
25
2.2 Molecular Biology of the Cell

2.2.2
Functional Groups in Biological Molecules
As outlined before, a major characteristic of life are the physiological processes in
which nutrients from the outside are converted by the organism to maintain a ther-
modynamically open system with features such as development or behavior. These
physiological processes are realized on the metabolic level by myriads of reactions in
which specific molecules are converted into others. These intra- or intermolecular re-
arrangements often take place at specific covalent bonds that can more readily be
disturbed than others. Such covalent bonds are often formed by certain intramolecu-
lar substructures called functional groups. Thus, functional groups often serve as re-
action centers that convert some molecules into others or link some molecular subu-
nits to form larger molecular assemblies, e. g., polypeptides or nucleic acids. The
functional groups most relevant in biological molecules are hydroxyl, carbonyl, car-
boxyl, amino, phosphate, and sulfhydryl groups (Fig. 2.2d).
Hydroxyl groups (–OH) are strongly polar and often enter into reactions that link
subunits into larger molecular assemblies in which a water molecule is released.
These reactions are called condensations. The reverse reaction, in which a water mo-
lecule enters a reaction by which a larger molecule is split into two subunits, is called
hydrolysis. The formation of a dipeptide from two amino acids is an example of a
condensation, and its reverse reaction is the hydrolysis of the dipeptide (Fig. 2.6a). If
the hydroxyl group is bound to a carbon atom, which in turn is bound to other hydro-
gen and/or carbon atoms, it is called an alcohol. Alcohols can easily be oxidized to
form aldehydes or ketones, which are characterized by their carbonyl group
(Fig. 2.2d). Aldehydes and ketones are particularly important for carbohydrates
(such as sugars) or lipids (such as fats). In aldehydes the carbonyl group occurs at
the end of a carbon chain, whereas in ketones it occurs in its interior. A carboxyl
group is strongly polar and is formed by an alcohol group and an aldehyde group.
The hydrogen of the hydroxyl part can easily dissociate as H
+
due to the influence of
the nearby carbonyl oxygen. In this way, it acts as an organic acid. The carboxyl
group (–COOH) is the characteristic group of organic acids such as fatty acids and
amino acids. Amino acids are further characterized by an amino group. Amino
groups (–NH
2
, Fig. 2.2d) have a high chemical reactivity and can act as a base in or-
ganic molecules. They are, for instance, essential for the linkage of amino acids to
form proteins and for the establishment of hydrogen bonds in DNA double strands.
Moreover, amino acids carrying NH
2
in their residual group often play a crucial role
as part of the catalytic domain of enzymes. Another group that has several important
roles is the phosphate group (Fig. 2.2d). As part of large organic molecules, this
group acts as a bridging ligand connecting two building blocks to each other, as is
the case in nucleic acids (DNA, RNA; see Section 2.2.3) or phospholipids. Further-
more, the di- and triphosphate forms in conjunction with a nucleoside serve as a
universal energy unit in cells, e.g., adenosine triphosphate (ATP, Fig. 2.7a). Phos-
phate groups are also involved in the regulation of the activity of enzymes, e.g.,
MAP kinases, which participate in signal transduction (see Section 6.3). Sulfhydryl
groups (Fig. 2.2d) are readily oxidized. If two sulfhydryl residues participate in an
26
2 Biology in a Nutshell

oxidization, a so-called disulfide bond is created (Fig. 2.6d). These linkages often oc-
cur between sulfhydryl residues of amino acids that form a protein. Thus they are re-
sponsible for the stable folding of proteins, which is required for their correct func-
tioning.
2.2.3
Major Classes of Biological Molecules
The structural and functional properties of an organism are based on a vast number
of diverse biological molecules and their interplay. The physicochemical properties
of a molecule are determined through their functional groups. In the following sec-
tions, four major classes of biological molecules that are ubiquitously present and
are responsible for fundamental structural and functional characteristics of living or-
ganisms will be introduced: carbohydrates, lipids, proteins, and nucleic acids.
2.2.3.1 Carbohydrates
Carbohydrates function as energy storage molecules and furthermore can be found
as extracellular structure mediators, e. g., in plants. The chemical formula of carbo-
hydrates is mostly C
n
(H
2
O)
n
. The individual building blocks of all carbohydrates are
the monosaccharides, which consist of a chain of three to seven carbon atoms. De-
pending on the number of carbon atoms, they are categorized as trioses, tetroses,
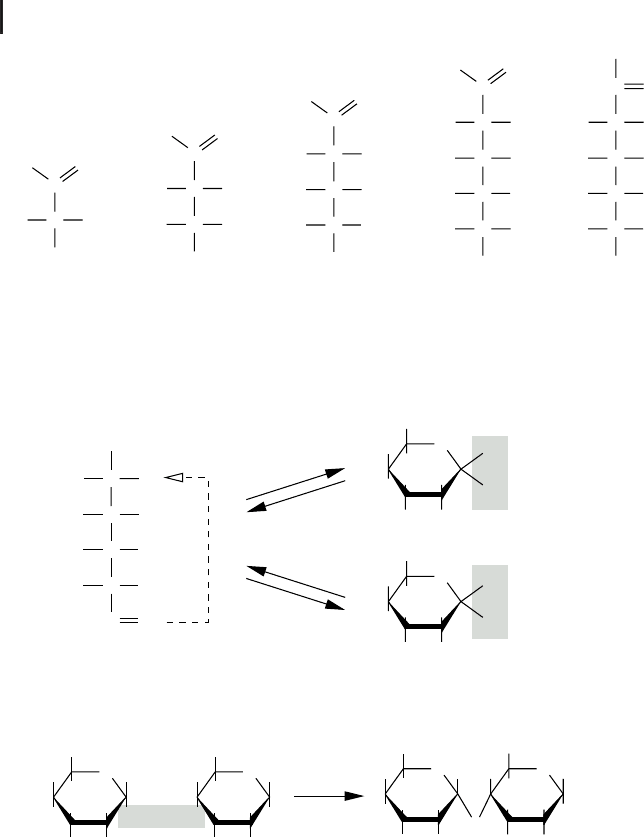
pentoses, hexoses, or heptoses (cf. Fig. 2.3a). All monosaccharides can occur in lin-
ear form, and with more than four carbons, they exist in equilibrium with a ring
form. In the linear form, all carbons of the chain, except for one, carry a hydroxyl
group (polyalcohol), which makes the carbohydrates hydrophilic. The remaining car-
bon carries a carbonyl group, and depending on its position–whether it is an alde-
hyde or ketone–it is called an aldose or a ketose. The circular configuration is at-
tained by an intramolecular reaction between the carbonyl group and one of the hy-
droxyl groups. Such a compound is called a hemiacetal. An example of the ring for-
mation for the six-carbon monosaccharide glucose is given in Fig. 2.3b, in which it
forms a so-called glucopyranose ring. Depending on the orientation of the hydroxyl
group at the 1-carbon, i.e., whether it points downwards (a-glucose) or upwards
(b-glucose), two alternate conformations exist. Glucose is one of the most important
energy sources for organisms. It is metabolized during glycolysis (see Section 2.3.3)
into ATP and reduction equivalents (e.g., NADH, NADPH, or FADH
2
).
The hydroxyl group at the 1-carbon position of the cyclic hemiacetal can react via a
condensation with the hydroxyl group of another monosaccharide. This linkage
forms a disaccharide from two monosaccharides (Fig. 2.3c). If this happens subse-
quently for several carbohydrates, polysaccharides that occur as linear chains or
branching structures are formed.
2.2.3.2 Lipids
Lipids are a very diverse and heterogeneous group. Since they are made up mostly of
nonpolar groups, lipids can be characterized by their higher solubility in nonpolar
solvents, such as acetone. Due to their hydrophobic character, lipids tend to form
27
2.2 Molecular Biology of the Cell
28
2 Biology in a Nutshell
CH
2
OH
C
C
O
H
HOH
CH
2
OH
C
C
O
H
HOH
CHOH
CHOH
CH
2
OH
C
C
O
H
HOH
CHOH
CH
2
OH
C
C
O
H
C
C
CH
H
HO
HO
H
H
OH
OH
CH
2
OH
CH
2
OH
C
C
C
COH
COH
HO H
O
OH
H
H
H
CH
2
OH
C
C
C
COH
OH
HC
H
H
H
HO H
OH
O
CH
2
OH
O
H
H
OH
H
H
HO
OH
OH
H
CH
2
OH
O
H
H
OH
H
H
H
OH
HO
OH
CH
2
OH CH
2
OH
O
H
H
OH
H
H
HO
OH
H
O
H
H
OH
H
H
OH
H
OH
O
CH
2
OH
O
H
H
OH
H
H
HO
OH
H
OH
CH
2
OH
O
H
H
OH
H
H
H
OHHO
OH
H
2
O
Sedoheptulose
(Heptose)
Mannose
(Hexose)
Ribose
(Pentose)
Erythrose
(Tetrose)
Glyceraldehyde
(Triose)
−Glucoseα
−Glucoseβ
A
B
C
++
Glucose Glucose Maltose (Disaccharide)
Fig. 2.3 Carbohydrates. (a) Some examples of
carbohydrates with a backbone of three to seven
carbon atoms. (b) Glucose, like other mono-
saccharides with more than four carbons in their
backbone, can form a circular structure, known
as hemiacetal, by an intramolecular condensa-
tion reaction that can occur in two different con-
formations. (c) By further condensation reac-
tions, such sugar monomers can form disac-
charides or even larger linear or branched mole-
cules called oligomers or polymers, depending
on the number of monomers involved.

nonpolar associations or membranes. Eventually, these membranes form cellular hy-
drophilic compartments. Furthermore, such hydrophobic regions offer a local area
for reactions that require a surrounding deprived of water. Three different types of
lipids are present in various cells and tissues: neutral lipids, phospholipids, and ster-
oids. Lipids can also be linked covalently to proteins or carbohydrates to form lipo-
proteins or glycolipids, respectively.
Neutral lipids are generally completely nonpolar and are commonly found as sto-
rage fats and oils in cells. They are composed of the alcohol glycerol (an alcohol with
three hydroxyl groups), which is covalently bound to fatty acids. A fatty acid is a lin-
ear chain of 4 to 24 or more carbon atoms with attached hydrogens (molecules like
this are well known as hydrocarbons) and a carboxyl group at one end (Fig. 2.4a).
Most frequent are chains with 16 or 18 carbons. Fatty acids can be either saturated or
unsaturated (polyunsaturated). Unsaturated fatty acids contain one or more double
bonds in their carbon chain and have a more fluid character than do saturated ones.
Linkage of the fatty acids to glycerol results from a condensation reaction of the car-
boxyl group with one of the alcohol groups of glycerol; this is called an ester binding.
If all three sites of the glycerol bind a fatty acid, it is called a triglyceride, which is the
most frequent neutral lipid in living systems. Triglycerides – in the form of fats or
oils – serve mostly as energy reserves.
Phospholipids are the primary lipids of biological membranes (cf. Section 2.3.1).
Their structure is very similar to that of the neutral lipids. However, the third carbon
of glycerol binds a polar residue via a phosphate group instead of a fatty acid. Polar
subunits commonly linked to the phosphate group are ethanolamine, choline, gly-
cerol, serine, threonine, or inositol (Fig. 2.4c). Due to their polar and unpolar parts,
phospholipids have dual-solubility properties termed amphipathic or amphiphilic.
This property enables phospholipids to form a so-called bilayer in an aqueous envir-
onment, which is the fundamental design principle of biological membranes
(Fig. 2.9 a). Polar and nonpolar parts of the amphipathic molecules are ordered side
by side in identical orientation and form a one-molecule thick layer (monolayer)
with a polar and a nonpolar side; the aqueous environment forces the lipophilic
sides of two such layers to each other, thus creating the mentioned bilayer.
Steroids are based on a framework of four condensed carbon rings that are modi-
fied in various ways (Fig. 2.4d). Sterols – the most abundant group of steroids – have
a hydroxyl group linked to one end of the ring structure, representing the slightly po-
lar part of the amphiphilic molecule; a nonpolar carbon chain is attached to the op-
posite end. The steroid cholesterol plays an important part in the plasma membrane
of animal cells. Among other things, cholesterol loosens the packing of membrane
phospholipids and maintains membrane fluidity at low temperatures. Other steroids
act as hormones (substances that regulate biological processes in tissues far away
from their own place of production) in animals and are, e.g., involved in regulatory
processes concerning sexual determination or cell growth.
In glycolipids, the lipophilic part is constituted of fatty acids bound to the 1-carbon
and 2-carbon of glycerol, as is the case with phospholipids. The 3-carbon is covalently
attached to one or more carbohydrate groups that confer an amphiphilic character to
the molecule. Glycolipids occur, e.g., in the surface-exposed parts of the plasma
29
2.2 Molecular Biology of the Cell
