Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


4
SPECIES BALANCES AND DESIGN
EQUATIONS
This chapter covers the fundamental concept that leads to the formulation of the
design equations of chemical reactors—conservation of mass. Since we deal here
with operations involving changes in composition, we carry out mass balances
for individual species. Also, since the operations involve chemical reactions, it is
convenient to express the amount of a species in terms of moles rather than
mass. In general, the species balances are carried out in one of two ways—as micro-
scopic balances or macroscopic balances. Microscopic species balances, often
referred to as the “species continuity equations,” are carried out over a differential
element and describe what takes place at a given “point” in a reactor. When inte-
grated over the volume of the reactor, they provide the species-based design
equations that describe the reactor operation. Macroscopic balances are carried
out over the entire reactor, or a large portion of it, and provide the design equations.
The general conservation statement for species j over a stationary system is
Molar flow
rate of
species j
into system
8
>
>
<
>
>
:
9
>
>
=
>
>
;
þ
Rate moles
of species j
formed
inside system
8
>
>
<
>
>
:
9
>
>
=
>
>
;
¼
Molar flow
rate of
species j out
of system
8
>
>
<
>
>
:
9
>
>
=
>
>
;
þ
Rate moles
of species j
accumulate
in system
8
>
>
<
>
>
:
9
>
>
=
>
>
;
(4:0:1)
Appendix B provides the derivation of the design equation from the species
continuity
equation. In Section 4.1, we carry out macroscopic species balances to
derive the species-based design equation of any chemical reactor. In Section 4.2,
Principles of Chemical Reactor Analysis and Design, Second Edition. By Uzi Mann
Copyright # 2009 John Wiley & Sons, Inc.
101
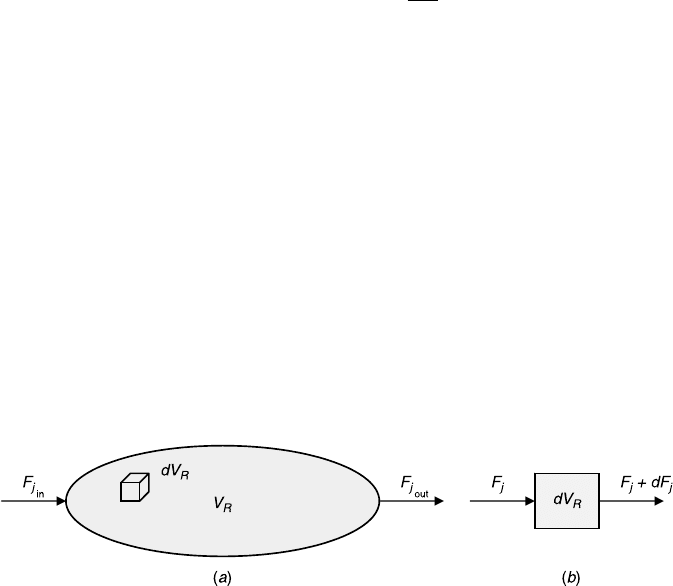
we apply the general species-based design equations to reactor configurations
commonly used in reactor analysis—ideal batch reactor, continuous stirred-tank
reactor (CSTR), and plug-flow reactor. In Section 4.3, the reaction-based design
equations are derived for the three ideal reactor configurations. In Section 4.4,
the reaction-based design equations are reduced to dimensionless forms.
4.1 MACROSCOPIC SPECIES BALANCES—GENERAL
SPECIES-BASED DESIGN EQUATIONS
To perform a macroscopic species balance, we conduct a species balance over an
entire reactor. Consider an arbitrary reactor with one inlet and one outlet and
volume V
R
, shown schematically in Figure 4.1a. Fluid flows through the system,
and chemical reactions take place inside the system. We impose no restrictions
on the system except the assumption that the chemical reactions are homogeneous
(i.e., they take place throughout the system).
The balance equation of species j over the reactor is
F
j
in
þ G
j
¼ F
j
out
þ
dN
j
dt
(4:1:1)
where G
j
is the rate species j is formed inside the entire reactor by chemical reactions.
To obtain useful relations, we should express the generation term, G
j
. When different
conditions exist at different points in the reactor, the species formation rates, (r
j
)’s,
vary from point to point in the reactor. Consider a differential volume element, dV,
and express the rate species j is generated in that element, dG
j
,by
dG
j
¼ (r
j
) dV
where (r
j
) is defined by Eq. 3.1.1a. Therefore, the rate species j is generated in the
entire reactor is
G
j
¼
ð
V
R
dG
j
¼
ð
V
R
(r
j
) dV (4:1:2)
Figure 4.1 Schematic description of a reacting system: (a) overall system and
(b) differential reactor.
102 SPECIES BALANCES AND DESIGN EQUATIONS
where V
R
is the volume of the reactor. When the same conditions (temperature and
concentrations) exist throughout the system (due to good mixing), the formation
rate of species j,(r
j
), is the same everywhere in the reactor, and Eq. 4.1.2 becomes
G
j
¼ (r
j
)V
R
Substituting Eq. 4.1.2, Eq. 4.1.1 reduces to
F
j
in
F
j
out
þ
ð
V
R
(r
j
) dV ¼
dN
j
dt
(4:1:3)
Equation 4.1.3 is the integral form of the general species-based design equation of
chemical reactors, written for species j.
The differential form of the general species-based design equation can be
derived either by a formal differentiation of Eq. 4.1.3 or by conducting a balance
for species j over a differential reactor shown schematically in Figure 4.1b. For a
differential reactor, the balance over species j is
F
j
(F
j
þ dF
j
) þ (r
j
) dV
R
¼
dN
j
dt
which becomes
(r
j
) dV
R
¼ dF
j
þ
dN
j
dt
(4:1:4)
Equation 4.1.4 is the differential form of the general, species-based design equation
for all chemical reactors.
For reactors with single chemical reactions, it has been customary to write the
species-based design equation for the limiting reactant A. Hence,
F
A
in
¼ F
A
out
þ
ð
V
R
(r
A
) dV þ
dN
A
dt
(4:1:5)
where (2r
A
) is the depletion rate of reactant A, defined by Eq. 3.1.1a. Note that
since A is a reactant, (2r
A
) is a positive quantity. Similarly, from Eq. 4.1.4, we
can readily obtain the differential form of the general species-based design
equation, written for limiting reactant A:
dF
A
¼ (r
A
) dV þ
dN
A
dt
(4:1:6)
To obtain useful expressions from the general species-based design equation, we
should know the formation rate of species j,(r
j
), at any point in the reactor. To
express (r
j
), the local concentrations of all species as well as the local temperatures
should be provided. To obtain these quantities, we should solve the overall conti-
nuity equation, the individual species continuity equations, and the energy balance
equation. This is a formidable task, and, in most situations, we cannot reduce those
4.1 MACROSCOPIC SPECIES BALANCES 103
equations to useful forms and have to resort to numerical or approximate solution
methods that are beyond the scope of this book. Therefore, in most instances, we
apply the design equations to simplified reactor configurations (or mathematical
models) that approximate the operations of actual reactors. In this text, we discuss
the application of the reactor design equations to the following reactor configurations:
†
Well-mixed batch reactor (ideal batch reactor)
†
Steady, well-mixed continuous reactor (CSTR)
†
Steady plug-flow reactor (PFR)
†
Certain other special reactor configurations
In Section 4.2, we derive the design equations for the first three ideal reactor
models. Other reactor configurations are discussed in Chapter 9.
4.2 SPECIES-BASED DESIGN EQUATIONS OF IDEAL REACTORS
4.2.1 Ideal Batch Reactor
For a batch reactor, F
j
out
¼ F
j
in
¼ 0, and Eq. 4.1.3 becomes
dN
j
dt
¼
ð
V
R
(r
j
) dV
If the reactor is well mixed, the same conditions (concentration and temperature) exist
everywhere; hence, (r
j
) is the same throughout the reactor. Thus, Eq. 4.1.3 becomes
dN
j
dt
¼ (r
j
)V
R
(t)(4:2:1)
Equation 4.2.1 is the species-based design equation of an ideal batch reactor,
written for species j. To obtain the operating time, we separate the variables and
integrate Eq. 4.2.1:
t ¼
ð
N
j
(t)
N
j
(0)
dN
j
(r
j
)V
R
(4:2:2)
Equation 4.2.2 is the integral form of the species-based design equation for an ideal
batch reactor, written for species j. It provides a relation between the operating time,
t, the amount of the species in the reactor, N
j
(t) and N
j
(0), the species formation
rate, (r
j
), and the reactor volume, V
R
. Note that when the reactor volume does
not change during the operation, Eq. 4.2.2 reduces to
t ¼
ð
C
j
(t)
C
j
(0)
dC
j
(r
j
)
(4:2:2a)
104 SPECIES BALANCES AND DESIGN EQUATIONS

For reactors with single chemical reactions, it has been customary to write the
species-based design equation for the limiting reactant A, and Eq. 4.2.1 reduces to
dN
A
dt
¼ (r
A
)V
R
(t)(4:2:3)
Using the conversion definition (Eq. 2.6.1a),
dN
A
dt
¼ N
A
(0)
df
A
dt
and Eq. 4.2.1 becomes
N
A
(0)
df
A
dt
¼ (r
A
)V
R
(t)(4:2:4)
To obtain the integral form of the design, we separate the variables and integrate
Eq. 4.2.4:
t ¼ N
A
(0)
ð
f
A
(t)
0
df
A
(r
A
)V
R
(4:2:5)
When the reactor volume does not change during the operation, V
R
¼ V
R
(0),
Eq. 4.2.5 reduces to
t ¼ C
A
(0)
ð
f
A
(t)
0
df
A
r
A
(4:2:5a)
4.2.2 Continuous Stirred-Tank Reactor (CSTR)
A CSTR is a reactor model based on two assumptions: (i) steady-state operation
and (ii) the same conditions exist everywhere inside the reactor (due to good
mixing). For steady operations, the accumulation term in the design equation
vanishes. Since the same conditions exist everywhere, the rate (r
j
) is the same
throughout the reactor and is equal to the rate at the reactor effluent, (r
j
)
out
.
Hence, the general, species-based design equation Eq. (4.1.3) reduces to
F
j
out
F
j
in
¼ (r
j
)
out
V
R
(4:2:6)
We can rearrange Eq. 4.2.6 as
V
R
¼
F
j
out
F
j
in
(r
j
)
out
(4:2:7)
Equation 4.2.7 is the species-based design equations for a CSTR, written for
species j. It provides a relation between the species flow rate at the inlet and
outlet of the reactor, F
j
in
and F
j
out
, the species formation rate (r
j
), and the reactor
volume, V
R
.
4.2 SPECIES-BASED DESIGN EQUATIONS OF IDEAL REACTORS 105

For a CSTR with single chemical reactions, the species-based design equation is
usually written for the limiting reactant A, and Eq. 4.2.7 reduces to
V
R
¼
F
A
in
F
A
out
(r
A
)
out
(4:2:8)
Using the conversion definition (Eq. 2.6.1b) and applying it to the reference stream,
F
A
0
:
F
A
in
¼ F
A
0
(1 f
A
in
)
F
A
out
¼ F
A
0
(1 f
A
out
)
and Eq. 4.2.8 becomes
V
R
F
A
0
¼
f
A
out
f
A
in
(r
A
)
out
(4:2:9)
Equation 4.2.9 is the species-based design equation of a CSTR, expressed in terms
of the conversion of reactant A.
4.2.3 Plug-Flow Reactor (PFR)
A PFR is a reactor model based on two assumptions: (i) steady-state operation and
(ii) a flat velocity profile (plug flow) with the same conditions existing at any given
cross-section area (no concentration or temperature gradients in the direction
perpendicular to the flow). Since the species compositions change along the
reactor and the species formation rate, (r
j
), varies along the reactor, we consider
a differential reactor of volume dV
R
. For steady operations, the accumulation
term vanishes, and Eq. 4.1.4 reduces to
dF
j
¼ (r
j
) dV
R
(4:2:10)
Equation 4.2.10 is the species-based differential design equations for a PFR, writ-
ten for species j. To obtain the reactor volume of a PFR, we integrate Eq. 4.2.10,
V
R
¼
ð
F
j
out
F
j
in
dF
j
(r
j
)
(4:2:11)
Equation 4.2.11 provides a relation among the species flow rate at the inlet and
outlet of the reactor, F
j
in
and F
j
out
, the species formation rate, (r
j
), and the
volume of the reactor, V
R
, for a plug-flow reactor.
106 SPECIES BALANCES AND DESIGN EQUATIONS

For PFRs with single chemical reactions, it has been customary to write the
species-based design equation for the limiting reactant A, and Eq. 4.2.10 reduces to
dV
R
¼
dF
A
(r
A
)
(4:2:12)
Differentiating the conversion definition (Eq. 2.6.1b),
dF
A
¼F
A
0
df
A
and Eq. 4.2.12 reduces to
dV
R
¼ F
A
0
df
A
(r
A
)
(4:2:13)
Equation 4.2.13 is the species-based differential design equation of a plug-flow
reactor, expressed in terms of the conversion of reactant A. To obtain the integral
form of the design equation, we separate the variables and integrate Eq. 4.2.13:
V
R
F
A
0
¼
ð
f
A
out
f
A
in
df
A
r
A
(4:2:14)
Equation 4.2.14 is the species-based integral design equation of a plug-flow reactor,
expressed in terms of the conversion of reactant A.
4.3 REACTION-BASED DESIGN EQUATIONS
To analyze and design chemical reactors more effectively and to obtain insight into
the operation, we adopt reaction-based design formulation. In this section, we derive
the reaction-based design equations for the three ideal reactor models. Reaction-
based design equations of other reactor configurations are derived in Chapter 9.
4.3.1 Ideal Batch Reactor
For an ideal batch reactor, the species-based design equation, written for species j,
is given by Eq. 4.2.1,
dN
j
dt
¼ (r
j
)V
R
(t)(4:3:1)
Using stoichiometric relation Eq. 2.3.3, N
j
(t) is expressed in terms of the extents of
the independent reactions by
N
j
(t) ¼ N
j
(0) þ
X
n
I
m
(s
j
)
m
X
m
(t)
4.3 REACTION-BASED DESIGN EQUATIONS 107

which, upon differentiation with time, becomes
dN
j
dt
¼
X
n
I
m
(s
j
)
m
dX
m
dt
(4:3:2)
Using Eq. 3.2.6 to express the formation rate of species j in terms of the rates of the
chemical reactions, we obtain
(r
j
) ¼
X
n
R
i
(s
j
)
i
r
i
(4:3:3)
Substituting Eqs. 4.3.2 and 4.3.3 into Eq. 4.3.1, the design equation becomes
X
n
I
m
(s
j
)
m
dX
m
dt
¼
X
n
R
i
(s
j
)
i
r
i
V
R
(t)(4:3:4)
Note that the summation on the left-hand side of Eq. 4.3.4 is over independent reac-
tions only, whereas the summation on the right-hand side is over all reactions that
take place in the reactor. Using Eq. 2.4.8, we write the right-hand side of Eq. 4.3.4
as a sum of dependent reactions and independent reactions,
X
n
I
m
(s
j
)
m
dX
m
dt
¼
X
n
I
m
(s
j
)
m
r
m
þ
X
n
D
k
(s
j
)
k
r
k
!
V
R
(t)(4:3:5)
The stoichiometric coefficient of species j in the kth-dependent reaction, (s
j
)
k
,
relates to the stoichiometric coefficients of species j in the independent reactions,
(s
j
)
m
, by Eq. 2.4.9,
X
n
I
m
a
km
(s
j
)
m
¼ (s
j
)
k
where a
km
is the multiplier of the mth-independent reaction to obtain the kth-
dependent reaction. Substituting this relation into Eq. 4.3.5, we obtain
X
n
I
m
(s
j
)
m
dX
m
dt
¼
X
n
I
m
(s
j
)
m
r
m
þ
X
n
D
k
X
n
I
m
a
km
(s
j
)
m
r
k
"#
V
R
(t)(4:3:6)
Since all multipliers a
km
are constants, we can switch the order of the two
summations in the second term in the parentheses and obtain
X
n
I
m
(s
j
)
m
dX
m
dt
¼
X
n
I
m
(s
j
)
m
r
m
þ
X
n
I
m
(s
j
)
m
X
n
D
k
a
km
r
k
"#
V
R
(t)(4:3:7)
Since the coefficients and summations of all the terms are identical, Eq. 4.3.7
reduces to
dX
m
dt
¼ r
m
þ
X
n
D
k
a
km
r
k
!
V
R
(t)(4:3:8)
108 SPECIES BALANCES AND DESIGN EQUATIONS
Equation 4.3.8 is the reaction-based, differential design equation of an ideal batch
reactor, written for the mth-independent reaction. As will be discussed below, to
describe the operation of a reactor with multiple chemical reactions, we have to
write Eq. 4.3.8 for each of the independent reactions. Note that the reaction-
based design equation is invariant of the specific species used in the derivation.
For an ideal batch reactor with a single chemical reaction, Eq. 4.3.8 reduces to
dX
dt
¼ rV
R
(t)(4:3:9)
When a single chemical reaction takes place, the extent of the reaction is pro-
portional to the conversion of reactant A, f
A
, given by Eq. 2.6.2:
f
A
(t) ¼
s
A
N
A
(0)
X(t)
where N
A
(0) is the mole content of reactant A initially in the reactor. Hence, we can
readily express the design equation in terms of the conversion of the limiting reac-
tant. We differentiate Eq. 2.6.2 and obtain
dX
dt
¼
N
A
(0)
s
A
df
A
dt
Also, using Eq. 3.2.5, the depletion rate of reactant A, (2r
A
), relates to the reaction
rate by
r
A
¼s
A
r (4:3:10)
Substituting these two relations into Eq. 4.3.9, we obtain
N
A
(0)
df
A
dt
¼ (r
A
)V
R
(t)
which is identical to Eq. 4.2.4, the differential, species-based design equations of
an ideal batch reactor with a single chemical reaction, expressed in terms of the
conversion of reactant A.
4.3.2 Plug-Flow Reactor
For a differential steady-flow reactor, the species-based design equation, written for
species j, is given by Eq. 4.2.10:
dF
j
¼ (r
j
) dV
R
(4:3:11)
We use Eq. 2.3.11 to express the local molar flow rate of species j, F
j
, in terms of
extents of the independent reactions:
F
j
¼ F
j
0
þ
X
n
I
m
(s
j
)
m
_
X
m
(4:3:12)
4.3 REACTION-BASED DESIGN EQUATIONS 109
