Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.


6.4 Many-electron atoms 173
lines due to spin–orbit coupling is called the fine structure of spectral lines, and
the splitting caused by the interaction between the intrinsic magnetic moments
of the electron and the nucleus is called the hyperfine structure of spectral lines.
6.4 Many-electron atoms
6.4.1 The Pauli exclusion principle and quantum states
Let us assume that in many-electron atoms the electron–electron interaction
can be neglected. This means that each electron moves independently in the
spherically-symmetric field of the nucleus:
U (r) =−
Zk
e
e
2
r
, (6.62)
where Z is the atomic number. In this approximation the energy of electron
stationary states in a many-electron atom should have the same form as the
energy (6.7) of the hydrogen atom:
E
n
= E
1
Z
2
n
2
=−13.6
Z
2
n
2
eV. (6.63)
If all electrons were in the state with n = 1, then the atom’s ionization energy
should be defined by the expression
E
i
= 13.6Z
2
eV.
Thus, for a mercury atom, Hg, with Z = 80 the ionization energy should be
equal to E
i
= 8.7 × 10
4
eV, whereas the correct value of the ionization energy
is E
i
= 10 eV. Therefore, such a big difference between the calculated and
experimental ionization energies shows that the approximation of non-interacting
electrons is not valid. The drawbacks of such an approximation are determined
by the fact that we do not take into account two important properties of the
behavior of a system of electrons in a many-electron atom.
First of all, when the atomic number, Z, increases, and therefore the number
of electrons increases, the filling of energy states by electrons occurs as follows.
As a new electron is added it does not occupy the ground state but instead
occupies the lowest of the available unoccupied energy states. The formation of
many-electron atoms happens according to one of the main principles of quantum
physics – the Pauli exclusion principle, according to which a particular state,
which is characterized by a given set of quantum numbers (in the case of an atom
the quantum numbers are n, l, m, m
s
), may be occupied only by one electron.
Because of this principle, the outer electrons, for example, in a mercury (Hg)
atom can occupy only states with n = 6.
Second, it is necessary to take into account that the energy levels in this
approximation are strongly degenerate, because their position depends only on
the principal quantum number, n, and does not depend on the other quantum
numbers. Electron repulsion lifts this degeneracy, and as a result the energy

174 Quantum states in atoms and molecules
levels depend not only on the principal quantum number, n, but also on the orbital
quantum number, l. Electrons with the same quantum number n may be grouped
in shells, and electrons with a given n, which have the same quantum number l,
may be grouped in subshells. Thus, the filling of energy levels by electrons in a
many-electron atom is reduced to their distribution over the shells and subshells.
Therefore, for n = 1 the values of the other quantum numbers are l = 0, m = 0,
and m
s
=±1/2. For the n = 1 shell, which has only a single subshell with l = 0,
we have that only two electrons with opposite spin projections may occupy it.
At n = 2, two subshells can exist, with l = 0 and l = 1. The quantum numbers
m = 0 and m
s
=±1/2 correspond to the first subshell, and two electrons may
occupy it. The second subshell, with l = 1, corresponds to the quantum numbers
m =−1, 0, 1 and each value of m corresponds to m
s
=±1/2. As a result this
subshell may contain six electrons. Therefore, the total number of electrons in
the shell with n = 2 is equal to 8. In the general case the capacity of the subshell
withagivenl is equal to 2(2l + 1) and for l = 0, 1, 2, 3, and 4 the capacity of
subshells is equal to 2, 6, 10, 14, and 18 electrons, respectively. Correspondingly,
the capacity of the shell is equal to
n−1
l=0
2(2l + 1) = 2[1 +3 +5 +···+(2n − 1)] = 2n
1 + (2n − 1)
2
= 2n
2
. (6.64)
For the principal quantum numbers n = 1, 2, 3, 4, and 5 the capacity of the shells
is equal to 2, 8, 18, 32, and 50, respectively. Thus, in accordance with the Pauli
exclusion principle, in many-electron atoms, after filling shells with lower n,
the electrons are distributed over the shells and subshells with increasing n and
l. The properties of atoms with closed shells drastically differ from those of
atoms with half-filled shells. It is difficult to remove an electron from atoms
with completely filled shells (for example, helium (He) or neon (Ne) with Z = 2
and 10, respectively). Atoms with one extra electron (for example, lithium (Li)
and sodium (Na) with Z = 3 and 11, respectively) easily lose their outermost
electron. Atoms lacking one electron for completion of their outer shell (for
example, hydrogen (H) and fluorine (F) with Z = 1 and 9, respectively) easily
acquire one more electron. In the next section we will consider the classification
of the quantum states of many-electron atoms.
6.4.2 The total angular momentum of an atom
For many-electron atoms the angular momenta and spin angular momenta are
defined by the expressions
M
L
= h
-
L(L + 1), (6.65)
M
S
= h
-
S(S + 1), (6.66)
where L and S are quantum numbers for the atomic angular momenta and spin
angular momenta. The quantum number L is always either an integer or equal

6.4 Many-electron atoms 175
Table 6.4. Spectroscopic classification of quantum states
L 012 34 5
SymbolSPDFGH
to zero. The quantum number S for an even number of electrons is an integer,
whereas for an odd number of electrons it is a half-integer.
The total angular momentum of an atom, M
J
, is a result of quantum-
mechanical summation of momenta, which depends substantially on the type
of interaction between the momenta. For the most common normal coupling the
angular momenta are added to give the resulting angular momentum, M
L
, and
the spin angular momenta to give the resulting spin angular momentum, M
S
.The
interaction of these momenta defines the total angular momentum of an atom,
M
J
, whose magnitude is equal to
M
J
= h
-
J (J + 1). (6.67)
Here, the quantum number J takes on one of the following values:
J = L + S , L + S − 1 , ..., |L − S|. (6.68)
This number takes on integer values if S is an integer, i.e., for an even number
of electrons in an atom, and it takes on half-integer values if S is a half-integer,
i.e., for an odd number of electrons.
The projection of the total angular momentum of an atom onto the chosen
direction is defined by the formula of space quantization:
M
Jz
= m
J
h
-
, (6.69)
where the quantum number m
J
takes on 2J + 1 values:
m
J
=−J, −J + 1, ..., J − 1, J. (6.70)
For the designation of different quantum states of many-electron atoms the
spectroscopic classification of quantum states is used. This consists of the follow-
ing: each value of the total orbital quantum number, L, corresponds to a certain
capital letter of the Latin alphabet (see Table 6.4). As a superscript to the left of
the Latin letter the multiplicity of states, which is equal to 2S + 1 and defines the
number of sublevels to which the level can be split, is written. The multiplicity
is related to the spin quantum number S (do not confuse this with the symbol of
state with L = 0). As a subscript to the right of the symbol the quantum number
J , which defines the total angular momentum of the atom, is written. Thus, the
ground state of the helium atom, He, is designated by the symbol
1
S
0
,forwhich
S = 0, L = 0, and J = 0. In an atom with two electrons, two types of states
with S = 0 (the electron spins are anti-parallel) and with S = 1 (electron spins
are parallel) may be possible. In the first case J = L = 0 and the multiplicity is
equal to 2S + 1 = 1. In the second case the multiplicity is equal to 2S + 1 = 3
176 Quantum states in atoms and molecules
Table 6.5. Symbolic representation of quantum states of
many-electron atoms
S 000 1 1 1
L 012 0 1 2
J 0 1 2 1,0 2,1,0 3,2,1
Symbol
1
S
0
1
P
1
1
D
2
3
S
1
,
3
S
0
3
P
2
,
3
P
1
,
3
P
0
3
D
3
,
3
D
2
,
3
D
1
Table 6.6. The total number of states in shells
Symbol of the shell K L M N O
Quantum number n 12 3 4 5
Total number of states 2n
2
2 8 18 32 50
and three values of J are possible: L + 1, L, and |L − 1|. The designations of
the above-mentioned states are listed in Table 6.5.
According to the Pauli exclusion principle and Eq. (6.64), the quantum state
with a given principal quantum number, n, corresponds to 2n
2
states with different
values of the quantum numbers l, m, and m
s
. Those electrons in an atom with
the same quantum number n form the so-called shell. In correspondence to the
values of n, the shells are designated by capital Latin letters. The first five atomic
shells are shown in Table 6.6. The shells are divided into subshells, which differ
only by orbital quantum number, l. The number of different states in a subshell
with different quantum numbers m and m
s
is equal to 2(2l + 1). The subshells
with l = 0, 1, 2, 3,...include the quantum states 2, 6, 10, 14, ..., respectively,
and may be designated as
1s, 2s 2p, 3s 3p 3d, 4s 4p 4d 4f,..., (6.71)
where the number designates the quantum number n, i.e., the affiliation with the
corresponding shell. Completely filled shells and subshells have L = 0, S = 0,
and J = 0. Each subsequent atom is built up from the previous one by changing
the charge of the nucleus by adding a positive elementary charge, e, and by
adding one electron, which according to the Pauli exclusion principle fills the
next vacant state with the minimal energy. Thus, the next element after hydrogen
(H), namely helium (He), fills the K shell by adding a second 1s-electron with
spin opposite to the spin of the first electron. The third element, lithium (Li), has
in addition to a filled K shell one more electron in the 2s subshell. The fourth
element, beryllium (Be), has a filled K shell and 2s subshell.
The distribution of electrons over the states is called the electron configuration.
Thus, the electron configuration of the ground state of silicon (Si), which contains
6.4 Many-electron atoms 177
14 electrons, has the form
1s
2
2s
2
2p
6
3s
2
3p
2
.
This means that the K and L shells and 3s subshell are completely filled, and
there are two electrons in the 3p subshell.
6.4.3 Hund’s rules
The fifth element of the Periodic Table of the elements, boron (B), has in its
2p subshell one electron with L = 1 and S = 1/2. These values correspond to
values of the quantum number J = 3/2 and 1/2, i.e., to the two quantum states
of an atom
2
P
3/2
and
2
P
1/2
. Which of these states is the ground state is decided
by Hund’s rules, which define the algorithm for the filling of subshells.
1. A given electron configuration has the smallest energy in the state with the largest
possible S and L, which corresponds to the value of spin, S.
2. The quantum number J is equal to J =|L − S| if the subshell is less than half-filled,
and to J = L + S if the subshell is more than half-filled.
Taking into account the second of Hund’s rules, it is clear that for the boron
atom (B) the ground state should be
2
P
1/2
since with one electron in the 2p
subshell the largest possible m
S
and m
L
are m
S
= 1/2 and m
L
= 1. Therefore,
S = 1/2, L = 1, and J =|L − S|=1/2. In Table 6.7 the result of the algorithm
for filling shells and subshells with electrons is shown for the first 36 elements.
6.4.4 Optical spectra
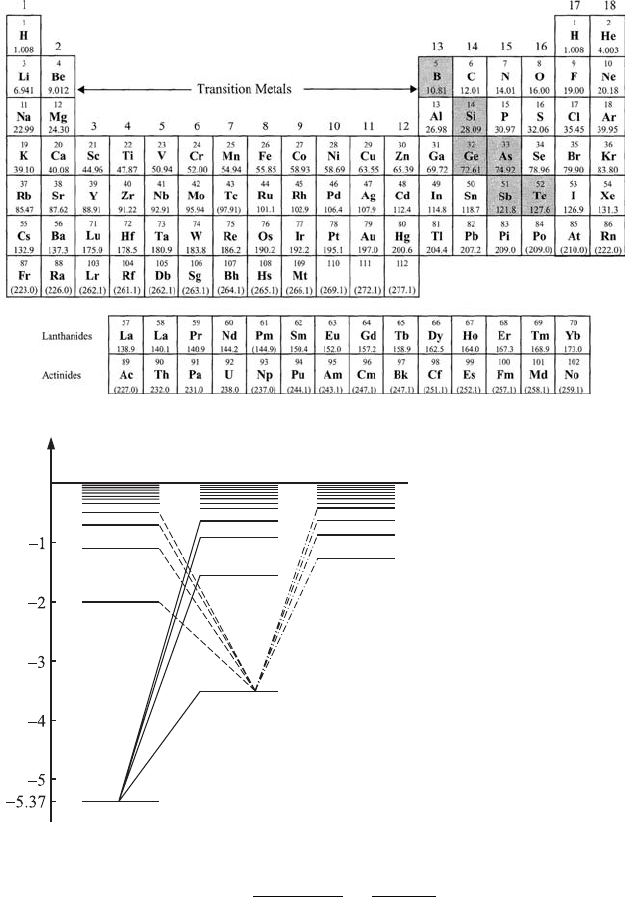
Atoms of the alkali elements, i.e., metals of the first group (Li, Na, and K) of
the Periodic Table of the elements (see Fig. 6.11), have one electron in their
outer orbits, which is called the valence electron. These atoms have the simplest
emission spectra. Their spectra, which are similar to the hydrogen-atom spectrum,
consist of a great number of spectral lines. The systematization of these lines
allowed one to group them in series, each of which is related to the transition of
the excited atom to a certain energy level.
An atom of an alkali element has Z electrons, of which Z − 1formafairly
strong core with the nucleus. The outer (valence) electron moves in the electric
field of this core and is weakly connected to it. Therefore, atoms of the alkali
elements can be considered as hydrogen-like atoms. However, in contrast to the
hydrogen atom, where the electron moves in a spherically-symmetric potential, in
the alkali elements the field of the core is not completely spherically symmetric.
The inner electrons deform the electron core and break the spherical symmetry
of its field. The violation of the potential’s symmetry leads to bound states whose
energy depends not only on the principal quantum number, n, as in the case of
the hydrogen atom, but also on the orbital quantum number, l. The solution of

178 Quantum states in atoms and molecules
Table 6.7. The filling of shells and subshells according to Hund’s rules
Number (Z ) Element K (1s) L (2s 2p) M (3s 3p 3d) N (4s 4p) Ground state
1H1
2
S
1/2
2He2
1
S
0
3Li21
2
S
1/2
4Be22
1
S
0
5B221
2
P
1/2
6C222
3
P
0
7N223
4
S
3/2
8O224
3
P
2
9F225
2
P
3/2
10 Ne 2 2 6
1
S
0
11 Na 2 2 6 1
2
S
1/2
12 Mg 2 2 6 2
1
S
0
13 Al 2 2 6 2 1
2
P
1/2
14 Si 2 2 6 2 2
3
P
0
15 P 2 2 6 2 3
4
P
3/2
16 S 2 2 6 2 4
3
P
2
17 Cl 2 2 6 2 5
2
P
3/2
18 Ar 2 2 6 2 6
1
S
0
19 K 2 2 6 2 6 1
2
S
1/2
20 Ca 2 2 6 2 6 2
1
S
0
21 Sc 2 26 261 2
2
D
3/2
22 Ti 2 26 262 2
3
F
2
23 V 2 26 263 2
4
F
3/2
24 Cr 2 26 264 1
7
S
3
25 Mn 2 26 265 2
6
S
5/2
26 Fe 2 26 266 2
5
D
4
27 Co 2 26 267 2
4
F
9/2
28 Ni 2 26 268 2
3
F
4
29 Cu 2 26 2610 1
2
S
1/2
30 Zn 2 26 2610 2
1
S
0
31 Ga 2 26 2610 21
2
P
1/2
32 Ge 2 26 2610 22
3
P
0
33 As 2 26 2610 23
4
S
3/2
34 Se 2 26 2610 24
3
P
2
35 Br 2 26 2610 25
2
P
3/2
36 Kr 2 26 2610 26
1
S
0
6.4 Many-electron atoms 179
Figure 6.11 The Periodic
Table of the elements.
5
E
, eV
l =2
l =1
l =0
6
4
3
2
2
3
3
4
4
5
5
6
6
s
p
d
0
Figure 6.12 A schematic
representation of energy
levels and transitions for a
lithium atom, Li: the
principal series is shown
by solid lines, the sharp
series by dashed lines,
and the diffuse series by
dash–dotted lines.
the Schr¨odinger equation gives the following expression for this energy:
E
nl
=−
k
2
e
m
e
e
4
2h
-
2
(n + b
l
)
2
=
E
1
(n + b
l
)
2
, (6.72)
where b
l
, which depends on l, is the quantum correction to the formula for the
energy levels in a hydrogen atom (see Eq. (6.7)).
The dependence of the electron energy in the alkali elements on the orbital
quantum number, l, is the principal difference of the energy spectrum of the alkali
elements from the spectrum of the hydrogen atom. This dependence means that
for the alkali elements the degeneracy of the orbital quantum number, l, is lifted.
Figure 6.12 shows schematically the energy levels and the corresponding tran-
sitions for lithium (Li). The ground state for lithium is the state 2s, for which n = 2
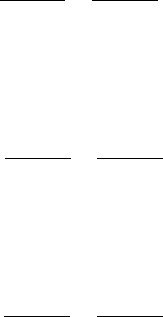
180 Quantum states in atoms and molecules
because the state with n = 1 is occupied by two electrons, which form the lithium
core. Three series of spectral lines of the lithium atom are shown in Fig. 6.12:
principal, sharp, and diffuse. The lines of the principal series correspond to the
transitions from p-levels to the 2s-level. The frequencies of the spectral lines of
the principal series, taking into account Eq. (6.72), can be found as follows:
ω = 2π cR
∞
1
(2 + b
s
)
2
−
1
(n + b
p
)
2
, n = 2, 3, 4,..., (6.73)
where R
∞
is the Rydberg constant. The lines of the sharp series correspond to
transitions from the s-levels to the 2p-level, and the frequencies of the spectral
lines of this series are given by the expression
ω = 2π cR
∞
1
(2 + b
p
)
2
−
1
(n + b
s
)
2
, n = 3, 4, 5,... (6.74)
The lines of the diffuse series correspond to the transitions from the d-levels to
the 2p-level. The frequencies of the spectral lines of this series are given by the
expression
ω = 2π cR
∞
1
(2 + b
p
)
2
−
1
(n + b
d
)
2
, n = 3, 4, 5,... (6.75)
We note that the quantum corrections b for each series are practically the same,
but they may change slightly from series to series.
As was the case for the hydrogen atom, the spectral lines of the lithium atom
have fine doublet structure, which is related to the splitting of its energy levels
due to the existence of the electron spin. The splitting of energy levels is caused
by the spin–orbit coupling, i.e., by interaction of magnetic moments µ
l
and µ
s
.
Since the core of the lithium atom has a completely filled 1s-level, its angular
momentum is equal to zero. Therefore, the angular momentum of the atom in
general is equal to the angular momentum of its external electron, which is
defined by the orbital quantum number l. The total momentum of this electron
is equal to the sum of the orbital magnetic and spin magnetic moments and is
defined by the quantum number j = l ± s, where l and s = 1/2 are the orbital
and spin quantum numbers. Thus, each energy level of p-states (l = 1) splits into
two sublevels with j = 1/2 and 3/2.
Each energy level of d-states (l = 2) splits into sublevels with j = 3/2 and
5/2. The levels of s-states (l = 0) do not split since they correspond to the single
value of j = 1/2.
Example 6.4. Find the maximum possible total angular momentum and corre-
sponding spectral symbol for the atomic state with the electron configuration
1s
2
2p 3d.
Reasoning. The total maximal angular momentum will be formed from the
maximal orbital magnetic and spin magnetic moments. Since for s-electrons

6.5 The wavefunction of a system of identical particles 181
l = 0, for p-electrons l = 1, and for d-electrons l = 2,
L
max
= 1 + 2 = 3.
For the filled 1s
2
shell spin s = 0, and therefore
S
max
=
1
2
+
1
2
= 1.
As a result, for the maximum quantum number which defines the total angular
momentum, we get
J
max
= L
max
+ S
max
= 4. (6.76)
The total maximal angular momentum of the atom is equal to
M
max
= h
-
J
max
(J
max
+ 1) =
√
20h
-
. (6.77)
Since the multiplicity of the state is 2S
max
+ 1 = 3, the symbol of this state
is
3
F
4
.
Example 6.5. Give the spectral symbol of the atomic state whose degeneracy
with respect to J is equal to seven, with multiplicity equal to five, and for which
the value of the orbital quantum number is maximal.
Reasoning. From the multiplicity of the state,
2S + 1 = 5,
we find spin quantum number S = 2.
From the order of degeneracy,
2J + 1 = 7,
we find J = 3. The quantum number J can take all the integer values from
L + S to |L − S|. Therefore, the values of J and S are J = 3 and S = 2. The
requirement for L to be maximal corresponds to
J = L − S.
Therefore, L = J + S = 5 and the spectral symbol of this state is
5
H
3
.
6.5 The wavefunction of a system of identical particles
For particles that obey the laws of classical physics, it is not important whether
they are identical or not. If we assign numbers to these particles we can track
their motion along each particle’s trajectory. In quantum physics the situation
radically changes since microscopic particles do not follow trajectories and thus it
is impossible to track their motion. Because of the uncertainty principle, identical
particles in the same region become indistinguishable and their numbering does
not make sense. Therefore, the description of the behavior of a system of identical
particles is subject to an additional requirement. Besides normalization and

182 Quantum states in atoms and molecules
orthogonality, for the wavefunction of a system of particles we must take into
account the fact that they are indistinguishable.
To illustrate this, let us consider a system of two identical particles. For a
complete description we need the set of variables that define the position of each
particle in space, r
j
, and the projection of spin on the chosen axis, σ
j
.Thewave-
function of the system, ψ (r
1
,σ
1
; r
2
,σ
2
), must depend on these variables. Since
the particles are indistinguishable, any permutation of the particles would have
no effect on the physical results. This means that permutation of the arguments
(r
1
,σ
1
) and (r
2
,σ
2
) can change only the phase factor of the wavefunction, i.e.,
ψ(r
1
,σ
1
; r
2
,σ
2
) = e
iθ
ψ(r
2
,σ
2
; r
1
,σ
1
). (6.78)
As a result of the second permutation of arguments on the right-hand side of this
equation, the wavefunction takes the initial form but with a new additional phase
factor, i.e.,
ψ(r
1
,σ
1
; r
2
,σ
2
) = e
2iθ
ψ(r
1
,σ
1
; r
2
,σ
2
). (6.79)
Since the second permutation of particles (or arguments) returns the system to
its initial physical state, the following equalities have to be satisfied:
e
2iθ
= 1 ⇒ e
iθ
=±1. (6.80)
Therefore, the permutation of the wavefunction’s arguments requires the satis-
faction of the following condition:
ψ(r
1
,σ
1
; r
2
,σ
2
) =±ψ (r
2
,σ
2
; r
1
,σ
1
). (6.81)
It follows from Eq. (6.81) that the wavefunction of a system of two identical
particles either does not change after permutation of its particles or changes its
sign. In the first case the wavefunction is called symmetric and in the second,
antisymmetric. Thus, because the particles are indistinguishable, the wavefunc-
tion of the system of particles must have some kind of symmetry with respect to
permutations.
Relativistic quantum theory shows that the symmetry of the wavefunction
is unambiguously defined by the spin of the particles. A system of particles
with integer spins (bosons) must be described by a symmetric wavefunction,
whereas a system of particles with half-integer spins (fermions) must be described
by an antisymmetric wavefunction. Since electrons have half-integer spin, the
wavefunction of a system of electrons must be antisymmetric.
Let us consider the algorithm for finding the wavefunction which satisfies
the above-mentioned condition, using the example of two free non-interacting
electrons. The wavefunction of a free electron without taking into account its spin
has the form (3.18). Taking into account the spin variables, the wavefunction of
the system of two electrons with momenta p
1
and p
2
must be represented (without
taking into account its dependence on time) as
ψ(r
1
,σ
1
; r
2
,σ
2
) = Ae
i
h
-
(p
1
r
1
+p
2
r
2
)
ϕ
1
(σ
1
)ϕ
2
(σ
2
), (6.82)
