Mitin V.V., Sementsov D.I., Vagidov N.Z. Quantum mechanics for nanostructures
Подождите немного. Документ загружается.


6.5 The wavefunction of a system of identical particles 183
where σ
1
and σ
2
are the projections of the spin of each of the electrons
onto the chosen axis. Together with the solution (6.82) of the corresponding
Schr¨odinger equation, three additional wavefunctions, which are also solutions
of the Schr¨odinger equation, may be obtained from Eq. (6.82) by permutations of
spatial variables r
1
↔ r
2
and spin variables σ
1
↔ σ
2
. These four functions give
us two linear combinations, which have the correct type of symmetry – two linear
combinations are made antisymmetric. These wavefunctions can be written in
the following form:
ψ
s
(r
1
,σ
1
; r
2
,σ
2
) = ψ
s
(r
1
, r
2
)ϕ
as
(σ
1
,σ
2
), (6.83)
ψ
as
(r
1
,σ
1
; r
2
,σ
2
) = ψ
as
(r
1
, r
2
)ϕ
s
(σ
1
,σ
2
). (6.84)
Here, the symmetric and antisymmetric parts of the coordinate dependence of
the wavefunction of the system of two free electrons are given by the expressions
ψ
s
(r
1
, r
2
) = A
e
i
h
-
(p
1
r
1
+p
2
r
2
)
+ e
i
h
-
(p
1
r
2
+p
2
r
1
)
, (6.85)
ψ
as
(r
1
, r
2
) = A
e
i
h
-
(p
1
r
1
+p
2
r
2
)
− e
i
h
-
(p
1
r
2
+p
2
r
1
)
, (6.86)
and the symmetric and antisymmetric parts of the spin dependence by
ϕ
s
(σ
1
,σ
2
) = ϕ
1
1
2
ϕ
2
1
2
, (6.87)
ϕ
as
(σ
1
,σ
2
) = ϕ
1
1
2
ϕ
2
−
1
2
. (6.88)
Thus, in the symmetric spin state the spins of the two electrons are parallel to
each other and the total projection of the spin of the system is equal to S
z
= 1.
In the antisymmetric spin state the spins of the two electrons are anti-parallel
to each other and S
z
= 0. The procedure for finding the correct wavefunctions
is sometimes called symmetrization. The operation of symmetrization must be
carried out in all cases of systems with identical particles.
If there are more than two identical particles, then the system has such states,
whose wavefunctions, for particles with integer spins, are symmetric with respect
to permutations of any pair of particles (i.e., the wavefunctions do not change their
sign), and, for particles with half-integer spins, are antisymmetric with respect
to the permutations of any pair of particles (i.e., the wavefunctions change their
sign to the opposite). One of the formulations of the Pauli exclusion principle
which relates to a system of electrons is that the total wavefunction of the system
must be antisymmetric, since electrons are particles with half-integer spin.This
formulation is closely connected with the fact that it is not possible to have more
than one electron occupying the same quantum state.
For complex particles (nuclei, atoms, and molecules) the following rule
approximately applies: if a particle consists of an odd number of fermions (elec-
trons, protons, or neutrons), then its spin is half-integer and it behaves as a
fermion. If the complex particle consists of an even number of fermions, then

184 Quantum states in atoms and molecules
its spin is integer and it behaves as a boson. Looking at a particular example,
individual helium atoms of types
2
He
3
and
2
He
4
are chemically indistinguish-
able. The atom
2
He
3
is a fermion since it contains two electrons and a nucleus,
which consists of two protons and one neutron. The atom
2
He
4
is a boson since
its nucleus contains one additional neutron. This difference substantially affects
the properties of a system consisting of a large number of the considered iso-
topes of helium. Liquid helium of type
2
He
4
at temperature T ≈ 2 K becomes a
superfluid, but liquid helium of type
2
He
3
does not exhibit such a property. This
is because the creation of a so-called Bose–Einstein condensate is possible only
in a system of bosons. Its main property is the transition of the entire ensemble
of particles to the lowest energy level when the phase-transition temperature is
reached, which for
2
He
4
is approximately equal to 2 K.
Example 6.6. Show that, if at some time a quantum system that consists of
identical particles is in the state described by the symmetric wavefunction
s
,
then it will be described by the symmetric wavefunction for all subsequent times.
Reasoning. Let us write the time derivative of the wavefunction as
d
s
dt
→
s
t
, (6.89)
where by
s
we understand the change of the wavefunction during the time t.
Let us substitute expression (6.89) into the time-dependent Schr¨odinger equation
(2.182), which describes the initial state of the system:
s
=
1
ih
-
ˆ
H
s
t. (6.90)
Since the Hamiltonian
ˆ
H is symmetric with respect to the system of coordinates of
the particles, the wavefunction
ˆ
H
s
is also a symmetric function of coordinates.
Therefore, in the process of evolution of the wavefunction, which is defined by
Eq. (6.90), its symmetry does not change. Let us note that the preservation of
the symmetry of the wavefunction is a universal property. The same argument
applies also to an antisymmetric wavefunction.
6.6 The hydrogen molecule
Let us consider one more important example where the Pauli exclusion principle
considerably affects the formation of stationary quantum states. This is the system
of two atoms of hydrogen, which form a hydrogen molecule. The wavefunctions
of each of the electrons in their atoms substantially differ from zero only in a
small area close to their nucleus. If the atoms are separated from each other by
such a distance that they can be considered to be independent, the wavefunctions
of electrons do not overlap and for this reason it is meaningless to make the
total wavefunction of the system of two electrons antisymmetric. If the atoms are
brought together to a distance such that the wavefunctions of individual electrons

6.6 The hydrogen molecule 185
a
b
R
r
a1
r
a
2
r
b
1
r
b2
r
12
1
2
Figure 6.13 The scheme of
the hydrogen molecule.
The nuclei are designated
as a and b, the electrons
as 1 and 2.
overlap, then the quantum states of such a system of electrons must be described
by a total antisymmetric wavefunction.
Let us place the nuclei a and b of two hydrogen atoms such that they are
separated by a distance R (see Fig. 6.13). Let us use an adiabatic approximation,
whereby the kinetic energy of the nuclei can be neglected and we can consider
the nuclei as motionless. In the zeroth approximation we can consider the atoms
as isolated and therefore the interaction between them can be neglected. Electron
1 near nucleus a and electron 2 near nucleus b are described by the atomic
wavefunctions of the 1s-state given in Table 6.2. Let us denote these functions as
ψ
a
(r
1
) ≡ ψ
a
(1), (6.91)
ψ
b
(r
2
) ≡ ψ
b
(2). (6.92)
These functions satisfy the following equations:
ˆ
H
(0)
a
ψ
a
(1) = E
1
ψ
a
(1), (6.93)
ˆ
H
(0)
b
ψ
b
(2) = E
1
ψ
b
(2), (6.94)
where the Hamiltonians of the unperturbed atoms are as follows:
ˆ
H
(0)
a
=−
h
-
2
2m
e
∇
2
1
−
k
e
e
2
r
a1
, (6.95)
ˆ
H
(0)
b
=−
h
-
2
2m
e
∇
2
2
−
k
e
e
2
r
b2
. (6.96)
Here E
1
, which is defined by Eq. (6.8), is the energy of the electron in the ground
state of the hydrogen atom. The total energy of the electron in the hydrogen
molecule in the zeroth approximation is equal to twice the energy of the electron
in the hydrogen atom, i.e.,
E
(0)
e
= 2E
1
=−
k
2
e
m
e
e
4
h
-
2
. (6.97)
If we consider that the atoms are not isolated we have to take into account all
the interactions in the hydrogen molecule: interaction of electron 1 with nucleus

186 Quantum states in atoms and molecules
b, interaction of electron 2 with nucleus a, and interaction of electron 1 with
electron 2. In this case the Schr¨odinger equation for the hydrogen molecule will
be the following:
[
ˆ
H
(0)
+
ˆ
W ]ψ(1, 2) = E
e
ψ(1, 2), (6.98)
where the unperturbed Hamiltonian,
ˆ
H
(0)
, is defined as
ˆ
H
(0)
=
ˆ
H
(0)
a
+
ˆ
H
(0)
b
. (6.99)
The perturbation term,
ˆ
W , is the potential energy of the interactions mentioned
above:
ˆ
W = W(1, 2) =
1
r
12
−
1
r
a2
−
1
r
b1
k
e
e
2
. (6.100)
Since the nuclei are considered motionless, their interaction is not included in the
total Hamiltonian, and therefore we will take into account this term by adding
e
2
/R to the total energy.
The wavefunctions of the unperturbed Hamiltonian of the hydrogen molecule,
ˆ
H
(0)
, can be constructed from two one-particle wavefunctions (6.3). In order for
the total wavefunction of the system of two electrons to be antisymmetric, the
radial part of this function must be symmetric in the case of the total spin equal
to S = 0 and antisymmetric in the case of the total spin equal to S = 1, i.e.,
ψ
(0)
s
(1, 2) = ψ
a
(1)ψ
b
(2) + ψ
a
(2)ψ
b
(1), (6.101)
ψ
(0)
as
(1, 2) = ψ
a
(1)ψ
b
(2) − ψ
a
(2)ψ
b
(1). (6.102)
These functions have the following property: after permutation of coordinates
(replacement r
1
↔ r
2
, i.e., 1 ↔ 2) the symmetric function stays the same and
the antisymmetric one changes its sign:
ψ
(0)
s
(1, 2) = ψ
(0)
s
(2, 1), (6.103)
ψ
(0)
as
(1, 2) =−ψ
(0)
as
(2, 1). (6.104)
Using the methods of perturbation theory for degenerate stationary states (see
Section 5.2), we can show that under the influence of perturbation,
ˆ
W ,the
unperturbed degenerate level E
e
splits into two. The one of these levels with lower
energy, E
s
e
, corresponds to the symmetric wavefunction, ψ
(0)
s
(1, 2), and the other
one, with higher energy, E
as
e
, corresponds to the antisymmetric wavefunction,
ψ
(0)
as
(1, 2). The expressions for the energies of the symmetric and antisymmetric
states have the following forms:
E
as
e
= 2E
1
+ W
as
(R), (6.105)
E
s
e
= 2E
1
+ W
s
(R), (6.106)

6.6 The hydrogen molecule 187
where
W
s
=
Q + A
1 + F
2
(6.107)
and
W
as
=
Q − A
1 − F
2
. (6.108)
Here we introduced the following quantities:
Q =
|ψ
a
(1)|
2
W (1, 2)|ψ
b
(2)|
2
dV
1
dV
2
, (6.109)
A =
ψ
∗
a
(1)ψ
b
(2)W (1, 2)ψ
∗
a
(2)ψ
b
(1)dV
1
dV
2
, (6.110)
F =
ψ
∗
a
(1)ψ
b
(1)dV
1
=
ψ
∗
a
(2)ψ
b
(2)dV
2
. (6.111)
The quantity Q is called the Coulomb integral since it includes the energy of
the electrostatic interaction of the electron clouds with each other and with the
atomic nuclei. The electron clouds have the following electric charge density:
ρ
a
(1) =−e|ψ
a
(1)|
2
, (6.112)
ρ
b
(2) =−e|ψ
b
(2)|
2
. (6.113)
The quantity A, which is called the exchange integral, is closely connected with
the wave nature of the electron. In this integral there are two “exchange charge
densities”:
ρ
ex
(1) =−eψ
∗
a
(1)ψ
b
(1), (6.114)
ρ
ex
(2) =−eψ
∗
a
(2)ψ
b
(2), (6.115)
which can be interpreted as if each of the electrons belonged to nucleus a and
nucleus b simultaneously.
The quantity F is called the overlap integral and it is a measure of the
overlap of the wavefunctions of atoms (i.e., overlap of electron clouds) at a given
distance between them, R. All the above-mentioned quantities are functions of
this distance, R, and consequently the corrections to the energy, E
e
(Eqs. (6.107)
and (6.108)), also depend on R.
In analyzing the expressions (6.105)–(6.111) for the energy of the system of
electrons E
s
e
and E
as
e
we emphasize that, when atoms approach each other,
the energy level E
e
= 2E
1
defined by Eq. (6.97) splits into two sublevels.
The magnitude of the splitting depends on the distance between nuclei and is
equal to
E(R) = E
as
e
(R) − E
s
e
(R) = W
as
(R) − W
s
(R) =−2
A − QF
2
1 − F
4
. (6.116)
Under the assumption of the adiabatic approximation, the energy of the electrons
plays a role as one of the components of the potential energy of interaction of the
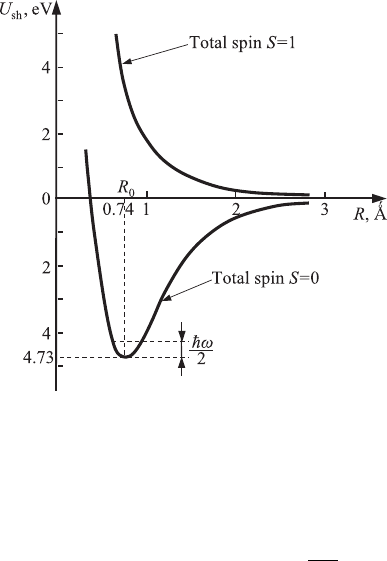
188 Quantum states in atoms and molecules
−
−
−
Figure 6.14 Tw o
dependences of the
potential energy U on R:
(1) the dependence with
the correction W
as
(total
spin equal to S = 1) and
(2) the dependence with
the correction W
s
(total
spin equal to S = 0). Here
U
sh
= U(R) − 2E
1
and
R
0
= 0.74
˚
A.
atoms. The total potential energy of interaction of the hydrogen atoms, including
the energy of the Coulomb repulsion of the nuclei, is defined by the following
expression:
U (R) = 2E
1
+
k
e
e
2
R
+
W
as
(R),
W
s
(R).
(6.117)
The total potential energy of interaction of the hydrogen atoms strongly
depends on the total spin of the system of electrons. Figure 6.14 shows two
dependences of the potential energy U(R): (1) the dependence that corresponds
to the correction W
as
and to the total spin S = 1 and (2) the dependence that
corresponds to the correction W
s
and to the total spin S = 0. The monotonic
decrease of energy with increasing R in the state with parallel spins means that
atoms repel each other. Thus, it is impossible to form the hydrogen molecule
with S = 1. The minimum potential energy for the hydrogen molecule, U (R
0
) =
−4.73 eV, which occurs at R
0
= 0.74
˚
A ≈ 1.4r
1
, occurs only for the state with
opposite spins. Therefore, for this state (S = 0) the bonding between hydrogen
atoms, which is called covalent bonding, occurs. This type of bonding plays a
significant role in the formation of molecules as well as of numerous crystals.
Example 6.7. Estimate the oscillation frequency of the nuclei of the hydrogen
molecule in the ground state and the dissociation energy of the hydrogen molecule
(i.e., the energy required for the breakup of the hydrogen molecule into separate
atoms).
Reasoning. The adiabatic approximation can be used for the solution of the
problem of oscillations of nuclei in the hydrogen molecule. To find the wave-
function of the nuclei (R) in a given quantum state we will take the energy of
6.6 The hydrogen molecule 189
the electrons, E
e
(R), as the potential energy of the interaction of nuclei. Let us
consider the motion of the nuclei in the system of their center of mass. Then, the
wavefunction of the nuclei, (a, b), will depend only on the relative distance,
R, between them and define their relative motion under the influence of internal
forces. To find this function it is necessary to solve the following Schr¨odinger
equation:
−
h
-
2
2m
ab
d
2
dR
2
+U (R)
(R) = E(R), (6.118)
where the reduced mass of two identical nuclei, m
ab
, is equal to the half-mass of
the proton,
m
ab
=
m
a
m
b
m
a
+ m
b
=
m
p
2
, (6.119)
and E is the total energy of the hydrogen molecule, taking into account the motion
of the nuclei. The magnitude U (R) is the total potential energy of the nuclei and
it is defined by Eq. (6.117). It includes the energy of the Coulomb interaction of
the nuclei and the energy of the electrons, E
e
(R), in the given quantum state. To
find the frequency of the small-amplitude oscillations of the nuclei let us expand
the function U (R) near the equilibrium position of the nuclei R
0
in series over the
displacement R − R
0
and let us keep only the first two terms of the expansion:
U (R) = U (R
0
) +
β
2
(
R − R
0
)
2
, (6.120)
where
β =
d
2
U
dR
2
R=R
0
. (6.121)
Taking into account the relation of the elastic constant with frequency, we get
ω =
β
m
ab
=
2
m
p
d
2
U
dR
2
R=R
0
. (6.122)
The expression for the energy U(R) can be rewritten in the form
U (R) = U (R
0
) +
m
ab
ω
2
2
(
R − R
0
)
2
. (6.123)
On choosing R
0
as the origin of coordinates and U (R
0
) as the reference point for
energy, i.e., taking R
0
= 0 and U(R
0
) = 0, we get the equation
−
h
-
2
2m
ab
d
2
dR
2
+
m
ab
ω
2
2
R
2
(R) = E(R), (6.124)
which coincides with the one-dimensional Schr¨odinger equation for an electron
in the parabolic potential (4.74). On solving this equation we get the quantized
harmonic oscillator states for the system of the nuclei, whose energy according

190 Quantum states in atoms and molecules
to Eq. (4.88) is given by the expression
E
osc
= h
-
ω
n +
1
2
, (6.125)
where n is the quantum number of the oscillator. The ground state of the hydrogen
molecule corresponds to the energy of the n = 0 oscillator state:
E
osc
=
h
-
ω
2
= 0.27 eV. (6.126)
This value of oscillatory motion energy was established from measurements of
the optical spectra of the hydrogen molecule. From Eq. (6.126) it follows that
the oscillations of the nuclei happen with frequency ω = 8.6 ×10
14
s
−1
.The
oscillatory motion of the nuclei splits the electron levels into closely spaced
oscillatory sublevels. Then, the energy levels of the hydrogen molecule are given
by the expression
E(R) = E
e
(R
0
) +
k
e
e
2
R
0
+ h
-
ω
n +
1
2
. (6.127)
The energy of dissociation, E
dis
, of the hydrogen molecule is defined by the
minimal energy necessary for dividing the hydrogen molecule into two individual
atoms:
E
dis
=
U (R
0
) +
h
-
ω
2
=|−4.73 +0.27| eV = 4.46 eV. (6.128)
We note that a more precise solution of the problem for the hydrogen molecule
requires the taking into account of two more factors – rotational motion of
the molecule and the fact that the nuclei are identical. Under the influence of
rotational motion the oscillatory energy levels split into a system of rotatory
sublevels. The distance between these sublevels is of the order of 8 × 10
−3
eV.
6.7 Summary
1. The electron state in the hydrogen atom is defined by the set of four quantum numbers:
the principal quantum number, n, orbital quantum number, l, orbital magnetic quantum
number m, and spin magnetic quantum number m
s
. The electron energy spectrum of
the hydrogen atom, which is defined only by the principal quantum number, n,is
discrete and all quantized levels are in the region of negative energy values.
2. One of the main principles of quantum physics is the Pauli exclusion principle, accord-
ingtowhichaparticular state, which is characterized by a given set of quantum num-
bers (in the case of an atom the quantum numbers are n, l, m, m
s
), may be occupied
by only one electron.
3. Considering the system of identical particles, we have to take into account that in
a quantum description the particles are distinguishable. Systems of particles with
integer spin (bosons) must be described by symmetric wavefunctions, whereas systems
6.8 Problems 191
of particles with half-integer spin (fermions) must be described by antisymmetric
wavefunctions. Since electrons have spin s = 1/2, the wavefunction of the electron
system must be antisymmetric.
4. All electrons of a many-electron atom with the same principal quantum number, n,
form a shell, which has 2n
2
states. Shells consist of subshells, which differ by the orbital
quantum number l. The number of different states in a subshell with different quantum
numbers m and m
s
is equal to 2(2l + 1). Completely filled shells and subshells have
the resulting quantum numbers of the orbital, spin, and total moment equal to zero:
L = 0, S = 0, and J = 0.
5. The order of filling by electrons of the atom’s subshells is defined by Hund’s rules:
(1) the electron configuration has the minimal energy in the state with the largest spin
S and with the largest (for such S) L; (2) for a less than half-filled subshell the quantum
number J is equal to J =|L − S|, whereas for a more than half-filled subshell J is
equal to J = L + S.
6. The covalent bonding between hydrogen atoms is caused by the exchange interaction,
and this type of bonding occurs for the state with anti-parallel spins (S = 0). This
type of bonding plays an important role in the formation of molecules and numerous
crystals.
6.8 Problems
Problem 6.1. Using Bohr’s model of the hydrogen atom and his postulates, find
the radii of electron stationary orbits, r
n
, electron velocities, v
n
, orbital periods,
T
n
, the electron energy on the corresponding orbit, and the circular frequency of
a photon emitted during electron transition from the nth to the mth orbit. Find
the radius and velocity, r
1
and v
1
, for the first electron orbit. Find the angular
frequency, ω
21
, of the electron transition from the second to the first orbit.
(Answer: r
1
≈ 0.53
˚
A, v
1
≈ 2.19 × 10
6
ms
−1
, and ω
21
≈ 1.53 × 10
16
s
−1
.)
Problem 6.2. Find the minimum, λ
min
, and maximum, λ
max
, wavelength of the
hydrogen spectral lines in the visible range of the spectrum. What is the minimum
speed, v
min
, of electrons incident on a hydrogen atom to excite these spectral
lines? (Answer: λ
min
≈ 365 nm, λ
max
≈ 656 nm, and v
min
≈ 8.2 × 10
6
ms
−1
.)
Problem 6.3. The electron in the hydrogen atom is in the ground state described
by the following wavefunction:
ψ
1s
(r) =
πr
3
1
−1/2
e
−r/r
1
. (6.129)
Find the average electrostatic potential at the distance r from the nucleus.
Problem 6.4. Find the spectrum of the vibrational levels of a diatomic molecule.
For the diatomic CO molecule, estimate the distance between vibrational energy
levels, E
n+1,n
, and the emission wavelength, λ, during the corresponding
transitions. The carbon atom mass m
1
= 1.99 × 10
−26
kg, the oxygen atom

192 Quantum states in atoms and molecules
mass m
2
= 2.66 × 10
−26
kg, and the force constant β = 190 kg s
−2
.(Answer:
E
n+1,n
≈ 8.44 × 10
−2
eV and λ ≈ 24 µm. Hint: use Eq. (6.122).)
Problem 6.5. Find for the first-order perturbation theory the energy, E
1
,ofthe
ground state of the helium, He, atom. Use as the perturbation the energy of
interaction between electrons. (Answer: E
1
≈−18.7 eV.)
Problem 6.6. In the alkaline atoms the nucleus with charge eZ and the first Z − 1
electrons form a core with positive charge equal to the elementary charge, e.A
valence electron rotates in the field of this core. Find the energy spectrum of the
valence electron if its potential energy is defined by the following expression:
U (r) =−
k
e
e
2
r
1 +
C
r
, (6.130)
where we can consider C/r
1
1(r
1
is the Bohr radius).
Problem 6.7. Find the angular momentum for electrons in an atom in 3s, 4d, and
5g states and the maximum number of electrons in an atom with the same sets
of quantum numbers n, l, and m.
