Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.


5.6 Problems 153
is subjected to a perturbation of the following form:
W (x) =
W
0
, b ≤ x ≤ L − b,
0, x < b, x > L − b.
Find the energy of the electron in stationary states, taking into account
corrections arising from the first-order perturbation theory.
Problem 5.4. Find the energy of the ground state of a one-dimensional harmonic
oscillator that is subjected to an anharmonic potential perturbation W (x) = γ x
3
.
Problem 5.5. A charged one-dimensional oscillator is placed in a perturbation
homogeneous electric field directed along the axis of oscillations. Find the
energy of the oscillator, taking into account the first two orders of correction of
perturbation theory. The charge of the particle is positive and equal to e and its
mass is equal to m.
Problem 5.6. The electron is in a spherically-symmetric potential well (4.41):
U (r) =
0, r ≤ a,
∞, r > a.
The unperturbed magnitudes of the electron energy in the well are E
nl
. Find, in
the first order of perturbation theory, the electron energy and wavefunction in the
magnetic field directed along the z-axis. The perturbation operator
ˆ
V is defined
as
ˆ
V =
ih
-
eB
2m
e
∂
∂θ
,
where θ is the polar angle in the plane perpendicular to the z-axis. Hint: use the
spherical coordinate system.
Problem 5.7. An electron, which is in a one-dimensional potential U(x) = βx
2
/2
(a linear harmonic oscillator), is placed in a homogeneous electric field. The
electric field is directed along the axis of oscillations and changes with time as
E(t) = E
0
e
−t
2
/τ
2
.
Before the electric field is turned on (t →−∞) the electron was in one of its
unperturbed states. Find, in the first order of perturbation theory, the probability
of electron excitation to the higher states at t →∞.
Problem 5.8. The motion of a plane harmonic oscillator takes place in the
xy-plane in the following potential:
U (x , y) =
β(x
2
+ y
2
)
2
.
Find, in the first order of perturbation theory, the splitting of the first excited
energy level of the oscillator under the perturbation potential W = γ xy.
154 Approximate methods of finding quantum states
Problem 5.9. Find, in the quasiclassical approximation, the probability of electron
transmission, P, through the potential barrier
U (x ) =
0, x < 0,
U
0
e
−x /b
, x ≥ 0,
for an electron whose energy is E < U
0
.
Chapter 6
Quantum states in atoms and molecules
The main characteristics of atoms and molecules are their structure and their
energy spectrum. Under the term structure of an arbitrary particle we usually
understand the size and the distribution of its mass and its charge in space. In
quantum physics such a distribution is defined by the square of the modulus of
the wavefunction of a particle. The particle itself may consist of a system of
other particles that are bound by a certain type of coupling. For example, an atom
consists of a nucleus and a system of interacting electrons, a molecule consists
of a system of interacting atoms, and so on. If we consider an atom, the nucleus
of the atom is assumed to be at rest (the so-called adiabatic approximation). This
assumption can be made because the nucleus has a much larger mass than that of
an electron (a proton’s mass is 1836 times larger than the mass of an electron).
Then, the square of the modulus of the wavefunction, |ψ(r
1
, r
2
,...,r
n
)|
2
,for
the system of electrons defines the probability density of finding the jth electron
at the point r
j
. Graphically it is very convenient to depict |ψ |
2
in the form
of an electron cloud, which can be considered as an averaged distribution of
matter added to the mass of the nucleus located at the center of an atom. Since
electrons have not only mass, m
e
, but also the electric charge, −e, the electron
cloud should be considered as the averaged density of the negative electric charge,
ρ
e
=−e|ψ(r
1
, r
2
,...,r
n
)|
2
, which compensates for the nucleus’ positive charge.
The current chapter is devoted to the study of the wavefunction and the geometry
of the electron cloud, as well as the energy spectra of the simplest atoms and
molecules.
6.1 The hydrogen atom
6.1.1 An electron in a Coulomb potential
As has already been shown, the de Broglie wavelength of an electron in an atom is
comparable to the atomic size. Therefore, we cannot neglect the wave properties
of the electron and we have to describe its behavior in an atom on the basis of
the Schr¨odinger equation.
155
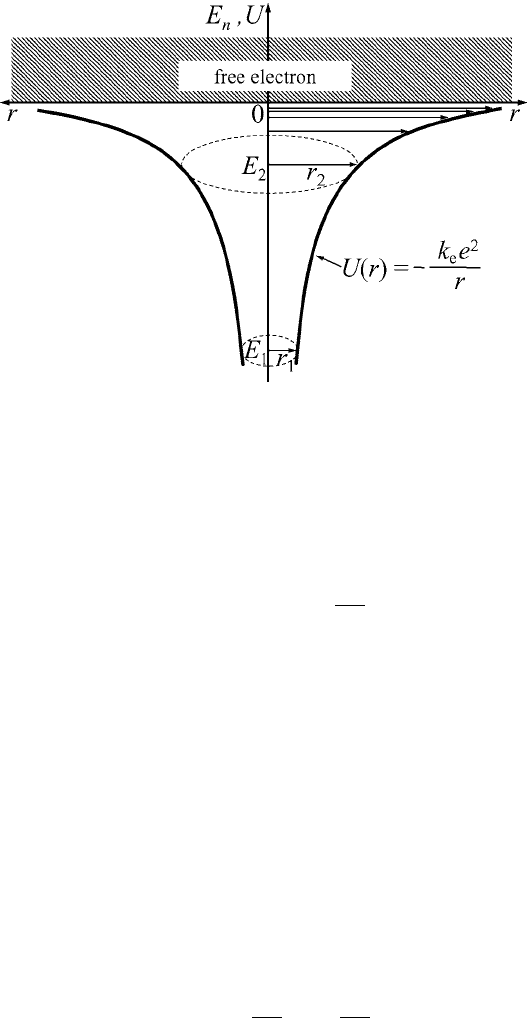
156 Quantum states in atoms and molecules
Figure 6.1 Quantization of
an electron in the
potential well U (r).
The hydrogen atom is the simplest atomic system, where the confined electron
with electric charge, −e, moves in the spherically-symmetric electrostatic field
of a positively charged nucleus (proton) (the charge of the proton is +e). The
potential energy of an electron in such a system is defined by its Coulomb
interaction with the nucleus and is equal to
U (r) =−
k
e
e
2
r
, (6.1)
where r is the distance between the electron and the nucleus. The nucleus, in
the first order of approximation, can be considered as a point (the proton size
a
p
≈ 10
−15
m while the distance between the electron and the proton is of the
order of 10
−10
m =1
˚
A). The motion of an electron in the potential (6.1) can
be considered to occur in a spherically-symmetric well of unlimited size (see
Fig. 6.1). However, the size of the atom itself, i.e., the average diameter of an
electron orbit in a stationary state, is finite and is of the order of 10
−10
m.
For the description of possible electron quantum states in an atom we will
first consider the hydrogen atom (H) and assume that its nucleus is motionless.
Because of the large difference between proton and electron masses, m
p
and m
e
(m
p
/m
e
= 1836), their center of mass coincides with the proton’s center of mass.
The stationary Schr
¨
odinger equation (4.26) in the case of the potential defined
by Eq. (6.1) can be written as
∇
2
ψ(r) +
2m
e
h
-
2
E +
k
e
e
2
r
ψ(r) = 0. (6.2)
Since the potential within which the electron is confined is spherically-symmetric,
it is convenient to seek the solution of Eq. (6.2) in spherical coordinates, whose

6.1 The hydrogen atom 157
origin (r = 0) coincides with the center of mass of the nucleus. In such a coordi-
nate system the electron wavefunction will depend on the distance of the electron
from the atom’s center, r , as well as on the azimuthal and polar angles, ϕ and θ,
respectively.
We will write the wavefunction for Eq. (6.2) in the form of a product of
two functions, as we have done already in the case of electron motion in a
spherically-symmetric potential well (see Eq. (4.27)):
ψ(r,ϕ,θ) = X(r)Y (ϕ, θ). (6.3)
According to Eq. (4.28), for a spherically-symmetric potential the function
Y
lm
(ϕ, θ) is the eigenfunction of the operator
ˆ
L
2
with the eigenvalues of this
operator equal to l(l + 1) h
-
2
, where l is the orbital quantum number. This quan-
tum number can have values l = 0, 1, 2,... and the orbital magnetic quantum
number is equal to m = 0, ±1,...,±l. The forms of several functions Y
lm
(ϕ, θ)
for l = 0, 1, 2 and m = 0, ±1, ±2 are given by the relations (4.37).
Let us substitute the wavefunction (6.3) into Eq. (6.2) and separate variables.
As a result we find the following equation for the radial function, X(r):
1
r
2
d
dr
r
2
dX
dr
+
2m
e
h
-
2
E −
l(l + 1) h
-
2
2m
e
r
2
+
k
e
e
2
r
X = 0. (6.4)
The solution of this equation depends on two quantum numbers, l and n, and can
be written as
X
nl
(ρ) = ρ
l
e
−ρ/n
n−l−1
j=0
a
j
ρ
j
, (6.5)
where we introduced the variable ρ = r/r
1
, with
r
1
=
h
-
2
k
e
m
e
e
2
= 0.53 × 10
−10
m (6.6)
the radius of the first Bohr orbit in the hydrogen atom for the ground state. The
distinctive feature of Eq. (6.5) is that it has a finite number of terms a
j
ρ
j
.Only
in this case will the exponential factor in Eq. (6.5) provide the vanishing of the
square of the modulus of the wavefunction at infinity, i.e., at ρ →∞.
The principal quantum number, n, which defines the energy state of an electron
in an atom, can take on the values n = 1, 2, 3,...The total energy of the electron
is quantized (see Fig. 6.1) and is equal to
E
n
=−
k
2
e
m
e
e
4
2h
-
2
n
2
=
E
1
n
2
, (6.7)
where E
1
is defined as
E
1
=−
k
2
e
m
e
e
4
2h
-
2
=−13.6eV. (6.8)
Here, E
1
is the energy of the electron ground state in the hydrogen atom (i.e., the
electron energy for the first Bohr orbit). The magnitude |E
1
|=13.6eVisthe

158 Quantum states in atoms and molecules
ionization energy of the hydrogen atom. This energy is required to transfer the
electron from the ground energy state, E
1
(shown schematically in Fig. 6.1), to
the continuous energy spectrum (denoted by “free electron” in Fig. 6.1).
The coefficients a
j
of the power series (6.5)for j > 0 can be found from the
following relation (we will not show here how to derive this relation because of
the complexity of the derivation):
a
j+1
= 2a
j
√
( j +l + 1) −1
( j + l + 2)( j + l + 1) −l(l + 1)
. (6.9)
Here, = E/E
1
is a positive dimensionless parameter for the confined states of
an electron, i.e., for the region of the discrete spectrum with E < 0. Relation
(6.9) is called recursive since it allows us to find the coefficient a
1
knowing the
coefficient a
0
, to find the coefficient a
2
knowing the coefficient a
1
, and so on.
The coefficient a
0
is found from the normalization of the total wavefunction of
the hydrogen atom’s ground state, i.e.,
∞
0
2π
0
π
0
|
ψ
100
(r,ϕ,θ)
|
2
r
2
sin θ dr dϕ dθ = 1. (6.10)
It follows from Eq. (6.5) that the last term of the power series has the index
j = n − l − 1. Then, the coefficient a
j+1
with j = n − l − 1 must be equal
to zero. According to Eq. (6.9), a
j+1
is equal to zero if the numerator in Eq. (6.9)
is equal to zero:
√
( j +l + 1) −1 = 0, or n
√
− 1 = 0. (6.11)
Thus,
n
√
= 1, i.e., n
E/E
1
= 1. (6.12)
From the last expression we get Eq. (6.7). The solution of Eq. (6.4) gives us the
eigenfunctions, X, and the corresponding eigenvalues, E. Equation (6.5)shows
that eigenfunctions X
nl
depend on l for any particular n. In the general case
each specific eigenvalue, E
nl
, must correspond to a certain eigefunction, X
nl
.
However, the solution of Eq. (6.4) gives a different result and the eigenvalue
E depends solely on n and does not depend on l at all (see Eq. (6.7)). Since
the total energy, E
n
, does not depend on the orbital quantum number, l, the total
energy, E
n
, corresponds to several radial wavefunctions X
nl
, i.e., the energy level
E
n
is degenerate. We have considered so far an individual hydrogen atom and
determined the energy levels for a single electron. If this hydrogen atom were to
interact with other atoms or were placed in an external electric or magnetic field
then this degeneracy would be lifted and the total energy E
nl
would depend on
the quantum number l as well.
For a given principal quantum number, n, the orbital quantum number, l, can
take on the following values:
l = 0, 1, 2,...,n − 1.

6.1 The hydrogen atom 159
Table 6.1. Notation of different quantum states
Quantum number l 012345
Symbol of state s p d f g h
The orbital magnetic quantum number, m, for each value of l can take on 2l + 1
values, and the total energy E
n
depends only on the principal quantum number,
n. Thus, all the electron states with n > 1 are degenerate, i.e., more than one
eigenfunction corresponds to the nth energy state. The order of degeneracy, g,is
defined by the number of different electron states for a given energy E
n
:
g =
n−1
l=0
(2l + 1) = 1 +3 +5 +···+(2n − 1) = n
2
. (6.13)
Thus, for the state described by the wavefunction ψ
100
the order of degeneracy is
equal to unity and therefore the energy E
1
is non-degenerate. For the energy E
2
,
g is equal to four and corresponds to four different electron states in the atom
described by the wavefunctions
ψ
200
,ψ
210
,ψ
21−1
,ψ
21+1
.
As will be shown later, the order of degeneracy of the nth energy level is equal
to g = 2n
2
because the electron has intrinsic angular momentum (spin), which
can take on two values (we will talk about spin in Section 6.3).
6.1.2 Symbols defining states and probability density
Different states of electrons in atoms, which correspond to different orbital
quantum numbers, l, are denoted by lower-case Latin letters. Table 6.1 shows
the correspondence of numbers and letters. When considering energy states we
usually talk about s-states (or s-electrons) for l = 0, p-states (or p-electrons) for
l = 1, and so on. In this notation of a quantum state the value of the principal
quantum number, n, is given before the symbol of the state with a given orbital
quantum number, l. Let us write the first four groups of states for the hydrogen
atom: (1) 1s; (2) 2s, 2p; (3) 3s, 3p, 3d; and (4) 4s, 4p, 4d, 4f (see Table 6.1). Thus,
an electron in state 4f has n = 4 and l = 3. The wavefunctions ψ
nlm
(r,ϕ,θ)for
the first two groups are given in Table 6.2.
The probability distribution of an electron’s location in an atom in the corre-
sponding stationary state is defined by the magnitude |ψ
nlm
|
2
. Its spatial distri-
bution tells us about the form of the electron cloud.
Taking into account that an electron possesses not only mass but also charge,
the parameter
ρ
e
(r) =−e
|
ψ
nlm
|
2
(6.14)
can be interpreted as the electron charge density. The spatial motion of an electron
is equivalent to the existence of a current. The density of this current can be found
160 Quantum states in atoms and molecules
Table 6.2. The wavefunctions ψ
nlm
(r,ϕ,θ) for the first two
groups of states (here, Z is the atomic number)
Wavefunction Expression State
ψ
100
1
√
π
Z
r
1
3/2
e
−ρ
1s
ψ
200
1
4
√
2π
Z
r
1
3/2
(2 −ρ)e
−ρ/2
2s
ψ
210
1
4
√
2π
Z
r
1
3/2
ρe
−ρ/2
cos θ 2p
ψ
21−1
1
8
√
π
Z
r
1
3/2
ρe
−ρ/2
sin θ e
−iϕ
2p
ψ
21+1
1
8
√
π
Z
r
1
3/2
ρe
−ρ/2
sin θ e
iϕ
2p
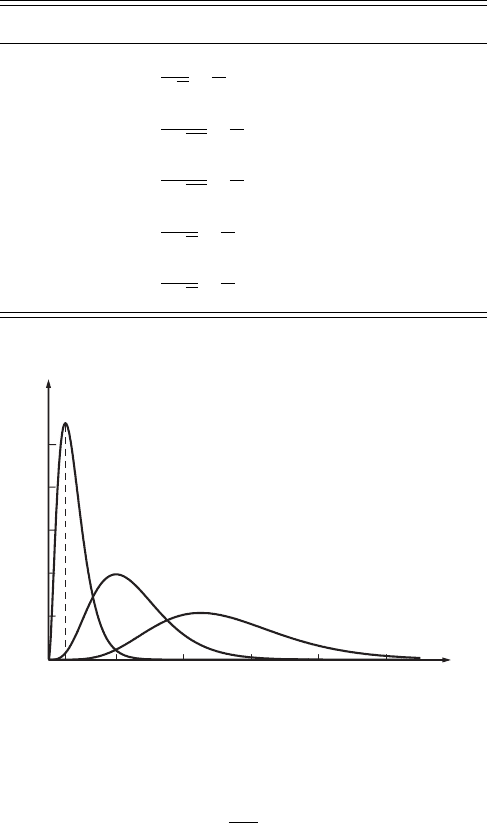
048121620
r
2
X
nl
2
(a.u.)
r/r
1
1s
2p
3d
1
0.1
0.2
0.3
0.4
0.5
Figure 6.2 Distributions of
the radial functions r
2
X
2
nl
for the 1s, 2p, and 3d
quantum states of the
hydrogen atom.
if we multiply the probability current density (2.190) in a given state by the charge
of the electron:
j
e
=−e
ih
-
2m
e
(ψ
nlm
∇ψ
∗
nlm
− ψ
∗
nlm
∇ψ
nlm
). (6.15)
In the 1s-state the orbital quantum number, l, and angular momentum, L,are
equal to zero. Therefore, the electron cloud in this state is spherically symmetric.
From the classical point of view this corresponds to electron motion only along
the radial direction – along r. The electron would have to cross the region
occupied by the nucleus, which is impossible in classical physics. To make this
result of quantum theory more understandable, let us consider the probability
of finding the electron within the volume dV :
dP =|ψ
nlm
|
2
dV.

6.1 The hydrogen atom 161
1s 2p, m = 0 2p, m = ±1
x
y
z
y
z
y
x
Figure 6.3 Apictorial
representation of the
electron clouds, |ψ
nlm
|
2
,
for the 1s and 2p states in
the hydrogen atom.
Let us choose a spherical shell of radius r, width dr, and volume dV = 4πr
2
dr.
Then, for the 1s-shell the magnitude
dP = 4πr
2
|ψ
100
|
2
dr (6.16)
defines the probability of finding an electron in this shell. The probability density,
dP/dr, is the probability of finding an electron in the spherical shell around the
radius r with width equal to unity. The radial dependences of r
2
X
2
nl
of the three
states 1s, 2p, and 3d for a hydrogen atom are shown in Fig. 6.2. We see that the
maximum probability density for the quantum state 1s corresponds to r = r
1
,
which coincides with the radius of the first Bohr orbit of the hydrogen atom. As
r goes to zero the probability density, dP/dr, also tends to zero, which means
that the electron is absent from the region occupied by the nucleus. The electron
clouds, |ψ
nlm
|
2
, corresponding to the 1s and 2p states in the hydrogen atom are
shown in Fig. 6.3. Note that |ψ
21±1
|
2
has a doughnut shape and Fig. 6.3 shows it
from the top.
Example 6.1. For the ground state of the hydrogen atom find the average, mean-
square, and most probable distance between the electron and the nucleus.
Reasoning. The wavefunction of the ground state (n = 1, l = 0, m = 0) for the
hydrogen atom (Z = 1) is given by the expression
ψ
100
(r) =
1
πr
3
1
e
−r/r
1
(6.17)
(see Table 6.2). The average distance of an electron from the nucleus according to
the quantum-mechanical definition of the average value of the physical magnitude
(see Eq. (2.173)) can be obtained as follows:
r
=
∞
0
r
|
ψ
100
|
2
4πr
2
dr =
4
r
3
1
∞
0
r
3
e
−2r/r
1
dr. (6.18)

162 Quantum states in atoms and molecules
In order to calculate the integral, let us change variables from r to x:
x =
2r
r
1
. (6.19)
As a result the integral takes the form
r
=
4
r
3
1
r
1
2
4
∞
0
x
3
e
−x
dx. (6.20)
Taking into account that
∞
0
x
n
e
−x
dx = n!, (6.21)
we get the final result
r
=
3r
1
2
. (6.22)
In order to find the mean-square distance between the electron and the nucleus,
let us find first the following magnitude:
r
2
=
∞
0
r
2
|
ψ
100
|
2
4πr
2
dr =
4
r
3
1
∞
0
r
4
e
−2r/r
1
dr = 3r
2
1
. (6.23)
Thus, the mean-square distance, r
ms
, is equal to
r
ms
=
r
2
=
√
3r
1
. (6.24)
Note that the integral in Eq. (6.23) was evaluated by using the same procedure
as for Eq. (6.18). To find the most probable distance, r
mp
, of an electron from the
nucleus, it is necessary to equate the derivative of the probability density defined
by Eq. (6.16) to zero:
d
dr
dP
dr
=
d
dr
4πr
2
1
πr
3
1
e
−2r/r
1
=
8
r
3
1
re
−2r/r
1
1 −
r
r
1
= 0. (6.25)
From Eq. (6.25) it follows that r
mp
= r
1
. This distance corresponds to the maxi-
mum of the probability density, dP/dr.
6.1.3 Orbital magnetic moment and orbital magnetic
quantum number
Since an electron is a charged particle, the current, I
e
, must be connected with
the electron motion around the atomic nucleus. The motion of an electron with
velocity v along a circular orbit of radius r with period T is equivalent to a
circular current, whose magnitude is given by the expression
I
e
=−
e
T
=−
ev
2πr
, (6.26)
where v is the orbital velocity of the electron. The minus sign in Eq. (6.26) indi-
cates that the electron velocity and current have opposite directions because
