Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Polarization
663
Figure
22.3.
A dielectric sphere placed in a uniform electric
field
E
0
counteracts the field
by developing a uniform polarization in the opposite direction, as if rigid spheres of pos-
itive and negative charge move slightly apart. The reduction of
EQ
to £o
~~ 4irP/3
results
from the uniform field created by the surface charges at the top and bottom of
the
sphere.
due to all the dipole moments of the other unit
cells.
By symmetry, E\ must vanish,
as shown in Problem 1, so E
œ
\\ = £o- But if this result holds for the center of the
sphere, it must hold elsewhere as well, because all parts of the sphere have the
same dipole moment. The polarization p in each unit cell is
p
= Q.E
C
e\\ ^r~ B —
OLEQ.
The susceptibility a is defined to be the
coef-
(22.5)
ficient
of proportionality
in this relation.
So when the spatial density of dipoles is n,
P
=
tlOtEn.
The ambiguities about choice of unit cell and (22.6)
definition
of P are resolved by requiring P to
vanish
when £
0
vanishes.
To adopt the language of dielectric media, one must consider the electric field
and polarization density on scales very large compared to the lattice spacing. The
spatial average of the polarization is P, while the spatial average of the electric
field E is given by Eq. (22.3) and is the sum of the external field
EQ
with the field
—4irP/3 produced by surface charges. Finally, to compute the dielectric constant,
one has to find the ratio of E +
4TTP
to E. This ratio is
E
*-?'
3 + 87rna
=ȣ=
. Using Eq.
(22.6).
(22.8)
3
—
Anna
Equation (22.8) is the Clausius-Mossotti relation.

664
Chapter 22. Optical Properties of Insulators
A sphere is not the only shape that adopts a uniform internal electric field when
placed in a uniform external electric field. Any ellipsoid has the same property, as
does a thin slab. In general, the electric field inside one of these samples has the
form
È
= ÈQ- NP, (22.9)
where Ji is the depolarization factor and equals 4ir/3 for a sphere, equals 4ir for a
slab,
and vanishes for a long thin rod whose axis lies along the field. The electric
field
£
ce
ii
acting upon a unit cell in one of these samples must equal the electric
field it would have seen if the sample had been a sphere, plus the additional field
due to the different arrangement of surface charges. So the field acting upon a unit
cell is
The field on the cell varies according to (22.10)
Eq. (22.9), but must equal
EQ
for a
sphere when
74
= 4n/3.
Use£ = 47r/V(e-1). (22.11)
(22.12)
(22.13)
Reproducing Eq. (22.6) as expected. (22.14)
22.3 Optical Modes in Ionic Crystals
In modeling the phonon dispersion relations of silicon in Section 13.2.4, ions were
taken to interact only with nearest neighbors. Despite the fact that models of this
type may produce good agreement with experiment, they are misleading. When-
ever an ion moves, it interacts with other ions throughout the crystal with a force
that dies off only as 1/r
2
. In an insulator, the absence of free charge means that
these long-range interactions are not screened. Therefore a realistic calculation
of phonon frequencies in an insulator should take long-range electrostatic effects
into account. Furthermore, because ionic motion and the accompanying motion of
charge are inseparable, the phonons should strongly couple to external electromag-
netic radiation. Optical phonons acquired their name for a reason.
A truly realistic calculation would be very difficult to perform, but a good ap-
proximate calculation can be obtained by mixing together three ingredients.
1.
Nearby ions interact through short-range forces, needed to stabilize the crys-
tal.
These forces will be modeled as linear forces between nearest neighbors.
2.
Ions also interact through long-range Coulomb forces that will be treated by
using the Clausius-Mossotti relation.
„„-.
4TÏ-. - 47T-
£
ce
„
= E
0
-
N/>
+ — P = E + — P
4ir
e
+
2
-.
£cell = -Z 7P
3
e —
1
4TT
e
+
2
-
= ^ r«a£
ce
ii
3
e
—
1
3 /e-1
4™
Ve
+ 2
3 + Snna
3
—
4nna

Optical Modes in Ionic Crystals 665
3.
The individual ions are polarizable, and they develop internal dipole moments
in response to electric fields.
Mechanical Model for Near Neighbors. As
a
mechanical model for nearest
neighbors, imagine that every unit cell contains two ions, of mass M\ and M2,
at
positions u\ and «2 relative to equilibrium. This problem has already been studied
in Section
13.2.1.
The equation of motion for the ions is Eq. (13.7), and the fre-
quency with which they oscillate is given by Eq. (13.9). According to Figure 20.1,
when the frequency of light is in the vicinity
of
10 THz characteristic of optical
phonons, its wavelength is on the order of 10
6
Â, so only the k
—>
0 limit given by
Eq. (13.10) is important. In the long-wavelength limit, the ions have two dynam-
ical modes. In the acoustic mode, shown in Figure 13.4, the ions within the unit
cell move together. The polarization in the unit cell does not change, so electro-
magnetic radiation will not easily excite this mode, and it can be neglected. In the
optical mode, however, the ions move in opposite directions, creating oscillating
dipole moments that can be excited by oscillating electric fields. To describe this
interaction, let
U z= U\ — Ui Normal modes are found by inverting
a
ma- (22.15)
trix described by Eqs. (13.10).
be the normal mode described by Eq. (13.10b). It has a resonance frequency of
[zK
.
1#
M\M
2
LU
where M = ^
'
s
the spring constant of nearest neighbors
(22 16)
M' (Mi
+M
2
)'
defined in Eq. (13.7).
In the presence
of
an oscillating electric field
of
strength
£
ce
n,
suppose that the
optical mode oscillates according to
M'u=-Mü
2
ü-MÜ/T +
e*E
ceü
.
(22.17)
e*
=^
U — ———-,—^
zr^i
',—r^cell- Fourier transforming with
\ dt
expfi'u;/]. (22.18)
M(LO
Z
—
co
z
+
VJJ/T)
The relaxation time
r
describes how long ions keep oscillating once the exter-
nal field is turned off. A reason to employ an effective charge e* is that relative
motion
of
the ions may well be accompanied by
a
readjustment in the electron
clouds around them, so that the net motion of charge is less or greater than one
would expect based on the distance the ions have moved.
Dipole Moments. The dipole moment in each cell is made of two contributions.
First, ionic motion produces
a
dipole moment e*u. Second, the electron cloud
around each of the ions can polarize, producing a second moment of size a°°.E
C
eii.
The electrons adjust much more quickly than the ions to external fields. The total
polarization is therefore
n
=
e*U
+
a°°£
ce
ii.
a
°°
comes from redistribution of charge around
(22.19)
individual ions, and is different from
a
caused
by ionic motion
in Eq.
(22.14).

666
Chapter 22. Optical Properties of Insulators
The long-range interactions between dipoles can be described by the same ar-
guments that lead to the Clausius-Mossotti relation. In particular, the field Z?
ce
ii
acting on the dipole in each cell is given by Eq. (22.10). Therefore
,*\2
(«*)
-ur
M(ü
2
3 e(w)-l
47reM+2
■
iuj/r)
+ a°
£
cdl
Put Eq. (22.18) in Eq. (22.19).
= n
„*\2
M(u>
2
—
to
2
- iuj/r)
+ a°
(22.20)
(22.21)
The quantity on the right is just P/E
ce
\\, while the left comes from Eq. (22.13).
There are four phenomenological quantities in Eq. (22.21): e*,T, a°°, and
Co.
Figure 22.4 shows that the dielectric function e(uj) has definite low- and high-
frequency limits, and these can be used to eliminate two of the phenomenological
constants. For frequencies u much greater than ü, but still small enough that elec-
tron charge responds quasi-statically, one can use Eq. (22.21) with u « 0. Denoting
the dielectric function in this limit by e°°, Eq. (22.21) gives
or
3
Aim
-1
'
+
2
(22.22)
On the other hand, for frequencies
uo
much less than
UJ
one can take
e*E
ce
\\
Mui
2
and find
(e*Y
9Müi
2
Aim
(e°
+ 2)
' + 2)
(22.23)
(22.24)
With these values for
e*
and a°°, one can now solve Eq.
(22.21 )
for e(u) and obtain
e
M
=
e°°
+
UJ . U!
UJ
2
TÜJ
2
e°
+ 2
N
;°°+2
(22.25)
1
Equation (22.25) goes to the correct limits as
ui —>
0 and
UJ
—*•
cxo.
22.3.1 Polaritons
To simplify the dispersion relation (22.25), it is useful to define the transverse
optical frequency
coj
by
UJ
7
' + 2
e°
+ 2
(22.26)
and the longitudinal optical frequency
C^L
by
Optical Modes in Ionic Crystals 667
"£
=
e(u)-.
-UJj
OJ
2
+ ÎÙJ/T — Lü
2
^
LÜ
2
+ iu/r
—
LÜ\
(22.27)
(22.28)
The relationship (22.27) between
O>L
and
U>T
is called the Lyddane-Teller-Sachs
equation.
The dispersion relation Eq. (22.28) allows both transverse and longitudinal
propagating modes within the crystal. According to Eq. (20.17), the transverse
modes satisfy
u)
2
e(uj)
while according to Eq. (20.23) the longitudinal mode satisfies
e(w) = 0.
(22.29)
(22.30)
In the limit r
—>
oo, the dielectric function vanishes for u =
O>L,
explaining the
name given to
WL-
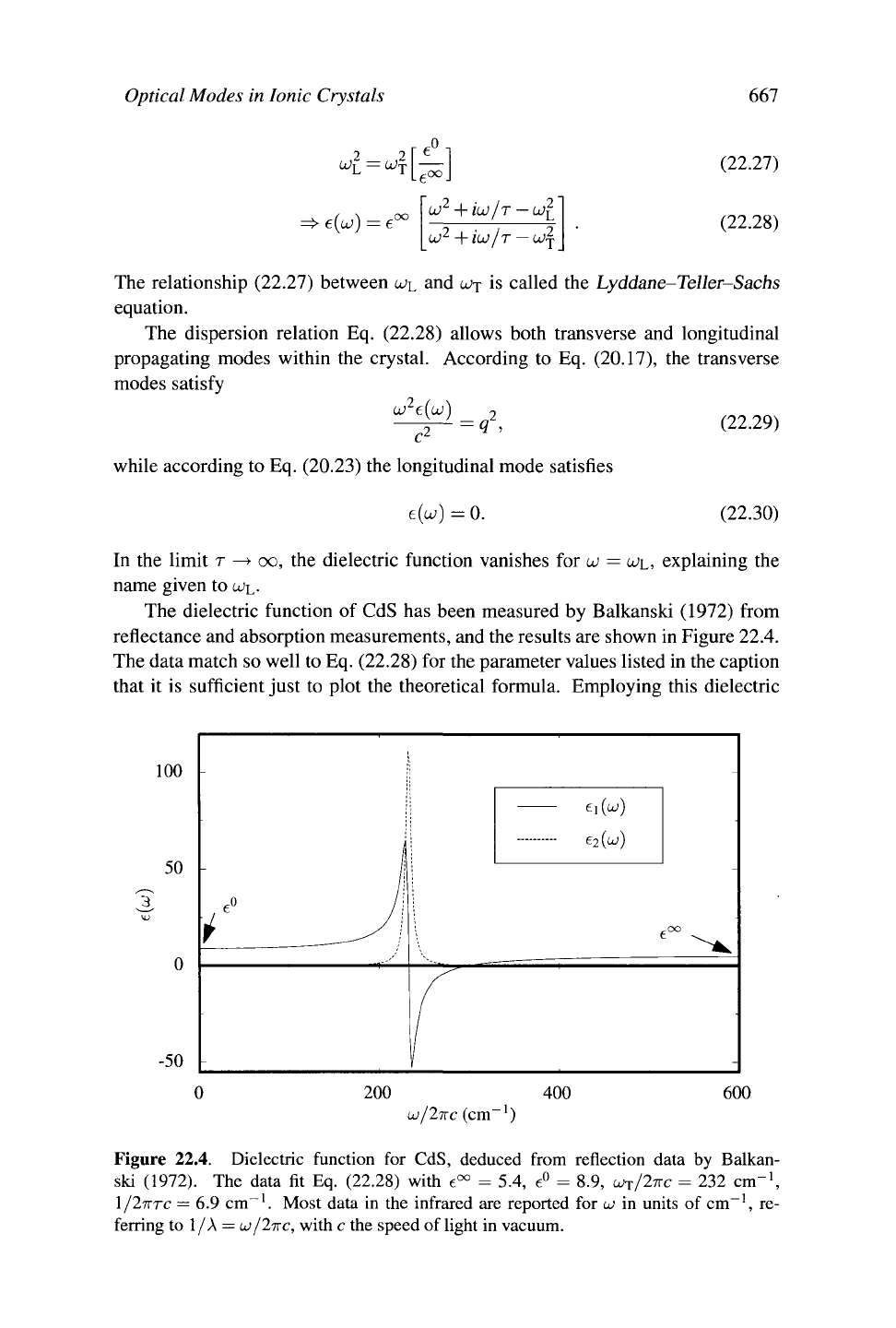
The dielectric function of CdS has been measured by Balkanski (1972) from
reflectance and absorption measurements, and the results are shown in Figure 22.4.
The data match so well to Eq. (22.28) for the parameter values listed in the caption
that it is sufficient just to plot the theoretical formula. Employing this dielectric
100
50
-50
-
/ J
e
2
(o>)
.-OO
\"
200 400
LÜ/2TTC
(cm
-1
)
600
Figure 22.4. Dielectric function for CdS, deduced from reflection data by Balkan-
ski (1972). The data fit Eq. (22.28) with e°° = 5.4, e° = 8.9, LÜJ/ITTC = 232 cm"
1
,
1/27TTC
= 6.9 cm"
1
. Most data in the infrared are reported for
u)
in units of cm
-1
, re-
ferring to 1/A
—
lojlixc, with c the speed of light in vacuum.

668 Chapter 22. Optical Properties of Insulators
2.0 x 10
500 1000 1500 2000 0
Re[q] (cm
-1
)
500 1000 1500
lm[q] (cm
-1
)
2000
Figure 22.5. Frequency
UJ
of transverse waves as a function of complex wave vector q
resulting from data of Figure 22.4.
function in Eq. (22.29) produces the plot of u versus real and imaginary parts of q
that appears in Figure 22.5.
The dispersion relation has three branches, which are most easily distinguished
by sending r
—>
oo to identify the physical significance of
LO^
and
^T^
Lower Branch: At low wave numbers, light behaves as if traveling in an ordi-
nary dielectric medium of dielectric constant e°. As q increases, the dispersion
curve bends over and finally approaches the transverse frequency
LOJ.
The di-
electric function becomes very large along this branch, which means that a
small amplitude of external electromagnetic radiation is accompanied by an
extremely large polarization. Purely light-like at low frequencies, the radia-
tion becomes almost completely phonon-like near
UJJ.
The mode created by
the coupling of light and phonons is called a polariton.
Central Branch: Between
u>j
and
U>L,
propagating modes are heavily damped,
and any incident radiation is almost completely reflected from the crystal.
The central region is fairly narrow, and collecting radiation reflected from a
crystal transforms broadband radiation into a fairly monochromatic residual or
Restrahl band. Einstein (1907) used this frequency in calculating the specific
heat of diamond, as mentioned in Section
13.3.1.
Upper Branch: Well above
U;L
light propagates as if in a medium of dielectric
constant e°°. There are propagating modes in the vicinity of frequency
WL
that
are transverse modes and should not be confused with the distinct longitudinal
mode that exists right at
LJL-

Optical Modes in Ionic Crystals 669
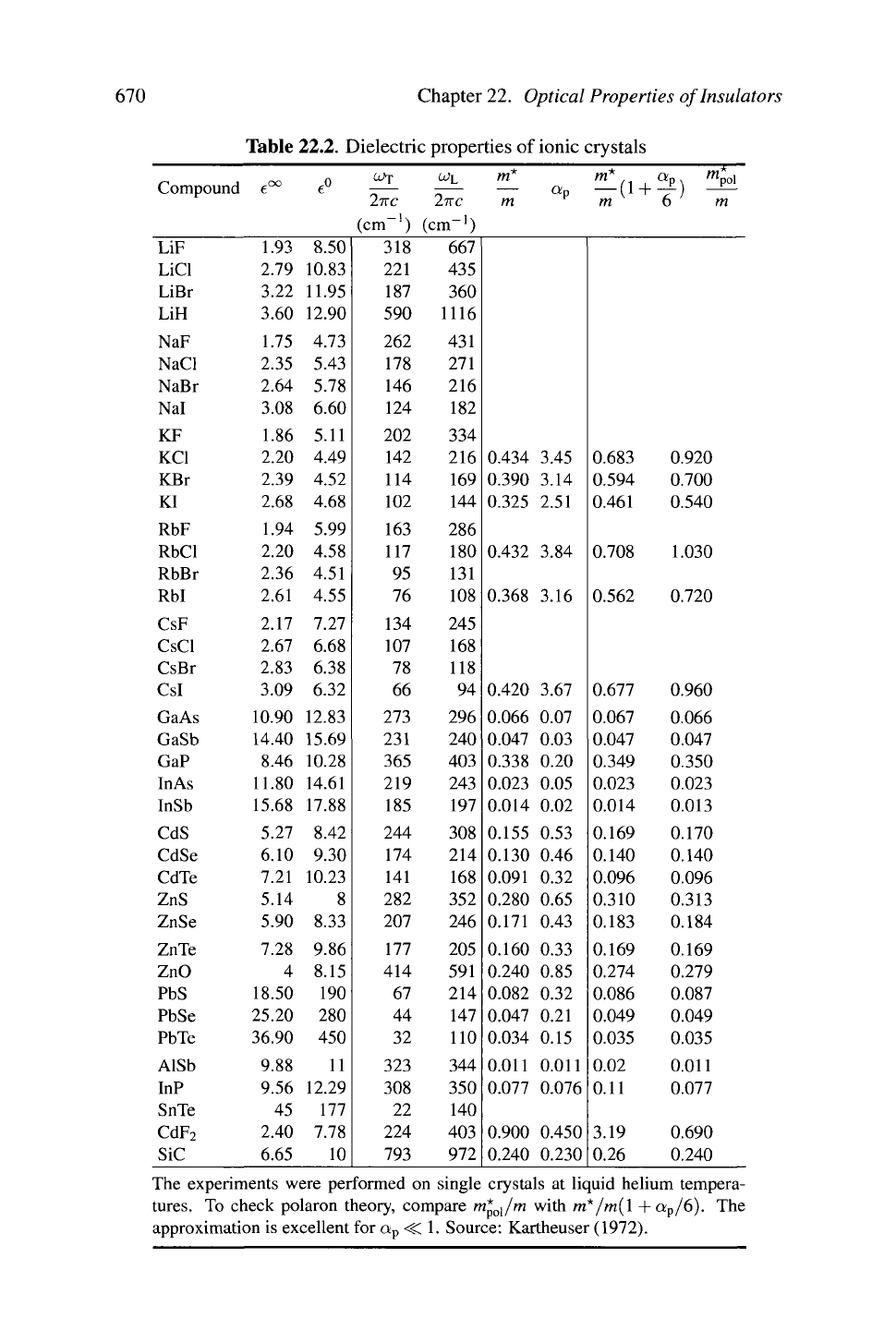
Values
of
dielectric constants and transverse and longitudinal frequencies appear
in Table 22.2.
22.3.2 Polarons
Electromagnetic radiation traveling in the vacuum is always transverse, so it tends
to generate transverse waves when
it
impinges upon
a
crystal.
It
provides no op-
portunity to excite the longitudinal mode where e
=
0.
If,
however, an electron flies
into a polar crystal, it excites mainly the longitudinal modes, while it pushes charge
out
of
the way like
a
snow plow. A polarization cloud surrounds the electron as
it
travels, changing its effective mass. The resulting quasi-particle is called a polaron.
The polaron is not an easy particle to find experimentally. The theoretical atten-
tion
it
has received might therefore seem difficult to understand. One explanation
is that the interaction between electrons and phonons needed to attack the polaron
problem is also the basic interaction lying behind superconductivity of metals and
alloys. Superconductivity brings new layers of complexity to the problem, so there
is value in studying the simpler case of the polaron first.
Relation between Polarization and Displacement. Because the interaction be-
tween electrons and phonons excites longitudinal modes,
it is
useful
to
begin by
finding
a
relation between the polarization P and ionic displacement ü for the lon-
gitudinal mode. Start with
P
=
n[e*Ü+a°°E
ce
n]. From Eq. (22.19).
(22.31)
Then
E —
—4TTP,
Because
e
=
0
=>
D
=
0. (22.32)
which holds for the longitudinal mode implies in conjunction with Eq. (22.10) that
£ceii
= ^ = -y^
(22.33)
ne*
l+na°°87r/3
The expressions (22.22) and (22.24) for
e*
and a°° give
U.
PutEq. (22.33) in
Eq.
(22.31). (22.34)
0
^oo
9Mco
2
e
u
-e
ne*
V 47m
(e°
+
2)(€°°+ 2)
:
"
, ,..00—TVTT^T^^ (22.35)
l+na-87r/3
1
+2(e°°
-
l)/(e°°
+
2)
JMu>ln
f 1 1
4TT
V
e°°
e°
(22.36)
Therefore
P = ßu, (22.37)
670 Chapter 22. Optical Properties of Insulators
Table 22.2. Dielectric properties
of
ionic crystals
^
A
oo o ^T w
L
m* m* a
p
m
ol
Compound e°° e
u
-— -— — a„ —(l + -r) —
t
-
2nc
27TC
m m 6 m
(cm
-1
) (cm
-1
)
LiF
LiCl
LiBr
LiH
NaF
NaCl
NaBr
Nal
KF
KC1
KBr
KI
RbF
RbCl
RbBr
Rbl
CsF
CsCl
CsBr
Csl
GaAs
GaSb
GaP
InAs
InSb
CdS
CdSe
CdTe
ZnS
ZnSe
ZnTe
ZnO
PbS
PbSe
PbTe
AlSb
InP
SnTe
CdF
2
SiC
1.93
2.79
3.22
3.60
1.75
2.35
2.64
3.08
1.86
2.20
2.39
2.68
1.94
2.20
2.36
2.61
2.17
2.67
2.83
3.09
10.90
14.40
8.46
11.80
15.68
5.27
6.10
7.21
5.14
5.90
7.28
4
18.50
25.20
36.90
9.88
9.56
45
2.40
6.65
8.50
10.83
11.95
12.90
4.73
5.43
5.78
6.60
5.11
4.49
4.52
4.68
5.99
4.58
4.51
4.55
7.27
6.68
6.38
6.32
12.83
15.69
10.28
14.61
17.88
8.42
9.30
10.23
8
8.33
9.86
8.15
190
280
450
11
12.29
177
7.78
10
318
221
187
590
262
178
146
124
202
142
114
102
163
117
95
76
134
107
78
66
273
231
365
219
185
244
174
141
282
207
177
414
67
44
32
323
308
22
224
793
667
435
360
1116
431
271
216
182
334
216
169
144
286
180
131
108
245
168
118
94
296
240
403
243
197
308
214
168
352
246
205
591
214
147
110
344
350
140
403
972
0.434
0.390
0.325
0.432
0.368
0.420
0.066
0.047
0.338
0.023
0.014
0.155
0.130
0.091
0.280
0.171
0.160
0.240
0.082
0.047
0.034
0.011
0.077
0.900
0.240
3.45
3.14
2.51
3.84
3.16
3.67
0.07
0.03
0.20
0.05
0.02
0.53
0.46
0.32
0.65
0.43
0.33
0.85
0.32
0.21
0.15
0.011
0.076
0.450
0.230
0.683
0.594
0.461
0.708
0.562
0.677
0.067
0.047
0.349
0.023
0.014
0.169
0.140
0.096
0.310
0.183
0.169
0.274
0.086
0.049
0.035
0.02
0.11
3.19
0.26
0.920
0.700
0.540
1.030
0.720
0.960
0.066
0.047
0.350
0.023
0.013
0.170
0.140
0.096
0.313
0.184
0.169
0.279
0.087
0.049
0.035
0.011
0.077
0.690
0.240
The experiments were performed on single crystals at liquid helium tempera-
tures.
To check polaron theory, compare m*
ol
/m with m*/m(l +a
p
/6). The
approximation is excellent for a
p
<C
1. Source: Kartheuser (1972).

Optical Modes in Ionic Crystals
671
with
'-HR^ÏJ-
Interaction between Electron and
Polarization.
When an electron interacts with
a polarized medium, the energy of interaction is
£4l-phon
—
e
I dr P(7 )
-VV'—^
:•
R is the position operator for the electron. (22.39)
./
\R —
r'\
Making use of Eq. (13.43a), which expresses û in terms of creation and annihila-
tion operators, and Eq. (22.37), which gives the polarization in terms of the lattice
displacements û, leads to
^'-^wW^bç^
V,
'
r'-R\
[e
ik
-
7
âr +
e-
ik
-
7
âl]
(22.40)
= -eß
fdfJ—^—y——
i
-^[e
il7
'â
l
-e-
ilT
'ât}. (22.41)
J ]j
2MOJ
L
N
^ k
\r'-R\
k k
In Eq. (22.40), only the longitudinal mode, where the polarization equals k/k, was
retained. The reason is that this mode
is
the only one to survive the dot product
with
k
that emerges in Eq. (22.41). Electrons moving through a charged medium
naturally push
at
material in front of them, creating compression waves, and are
less likely to induce the side to side wiggling of transverse waves. Finally carrying
out the integral over V the electron-phonon interaction becomes
t/d-ph«
= */W™
Ç J^±^
l
-[e-^-^%].
(22.42)
To characterize the strength
of
the electron-phonon interaction, define
a
di-
mensionless parameter comparing the typical energy of
phonons,
HLOL,
to a charac-
teristic electrostatic energy. Forming a distance from
^H/2m*LOL,
where m* is the
electron effective mass gives a dimensionless ratio
a
p
of electron energy to phonon
energy defined by Fröhlich (1952):
ap
e
2
2m*u>
L
1/1
1
\ « / 1
1
\ m*/m
—
i/—r--—
„
=l.44-10
8
jr) \
—'—. (22.43)
2 V
n
hoj
L
\e°° e°J \e°° e°J V
u>
L
-s
Finally, the ion mass M cancels between Eqs. (22.42) and (22.38), so
'»VN
1/4
£/el-phon
=
V
4
™P^
(ä^J
Efc^'M-^'^-
(2244)

672
Chapter 22. Optical Properties of Insulators
The second quantized form
of
(22.44) will
be
useful.
It is
obtained through the
identity (C.7),
^el-phon
= Y] et
(q"\Û
e
i-
phon
\q)cg The
c's
are creation and annihilation opera- (22.45)
r
* ■* H
r i
tors tor p|p.rtrnrK in nlane. wave, states
<?<?
tors for electrons in plane wave states.
and is
^el—phon
=
I
r—
1
/^WVt
~
v
\ / £"fc
it
it
^"V+/?"^'
(22
-
46)
* ç"+it
Polaron Dispersion Relation. The operator for electron-phonon interactions can
be used
to
search for the change
in
effective mass
of
an electron moving through
an ionic crystal.
If
the coupling parameter
a
p
is
small, then this task may
be
ac-
complished by finding the change in energy
of
an electron to leading order in per-
turbation theory. To make things simple, assume that in the absence of polarization
effects, the electron would travel
as a
plane wave with energy H
2
q
2
/2m*. The
ef-
fective mass here results from interactions of the electrons with the static lattice, as
in Section
16.2.3,
and must be distinguished from the additional effective mass the
electron will acquire when
it
drags around
a
cloud of phonons.
At first order in perturbation theory, the expectation value
of
(22.46) vanishes,
so to obtain
a
nonzero result one must move to the second-order term, which
is
A£
(2)
£(?,*o) -£(?,*')
$o refers to the ground state of the
phonons, and
$'
is
a
state with
longitudinal optical phonons present;
q
and q' are electron wave numbers.
(22.47)
Placing Eq. (22.46) into Eq. (22.47), the only intermediate phonon state <&' that
survives
is the
one with precisely one phonon
of
wave number
k = q
—
q' and
energy hoj^. All possible electron momenta
q' are
allowed,
but
states
q
and
q'
must have the same spin, so when the sum over
q
is converted into an integral, the
density
of
states is V/(2vr)
3
, not 2V/(2n)
3
. One has
A£
(2)
=4vra
r
1
//Pu;
5,
,3
'VV
2m
^EÛ
1
q-q
712
1
j-2
2
n
q
In?
2m*
+
hujL
(22.48)
= 47ra
r
lh
5
Lül 1
W
2m*
a
n
7T
5,
,3
h
, d(cos
9)
2vrV
(27T)
iH^H
2m*
J-\ Jo
1
,2J2
h
z
q
2m*
n
2
(q'
2
+ q
2
+ 2qq's)
2m*
+HU;L
= -CKr
m*hoj
3
— sin
q
2„2
h
l
q
2m*hwi
J
(22.49)
(22.50)
(22.51)
Perform first the
q'
integral, and then s.
q'
can safely be integrated to infinity because the
integral is convergent. One of the terms following the
q'
integral looks hopeless, but
it is
odd in
i
and vanishes by symmetry.
