Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Hartree and Hartree-Fock Equations 243
that "when the electrons oscillated, the positive ions behave like a rigid jelly with
uniform density of positive charge ne." Jellium is just a collection of electrons, into
which ions are introduced as a spatially uniform background to maintain overall
charge neutrality. The Hartree-Fock equations are in this case solved by plane
waves. For N electrons in a volume V they take the form
Kinetic energy Interaction with ions
—h
2
V
2
N f e
2
2m V J \r—r2\
+Φι
(7)
f
dr
2
E
iJMif-
- £
δ
ΧιΧ
^(7)
f
dn
Coulomb interaction Exchange interaction
Insert a plane wave of the form
„iki-r
e
V
(9.39)
(9.40)
The allowable set of k's as in Section 6.3 is given by imposing periodic boundary
conditions on the system, producing the usual conclusion that the density of k states
per volume and per spin is
1/(2π)
3
.
The kinetic energy term in Eq. (9.39) produces
-^Φι^)-
(9.41)
The interaction with the ions and the Coulomb interaction cancel against each other
because \φ]{τ2)\
2
— 1/"V. The only calculation remaining is the exchange interac-
tion, which becomes
e
J^
e*J
7
f dfi
e'^i-^'
72
;
=1
VV J V \r-r
2
= e\
N / S^ _, Changing the variable of integration to (9 43)
4^
J V r'
XhXi
r' = r
2
-r.
V
' '
7=1
N
=
β
2
φι
y^ — — =i δ
χι χ
■
The Fourier transform of 1/r is Α-κ/k
2
. (9.44)
j=x
V\k-kj\
2
/
k
F
SÎ,
Λ
__
Assume that states are occupied up to a Fermi
wave vector and turn the sum over j into an (Q
Λ
C\
(27r)3 1,2 I I,2 _ yi, Ί, integral. However, the restriction that χι =
\ ) K
l
-f K ΔΚ- Kj
cuts in
half
the
density of states in Section
6.3.
= e φι (r) ——- ί k
F
— kf J In < — > + 2kikf . Write k-k,= kk, cos
Θ,
integrate
2νΓ
*/ L
v
' ykp-ki) J
firstdcos0ythenk
2
dk
(9.46)

244
Chapter 9. Electron-Electron Interactions
So plane waves solve the Hartree-Fock equations, and the energy of state / is
(9.47)
vP-iP- 1P
2
E
l
= ^-—k
F
F(k
l
/k
F
),
2m π
where the Lindhard dielectric function F(x) is
1
F(x)
4x
('-*>{^H
(9.48)
This expression has certain peculiarities. In particular, dE/dk is infinite at the
Fermi surface, which means that the group velocity of electrons at the Fermi sur-
face should be infinite, a conclusion that would have catastrophic consequences
for transport properties were it not both unphysical and incorrect. The source of
the difficulty is visible in Eq. (9.44); it arises from the divergence when ki
—>
kj,
which in turn arises from the very slow decay at long distances of the Coulomb
interaction
1
/1r
—
r'|. An effect that the Hartree-Fock approximation has missed is
screening. The effective interaction of two distant electrons actually falls off much
faster than \/\r
—
7*21
because the many electrons in between them adjust their po-
sitions to hide the distant electrons from one another, but Hartree-Fock misses this
fact because the nature of the approximation leads it to treat only two electrons at
a time. Screening is the subject of Problem 3 and is reviewed by Echenique et al.
(1990).
The energy of a collection of independent electrons is given by a sum of the
one-particle energies. This simple relation does not hold for the energies £/ of
Hartree-Fock theory. The sum of all £; counts terms resulting from the Coulomb
and exchange interactions twice. The correct expression for the total energy is
therefore, as shown in Problem 2,
ε
=
Σ
tfk]
2m
-k
F
F
7Γ
F from Eq. (9.48).
(9.49)
-N
!*-4
3A
f
7Γ
The integration is straightforward, although (9.50)
care needs to be taken as k
—>
kp.
9.3 Density Functional Theory
While in the end it will produce formulae that look a great deal like Hartree-Fock,
density functional theory has a rather different flavor. In principle, it is an exact
account of the many-body wave-function, while in practice it spurs on varied types
of daring approximations.
The starting point of the theory is the observation of Hohenberg and Kohn
(1964) that electron density contains in principle all the information contained in
a many-electron wave function. The electronic density of a many-electron system

Density Functional Theory
245
at point 7 is defined
to be
N
n(r) =
(^\J2ô(r-R
l
)\^)
(9.51)
=
N
J
d?\...
dr
N
**(r
u
h . . .
ΓΛ,)5(Γ-Γ,)Φ(ΓΙ
. . .
r
N
).
(9.52)
Hohenberg
and
Kohn pointed
out
that
if
one knows
the
density
of
the ground state
of
a
many-electron system,
one can
deduce from
it the
external potential
in
which
the electrons reside,
up to an
overall constant.
It
must
be
kept
in
mind that the only
ways
in
which two many-electron problems
can
differ are
in the
external potentials
U
and in the
number
of
electrons that reside
in the
potentials. According
to
this
result, both
of
these external parameters
are
determined
by the
electron density,
so
one
can say
that
the
density completely determines
the
many-body problem. This
statement
is
surprising, because
the
density
is a
real function
of a
single spatial
variable, while the complete quantum mechanical wave function needs
N
variables
for
its
description.
To prove
the
claim, suppose that
it is
false. Suppose that there exist
two ex-
ternal potentials
U\ (?)
and
t/
2
(?)
that result
in the
same charge density. Call
the
Hamiltonians that result from them
3ΐι
and
Ä
2
,
and
let
the ground state wave func-
tions
for
the two Hamiltonians
be Φι and Φ
2
.
Assume that the ground states
of
the
two Hamiltonians
are
nondegenerate; this
is
a
technicality that will
be
avoided
completely
by an
improved version
of
the argument
to be
presented shortly. Then
the ground-state energy
of
"K\
is
realized only
by Φι. So
£ι
=(Φι|ίΚι|Ψι)
<
(Φ2ΙΉ1ΙΦ2) Because
Ψ
2
is not the
ground (9.53)
state
of
"K\.
=>
ε,
<
(Φ2|Ή
2
|Φ
2
>
+
<Φ
2
|(ΛΙ -Λ
2
)|Φ
2
)
(9.54)
=>
ε
ι
< £
2
+ / drnir)
\U\ (?)
-
U
2
(?)}
.
Two
Hamiltonians with
the
same (9.55)
J
number
of
electrons
can
differ only
in
the potential.
However,
one can
equally well switch indices
1
and 2 to
obtain
£
2
<
£i +
j
d?
n(?)
[U
2
(?)
-
U\ (?)]
.
(9.56)
Adding Eqs. (9.55)
and
(9.56) gives
ει + ε
2
<ε
1
+ ε
2
, (9.57)
which
is a
contradiction. Therefore
the
potentials U\
and i/
2
must
be the
same.
As
a
result
of
these observations, one can imagine being handed many different
solid systems, told that they consist
of
electrons interacting with
one
another
via
Coulomb potentials, moving
in
potential U, and obeying Schrödinger's equation.
If
in each case one
is
given the charge density, as
a
function
of
space,
then
in
principle
one
can
deduce
U and
solve
for all
properties
of
the system. Thus
one can
think
of

246
Chapter 9. Electron-Electron Interactions
the ground-state energy £, kinetic energy T, and so on as being functionals of the
density, and can write the following, to indicate this fact:
Here n really means n(r), a function of space,
ε\η] = Τ\n] +
U\n}
+
U
ee
In].
T
'?
the
^c
ener
gy*v
is
,
the
P°
tentialdue
(9.58)
L
'
L J L J ee
L
' to ions, and
Uee
is the Coulomb interaction
v
'
between electrons.
Hohenberg and Kohn next observed that if one can find the functional
£
[n],
then the true ground-state density n{r) minimizes it, subject only to the constraint
that
/
dr n(r) = N. (9.59)
This assertion is proved by noting that if
one
starts with the "wrong" density,
AÎ2,
for
Hamiltonian Ή\, then «2 should really be associated with a different Hamiltonian
Ä2,
which has ground-state wave function Φ2 and which does not minimize
<φ
2
|:Κι|Φ2> = ει[«2]. (9.60)
Only n\ minimizes
£1
[n{\,
and this
is
just what needed to be shown.
The most intriguing feature of this view of the many-body problem is that one
can write the energy functional £ as
E[n]
= j drn(r)U(r)+F
HK
[n}, (9.61)
where
FHK
is the sum of kinetic and Coulomb energies:
F
H
K[n}
=
T[n]
+
U
ee
[n}.
(9.62)
The functional
FHK
does not depend upon the potential U(r), and so it constitutes
a universal functional for all systems of N particles; if one only could find this
functional, it would solve all many-body problems for all external potentials U.
Second Derivation of Energy Functional. It is desirable to have a second demon-
stration of the existence of the energy functional £ for two reasons. First, the
demonstration given so far relies crucially upon assuming that ground states are
nondegenerate. It is worth knowing whether this requirement is a dull technicality
or a fatal flaw. Second, it might be possible to find a charge density n(r) that cannot
result from any conceivable potential U(r). In this case, the functional
£[n]
is not
yet even properly defined. Both these points are answered by defining a functional
F[n]
that is the minimum over all wave functions producing density n(r):
F[n]
= min (*|r +
t/
ee
|*).
(9.63)
This functional F can be defined even if there does not exist a potential U that
would produce density n for some quantum-mechanical ground state. The process

Density Functional Theory
247
of finding the ground state £o of a many-body system may therefore be carried out
in the following way:
£
0
= min(*|7
,
+ £/ + t/ee|1') (9.64)
Φ Minimize over all wave functions
•
r ·
/,τ,ΐτ
i H i H
Ι,ΤΛΙ
Ψ that produce density n, and
/ri
^c\
min[min(*|r + i7 + i/ee|*)J
then minimize over all densities. (9-65)
πιΐη(Φ|7' + £/
β
β|Φ)
+ [
U(r)n(r)d.
f>]
+ [
U{r)n(r)dr
n
ψ^„
min
Because the potential U depends (9.66)
only upon the density.
mm
n
(9.67)
= mia E\n]. (9.68)
The problems
of
defining
£
in general and accommodating degenerate ground
states are therefore solved simultaneously.
In principle, what has been accomplished here is enormous. In principle, there
exists a universal functional
F[n]
that needs to be found once and for all. One adds
to it any particular set of
nuclei,
in the form of
the
potential £/(?), and then has only
to find the function n(r) that minimizes it in order to solve the full complexities of
Schrödinger's equation. As is often the case, the gap between accomplishments in
principle and in practice is also enormous. The functional F is a magical lookup ta-
ble that is supposed to solve all of quantum mechanics upon request. No one knows
the true F, and no one ever will, so it is replaced by various uncontrolled approx-
imations. The decades-long process of discarding approximations that disagree
with experiment and improving on the ones that are more successful has encoded
a great deal of accumulated physical insight within apparently simple functional
forms.
9.3.1 Thomas-Fermi Theory
The simplest approximation providing an explicit form for the functionals F
[n]
or
£[n]
is Thomas-Fermi theory. The basic idea of the theory is to find the energy of
electrons in a spatially uniform potential as a function of density. Then one uses
this function of the density locally even when the electrons are in the presence of
an external potential.
The problem of electrons interacting by
a
Coulomb interaction in
a
uniform
background (jellium) is unfortunately not possible to solve exactly, except in the
limit of high density. It was solved approximately with the Hartree-Fock approxi-
mation in Section 9.2.4. The strategy here will be to use the results of the Hartree-
Fock approximation to obtain an approximate account of any term in Schrödinger's
equation that cannot automatically be expressed in terms of density. The first term
of this sort is the kinetic energy; the kinetic energy of a single k state is H
2
k
2
/2m,
and one must sum over all available k, so that the total kinetic energy is
f
-*
H
2
k
2
T = V [dk\—
See Eq. (6.15) for the definition of
[dk]
(9.69)
J
2m

248
Chapter 9. Electron-Electron Interactions
=
V
^-=V— -(3ττψ\
5
/\ Using Eq. (6.29).
(9
.70)
2m5ir
z
2m
5
The electron-electron interaction results within the Hartree-Fock approxima-
tion
in two
terms.
The
first part,
the
classical piece,
is a
functional
of
electron
density automatically, because
it
equals
-
ί
dr
2
dr/
n
^
)n
}\
2)
.
(9.71)
2
J
\r\
-r
2
\
The second piece, the exchange term, was evaluated in Eq. (9.50) for jellium, where
it was shown to
be
3e
2
k
F
3
/3\'/
3
,
4/
,
—TV
= —V— — ) en.
J
ust
rewrite
Λ?
and
kp in
terms
of
density. (9.72)
4
π 4 \πJ
The idea
of
Thomas-Fermi theory
is to
assume that
in a
system where
the
charge density
is not
uniform,
but
varies slowly, the kinetic energy
and
exchange
term will
be
given
by
the same expressions given above, but evaluated locally and
integrated over all space. The kinetic energy
is
T[n]
=
jdr^
n
3
-{3n
2
)
2/3
n
5
/\r), (9.73)
and the exchange energy
is
i
xc
= - j
dr\ß\
ß
e
2
n^(r).
(9.74)
So finally
a
first explicit density functional
is
£[n}
= J-ï
(3π
2
)
2/3
ί
drn
5
l
3
{?)+
ί
drn{r)U(r)
+
' /
M
ipm -far 1β)
Φ
Mfiffl.
<«5)
2
J
\r\
-r
2
\ J 4 \ιτ
J
Actually, this functional constitutes Thomas-Fermi theory only
if
the last term,
due to exchange,
is
simply omitted; with the exchange term included, the theory
is
called Thomas-Fermi-Dirac.
The conventional Thomas-Fermi-Dirac equation
is
obtained by writing down
the condition that the functional (9.75) be minimized by n, subject
of
course to the
constraint that the integral
of
n
be N. One has
δε
u
The
chemical potential
μ
arises
as the La-
(9.76)
δη
Cf) grange multiplier enforcing the constraint that
density be conserved.
£
M
2/
\y\r)
+
U(r)+
[α7
2
^-(
3
-)
1/
\\^(7)
=
μ.
(9.77)
2m
V
/ J
\r
—
r
2
\
\πJ

Density Functional Theory 249
Omitting the last term on the left-hand side of
Eq.
(9.77) gives the Thomas-Fermi
equation.
Thomas-Fermi theory is simple, but not particularly accurate, so its solution
is relegated to Problem 4. One result of solving Eq. (9.77) is that the energy of
an atom of nuclear charge Z is approximately
—
1.5375Z
7
/
3
Ry. For small atoms,
this result is large by a factor of two when compared with Hartree-Fock; even for
an atom as large as Xe (Z = 54), where one has better hope for an approxima-
tion based on slowly varying charge distributions, the equation is still in error by
20%.
The Thomas-Fermi-Dirac equation gives energies that deviate even further
from reality. Another disturbing feature of the equations is that Thomas-Fermi
and Thomas-Fermi-Dirac equations predict that atoms never bind into molecules;
the energy of a supposed molecule is always lowered by pushing the nuclei further
apart. This subject is discussed by Lieb (1981).
Thomas-Fermi theory smooths out the charge distribution, because it has no
way to know that electrons arrange themselves into separate shells. Thomas-
Fermi-Dirac is even less physical; it predicts that at some finite radius the charge
distribution drops instantaneously to zero. There have been attempts to develop
improved theories of this type by bringing in dependence upon gradients of the
charge distribution. The original Thomas-Fermi theory is most accurate for nearly
uniform charge distributions, so it is natural to work out the corrections that would
occur for an electron gas in a linearly varying potential, a quadratically varying
potential, and so on, using these results to construct an expansion in terms of gra-
dients of the density. However, none of the theories of this type has gained wide
usage.
9.3.2 Stability of Matter
A different type of application of density functional theory is to address the ques-
tion of the stability of matter. That is, why does the attraction between electrons
and nuclei not lead to a collapse in which the electrons crowd in upon the nuclei,
producing solids with an atom every 10^
15
m rather than every 10~
10
m? On this
point there is no need even to turn to experiment; common experience says that
elements and compounds are stable. However, it is interesting to find the features
of quantum mechanics that make the obvious possible.
To the obvious fact corresponds an obvious answer; the Heisenberg uncertainty
principle forbids electrons to come too close to nuclei. The momentum of an elec-
tron confined within a box of radius a must scale as h/a, so the kinetic energy must
scale as h
2
/ma
2
. However, the potential energy to be gained by coming close to
the nucleus only scales as
—e
2
ja, so the kinetic energy term seems to win. While
this argument is essentially correct, it does not provide the formal tools needed to
show that matter is stable. The tools need to provide a good estimate of the kinetic
energy of electrons based upon their density, and the Heisenberg uncertainty prin-
ciple does not do a good job of it. The precise statement of Heisenberg's principle
250 Chapter 9. Electron-Electron Interactions
is that
/
drh
2
\V^Y
I
drr
2
\^\
2
t2
The root mean square momentum times the
y.
. root mean square fluctuation in position of /n ηο\
— 4 ' a wave function must have a product greater ^ ' '
than
fi/2; ψ is normalized, and is assumed to
be
centered at the origin.
it can be used to bound electron kinetic energies T through
T[n}>
h
l
%m
J dr r
2
n(r)
(9.79)
-$-
=^
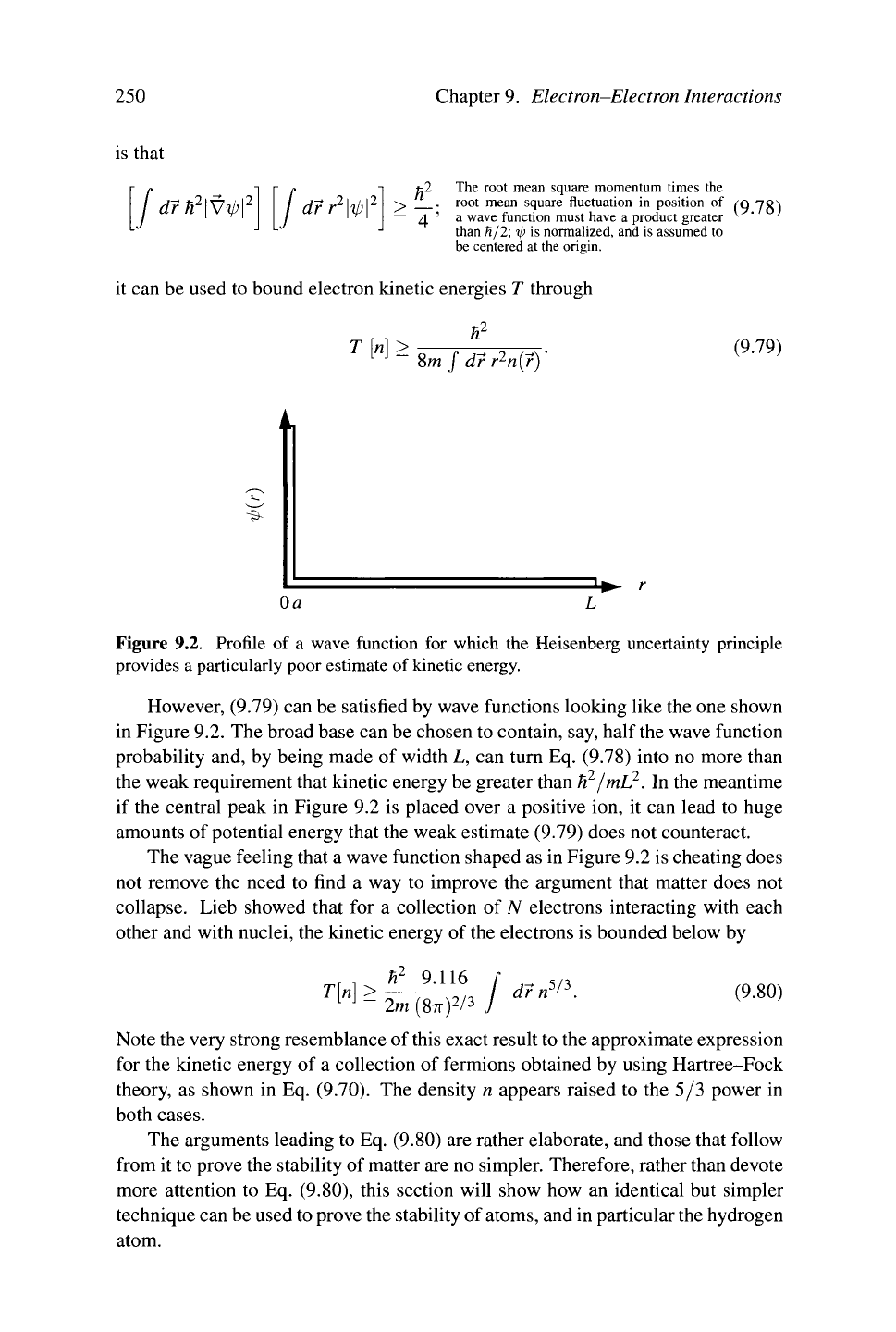
Figure 9.2. Profile of a wave function for which the Heisenberg uncertainty principle
provides a particularly poor estimate of
kinetic
energy.
However, (9.79) can be satisfied by wave functions looking like the one shown
in Figure 9.2. The broad base can be chosen to contain, say, half the wave function
probability and, by being made of width L, can turn Eq. (9.78) into no more than
the weak requirement that kinetic energy be greater than H
2
/mL
2
. In the meantime
if the central peak in Figure 9.2 is placed over a positive ion, it can lead to huge
amounts of potential energy that the weak estimate (9.79) does not counteract.
The vague feeling that a wave function shaped as in Figure 9.2 is cheating does
not remove the need to find a way to improve the argument that matter does not
collapse. Lieb showed that for a collection of yv electrons interacting with each
other and with nuclei, the kinetic energy of the electrons is bounded below by
T[n]>
W- 9.116
drn
5/3
.
(9.80)
2m (8TT)
2
/3
Note the very strong resemblance of
this
exact result to the approximate expression
for the kinetic energy of a collection of fermions obtained by using Hartree-Fock
theory, as shown in Eq. (9.70). The density n appears raised to the 5/3 power in
both cases.
The arguments leading to Eq. (9.80) are rather elaborate, and those that follow
from it to prove the stability of matter are no simpler. Therefore, rather than devote
more attention to Eq. (9.80), this section will show how an identical but simpler
technique can be used to prove the stability of
atoms,
and in particular the hydrogen
atom.

Density Functional Theory 251
As shown
in
Problem
6, the
kinetic energy
of
any wave function
h
2
r
T\n}
= — Jdr\V^\
2
(9.81)
obeys
the
inequality
where
T
\n]>^f*
j
drn
5
'\
(9.82)
ΛΓ
5
= 3(ΤΓ/2)
4
/
3
,
(9.83)
and with
the
density
n
given
by
η(7)
=
\ψ(7)\
2
.
(9.84)
Therefore,
for
the hydrogen atom,
the
energy
of
a wave function
ψ is
bounded
below
by
*-K, [
drn^-i
dr^à.
(9.85)
2m
J J r
The variational equation that
can be
used
to
minimize this functional subject
to the
constraint
λ(1-
f
drn)=0
(9.86)
is
s2
5
h
K_
n I (7)
—
g /r + λ = 0
Here
λ is a
Lagrange multiplier enforcing the
(9 87)
3
2m '
constraint
(9.86).
There
is
also
the
constraint that
n has to be
positive.
One way to
enforce this
constraint
is to
define
n = σ
2
and
carry
out
variations with respect
to σ. One
for-
mally finds
in
this
way
that
n = 0 is an
acceptable local minimum
of
the functional
Eq. (9.87);
of
course
one
can't have
n
—
0
everywhere
or
Eq. (9.86)
is
violated,
but
instead
n{7)=
i{6m[e
2
/r-X}/(5K
s
h
2
)}
3/2
forr<e
2
/X
(g gg)
[
0
else
One fixes
A
by
requiring
the
integral
of
n
to be 1, and one
finds that
3me
4
.
7Γ
4
.
i /-,
λ
=
5ΡΕ
(
Τ>
,/3:
<989)
inserting this form
of
n
into Eq. (9.85) gives
a
lower bound
to the
energy
of
9me
4
, -Ko«
6me
4
12„
-
(2ττ
2
)
2
/
3
= --— = - -Ry.
(9.90)
\0h
2
K
s
5 h
2
5
The correct answer
is of
course that
the
ground-state energy
is
— 1
Ry, so the
esti-
mate
is not so bad.

252 Chapter 9. Electron-Electron Interactions
In this way, without explicit solutions of Schrödinger's equation, one can place
limits upon the total energy to be gained through compression in the presence of
Coulomb forces. Similar arguments prove the stability of matter using Eq. (9.80).
What is really crucial is the fact that since one is considering an assembly of elec-
trons,
the Pauli exclusion principle forbids them from occupying a single lowest-
energy state together. As a result, the kinetic energy penalty for compacting elec-
trons grows as w
5
/
3
rather than just as n. The arguments by Lieb (1976) and Lieb
et al. (1997) showing that many-electron systems are stable are rather elaborate,
but the basic flavor is contained in use of the inequality for the very simple case of
the hydrogen atom, and the end result is the same; matter does not implode because
it would cost too much in kinetic energy.
9.4 Quantum Monte Carlo
9.4.1 Integrals by Monte Carlo
Despite the intrinsic difficulties of solving Schrödinger's equation for many parti-
cles,
there has long been a tenacious effort to solve it with as few approximations
as possible. The most impressive progress has come from quantum Monte Carlo
methods. The starting point is the Monte Carlo method (Section 5.4.1), now ap-
plied to the computation of integrals in large-dimensional spaces.
The Monte Carlo idea can be used to compute the average of any function in a
probability distribution. Consider a function g(x) where x may be a vector in a n-
dimensional space. Suppose that the probability of being at point x is iP(x), where
to be a legitimate probability distribution
3>(x)
> 0; / d
n
x ?(x) = 1. (9.91)
The average of g is
g =
Jd
n
xg(x)nï)-
(9-92)
To compute this average, follow the same steps as in Section 5.4.1
:
1.
Pick a starting value, xo, and compute
go
= g(xo)-
2.
Pick a random vector Δχ, all of whose components range between — e and e,
where e is some constant.
3.
Compute 7
(xo
+ Ax).
(a) If 7(xo + Ax?)/!P(jeo) >
1
then accept the move: 2\
—
XQ
+ Δχ.
(b) If ^(fo +
AX)/7(XQ) < 1, choose a random number p between 0 and 1.
i. If
iP(xo
+
Ax)/CP(Jco)
< p, then again accept the move so that x\
=XQ
+
AX.
ii.
Otherwise, reject the move and take 1\ =
XQ.
