Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Vibrations
of a
Quantum—Mechanical
Lattice
353
|>.
Jï-R'
,
5t „-i*-J?l „,
;Λ
â. _
;Α
ηΜω
Ιν
î*
=
±=T\he
ik
*+P\e-
&
*
VN
^ ί
ku
k
"
^*^
= -'ν-2
^^·
(13
-
43b)
*!/
From the commutation relation [Ρ', Λ']
=
—ifô, one finds for the creation and
annihilation operators the commutation relation
\ä
lv
J
lv
\
= \. (13.44)
In moving between Eqs. (13.42) and (13.43), the frequencies
UJ^
V
and the unit
vectors e-^ may in principle be chosen arbitrarily, just so long as the three unit
vectors e^ at every value of k are orthonormal. However, the only choice one
would sensibly make for these quantities is the one that diagonalizes the Hamil-
tonian (13.41), which means choosing
e-r.
to be the eigenvectors of the dynamical
matrix Φ and choosing
ω^
to be related to the eigenvalues by Eq. (13.18). Also
notice from Eq. (13.17) that Φ(ϋ) =
Φ(—k),
because one is related to the other just
by changing order of summation, and therefore
<*„
=
"-*»■
(13.45)
Making use of
Eq.
(13.45) gives
Σ
Ρ
1
τ-^
hcor
I
[âr
al
+ âl âr 1
I
£_
=
y-_^
J
L ku
kv kv
kui
I
(1346a)
2M
^ 4 Ì -[âr âr er ·€ r +4 âl S
· e*
-.
] f
U
^°
d
'
/
ku y y ku
kv ku
-ku
ku ku ku -ku
1
)
Σ
,
,,, „
,—. Hutr
I [âr âl +âl âr
1
I
ίΰ
ι
Φ"ΰ'=Υ—^<
\
k
» ku
ku ku\ i.(13.46b)
.„
2
4^ 4 |
+ [âr
âr
er
·? r
+ âl ô!
£
■
e* -
f
//'
*„ (
<■
kv
ku ku
-ku
ku ku ku -ku'
)
In order to arrive at Eqs. (13.46), one employs the facts that eï
·
e-^, = δ
νν
' and that
e^
·
~e_~
kv
,
vanishes unless v = v'. Although one has the freedom in defining e^
v
to request that ë|
—
e_-
ku
such a choice is very confusing for longitudinal modes,
where ë3
=
—e
7
is more natural, so no relation between ei and e
7
will be
ku
-ku ku -kv
assumed.
Summing Eqs. (13.46a) and (13.46b) gives for the Hamiltonian of (13.41) that
A = E^M,
+
4ÂJ = EH,(«L^ +
5)·
UseE
^
13
-
44
>· (13.47)
ku
ku
For a lattice with a basis, Eq. (13.47) still holds true, but the summation ac-
quires an additional index corresponding to the branch of the phonon mode, which
ranges over the number of atoms in each unit
cell.
If the number of indices becomes
too cumbersome, one can adopt the abbreviated notation
5ΐ
=
2_^ ^W/(«
(
-+ 2)· The number operator
«,
=â/â;. (13.48)
where the sum over reciprocal vectors k, polarizations i/, and modes is subsumed
into the single index i.

354
Chapter 13. Phonons
Time Evolution. In the study
of
neutron scattering,
it
will also be necessary
to
know how the operators
â
and
û
evolve
in
time.
In
the Heisenberg picture,
the
annihilation operators evolve according to
â
lv
{t) = ê™l
h
â
lv
e~
i
™l
h
(13.49)
so that
^Ι^
=
j™/*i[ÔÎ,
â^e-^'K/n
(13.50)
^à
iv
(t)
=
â
iu
e-
i
^
t
.
(13.52)
As for the time evolution of û
l
, Eq. (13.43a) generalizes to
"'(0
=
j=
Σ^/^'^'
+
ûle-
iU
'
+
'^}.
(13.53)
13.3.1 Phonon Specific Heat
The specific heat of solids was one of the "19th century clouds" over 19th-century
physics described by Kelvin (1904). Solid objects were clearly built by connect-
ing small massive objects together with some form
of
spring. According
to
the
equipartition theorem
of
statistical mechanics, the specific heat must be
ke/2
per
degree
of
freedom. Each mass point must have three kinetic and three potential
degrees of freedom, and therefore a specific heat of Cy = 3Nke, the law of Dulong
and Petit. Not many data were available to test this law thoroughly, but by 1907,
specific heat had been measured down
to
around 50
K in
diamond. Instead
of
remaining constant, as shown in Figure 13.8,
it
declines precipitously at low tem-
peratures. For some reason, oscillators within the solid refused to take in energy at
low temperatures.
Einstein conceived the remarkable hypothesis that one might employ Planck's
radiation law, which stated that for black-body radiation the probability of exciting
a mode of frequency ω varied as
1
/(exp(hßu>)
—
1). This formula was still purely
empirical,
so
there was not
as
yet the possibility
of
deriving the specific heat of
solids from firm underlying principles. However, one could guess that
if
the solid
were built of N oscillators of frequency
ωο,
then the mean energy at temperature T
would be given by
3ΝΗωο
β
Ηβω
0
_ ]
(13.54)
d£
_
3N(hLü
0
)
2
e
Hßu}
o/{k
B
T
2
ßf
lv-
ighßwo
_
112
C
V=T^
lv= rSI TT5 ■
(13.55)
A particular strength
of
this formula was the fact that one was not condemned
to
fit
ωο from the specific heat data alone.
If
the solid were truly built
of
oscil-
lators concentrated
at a
certain frequency, then
it
should absorb radiation
at
this
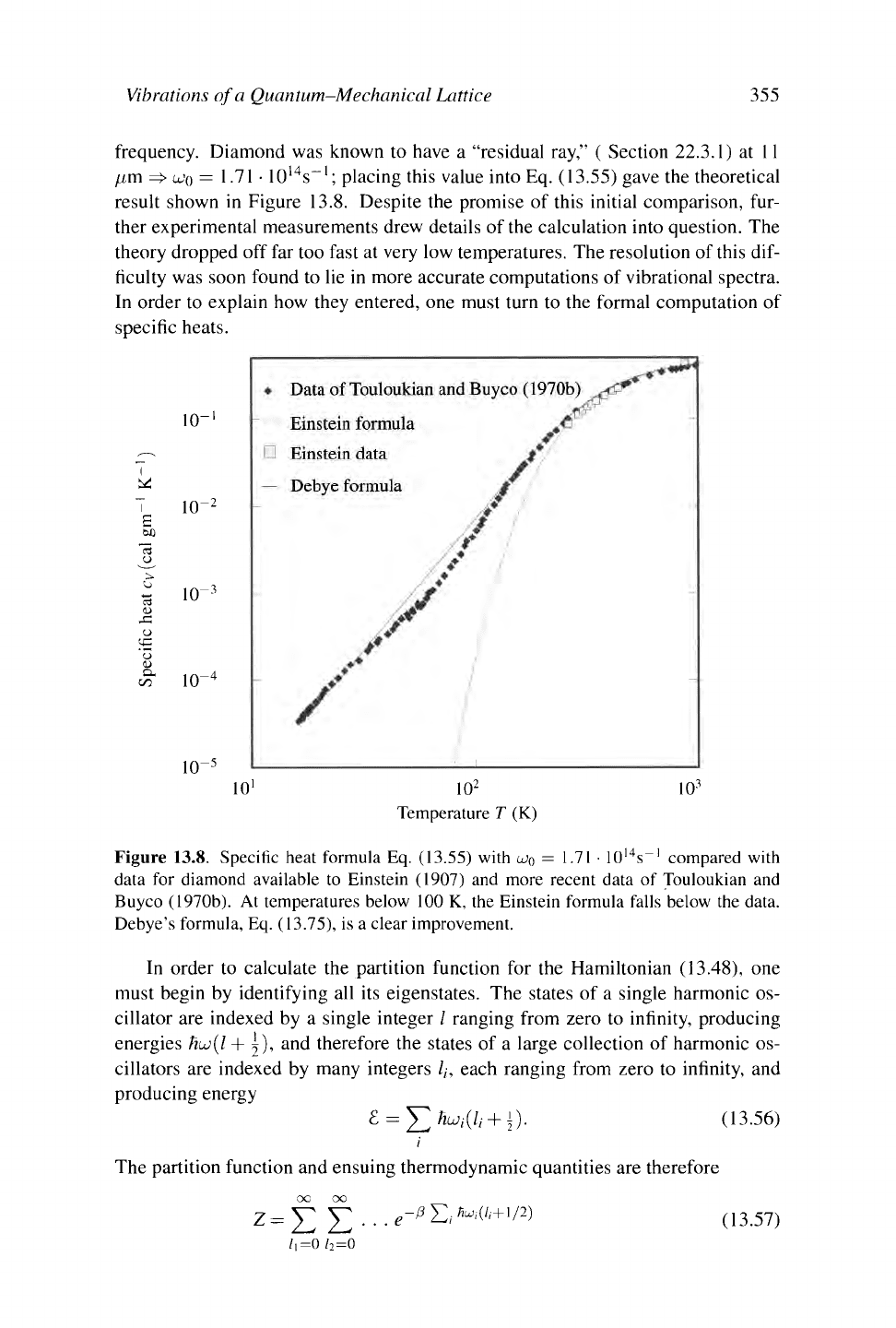
Vibrations of a Quantum-Mechanical Lattice 355
frequency. Diamond was known to have a "residual ray," ( Section 22.3.1) at 11
μνη
=>
ωο = 1.71 · 10
14
s"'
;
placing this value into Eq. (13.55) gave the theoretical
result shown in Figure 13.8. Despite the promise of this initial comparison, fur-
ther experimental measurements drew details of the calculation into question. The
theory dropped off far too fast at very low temperatures. The resolution of this
dif-
ficulty was soon found to lie in more accurate computations of vibrational spectra.
In order to explain how they entered, one must turn to the formal computation of
specific heats.
IO"
1
io-
2
io-
3
io-
4
io-
5
io
1
io
2
io
3
Temperature T (K)
Figure 13.8. Specific heat formula Eq. (13.55) with
U>Q
= 1.71 · 10
l4
s~' compared with
data for diamond available to Einstein (1907) and more recent data of Touloukian and
Buyco (1970b). At temperatures below 100 K, the Einstein formula falls below the data.
Debye's formula, Eq. (13.75), is a clear improvement.
In order to calculate the partition function for the Hamiltonian (13.48), one
must begin by identifying all its eigenstates. The states of a single harmonic os-
cillator are indexed by a single integer / ranging from zero to infinity, producing
energies
Ηω(1
+ |), and therefore the states of a large collection of harmonic os-
cillators are indexed by many integers
/,·,
each ranging from zero to infinity, and
producing energy
£ = ÇAU;
/
(/
/
+ I). (13.56)
The partition function and ensuing thermodynamic quantities are therefore
oc oo
Ζ
=Σ Σ···
β
~
βΣίΛωί{1ί+ι/2)
<
13
·
57
)
/l=0 /
2
=0
Ui
E
u
C3
o
Έ
'ΰ
Q.

356 Chapter 13. Phonons
OC I OO
=
Π Σ^^'
(/+
'
/2)
Π3.58)
ι=1
l/=0
J
= n(^
expW
} 03.59)
=>J=-yt
ß
rinZ = ^ ^+Α:
β
Γ1η(1-ί>-^
ω
') (13.60)
" ί' I
with 1
/ΐ
;
· = This expression defines the (13.62)
gßhwj _ ] Bose-Einstein factor m.
=►
c
v=§
iv =
Σ
G
=
Σ «^-
(13
·
63)
Phonon Density of States. Just as the density of electronic states plays a crucial
role in determining the thermal properties of the electron gas, similarly the density
of phonon states encodes the information needed to deduce thermal properties of
lattices. This density of states is defined by
' kv
υ
and one may use it, for example, to write
f°° d Hui
C
V
= V duDUjj)- s= . (13.65)
,/o
y
' dT
β?
ηω
- 1
Before presenting the simple models frequently used to represent densities of
states and thereby extract thermodynamic quantities, it is best to present some ac-
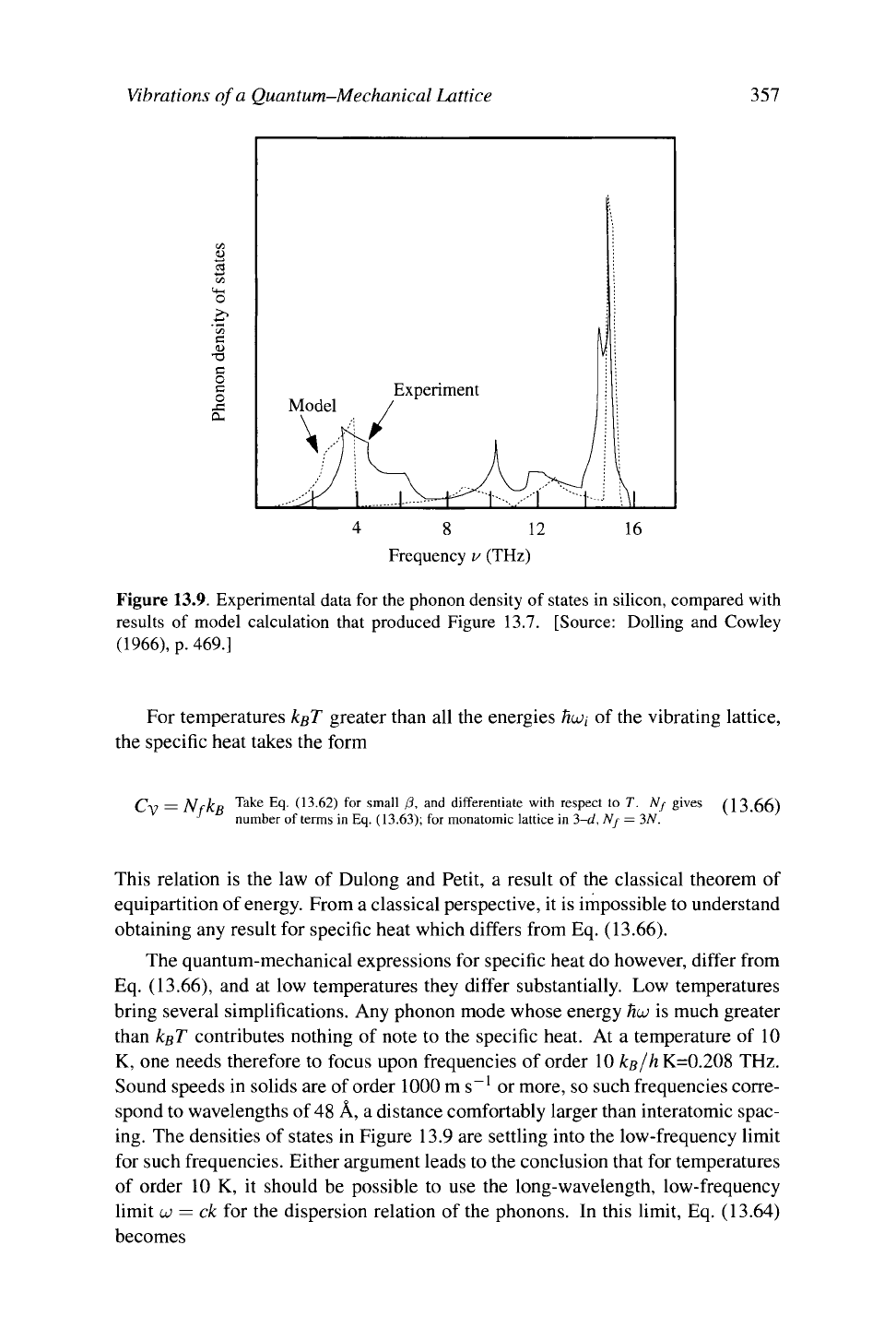
tual data derived from experiment. Figure 13.9 presents representative data for
silicon.
Two qualitative lessons are apparent in these data.
1.
The characteristic upper frequency at which the phonon modes terminate is
16 teraherz; this is the characteristic frequency of the optical modes and is the
upper limit of the acoustic modes.
2.
The densities of states are littered with cusps. These cusps are precisely the
van Hove singularities discussed in Section 7.2.5. They arise at any point in
frequency space where one of the phonon modes passes through an extremal
value. In three-dimensional solids the result is a cusp, in two-dimensional
solids it is a logarithmic divergence, and for one-dimensional chains it is a
square root divergence.
Vibrations
of a Quantum-Mechanical Lattice 357
V3
u
id
ΙΛ
4-H
O
*—>
C
Ό
C
o
c
o
0-
f
1
Experiment
Model /
..-■■y
\
.A-^K/i'Si
4 8 12
Frequency ^ (THz)
|
I
16
Figure 13.9. Experimental data for the phonon density of states in silicon, compared with
results of model calculation that produced Figure 13.7. [Source: Dolling and Cowley
(1966),
p. 469.]
For temperatures kßT greater than all the energies /ϊω, of the vibrating lattice,
the specific heat takes the form
C-O = NflCR Take ^Ί- (13.62) for small β, and differentiate with respect to T. Nf gives M 3 foß\
number of terms in Eq. (13.63); for monatomic lattice in 3-d, Nf = 3/V.
This relation is the law of Dulong and Petit, a result of the classical theorem of
equipartition of
energy.
From a classical perspective, it is impossible to understand
obtaining any result for specific heat which differs from Eq. (13.66).
The quantum-mechanical expressions for specific heat do however, differ from
Eq. (13.66), and at low temperatures they differ substantially. Low temperatures
bring several simplifications. Any phonon mode whose energy
Ηω
is much greater
than kgT contributes nothing of note to the specific heat. At a temperature of 10
K, one needs therefore to focus upon frequencies of order 10 kß/h K=0.208 THz.
Sound speeds in solids are of order 1000 m s
-1
or more, so such frequencies corre-
spond to wavelengths of
48
Â, a distance comfortably larger than interatomic spac-
ing. The densities of states in Figure 13.9 are settling into the low-frequency limit
for such frequencies. Either argument leads to the conclusion that for temperatures
of order 10 K, it should be possible to use the long-wavelength, low-frequency
limit ω = ck for the dispersion relation of the phonons. In this limit, Eq. (13.64)
becomes
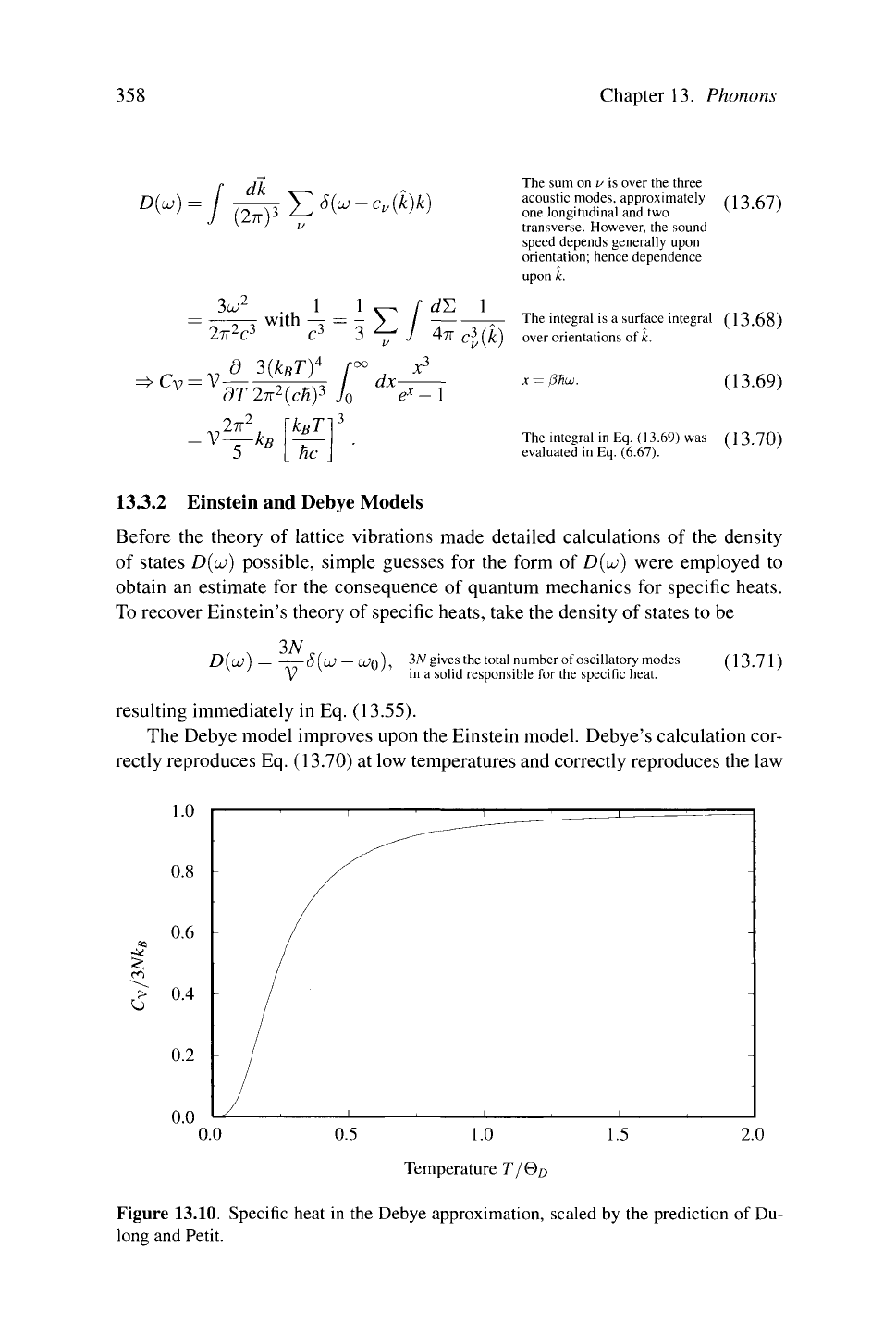
358 Chapter 13. Phonoris
DM
3ω
2
2π
2
ϋ
3
with
C
V
= V
d 3(k
B
T)
4
dT2n
2
(cH)
3
k
B
T
V—k
B
he
Σ/
1/
J
ί/Σ 1
dx-
The sum on u is over the three
acoustic modes, approximately /1
-3
f.n\
one longitudinal and two ^ ' '
transverse. However, the sound
speed depends generally upon
orientation; hence dependence
upon k.
The integral is a surface integral (13.68)
over orientations of k.
x =
β%ω.
(13.69)
The integral in Eq. ( 13.69) was (13.70)
evaluated in Eq. (6.67).
13.3.2 Einstein and Debye Models
Before the theory of lattice vibrations made detailed calculations of the density
of states D(uj) possible, simple guesses for the form of Ό{ω) were employed to
obtain an estimate for the consequence of quantum mechanics for specific heats.
To recover Einstein's theory of specific heats, take the density of states to be
3iV
D(u)) = δ(ίϋ —
U>o),
3iV
gives the total number of oscillatory modes
V in a solid responsible for the specific heat.
(13.71)
resulting immediately in Eq. (13.55).
The Debye model improves upon the Einstein model. Debye's calculation cor-
rectly reproduces Eq. (13.70) at low temperatures and correctly reproduces the law
0.5 1.0 1.5
Temperature Τ/θο
2.0
Figure 13.10. Specific heat in the Debye approximation, scaled by the prediction of Du-
long and Petit.
Vibrations of a Quantum-Mechanical Lattice
359
150
"o
Ë
5
100
D
Sodium
o Silver
200
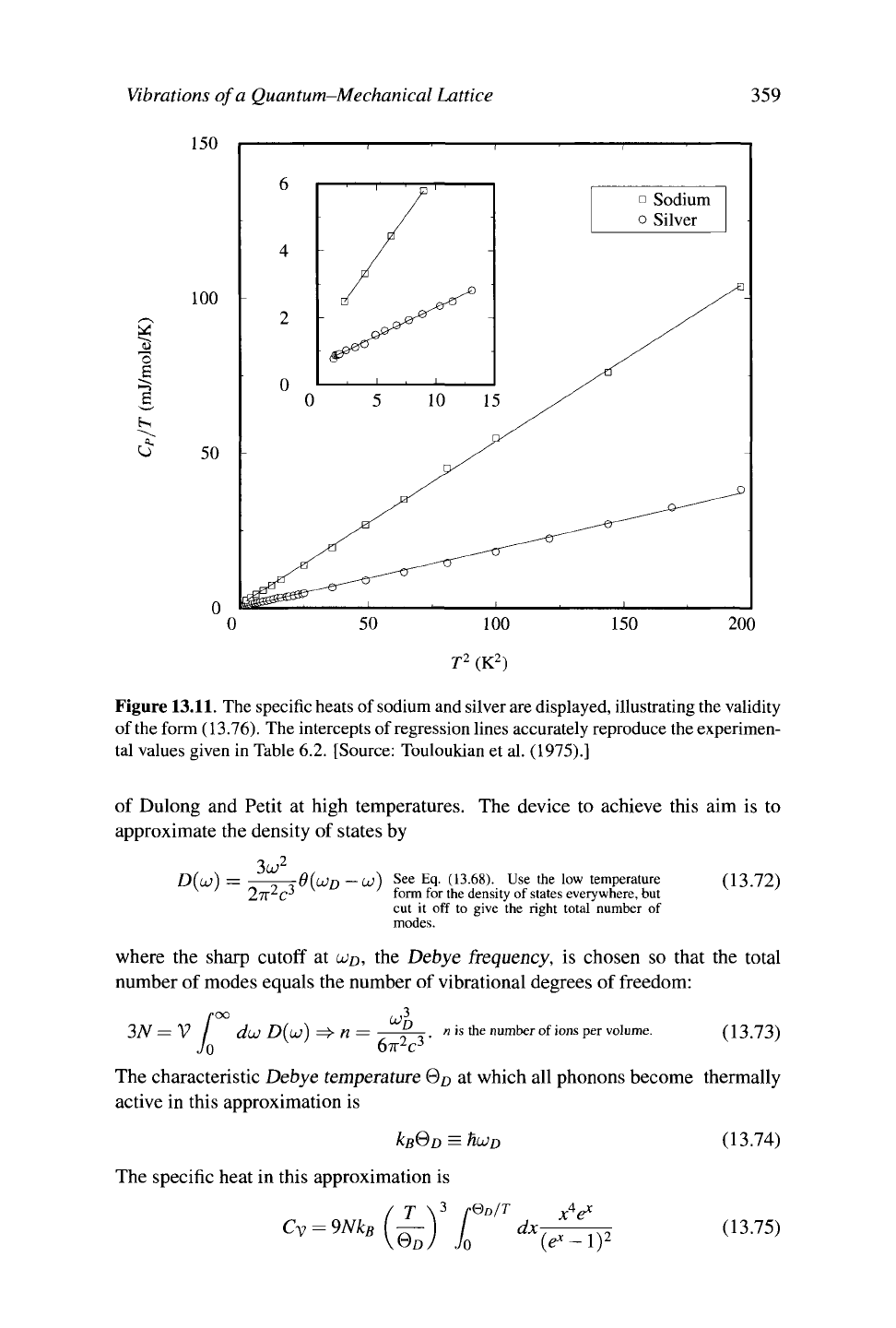
Figure
13.11.
The specific heats of sodium and silver are displayed, illustrating the validity
of
the
form (13.76). The intercepts of regression lines accurately reproduce the experimen-
tal values given in Table 6.2. [Source: Touloukian et al. (1975).]
of Dulong and Petit at high temperatures. The device to achieve this aim is to
approximate the density of states by
3a;
2
D(uj) = θ(ϋϋη — w) See Eq. (13.68). Use the low temperature
27r
2
C
J
form for the density of states everywhere, but
cut it off to give the right total number of
modes.
(13.72)
where the sharp cutoff at
UJD,
the Debye frequency, is chosen so that the total
number of modes equals the number of vibrational degrees of freedom:
roo
3N = V dujD{uj)^n-
Jo
Wr
67T
2
C
3
n is the number of ions per volume.
(13.73)
The characteristic Debye temperature Θο at which all phonons become thermally
active in this approximation is
k
B
®D = hu
D
The specific heat in this approximation is
T \
3
/-θο/Γ v-V
(13.74)
(13.75)

360 Chapter 13. Phonons
Table 13.1. Debye temperatures of elements
El.
Am
Ar
Ag
Al
As
Au
Ba
Be
Bi
B
C(gr)
C(dia)
Ca
Cd
Ce
Co
Cr
Cs
Cu
Dy
Er
e
D
121
92
227
433
282
162
111
1481
120
1480
412
2250
229
210
179
460
606
40.5
347
183
188
El.
Eu
Fe
Ga
Ge
Gd
H
He
Hf
Hg
Ho
I
In
Ir
K
Kr
La
Li
Lu
Mg
Mn
Mo
QD
118
All
325
373
182
122
34-108
252
72
190
109
112
420
91.1
71.9
150
344
183
403
409
423
El.
Na
Nb
Nd
Ne
Ni
Np
Os
Pa
Pb
Pd
Pr
Pt
Pu
Rb
Re
Rh
Ru
Sb
Sc
Se
Si
e
D
157
276
163
74.6
477
259
467
185
105
271
152
237
206
56.5
416
512
555
220
346
153
645
El.
0
D
Sm 169
Sn 199
Sr 147
Ta 245
Tb 176
Te 152
Th 160
Ti 420
TI 78.5
Tm 200
U 248
V 399
W 383
Xe 64.0
Y 248
Yb 118
Zn 329
Zr 290
Not available for all elements. Source: Stewart (1983).
and is plotted in Figures 13.8 and 13.10.
Because the low-temperature lattice contribution to the specific heat in
Eq. (13.70) varies as Γ
3
, it should disappear in metals beneath the electronic con-
tribution, which is linear in temperature. However, putting in numbers, one finds
that very low temperatures, on the order of a few degrees kelvin, are needed before
the electronic contribution stands out. The reason is that the natural temperature
scale for the electrons is the Fermi temperature, which is on the order of 20000
K, and the electronic contribution goes as T/Tf. However, the natural tempera-
ture scale for the phonons is the Debye temperature, given by Θο =
Hujo/kß-
The
contribution of the phonons to the specific heat goes as (Γ/θο)
3
. So the electron
and phonon contributions are roughly equal when T = θο
^Θο/Tf.
Debye tem-
peratures are tabulated in Table 13.1. Because they are typically on the order of a
few hundred degrees, the electronic specific heat has barely become visible even
at 10 K. Adding together electron and phonon contributions, the low temperature
specific heat at constant pressure
Cp
behaves quite accurately as
Cp ~ ^Τ + βΤ The constant 7 is the Sommerfeld parameter. ( 13.76)

Vibrations
of a Quantum-Mechanical Lattice 361
a claim best checked experimentally by plotting Cp/T versus T
2
, as in Figure
13.11.
Debye temperatures are usually 30% to 50% percent of the melting tempera-
ture of the element, so that by the time one gets to temperatures high enough to
see the fully classical specific heat of \kßT per degree of freedom, the harmonic
approximation for phonons is beginning to break down.
13.3.3 Thermal Expansion
Figure 13.12. Thermal expansion of
a
molecule.
The change of objects' size and shape as they heat and cool often poses an
unpleasant challenge for engineers. Road sections swell and buckle in the summer;
engine parts designed to work at high temperatures barely mesh when they are cool.
Yet it is a curious fact that the general framework allowing calculation of so many
other mechanical properties of condensed matter fails completely to predict the
possibility of thermal expansion. A solid whose energy changes only to quadratic
order when its atoms move does not change size or shape with temperature at all.
The reason for this somewhat unexpected result is most easily seen in the small-
est possible solid, one consisting only of two identical atoms (see Figure 13.12).
In a reference frame tied to their center of
mass,
the energy of the two atoms is
„ 1„ 2
Λ:
= «i
—
«2 is the difference of the deviation
t
=
~Ζ·^Χ · of the two atoms from equilibrium,
OC
some (13. / /)
•^ spring constant.
In any thermal average over the locations of the atoms, positive and negative
values of x occur with equal frequency, and the mean distance between the two
atoms does not change no matter how much the molecule may be heated. If there
were any solid in which atomic interactions were really only present to quadratic
order, it would similarly refuse to change size in response to temperature.
Suppose now that the interaction energy of the molecule is a general function
1 -,
ε(χ) = ε
0
+ -Χχ
2
+
.
. . (13.78)
and ask under what conditions the mean size x of the molecule changes. The ther-
mal average of x is
f dxe-W*) dA V J
A can be viewed as a purely
formal quantity, but also
can be interpreted as an (13.79)
A=0 external force acting on the
system.
/ dxe-P^x) dA
In /
dxe
Ax-ßS.(xa)-ߣ.'(xo)(x-xo)-ßZ"(xo)(x-xo)
2
/2\
A=Q
_
(13.80)
d_
dÄ

362
Chapter 13. Phonons
According to the technique of steepest descents (Appendix B.4),
XQ
should be cho-
sen so that the linear term inside the exponential in Eq. (13.80) vanishes;
XQ
is
determined by
A = ß E '
(XQ
)
= ß%XQ Maximizing the integrand; A can be (13.81)
taken very small because it is on the
verge of being set to zero, and
XQ
should be viewed as a function of
A.
d_
dA
ln
./^Zl_
e
A*ö-/3£(*,)
ßV'ixo)
k
B
T d
% dxç,
kßT δω
In W
2π
e
ßx4/2-0£o
Λ=Ο
(13.82)
,ο=ο (13.83)
U=o
with μω
2
(χ) =
E"(x).
ß is the reduced mass of the (13.84)
OCui dx ' molecule
The possibility of thermal expansion therefore rests upon a nonzero third derivative
of the energy £ about equilibrium, or equivalently upon the change of vibrational
frequency ω if the molecule is forced to expand or contract.
General Theory. The general theory of thermal expansion begins with a collection
of thermodynamic identities. One wishes to calculate
„,~ „ ,„ I An identity from thermodynamics concern-
er) I
θΓ/ΟΙ\·γ
ing partial derivatives; the minus sign is cor- ,..„->
*ΡΤ = O-JÏ \p ilo /imi rect. Do not confuse the coefficient of volume (,lJ.oJj
Ol -Or/OV\T expansion/3
r
with
\/k
B
T.
V d
2
3
= The bulk modulus B =
-VdP/dV.
(13 86)
so the formal aim can be achieved by calculating derivatives of the free energy
with respect to temperature and volume. Using Eq. (13.60) for the free energy of
a collection of interacting ions and using the definition of the Bose-Einstein factor
in Eq. (13.62), one has
d
1
5 v-^ dtii δΗω,
dVdT 4- dT OV
(13.88)
Comparing with Eq. (13.63) for the specific heat, define the Grüneisen parameter
IT
V
dn
'( ν
9Ηω
'<
A/y.
=
_ The Griincisen constant is an average over the (13.89)
OYli volume rate of change of the frequencies of
dT
l
d
2
3 7rC
v
the phonon modes, weighted by the contribu-
tion of each mode to the specific heat.
The denominator of Eq. (13.89) is just the (13.90)
d*V0T V specific heat; see Eq. (13.63).
JT C-
β^
_ΊΤ V
^
Combining Eqs. (13.90) and Eq.
(
13.86).
(13.91)
