Klipp E., Herwig R., Kowald A., Wierling C., Lehrach H. Systems Biology in Practice: Concepts, Implementation and Application
Подождите немного. Документ загружается.

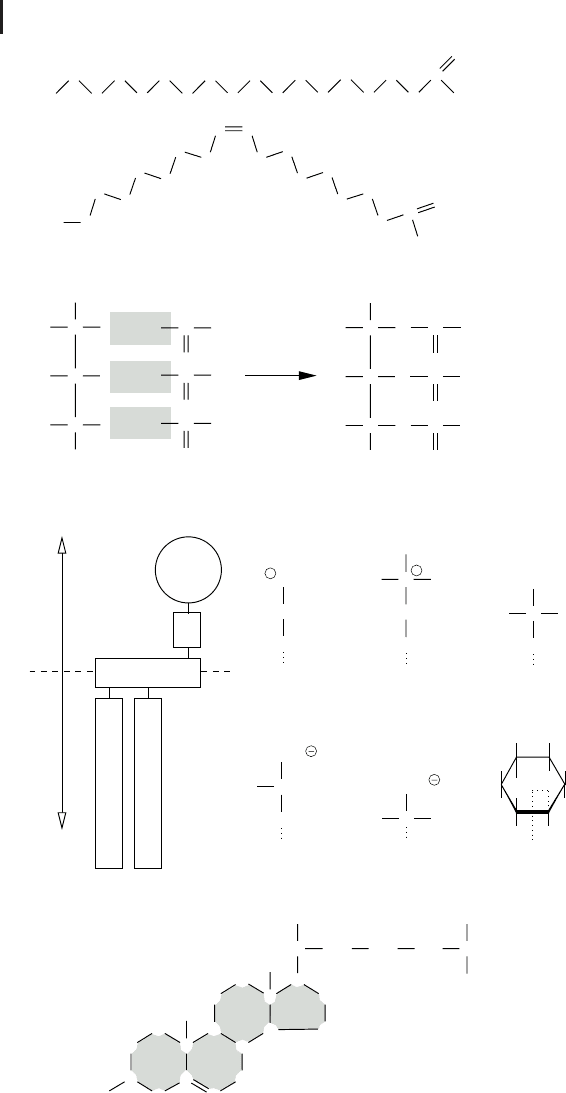
30
2 Biology in a Nutshell
H
3
C
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
C
O
OH
H
3
CCH
2
CH
2
CH
2
CH
2
H
2
C
H
2
C
H
2
C
H
2
C
H
2
C
H
2
C
H
2
C
CH
2
CH
2
CH
2
OH
C
O
CHHC
CH
3
CH
3
CH
2
H
3
C
CH
2
N
NH
3
CH
2
CH
2
CH
2
OH
CH
2
CHOH
H
3
N
COO
CH
CH
2
H
3
N
COO
CH
H
H
H
H
H
H
OH
OH
OH
HO
CH
2
CH
3
CH
3
H
2
C
CH
2
H
2
CCH
2
CH
2
H
2
H
2
H
2
CH
2
CH
3
CH
3
CH
2
CH
3
HO
HC
C
C
C
C
CC
H
C
C
H
C
H
C
C
H
CH
H
C
H
C
C
C
OH
OH
OH
H
H
H
H
H
HO C
O
R
HO C
O
R
HO C
O
R
C
C
C
H
H
H
H
H
O
O
O
C
C
C
R
R
R
O
O
O
H
2
O3
F
a
t
t
y
a
c
i
d
F
a
t
t
y
a
c
i
d
Polar
group
p
o
l
a
r
n
o
n
p
o
l
a
r
CholineEthanolamine Glycerol
Serine Threonine
Inositol
C
P
Glycerol
D
B
+
+
+
+
Glycerol Fatty acids Triglyceride
+
+
A
Stearic acid
Oleic acid

membrane bilayer of animal cells that are subject to physical or chemical stress.
Furthermore, among several other things, they are responsible for the AB0 blood
system of humans.
2.2.3.3 Proteins
Proteins fulfill numerous highly important functions in the cell, of which only a few
can be mentioned here. They build up the cytoskeletal framework, which forms the
cellular structure and is responsible for cell movements (motility). Proteins are also
part of the extracellular supportive framework (extracellular matrix), e.g., as collagen
in animals. As catalytic enzymes for highly specific biochemical reactions, they rule
and control the metabolism of a single cell or whole organism. Furthermore, pro-
teins regulated by transient modifications are relevant for signal transduction, e.g.,
proteins controlling cell division such as cyclin-dependent protein kinases (CDK).
A further highly important function of proteins is their ability to control the tran-
scription and translation of genes as well as the degradation of proteins (see Sec-
tion 2.4).
Proteins consist of one or more polypeptides. Each polypeptide is composed of
covalently linked amino acids; these covalent bonds are called peptide bonds. Such a
bond is formed by a condensation reaction between the amino group of one amino
acid and the carboxyl group of another (Fig. 2.6a). The primary structure of a poly-
peptide is coded by the genetic information that defines in which order amino acids
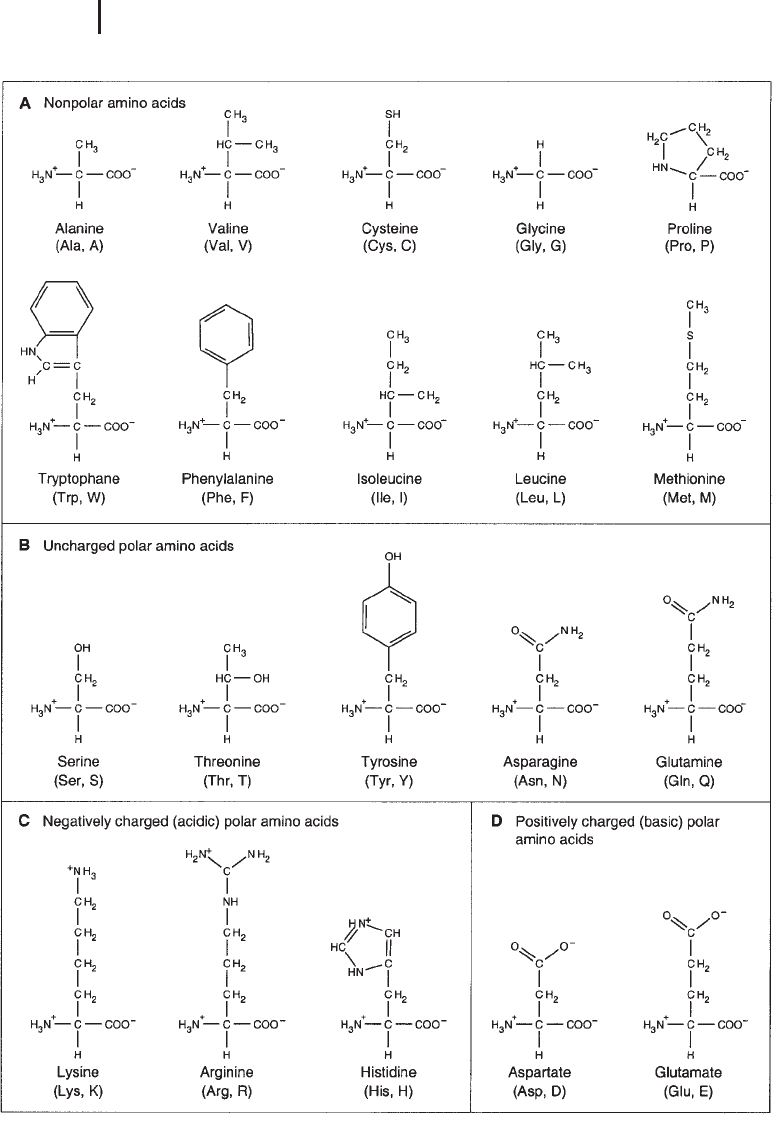
– chosen from a set of 20 different ones – appear. Figure 2.5 shows the chemical
structures of these amino acids. Common to all amino acids is a central carbon
(a-carbon), which carries an amino group (except for proline, where this is a ring-
forming imino group), a carboxyl group, and a hydrogen. Furthermore, it carries a
residual group with different physicochemical properties, due to which the amino
acids can be divided into different groups, such as amino acids that carry (1) nonpo-
lar residues that can grant lipophobic characteristics, (2) uncharged polar residues,
(3) residues that contain a carboxyl group that is negatively charged at physiological
pH and thus act as acids, and (4) residues that are usually positively charged at com-
mon pH ranges of living cells and thus show basic characteristics. Due to the combi-
nation of possibilities of these amino acids, proteins are very diverse. Usually, pro-
teins are assembled from about 50 to 1000 amino acids, but they might be much
smaller or larger. Except for glycine, the a-carbon of amino acids binds four different
residues and therefore amino acids can occur in two different isoforms that behave
like an image and its mirror image. These two forms are called the L-isoform and
the D-isoform, of which only the L-isoform is used in naturally occurring proteins.
Furthermore, amino acids of proteins are often altered posttranslational. For in-
31
2.2 Molecular Biology of the Cell
3 Fig. 2.4 (a) Fatty acids represent one part of
fats and phospholipids. They are either saturated
or unsaturated. (b) Triglycerides are formed by
condensation reactions of glycerol and three
fatty acids. (c) In phospholipids the third carbon
of glycerol is bound to a polar group via a phos-
phate group (P), which is usually ethanolamine,
choline, glycerol, serine, threonine, or inositol.
(d) Steroids constitute another major lipid class.
They are formed by four condensed carbon
rings. Cholesterol, shown here, is important,
e.g., for membrane fluidity of eukaryotic cells.
32
2 Biology in a Nutshell

stance, proline residues in collagen are modified to hydroxyproline by addition of a
hydroxyl group.
The primary structure of a protein is given by the sequence of the amino acids
linked via peptide bonds. This sequence starts at the N-terminus of the polypeptide
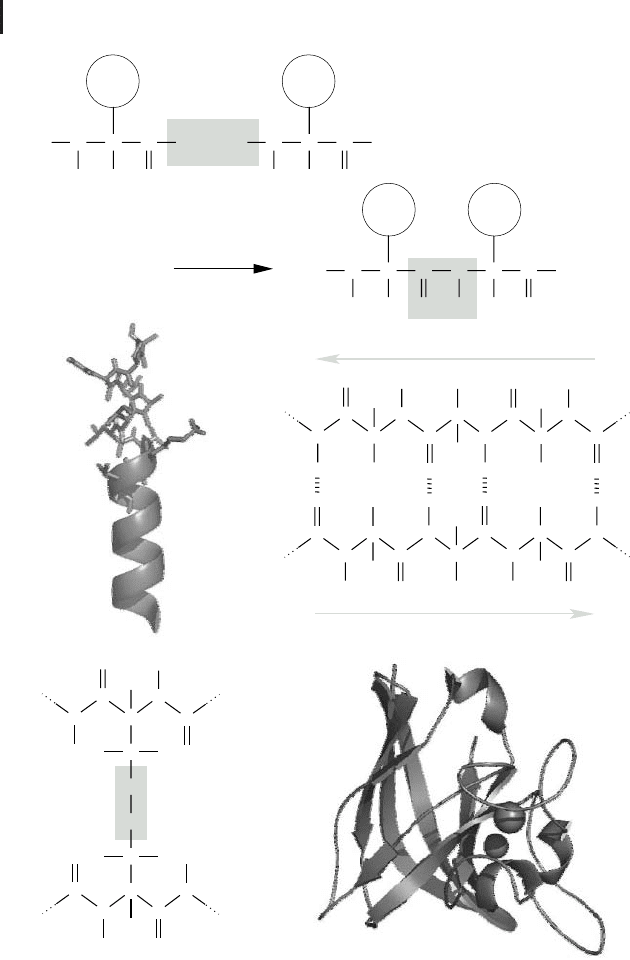
and ends at its C-terminus (cf. Fig. 2.6a). In the late 1930s Linus Pauling and Robert
Corey elucidated the exact structure of the peptide bond. They found that the hydro-
gen of the substituted amino group almost always is in opposite position to the oxy-
gen of the carbonyl group, so that both together, with the carbon of the carbonyl
group and the nitrogen of the amino group, build a rigid plane. This is due to the
fact that the bond between carbon and nitrogen has a partial double-bond character.
In contrast to this, both the bonds of the a-carbon with the nitrogen of the substi-
tuted amino group and the carbon of the carbonyl group are flexible since they are
pure single bonds. The free rotation around these two bonds is limited only by steric
interactions of the amino acid residues. Based on this knowledge, Pauling and Corey
proposed two very regular structures: the a-helix and the b-strand. Both are very
common in proteins. They are formed by the polypeptide backbone and are sup-
ported and stabilized by a specific local amino acid sequence composition. Such reg-
ular arrangements are called secondary structures. An a-helix (Fig. 2.6b) has a cy-
lindrical helical structure in which the carbonyl oxygen atom of each residue (n) ac-
cepts a hydrogen bond from the amide nitrogen four residues further in sequence
(n + 4). Amino acids often found in a-helices are Glu, Ala, Leu, Met, Gln, Lys, Arg,
and His. In a b-sheet, parallel peptide strands – b-strands that may be widely sepa-
rated in the linear protein sequence – are linked side by side via hydrogen bonds be-
tween hydrogen and oxygen atoms of their backbone (Fig. 2.6c). The sequence direc-
tion (always read from the amino/N-terminus to the carboxy/C-terminus of the poly-
peptide) of pairing b-strands can be either parallel or antiparallel. The residual
groups of the amino acids point up and down from the b-sheet. Characteristic amino
acids of b-sheets are Val, Ile, Tyr, Cys, Trp, Phe, and Thr. The regular secondary struc-
ture elements fold into a compact form that is called the tertiary structure of a pro-
tein. Its surface topology enables specific interactions with other molecules. Fig-
ure 2.6e shows a model of the three-dimensional structure of the superoxide dismu-
tase (SOD), which detoxifies aggressive superoxide radicals (O
2
Q
–
). Sometimes the ter-
tiary structure is stabilized by posttranslational modifications such as disulfide
bridges (Fig. 2.6d) or by metal ions such as calcium (Ca
2+
) or zinc (Zn
2+
). Some pro-
teins are fibrous, i.e., they form filamentous structures (e.g., the keratin of hair).
But most proteins fold into globular, compact shapes. Larger proteins often fold into
several independent structural regions: the domains. Domains frequently consist of
50 to 350 residues and are often capable of folding stably enough to exist on their
own. Often proteins are composed of assemblies of more than one polypeptide
33
2.2 Molecular Biology of the Cell
3 Fig. 2.5 Amino acids are formed by carbon that is bound to an amino
group, a carboxy group, a hydrogen, and a residual group. Depending
on the physicochemical characteristics of the residual group, they can
be categorized as (a) nonpolar, (b) uncharged polar, (c) acidic, or
(d) basic amino acids.
34
2 Biology in a Nutshell
H
H
N
H
CC
O
OH H
H
N
H
CC
O
OH
H
HH HHO
NNCC CCOH
O
H
2
O
O
O
H
H
N
N
H
C
C
C
CC
CCN
N
O
O
H
H
H
H
O
O
H
H
N
N
H
C
C
C
CC
N
O
H
H
N
H
R
R
R
R
R
R
H
O
C
C
S
S
C
C
H
H
H
H
O
O
O
O
H
H
H
H
N
N
N
N
H
H
C
C
C
C
C
C
Side
chain
Side
chain
Side
chain
Side
chain
A
B
D
+
+
C
E
Fig. 2.6 (a) Formation of a peptide linkage by a
reaction between the carboxyl group of one
amino acid with the amino group of a second.
(b) The molecular structure of an a-helix, as
shown in the upper part of the image, is often il-
lustrated by a simple helical structure as shown
below. (c) An antiparallel b-sheet. (d) A disulfide
bridge is formed by oxidation of the SH groups
of cysteine residues belonging to either the
same or different polypeptides. (e) Three-dimen-
sional illustration of the copper zinc superoxide
dismutase (CuZnSOD) of E. coli (PDB: 1EOS).
a-helices are depicted as helical structures and
b-strands are illustrated by arrows. The two
metal ions are shown as spheres.

chain. Such a composition is termed the quaternary structure. The subunits can be
either identical or different in sequence and the protein is thus referred to as a
homo- or heteromer, e. g., a protein composed of four identical subunits such as the
lac repressor is called a homotetramer.
2.2.3.4 Nucleic Acids
Deoxyribonucleic acid (DNA) is present in all living organisms and is the molecule
storing hereditary information, i.e., the genes. Another molecule, ribonucleic acid
(RNA), takes part in a vast number of processes. Among these, the transfer of the
hereditary information leading from DNA to protein synthesis (via transcription and
translation; see Section 2.4) is the most important. Both DNA and RNA are nucleic
acids. Nucleic acids are polymers built up of covalently bound mononucleotides.
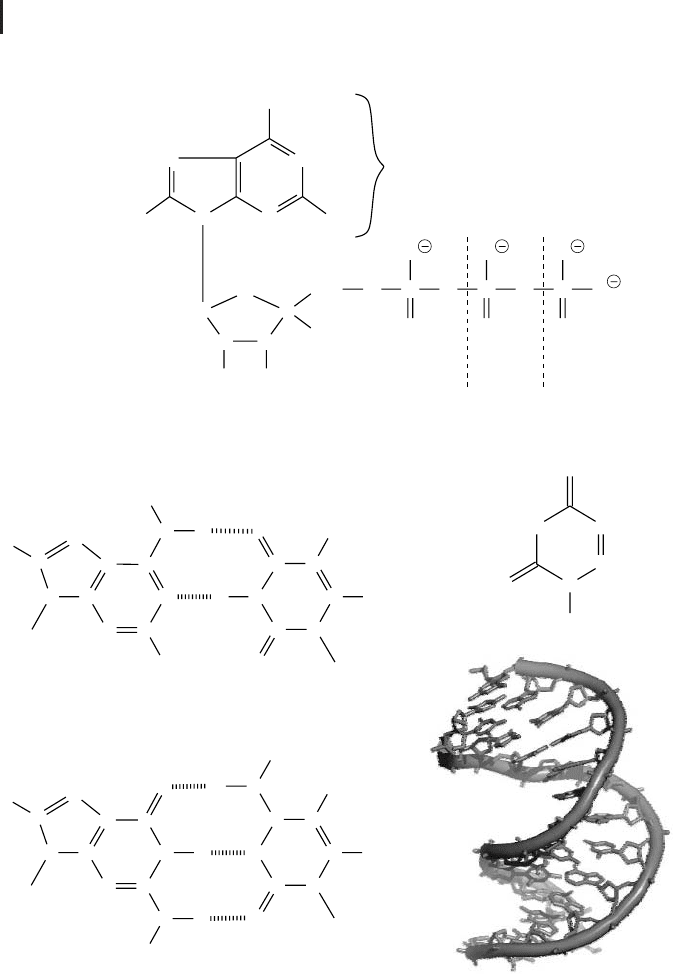
A nucleotide consists of three parts: (1) a nitrogen-containing base, (2) a pentose,
and (3) one or more phosphate groups (Fig. 2.7 a). Bases are usually pyrimidines
such as cytosine (C), thymine (T), or uracil (U) or purines such as adenine (A) or
guanine (G) (Fig. 2.7 b). In RNA the base is covalently bound to the first carbon
(1'-carbon) of the circular pentose ribose. In DNA it is bound to the 1'-carbon of
deoxyribose, a pentose that lacks the hydroxyl group of the 2'-carbon. A unit consist-
ing of these parts – a base and a pentose – is named nucleoside. If it furthermore car-
ries a mono-, di-, or triphosphate, it is called a nucleotide. Nucleotides are named ac-
cording to their nucleoside, e.g., adenosine monophosphate (AMP), adenosine di-
phosphate (ADP), or adenosine triphosphate (ATP); prepending deoxy to the name
(or d in the abbreviation) indicates the deoxy form (e. g., deoxyguanosine tripho-
sphate or dGTP). Nucleotides are not only relevant for nucleic acid construction but
also are responsible for energy transfer in several metabolic reactions (e. g., ATP and
ADP) or play certain roles in signal transduction pathways, such as 3'-5'-cyclic AMP
(cAMP), which is synthesized by the adenylate cyclase and is involved, for instance,
in the activation of certain protein kinases.
In DNA and RNA, the 3' -carbon of a nucleotide is linked to the 5'-carbon of the
next nucleotide in sequence via a single phosphate group. These alternating sugar
and phosphate groups form the backbone of the nucleic acids. Both DNA and RNA
can carry the bases adenine, guanine, and cytosine. In DNA thymine can also be pre-
sent. The sequence of the different bases has a direction – because of the 5’-3’-link-
age of its backbone – and is used in living organisms for the conservation of infor-
mation. DNA contains millions of nucleotides, e.g., a single DNA strand of human
chromosome 1 is about 246 million nucleotides long. Each base of the sequence is
able to pair with a so-called complementary base by hydrogen bonds. Due to the
number and steric arrangement of hydrogen bonds, only two different pairing types
are possible (Fig. 2.7 b): adenine can bind thymine (A-T, with two hydrogen bonds)
and guanine can bind cytosine (G-C, with three hydrogen bonds). In RNA, thymine
is replaced by uracil. In 1953, Watson and Crick proposed a double strand for DNA,
with an antiparallel orientation of the backbones. Each of the bases of one strand
binds to its complementary base on the other strand, and together they form a heli-
cal structure (Fig. 2.7d). This so-called double helix is the usual conformation of
DNA in cells. RNA usually occurs as a single strand. Occasionally it is paired to a
35
2.2 Molecular Biology of the Cell
36
2 Biology in a Nutshell
CH
3
O
O
CC
C
C
HH
N
C
C
C
C
C
N
N
N
N
N
H
H
H
H
N
O
CC
C
C
H
N
C
C
C
C
C
N
N
H
H
N
H
H
O
H
NH
H
N
N
N
NH
2
CH
2
OPOPOP O
OOO
OOO
N
NN
N
HH
OH
O
X
HC
H
CC
C
O
O
HN
N
H
CH
CH
Adenine Thymine
Guanine Cytosine
Ribose (X = OH)
or
Deoxyribose (X = H)
Bases are one of the following:
adenine, guanine, cytosine,
thymine (in DNA), or
uracil (in RNA)
A
BC
D
Uracil
(Deoxy)adenosine triphosphate (ATP or dATP)
1’
2’
4’
5’
3’
(Deoxy)ribose
Mono− Di− Triphosphate
Fig. 2.7 (a) Nucleoside phosphates are com-
posed of a ribose or deoxyribose that is linked at
its 1’-position to a purine or pyrimidine base.
Purines are adenine and guanine, and pyrimi-
dines are thymine, cytosine, or uracil. (b) In
DNA, adenine is bound to its complementary
base thymine by two hydrogen bonds, and gua-
nine is bound to cytosine by three hydrogen
bonds. (c) In RNA, thymine is replaced by uracil.
(d) The DNA double helix (PDB: 140D).
DNA single strand, as during mRNA synthesis (see Section 2.4.1), or complemen-
tary bases of the same molecule are bound to each other, e.g., as in tRNA.
2.3
Structural Cell Biology
This section gives a general introduction to the structural elements of eukaryotic
cells. Fundamental differences between prokaryotic and eukaryotic cells have already
been mentioned and are summarized in Table 2.1.
The first microscopic observations of cells were done in the 17th century by Robert
Hooke and Anton van Leeuwenhoek. The general cell theory was developed in the
1830s by Theodor Schwann and Matthias Schleiden. It states that all living organ-
isms are composed of nucleated cells that are the functional units of life and that
cells arise only from preexisting cells by a process of division (see Section 2.5). Today
we know that this is true not only for nucleated eukaryotic cells but also for prokaryo-
tic cells lacking a nucleus. The interior of a cell is surrounded by a membrane that
separates it from its external environment. This membrane is called the cell mem-
brane or plasma membrane and it is semipermeable, i.e., the traffic of substances
across this membrane in either orientation is restricted to some specific molecule
species or is specifically controlled by proteins of the membrane that handle the
transport. Fundamental to eukaryotic cells – in contrast to prokaryotic cells – is their
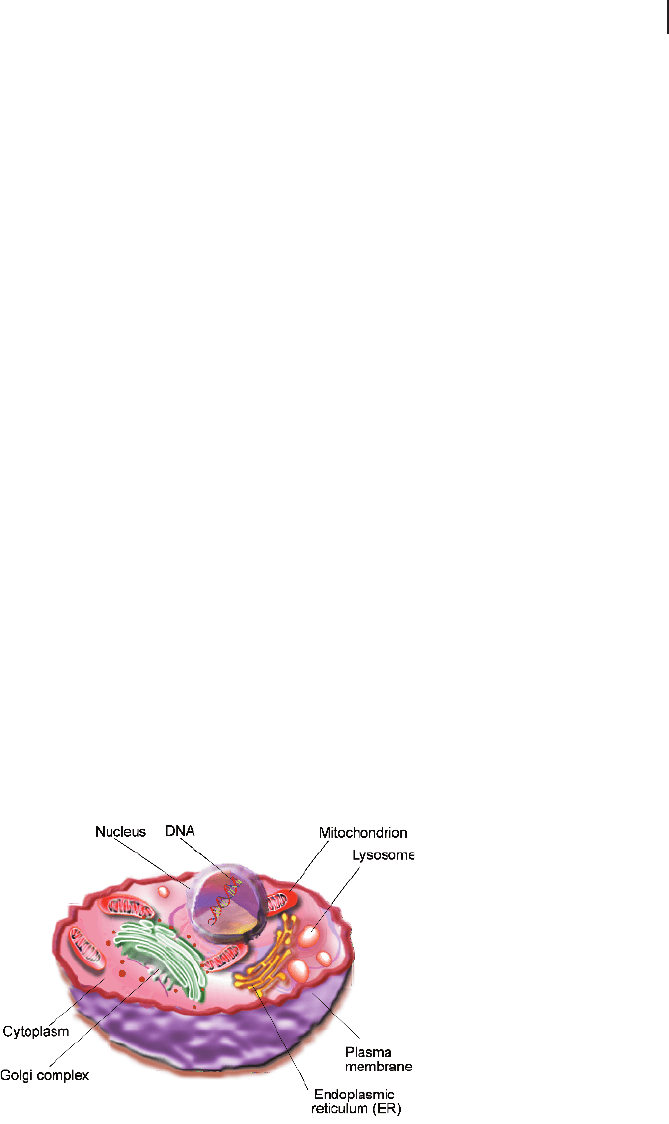
subdivision by intracellular membranes into distinct compartments. Figure 2.8 illus-
trates the general structure of a eukaryotic cell as it is found in animals. Generally,
one distinguishes between the storage compartment of the DNA, the nucleus, and
the remainder of the cell interior that is located in the cytoplasm. The cytoplasm con-
tains further structures that fulfill specific cellular functions and that are surrounded
by the cytosol. Among these cytoplasmatic organelles are the endoplasmatic reticu-
lum (ER), which forms a widely spread intracellular membrane system; the mito-
chondria, which are the cellular power plants; the Golgi complex; transport vesicles;
peroxisomes; and, additionally in plant cells, chloroplasts, which act as sunlight-har-
37
2.3 Structural Cell Biology
Fig. 2.8 Schematic illustra-
tions of an animal cell with its
major organelles.

vesting systems performing photosynthesis, and the vacuole. In the following sec-
tions, we will describe the structure and function of biological membranes and the
most important cellular compartments that are formed by them.
2.3.1
Structure and Function of Biological Membranes
All cells are surrounded by a plasma membrane. It not only separates the cell plasma
from its surrounding environment but also acts as a selective filter for nutrients and
byproducts. By active transport of ions, for which the energy source ATP is usually
utilized, a chemical and/or electrical potential can be generated across the mem-
brane that is essential, e.g., for the function of nerve cells. Furthermore, receptor
proteins of the plasma membrane enable the transmission of external signals that
enable the cell to react to its environment. As already mentioned, eukaryotes addi-
tionally possess an intracellular membrane system acting as a boundary for different
essential compartments.
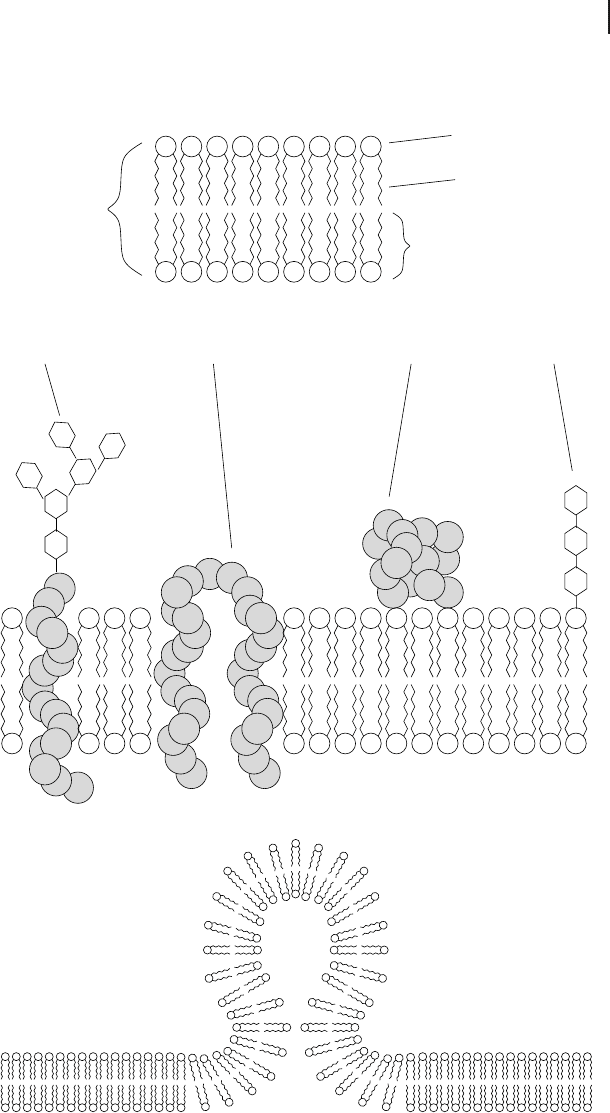
The assembly of a bilayer,which is the fundamental structure of all biological mem-
branes, is described in the section about lipids (Section 2.2.3; cf. also Fig. 2.9a). Biolo-
gical membranes are composed of this molecular bilayer of lipids (mainly phospholi-
pids, but also cholesterol and glycolipids) and membrane proteins that are inserted
and held in the membrane by noncovalent forces. Besides integral membrane pro-
teins, proteins can also be attached to the surface of the membrane (peripheral pro-
teins). This model of biological membranes is known as the fluid mosaic model and
was introduced by Singer and Nicolson (1972) (Fig. 2.9b). They further proposed a
possible asymmetric arrangement of adjoining monolayers caused by different lipid
composition and orientation of integral proteins, as well as specific occurrence of per-
ipheral proteins in either of the monolayers. In the plasma membrane, for example,
glycolipids always point to the exterior. While an exchange of lipid molecules between
the two monolayers – a so-called flip-flop – very seldomly occurs by mere chance, lat-
eral movement of lipid molecules takes place frequently. This can also be observed
with proteins as long as their movement is not prevented by interaction with other
molecules. Lateral movement of lipids depends on the fluidity of the bilayer. The fluid-
ity is strongly enhanced if one of the hydrocarbon chains of the phospholipids is unsa-
turated and the membrane contains a specific amount of cholesterol.
An important feature of biological membranes is their ability to form a cavity that
pinches off as a spherical vesicle, and the reverse process in which the membrane of a
vesicle fuses with another membrane and becomes a part of it (Fig. 2.9c). This property
is utilized by eukaryotic cells for vesicular transport between different intracellular
compartments and for the exchange of substances with the exterior. The latter process
38
2 Biology in a Nutshell
Fig. 2.9 (a) In a lipid bilayer the amphipathic
lipids are orientated to both aqueous compart-
ments with their hydrophilic parts. The hydro-
phobic tails point to the inner membrane space.
(b) The fluid mosaic model of a cellular mem-
brane. (c) Formation of a spherical vesicle that
is in the process of either pinching off from or
fusing with a membrane. Such vesicles are
formed during either endo- or exocytosis by
peripheral proteins inducing the process.
"
39
2.3 Structural Cell Biology
B
A
C
GlycolipidGlycoprotein Transmembrane protein
Bilayer
Monolayer
Hydrophilic head
Lipophilic tail
Peripheral protein
