Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Atomic Magnetism
763
electrons organize themselves into shells. The filled shells are relatively inert, and
Hund's rules concern themselves only with the organization of electrons in incom-
pletely filled shells. Electrons in these shells can be indexed by quantum numbers
L,
L
z
, S, and S
z
. In fact, only J = L + S is a true constant of the motion. Use of
L and S as independent quantum numbers is predicated upon assuming that spin
and angular momentum do not interact strongly with one another, which means
that the atom does not interact strongly with the magnetic field it generates
itself.
Formally, the spin-orbit coupling, proportional to S
■
L, needs to be a small pertur-
bation. Given this assumption of Russel-Saunders coupling, Hund's rules are as
follows:
1.
Electrons in the incomplete shell first choose to maximize the total spin S.
For example, for an atom with two valence electrons in a shell with / > 1, the
electrons can choose either to occupy the same L
z
state, in which case they
must have opposite spins, or different L
z
states (all degenerate in energy), in
which case the spins can do what they want. If the electrons occupy differ-
ent orbitals and adopt a triplet spin state, which is symmetric, then they may
also have an antisymmetric spatial state. The Coulomb repulsion between the
electrons is then reduced because the wave function automatically vanishes as
they approach each other. For this reason, the two electrons prefer to stick to
different orbitals, and they take the triplet over the singlet state. One can think
of this rule as specifying that atoms on a single site develop ferromagnetic
correlation and want their spins to point together.
2.
Once S has been determined, the electrons choose the largest value of L con-
sistent with putting electrons in different orbitals whenever possible. For ex-
ample, when the shell is half full, all possible values of l
z
are occupied, and
the total L must be zero. This rule may be understood classically as a second
consequence of the desire to reduce Coulomb interactions. If one were re-
quired to set two electrons spinning about an atom with the same total angular
momentum, but otherwise as far apart as possible, one would put them in the
same orbit, but 180° out of
phase.
In this case, the electrons would actually be
in the same state quantum mechanically, just differing by a phase factor; the
Coulomb repulsion between them would be enormous, and such a state is not
favored. The electrons have to be in states of different L
z
. So the next guess
is that they rotate classically in the same direction, and about axes that differ
as little as possible. If, for example, they were to rotate in opposite directions,
they would encounter each other twice per orbit.
3.
Once L and S have been determined, the space of (2L + 1)(25+ 1) states
is split by the spin-orbit interaction, which has a magnitude on the order of
electronic orbital energies times (ZQ)
2
, where Z is the atomic number and a
is the fine structure constant. The electrons choose as the ground state
J=\L-S\
(25.8a)
if the shell is less than half full, and they choose
764 Chapter
25.
Magnetism of Ions and Electrons
J — L + S See Landau and Lifshitz (1977), p. 267 for a (25.8b)
discussion.
if the shell is more than half full. When the shell is half full, L = 0, so there is
no jump in J at half filling. For a shell that is one electron shy of half filling,
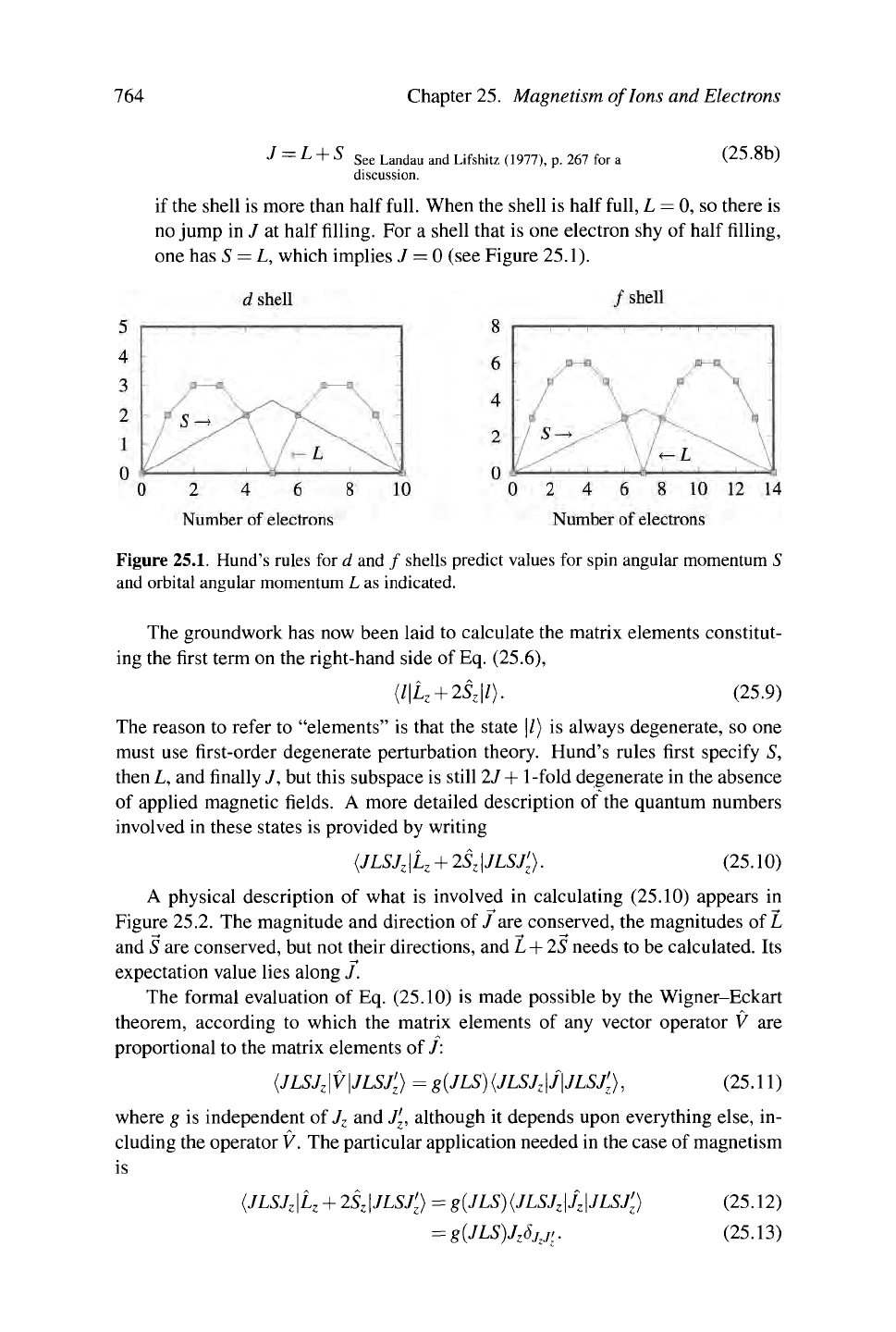
one has S = L, which implies 7 = 0 (see Figure 25.1).
Figure
25.1.
Hund's rules for d and / shells predict values for spin angular momentum S
and orbital angular momentum L as indicated.
The groundwork has now been laid to calculate the matrix elements constitut-
ing the first term on the right-hand side of
Eq.
(25.6),
(l\L
z
+
2S
z
\l).
(25.9)
The reason to refer to "elements" is that the state |/) is always degenerate, so one
must use first-order degenerate perturbation theory. Hund's rules first specify S,
then L, and finally J, but this subspace is still 2J
+
1-fold
degenerate in the absence
of applied magnetic fields. A more detailed description of the quantum numbers
involved in these states is provided by writing
(JLSJ
Z
\L
Z
+ 2S
Z
\JLSJ
Z
). (25.10)
A physical description of what is involved in calculating (25.10) appears in
Figure 25.2. The magnitude and direction of J are conserved, the magnitudes of L
and S are conserved, but not their directions, and L + 2S needs to be calculated. Its
expectation value lies along J.
The formal evaluation of Eq. (25.10) is made possible by the Wigner-Eckart
theorem, according to which the matrix elements of any vector operator V are
proportional to the matrix elements of J:
{JLSJ
Z
\V\JLSJ'
Z
) = g(JLS){JLSJ
z
\J\JLSJ'
z
), (25.11)
where g is independent of J
z
and
J'
z
,
although it depends upon everything else, in-
cluding the operator V. The particular application needed in the case of magnetism
is
(JLSJ
Z
\L
Z
+
2S
Z
\JLSJ'
Z
)
= g(JLS)(JLSJ
z
\J
z
\JLSJ
z
) (25.12)
= g(JLS)J
z
ôj
zJ
,. (25.13)

Atomic Magnetism
765
Figure 25.2. View the spin and orbital angu-
lar momentum of
an
atom as two gyroscopes,
linked end to end. The total angular momen-
tum J =
L
+
S
is conserved, but L and S pre-
cess about
one
another.
The expectation value
of
L
+
S
lies along 7.
Evaluation of g(JLS). The problem of finding the splitting induced by the
magnetic field is therefore reduced to the problem of finding the Lande g factor
g(JLS).
A trick enabling the evaluation begins by considering
{JLSJ
Z
\L
+
2S\JLSJ'
Z
)
= g(JLS)
(JLSJ
Z
\J\JLSJ'
Z
)
(25.14)
=>
{JLSJ
z
\L + 2S\j'L'S'J
z
)=g(JLS){JLSJ
z
\J\J'L'S'J
z
) (25.15)
Both matrix elements vanish unless J = J' and
Z,
= L' and S = S'.
=>
Y,
{JLSJ
Z
\L
+
2S\J'L'S'J'
Z
) ■
(J'L'S'J'
Z
\J\J"L"S"J'1)
L'J'S'J'.
= g(JLS)
Y,
(JLSJ
Z
\J\J'L'S'J'
Z
)
■
(J'L'S'J'
Z
\J\J"L"S"J'
Z
'). (25.16)
L'J'S'J',
Because the sum in Eq. (25.16) is taken over a complete set of states, it becomes
(JLSJ
Z
\(L + 2S)-J\JLSJ'
Z
) = g(JLS)(JLSJ
z
\J
2
\JLSJ'
z
). (25.17)
All of the matrix elements in (25.17) can be evaluated because
S
2
= (J-L)
2
=ß + L
2
-2L-J (25.18)
L
2
=
{J-S)
2
=J
1
+
S
2
-2S-J.
(25.19)
Therefore
^
=
iwi!ai«ia.
(2
,
20)
The final result is that an external magnetic field splits the 27 +
1-fold
ground
state of an isolated atom into 27+1 separate levels, and the energy difference
between them is
Mg
g[37(7+l)-L(L+l) + 5(5+l)]
2 7(7+1)
'
{
'

766 Chapter 25. Magnetism of Ions and Electrons
25.2.2 Curie's Law
Room temperature corresponds
to an
energy
of
0.025
eV, much larger than
the
basic energy scale
of
magnetic ions given in Eq. (25.7). Therefore magnetic spins
at room temperature find themselves
in a
statistical distribution
of
states. States
with spin
up
and spin down
are
almost equally populated,
so
the
net
magnetic
moments are fairly small.
It is usually, although not always, adequate to assume that only the lowest-lying
spin multiplet contributes to statistical mechanical sums. Making this assumption,
the partition function for
a
single magnetic ion is given by
j
Zion=
Y)
e-^»
BJ
i
(25.22)
J
Z
=~J
a
B
B{J+l/2)
_
e
-ßg
ßB
B{J+l/2)
e
ßg/J,
B
B/2
_
e
-ß
gfJ
,
B
B/2
(25.23)
The free energy
is
7 =
-k
B
T
In Zjon
+
i
/.
In
this thermodynamic
ensemble,
the
energy
/
j-p
D2 of the
external field
must
be
included.
If
the /9S
0A\
<
t
~
I '
potential
S
were
used
instead,
the
integral
over
^ ' '
the
external field
could
be
omitted
According to Eq. (24.26), with a density of
n
magnetic ions in a volume V, one
can now find the magnetization from
„
^d?
w
B 19J „
e
^
H
=
YTB^
M
=
A-,-VTB
(25
-
25)
^M =
nk
B
T--
In Z
ion
(25.26)
dB
= nn
B
gJ'Bj(ßti
B
gJB), (25.27)
*M=^
H*
(^'h-h
**(£)■
(25
-
28)
where
So long as [i
B
B <S k
B
T, one has
cothx«-
+
J
+
.
. . (25.29)
x
3
^'B
J
=
^^j
i
-ßß
B
gJB, (25.30)
so that
MW(
MB
)
2
-^^±H.
(25.31)
k
B
l
5
Equation (25.31) is Curie's
law.
It holds so long as the magnetic ions may be treated
as noninteracting and in isotropic environments. The law breaks down once inter-
actions between the different ions become important; even
at
high temperatures,
interacting ions are better described by Eq. (24.41).

Atomic Magnetism
767
Comparison with Experiment. Curie's law can be tested for a variety of magnetic
ions in nonmagnetic hosts. The comparison is particularly successful for lanthanide
compounds, where a rare earth ion sits in a nonmagnetic host. For example, cerium
compounds include CeCi3, CeF^, and Ce2Mg3(NC<3)i2 -24H20. Properties of a
magnetic ion vary by around 5% from one host to another.
Comparison with experiment is accomplished by writing the susceptibility in
the form
X
= n
3^f^
(2532)
where according to Eq. (25.31),
Meff = g(JLS) JJ{J +l).flß
For dilute ma
g
ne
i
ic
systems, ignore the dif- (25.33)
ference between B and H.
Equation (25.33) is to be compared to an experimental measurement of
Mexp
=
y—-—• (25.34)
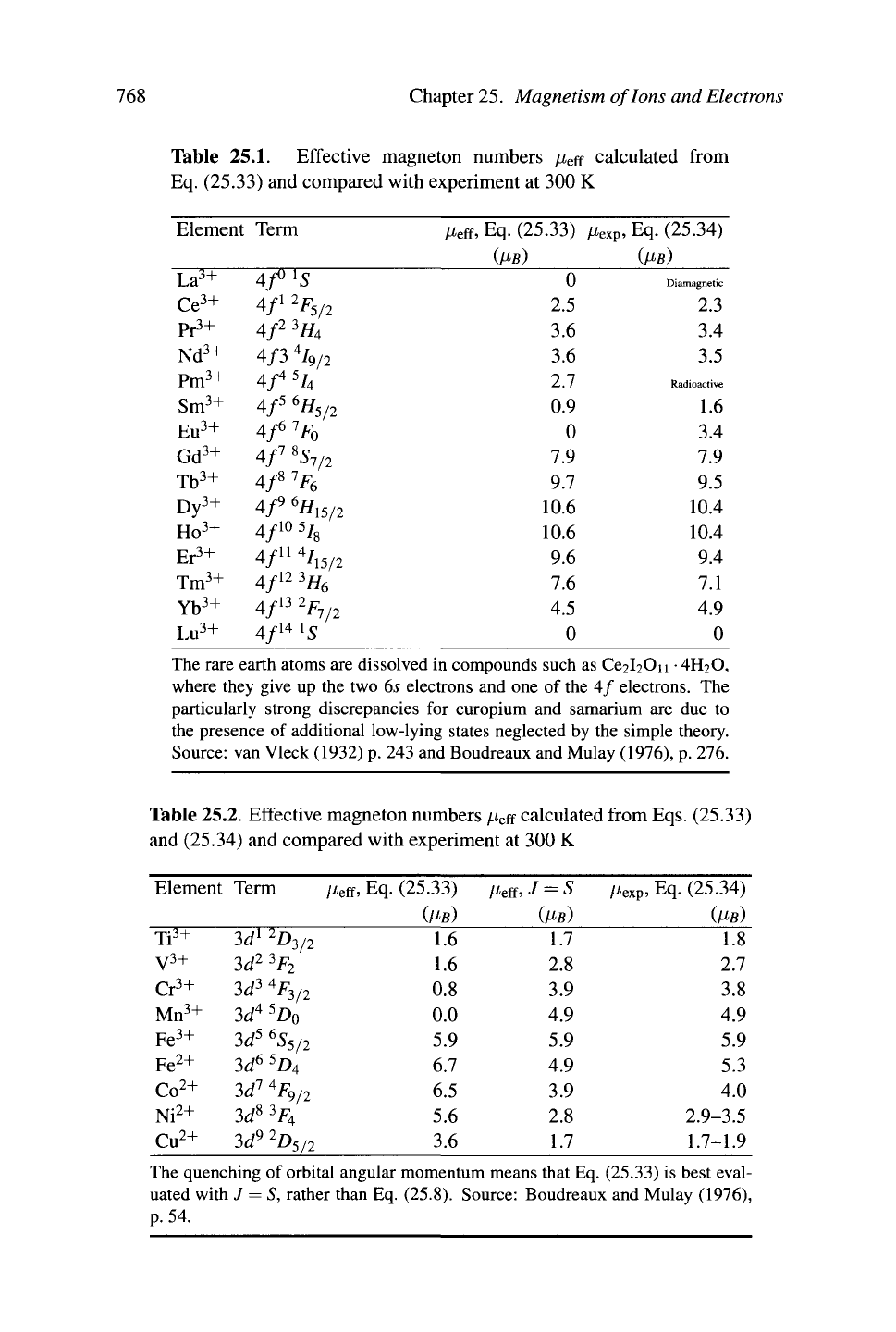
This comparison is performed for the lanthanides in Table 25.1. The agreement
with theory is impressive, except in the case of europium and samarium, where
magnetic multiplets lying above the ground state play
a
measurable role. Bohr
says that "[o]n the whole a consideration of
the
magnetic properties of the elements
within the long periods gives us a vivid impression of how a wound in the otherwise
symmetrical inner structure is first developed and then healed as we pass from one
element to another." [Bohr (1922), p. 107]
For magnetic transition metal ions the comparison is much less successful, as
shown in Table 25.2.
d
electron wave functions have a longer range than the
/
electron wave functions, and transition metal ions interact more strongly with their
environment than the lanthanides, so the third of Hund's rules is no longer correct.
However, the theory can be improved substantially by postulating that the orbital
angular momentum L is quenched by interactions with the crystal. Intuitively, one
can think of interactions with crystal fields causing L to precess, so that all matrix
elements of L vanish, although L
2
still has value L(L+1). In this case, Hund's third
rule is replaced by
J
—
S. Table 25.2 shows that this new rule is fairly successful
in explaining the experimental observations.
Larmor Diamagnetism and
Van Vleck
Paramagnetism. The leading contribution
to the susceptibility in Eq. (25.6) vanishes when 7
=
0, because 7
=
0 leads to
g(JLS)
=
0. J can vanish either for filled shells, or for shells that are one electron
shy of half filling, because in the latter case L = S, and J
=
\L —
S\
=
0 according
to the third of Hund's rules. The ground state |0) with J = 0 is nondegenerate, and
its change in energy with magnetic induction B is
j
/Y0
768 Chapter 25. Magnetism of Ions
and
Electrons
Table
25.1.
Effective magneton numbers /Lt
e
ff calculated from
Eq. (25.33)
and
compared with experiment
at
300
K
fits,
Eq.
(25.33)
p^,
Eq.
(25.34)
G"ß)
(VB)
Element
La
3
+
Ce
3+
Pr
3
+
Nd
3+
Pm
3+
Sm
3
+
Eu
3
+
Gd
3
+
Tb
3
+
Dy
3
+
Ho
3+
Er
3
+
Tm
3
+
Yb
3
+
Lu
3
+
Term
4/
)1
S
4/
1
2
F
5/2
4/
2
3
//
4
4/3
4
/
9/2
4/
45
/
4
4/
5
6
H
5/2
4/
6
7
F
0
4/
7 8
S
7/2
4/
8
7
F
6
4/
96
#i
5
/2
4/
10
5
/
8
4/
11 4
/
15/2
4/
12
3
//
6
4/
13 2
F
7/2
4f
14 l
S
0
2.5
3.6
3.6
2.7
0.9
0
7.9
9.7
10.6
10.6
9.6
7.6
4.5
0
Diamagnetic
2.3
3.4
3.5
Radioactive
1.6
3.4
7.9
9.5
10.4
10.4
9.4
7.1
4.9
0
The rare earth atoms
are
dissolved
in
compounds such
as
Ce2l20n
•
4H2O,
where they give
up the two 6s
electrons
and one
of
the
4/
electrons.
The
particularly strong discrepancies
for
europium
and
samarium
are
due
to
the presence
of
additional low-lying states neglected
by the
simple theory.
Source: van Vleck (1932) p. 243 and Boudreaux and Mulay (1976), p. 276.
Table 25.2. Effective magneton numbers /i
e
ff calculated from Eqs. (25.33)
and (25.34)
and
compared with experiment
at
300
K
Element
Ti
3
+
v
3
+
Cr
3
+
Mn
3+
Fe
3+
Fe
2
+
Co
2
+
Ni
2
+
Cu
2
+
Term
3d
1
2
D
3/2
3d
2
3
F
2
3d
3
4
F
3/2
3d
4
5
D
0
3d
5
6
S
5/2
3d
6
5
D
4
3d
74
F
9/2
3d*
3
F
4
3d
9
2
D
5/2
/i
eff
,
Eq.
(25.33)
(Ms)
1.6
1.6
0.8
0.0
5.9
6.7
6.5
5.6
3.6
Meff>
J = S
(VB)
1.7
2.8
3.9
4.9
5.9
4.9
3.9
2.8
1.7
/x
eX
p,
Eq.
(25.34)
(HB)
1.8
2.7
3.8
4.9
5.9
5.3
4.0
2.9-3.5
1.7-1.9
The quenching
of
orbital angular momentum means that Eq. (25.33)
is
best eval-
uated with
J =
S,
rather than Eq. (25.8). Source: Boudreaux
and
Mulay (1976),
p.
54.

Magnetism of the Free-Electron Gas 769
Table 25.3. Susceptibilities of noble gases compared with Eq. (25.36)
Element: He Ne Ar Kr Xe
-X, experiment (1(T
Ö
cm
3
mole"
1
): 1.88 7.02 19.18 28.49 43.33
X, Eq. (25.36) x0.35 (1(T
6
cm
3
mole"
1
): 0.99 14.82 20.54 23.74 27.95
The very rough agreement
is
slightly improved
by
multiplying
Eq.
(25.36) by
0.35.
For
helium, the factor of 6 in Eq. (25.36) is eliminated and a radius of
1
 is employed.
Otherwise, radii come from Table
11.1.
Experimental data from Table
24.1.
Because the magnetic susceptibility x =
—d
2
S/dH
2
ss —d
2
A8./dB
2
, the first
term on the left hand side of
Eq.
(25.35) leads to a magnetization against the applied
field and is therefore diamagnetic, Larmor diamagnetism. The second term on the
right-hand side of
Eq.
(25.35) is negative, because the denominator is negative and
therefore produces magnetic response that is paramagnetic—van Vleck paramag-
netism.
The second term vanishes for closed shells, where not only J but L and 5
are all separately zero; however, for shells one electron short of half filling, both
terms on the right of Eq. (25.35) may compete. Aside from observing that both
are very small, no general statement can be added. Curie paramagnetism, Larmor
diamagnetism, and van Vleck paramagnetism all feature a small magnetic suscep-
tibility proportional to
pb
2
B
.
However, the mechanisms are quite different. Curie's
law results from a matrix element that is linear in the magnetic field, and the sus-
ceptibility would vanish if it were not for the way that magnetic fields change the
statistical occupation of states at nonzero temperature.
The susceptibility of the noble gases can be estimated by employing atomic
radii from Table 11.1 and writing
r is the atomic radius, the factor of 6 comes
from the fact that there are 6 electrons in the ('yz T.f.\
outer shell, and the factor of 2/3 comes from ^ ' '
estimating x
2
+y
2
= Ir
2
/3.
As shown in Table 25.3, the comparison with experiment is only partially success-
ful;
agreement is improved by arbitrarily multiplying Eq. (25.36) by 0.35, presum-
ably because it is not legitimate to treat all electrons as occupying the outer radius.
25.3 Magnetism of the Free-Electron Gas
Having determined the magnetic properties of electrons when strongly bound within
ions,
it is valuable to consider the opposite extreme and examine their magnetic
properties as they move about nearly free in a metal.
There are essentially three terms in the response of free fermions to an external
magnetic field. The first is called Pauli paramagnetism, and it results from the
action of the magnetic field on the spins of the electrons. The second is called
Landau diamagnetism, after Landau (1930), and results from the moments created
by the circular motions of the electrons. The third (a series) was also calculated by
X
:
l
l,—ö
6
^
r
■
4mc
z
3

770
Chapter
25.
Magnetism of Ions and Electrons
Figure
25.3.
When subject to a magnetic induction
B,
the number of spin-down electrons
increases and the number of
spin-up
electrons decreases, to minimize the total free energy.
Landau (1939) and predicts the oscillations of magnetization with magnetic field
that lead to the de Haas-van Alphen effect.
25.3.1 Pauli Paramagnetism
Pauli paramagnetism can be understood as follows: In the absence of a magnetic
field, the ground state of a free electron gas has equal numbers of spin-up and spin-
down electrons. An induction B pointing up raises the energies of spin-up electrons
by the amount
The energy SS is the energy of electrons in the
CT = Oj+/igo. absence of a magnetic field. The g-factor, which (25.37)
equals almost exactly 2, cancels the factor of
1
/2
from the spin-1/2 quantum number.
Now consider a very weak magnetic induction B applied to the electron gas, as
shown in Figure 25.3, and calculate the net magnetic moment it induces. Remem-
bering that the definition of D(E) assumes up- and down-spin states to be equiva-
lent, and must be divided by two if they are considered separately, the number of
spin-up electrons is
N
up
= V
I d£°
I)
^p-f(£
0
+
f
i
B
B),
(25.38)
D(£°) denotes the density of states in the absence of a magnetic field, and /(£) is defined in
Eq. (6.49). It is not clear that the density of states D should not change to first order with the
magnetic field, but this guess will be verified in later calculations.
and the number of spin down electrons is
Wdown
= V I dl° ^-^/(£° -
HBB).
(25.39)
Expanding in the small quantity B gives
Notice that differentiating / with respect to
HBB
is the same as differentiating with respect to
£°,
which in turn is the same as differentiating by the negative of the chemical potential, —fi.
The derivative with respect to the chemical potential can then be brought outside the integral.
while

Magnetism of the Free-Electron Gas
111
„
d
~
+
^.
(25.41)
2
2. o\i
The magnetization M points in the direction opposite to the spins, because the
electron is negatively charged. Therefore, the magnetization per volume is
M=^(N
down
-N
up
)=
i
-^f^B
(25.42)
and the magnetic susceptibility
x
is
dM
dM . ,,
1 dN
«'«"»'WT*'
(25
'
43)
which at temperatures well below the Fermi temperature becomes
Y
=
njyD(Ep). This expression makes use of the Sommer- (25.44)
feld expansion—for example, by differentiat-
ing the expression for N exhibited in Eq. (6.72).
In the particular case of
the
free Fermi gas, the density of states at the Fermi surface
is given by Eq. (6.24), resulting in
y
=
——
=
4.757 • 10 (n/f 10
•
Cm
1) .
This expression is in cgs. For SI, multiply by
7T
2
tl
2
4OT
-
(25.45)
25.3.2 Landau Diamagnetism
A collection of noninteracting electrons would change their energy in response to
an applied magnetic field even if they did not have spin. This effect, first calculated
by Landau (1930), is entirely quantum mechanical in origin, because it vanishes in
any classical calculation. For classical electrons, electrons bouncing off bound-
aries cancel effects of electrons in the bulk. However, for quantum electrons, the
quantization of electron orbits destroys the perfect cancellation and allows an ef-
fect to remain. When thermal energies are large compared with magnetic energies
the resulting effect is diamagnetic, and precisely one-third as large as the Pauli
paramagnetism in magnitude.
Electron Energy Levels in a Magnetic Field. To calculate this effect, one must
find the energy levels and density of states for electrons in a magnetic field. This
task is left as Problem 4, and the results are as follows: The problem has a charac-
teristic frequency, the cyclotron frequency defined in Section 21.2,
eB
to
c
=
—,
(25.46)
mc
that gives the rate at which electrons spin about in their orbits. Energy states are
indexed by three quantum numbers: k
z
, a wave vector parallel to the field; k
y
,
a

772 Chapter 25. Magnetism of Ions and Electrons
wave vector perpendicular to the field which
is
related
to
the
x
coordinate
of
the
center of the orbit through
x
0
= ^;
(25.47)
mu
c
and an index
v
that describes how energetically the electron rotates about the center
of the orbit. The integer
v
labels Landau levels.
In a
box
of
side length
L
with
periodic boundary conditions, the allowed values of k
y
and k
z
are given by Eq. (6.7).
The energy levels corresponding to these three quantum numbers are
The rotation rates
of
the electrons are quantized in units of uv, and the energy
is
independent
of k
y
, which corresponds physically to the center of the orbit. There really should be an extra
term in this expression, aßaB/2, with
a
taking values ±1 according to the spin of the electron.
The effects of the term have already been included in the previous section, so drop it to simplify
things a bit.
By requiring that
xo
lie between 0 and the end of the sample at L, one can determine
the degeneracy of an energy state with given
v
and k
z
. One has that
0<X
0
<L=^0< — < L Using
Eq.
(6.7) for
the
allowed (25.49)
mui
c
L
values of k
y
, where
fe
is an
integer, and Eq. (25.47) forxo.
mcü
r
L
2
=►
0 > h > .
Tne
absolute value of the (25.50)
27l7z right-hand side gives the number
of allowed values of li-
BA
$
^AT=
— = —
(25.51)
with
,
$
0
= — = 4.14
•
10"
7
G Cm
2
; N is the total number of (25.52)
e
degenerate states,
<3>
is the total
magnetic flux, and A
=
L
2
is the
total area.
the number
of
degenerate electron states
N
in
a
Landau level
is
given by the ratio
of the total magnetic flux to the magnetic flux quantum
<£>o-
The density
of k
z
states
is
L/2TT.
Therefore
for
each
v,
the density
of
states
including spin degeneracy
is
The first factor of
two
comes from spin degeneracy, the next factor comes from Eq. (25.49),
and the final factor comes from the density of k
z
states. The spin degeneracy comes from
the fact that a spin-up electron in one level has the same energy as
a
spin-down electron in
an adjacent level.
Instead
of
the density
of k
z
states, one can instead consider the density
of
energy
states and obtains
2 &j
c
/2m\3/2
-1/2
D(£
'
^
)=
(27^)2
^(F)
[
£
-(^)H
"sing
Eq.
(25.48). (25.54)
