Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.

3.5 Tunneling 93
U
0
E,U
x
1
2
0
E < U
0
E > U
0
Incident
electron
Incident
electron
L
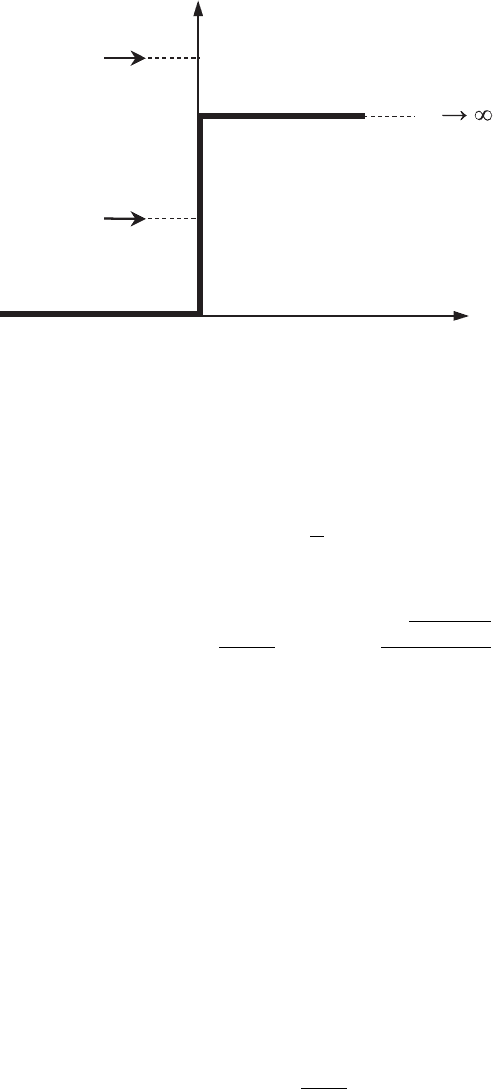
Figure 3.8 Reflection of an
electron from a potential
step with U = U
0
.
through the barrier becomes equal to zero, and the reflection coefficient becomes
equal to unity: R = 1. The case with finite U
0
and width L →∞corresponds to
the total reflection of the electron from the so-called potential step with a finite
height of the barrier U
0
(see Fig. 3.8). In this case a phase shift δ between the
wavefunctions of the incident and reflected waves occurs:
B
1
=
√
Re
iδ
A
1
. (3.161)
Here the phase shift δ is defined by the expression
δ = arctan
2kκ
k
2
− κ
2
= arctan
2
E(U
0
− E)
2E − U
0
. (3.162)
We see that at E = U
0
the phase shift is equal to zero, at E = U
0
/2 the phase
shift is equal to δ = π/2, and at E U
0
the phase shift is δ → π . In the case
of reflection from an infinitely high barrier (U
0
→∞) the phase shift between
incident and reflected wavefunctions of the electron is equal to δ = π as in
the case of the reflection of an electromagnetic wave from an optically denser
medium.
It is interesting to study the probability of finding an electron in the region
x > 0. In spite of the fact that E < U
0
and that the reflection coefficient of an
electron from the potential step is equal to unity, the probability of finding an
electron in the region x > 0 is not equal to zero! The electron wavefunction,
ψ
2
(x), in this region is equal to
ψ
2
(x ) = A
2
e
−κx
, (3.163)
where
A
2
=
2k
k − iκ
A
1
. (3.164)

94 Layered nanostructures
Therefore, |ψ
2
|
2
= 0. The effective penetration depth of the electron under the
potential step can be estimated by a quantity l
eff
= κ
−1
. At this distance from the
boundary x = 0 the probability of finding the electron decreases by a factor of
e
2
in comparison with its magnitude at x = 0. The probability that the electron
penetrates the potential step in this case is defined by the transmission coefficient,
D, through the boundary x = 0:
D =
κ
k
|A
2
|
2
|A
1
|
2
=
4kκ
k
2
+ κ
2
. (3.165)
For the reflection coefficient in this case we obtain the expression
R =
|B
1
|
2
|A
1
|
2
=
k + iκ
k − iκ
2
= 1. (3.166)
Thus, the electron penetrates through the boundary of the potential step into
the region x > 0, but then it returns back into the region with x < 0, reflecting
effectively from the potential step with reflection coefficient R = 1. Because of
such penetration there occurs a phase shift δ between the two wavefunctions
which correspond to reflected and incident waves.
3.5.2 Electron motion above the barrier
Let us write the general expressions for the electron reflection and transmission
coefficients in the case of electron motion with energy E > U
0
above the barrier
with finite width L (see Fig. 3.7):
R =
1 +
4E(E − U
0
)
U
2
0
sin
2
(κ L)
−1
, (3.167)
D =
1 +
U
2
0
sin
2
(κ L)
4E(E − U
0
)
−1
, (3.168)
where κ =
√
2m
e
(E −U
0
)/h
-
. In the case of electron motion above the potential
well (Eqs. (3.129) and (3.131)) as well as in the case of motion above the potential
barrier (Eqs. (3.152) and (3.153)) the change in electron energy leads to oscilla-
tions of both coefficients, R and D. The maxima of the transmission coefficient,
D (and minima of reflection coefficient, R), are defined by the condition
κ
n
L = π n, n = 1, 2, 3,... (3.169)
It follows from the last condition that
E
n
= U
0
+
π
2
h
-
2
n
2
2m
e
L
2
. (3.170)
The transmission and reflection coefficients are D = 1 and R = 0, and we are
dealing with the so-called resonant interaction of an electron with the potential
barrier. We note that the quantity E
n
= E
n
−U
0
coincides with the expression
3.5 Tunneling 95
3π/2π
π/2
1
0
kL
D
2π
0.5
0.1
0.2
0.5
1
0
0.01
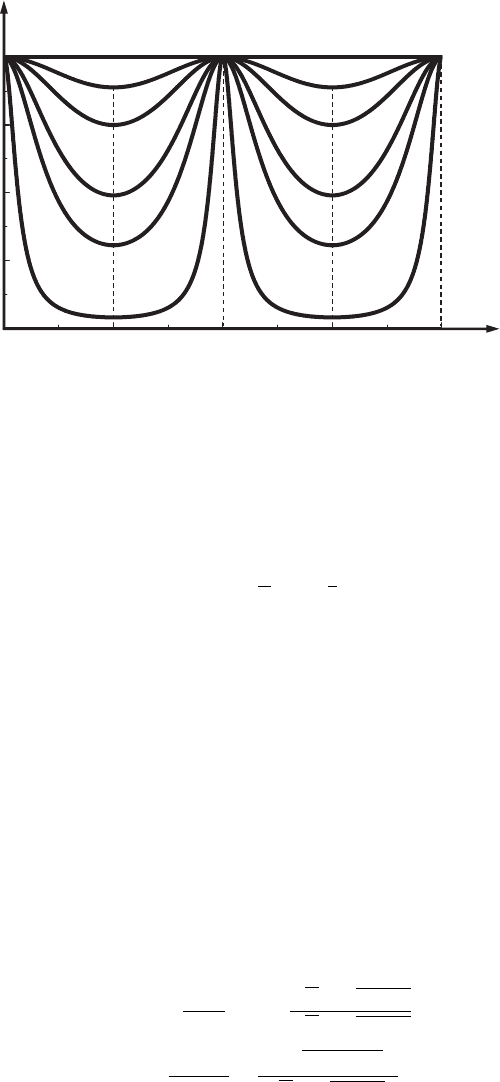
Figure 3.9 The
dependence of the
electron’s transmission
coefficient, D,onthe
magnitude of the
potential barrier, U
0
,and
κ L. The six curves
correspond to E
1
/U
0
=
0, 1, 0.5, 0.2, 0.1, and 0.01.
Here E
1
= E
1
−U
0
and
E
1
is the electron’s first
energy level.
(3.44) for the energy of the nth level of an electron localized inside of the potential
well with infinite barriers.
Since the wavenumber is equal to κ = 2π/λ
Br
, the condition (3.169) means
that the width of the potential barrier, L, contains an integer number of de Broglie
half-wavelengths, λ
Br
/2. Near values of the wavenumber of
κ
n
=
π
L
n +
1
2
(3.171)
we have maxima of the reflection coefficient and minima of the transmission
coefficient. The existence of oscillations in the coefficients R and D is the result
of the interference of waves reflected from the potential steps at the boundaries of
the rectangular potential barrier. The dependences of the transmission coefficient,
D,onκ L found for six values of the ratio E
1
/U
0
, namely 0, 1, 0.5, 0.2, 0.1, and
0.01, where E
1
= π
2
h
-
2
/(2m
e
L
2
), are shown in Fig. 3.9. We see that an increase
of the barrier height or an increase of the barrier width results in a decrease of the
amplitude of oscillations of the transmission coefficient, D. When the electron
energy, E, increases the transmission coefficient, D, quickly increases, tending
to unity, whereas the amplitude of oscillations decreases. The locations of the
maxima correspond to the resonance values of the energy, E
n
(Eq. (3.170)).
The corresponding expressions for the reflection and transmission coefficients
at E > U
0
for the potential step shown in Fig. 3.8 have the forms
R =
k − κ
k + κ
2
=
√
E −
√
E −U
0
√
E +
√
E −U
0
2
, (3.172)
D =
4kκ
(k + κ)
2
=
4
E(E −U
0
)
(
√
E +
√
E −U
0
)
2
. (3.173)

96 Layered nanostructures
1
0
E/ U
0
R
0.5
123
Figure 3.10 The
dependence of an
electron’s reflection
coefficient, R,onthe
barrier height, U
0
.
Figure 3.10 shows the dependence of the reflection coefficient, R,onthetotal
energy of an electron, E, for a given height of the potential step, U
0
. We see that
even when an electron moves above the potential step (E /U
0
> 1) the electron
still undergoes reflection. At E = U
0
reflection stays the same as at E < U
0
, i.e.,
R = 1.
Example 3.7. An electron traveling from the negative part of the x-axis is
incident on a rectangular potential step. The height of the potential barrier U
0
is
greater than the total energy of the electron E (see Fig. 3.8). Find the probability
of finding the electron “under the barrier” up to the distance a from the boundary
x = 0.
Reasoning. The probability density of an electron’s penetration under the barrier
depends on the coordinate x and can be written as
|ψ
2
(x )|
2
=
4k
2
A
2
1
(k
2
+ κ
2
)
e
−2κx
. (3.174)
The probability density, |ψ
2
(x)|
2
, of finding the electron at x > 0 exponentially
decreases with increasing x. If the distance from the barrier at which we can
find the electron is equal to a, then the total probability of finding the electron
between x = 0 and x = a is equal to
P =
a
0
|ψ
2
(x )|
2
dx =
2k
2
A
2
1
(k
2
+ κ
2
)κ
1 −e
−2κa
. (3.175)
Thus, with increasing a the total probability, P, of finding the electron at 0 ≤
x ≤ a increases, and at a →∞this probability tends to its limit:
P
max
=
2k
2
A
2
1
(k
2
+ κ
2
)κ
. (3.176)
T
3.5 Tunneling 97
Example 3.8. A rectangular potential barrier (Fig. 3.7) has a width L = 0.15
nm. Find the barrier height U
0
and the electron energy E for which the electron’s
total probability of transmission through the barrier is equal to P = 0.4 and the
coefficient D
0
from Eq. (3.156) is equal to unity.
Reasoning. Considered from the physical point of view, the probability P of
electron transmission through the potential barrier is equal to the transmission
coefficient D, which is defined by Eq. (3.156):
P = D = D
0
e
−2L
√
2m
e
(U
0
−E)/h
-
. (3.177)
From Eq. (3.157) and the condition D
0
= 1 we can find the ratio γ = E/U
0
:
16γ (1 −γ ) = 1. (3.178)
On solving this quadratic equation, we find two possible values of γ :
γ =
1
2
±
√
3
4
. (3.179)
Let us take the logarithm of Eq. (3.177). As a result we obtain
ln P =−
2L
h
-
2m
e
(U
0
− E). (3.180)
From the last equation, taking into account E = γ U
0
, we find the expression for
U
0
:
U
0
=
h
-
2
1 −γ
(ln P)
2
8m
e
L
2
. (3.181)
After the substitution of the given parameters we obtain two values of U
0
:
U
01
= 8.51 × 10
−19
J = 5.31 eV,
U
02
= 0.61 × 10
−19
J = 0.38 eV.
The corresponding values of the electron’s energy are
E
1
= 7.94 ×10
−19
J = 4.95 eV,
E
2
= 0.4 ×10
−19
J = 0.25 eV.
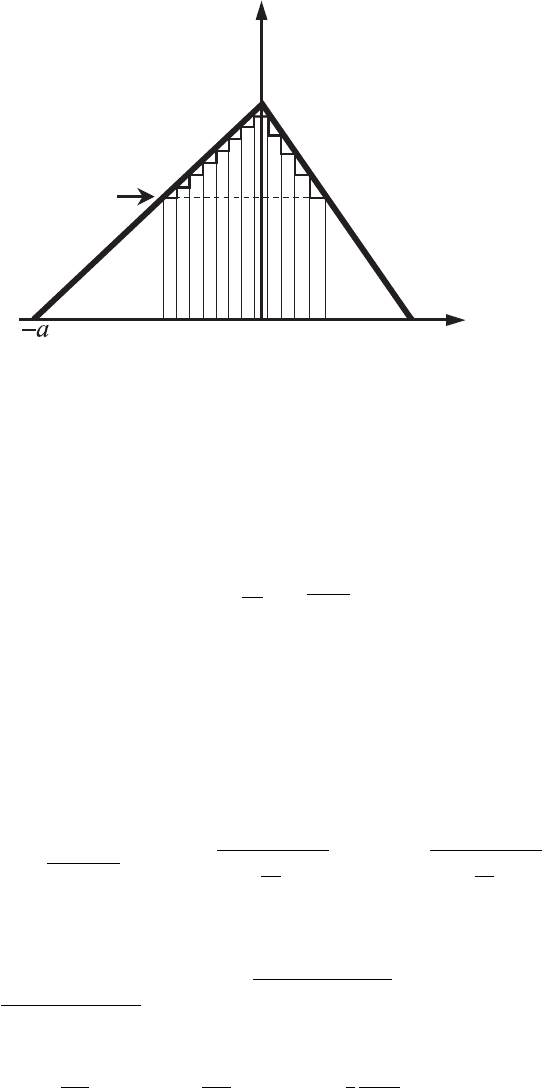
Example 3.9. Find the electron transmission probability through the potential
barrier of the following profile:
U (x ) =
0, x < −a,
U
0
(1 + x /a), −a ≤ x ≤ 0,
U
0
(1 − x /b), 0 < x ≤ b,
0, x > b,
(3.182)
where a and b are positive numbers. Assume that the electron is incident on the
barrier from the negative part of the x-axis and that the electron’s energy is less
than U
0
(E < U
0
).
98 Layered nanostructures
0
x
U
b
U
0
x
1
x
2
with energy E
Incident electron
Figure 3.11 An electron’s
transmission through the
triangular potential barrier
defined by Eq. (3.182).
Reasoning. Equation (3.156) for the probability of an electron’s transmission
through the rectangular potential barrier can be generalized for the case of a
potential barrier of arbitrary form. If the potential in the barrier region changes
slowly, i.e., its change is small at distances comparable to the de Broglie wave-
length, the potential of an arbitrary profile can be presented as a sum of a large
number of rectangular potentials (see Fig. 3.11). Then for the transmission coef-
ficient the following expression is valid:
D = D
0
e
−2
√
2m
e
x
2
x
1
√
U (x)−Edx/h
-
, (3.183)
where x
1
and x
2
are the points of the electron’s penetration under the poten-
tial barrier (see Fig. 3.11) and are defined from the solution of the following
equations:
U
0
(
1 + x /a
)
= E, x
1
=−
(
1 − E/U
0
)
a,
U
0
(
1 − x /b
)
= E, x
2
=
(
1 − E/U
0
)
b. (3.184)
Thus, to find D it is necessary to find the following integral:
I =
x
2
x
1
U (x ) − E dx =
0
x
1
U
0
+
U
0
a
x − E dx +
x
2
0
U
0
−
U
0
b
x − E dx.
(3.185)
In order to evaluate the integrals on the right-hand side of Eq. (3.185)itis
necessary to change variables: t =
√
U
0
+U
0
x/a − E in the first integral and
t =
√
U
0
−U
0
x/a − E in the second integral. As a result of integration and
taking into account the expressions for x
1
and x
2
, we obtain
I =
2a
3U
0
(U
0
− E)
3/2
+
2b
3U
0
(U
0
− E)
3/2
=
2
3
a + b
U
0
(U
0
− E)
3/2
. (3.186)
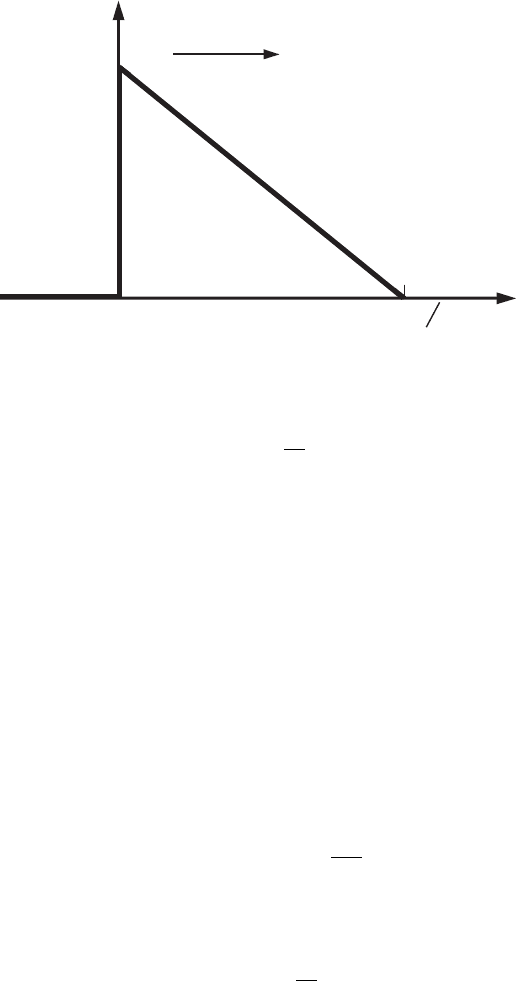
3.5 Tunneling 99
0
x
U
U
0
E
b =
(e|E|)U
0
metal vacuum
Figure 3.12 The triangular
potential barrier at the
metal–vacuum interface
under an applied electric
field, E.
On substituting the last expression into Eq. (3.183) we obtain
D = D
0
e
−4
√
2m
e
(a+b)(U
0
−E)
3/2
/(3h
-
U
0
)
. (3.187)
In practice very often a potential with one of the parameters, a or b,having
a value of zero is encountered. For example, at a = 0 the potential has the form
shown in Fig. 3.12. Such a potential is common for a metal–vacuum interface to
which an electric field, E, is applied. Without the electric field the interface of the
metal with vacuum is a rectangular potential step of finite height, i.e., U(x) = 0
at x < 0 and U(x) = U
0
at x ≥ 0. If an external electric field E is applied along
the electron’s direction of motion (perpendicular to the interface), then at x > 0
the electron’s potential energy becomes equal to
U (x ) = U
0
− e|E|x, (3.188)
and the coordinate for which U (b) = 0is
x = b =
U
0
e|E|
.
For this potential barrier the transmission probability for the electron is defined
by the expression
D = D
0
e
−4
√
2m
e
(U
0
−E)
3/2
/(3eh
-
|E|)
. (3.189)
It follows from the last equation that with increasing electric field the probability
of an electron’s transmission through the barrier rapidly increases. This is because
of the decrease of the barrier width.
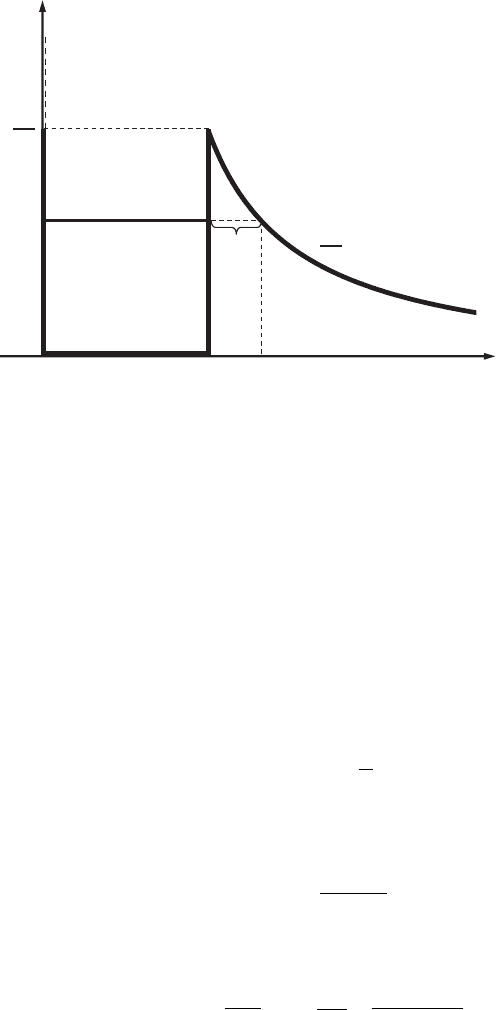
100 Layered nanostructures
0
x
E,U
L
U = ∞
a
a
L
x
x
0
d
E
Figure 3.13 Awellwith
the potential profile
(3.190).
Example 3.10. Find the dependence on energy of the probability of an electron
escaping a potential well through a potential barrier with the following profile:
U (x ) =
∞, x < 0,
0, 0 ≤ x ≤ L ,
α/x, x > L .
(3.190)
The graph of the potential well defined by Eq. (3.190) is shown in Fig. 3.13.
Reasoning. The probability of the electron escaping from the potential well is
defined by its transmission coefficient, D. Expression (3.156) was obtained for a
barrier of rectangular form. The main difference of the present problem from the
potential barrier with profile (3.141) is that the barrier width, d, now depends on
the energy, E , and, as follows from Eq. (3.190), it is equal to
d(E) = x
0
(E) − L =
α
E
− L , (3.191)
where the coordinate x
0
is the solution of the equation E = α/x. To calculate
the probability of an electron escaping from the well as a result of transmission
through the barrier, we have to use expression (3.183):
D = D
0
e
−2
x
0
L
√
2m
e
(
α/x−E
)
dx/h
-
, (3.192)
where the constant D
0
is close to unity and the upper limit of integration is the
coordinate x
0
. As a result of integration, we obtain the expression
D ≈ D
0
e
−2α
√
2m
e
/E
arccos
√
E/U
0
−
√
(E/U
0
)
(
1−E/U
0
)
/h
-
, (3.193)
where U
0
<α/L. In the case of a sufficiently high potential barrier, when U
0
E, this expression, taking into account that arccos(0) = π/2, transforms into the

3.7 Problems 101
simpler expression
D ≈ D
0
e
−πα
√
2m
e
/E/h
-
. (3.194)
From the last expression it follows that with increasing electron energy, E,the
transmission coefficient, D, increases. At E = U
0
, as follows from Eq. (3.193)
and from arccos(1) = 0, the transmission coefficient, D, is equal to unity.
3.6 Summary
1. The wavefunction of a free electron can be described by a de Broglie plane wave,
whose group velocity coincides with the electron velocity. The energy spectrum of
such an electron is continuous.
2. In the case of electron motion in a limited space the main important property of such
motion is the discreteness of the energy spectrum, i.e., quantization of electron energy.
All quantum states of an electron and the corresponding energy states in the case of
one-dimensional motion can be enumerated by one quantum number, n.
3. In a potential well with infinite barriers there is an infinite number of energy levels
that correspond to the stationary quantum states of an electron in the well. The energy
of corresponding levels is proportional to n
2
and the distance between levels is pro-
portional to 2n + 1. If we place the center of coordinates at the center of the well,
then the wavefunctions of an electron for quantum states with odd numbers of n are
symmetric functions of coordinate, and those for quantum states with even numbers
of n are antisymmetric functions of coordinate.
4. Higher electron energy corresponds to higher wavenumber. The lowest energy level
with n = 1 is called the ground state and it corresponds to the minimal electron energy.
However, this state is not the state of rest.
5. In confined quantum states the probability density of finding an electron, |ψ|
2
,has
maxima in the region of the potential well and exponentially decreases outside of the
well. This means that an electron can be outside of the well with a non-zero probability.
6. In a symmetric potential well with finite barriers there is always at least one energy
level, i.e., the energy level E
1
, which is lower than the barrier height U
0
.
7. An electron, just like any other quantum particle, can tunnel through a potential barrier
whose height is greater than the total energy of the electron. The probability of a particle
tunneling through a barrier exponentially decreases with increasing width and height
of the barrier and with increasing mass of the particle. The total energy of the particle
does not change during tunneling through the barrier.
3.7 Problems
Problem 3.1. Using the procedure of calculations of average values of physical
quantities, show that for a one-dimensional motion the operator of coordinate is
coordinate itself.
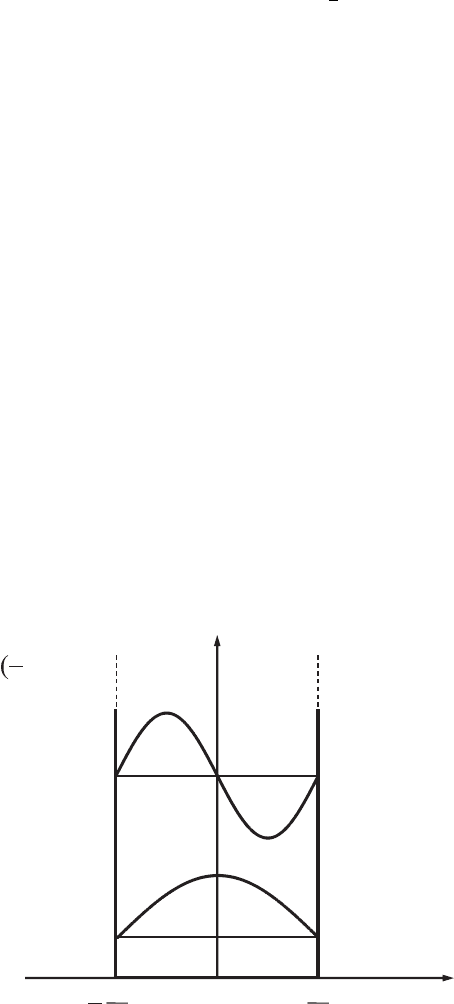
102 Layered nanostructures
Problem 3.2. A particle with mass m is moving in the region −L/2 ≤ x ≤ L/2.
The state of the particle is described by the wavefunction
ψ(x) = A exp
i
h
-
px
, (3.195)
where p is the particle’s momentum. Find the normalization constant, A, and
the average value of the particle’s kinetic energy, K .
Problem 3.3. An electron is in a one-dimensional rectangular potential well with
barriers of infinite height. Find the well width if the energy difference between
the fifth and fourth quantum states, E
5
− E
4
, is equal to the electron’s average
thermal energy at room temperature.
Problem 3.4. An electron is in a one-dimensional rectangular potential well
with barriers of infinite height. The width of the well is equal to L = 5nm.
Find the wavelengths of photons emitted during electronic transitions from the
excited states with quantum numbers n = 2, λ
21
, and n = 3, λ
31
, to the ground
state with n = 1. (Answer: λ
21
≈ 1.15 µm and λ
31
≈ 0.43 µm.)
Problem 3.5. An electron is in a one-dimensional rectangular potential well with
barriers of infinite height. Find the normalized wavefunctions and energy levels
if the width of the potential well is equal to L and the wavefunction is symmetric
with respect to the coordinate origin placed at the center of the well (see Fig. 3.14).
Problem 3.6. An electron is in a one-dimensional rectangular potential well with
barriers of infinite height and with well width equal to L as shown in Fig. 3.14.
Find the average values of the coordinate, the momentum projection, and the
squares of these magnitudes for an electron that is in quantum states described
by symmetric wavefunctions.
E
2
E
1
x
0
E
n
,U
ψ
1
U
L/2)=
∞
ψ
2
U(L/2)= ∞
L
2
L
2
Figure 3.14 A rectangular
symmetric potential well
with respect to the
coordinate origin with
barriers of infinite height.
