Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

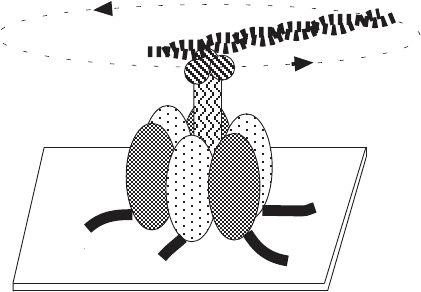
be further discussed. The F - ATPases typically synthesize ATP driven by a
proton motive force, while the V - type ATPases hydrolyze ATP to generate an
electrochemical gradient across a membrane. The latter two are operating on
similar principles. The discussion will focus on the mitochondrial complex V,
an F - ATPase. In the above model system, ATP hydrolysis by the a/ β subunits
caused the γ subunit to rotate, and one can therefore imagine this rotation to
be the reverse of the rotation caused by a proton fl ux driving ATP synthesis.
Following the original publication describing rotary motion, several more
elegant demonstrations of rotary motion have appeared (288 – 293) . Some of
these model systems have included the c subunits of the rotor, demonstrating
their rotation when attached to the γ subunit with the help of ε . These studies
are beautiful illustrations of the power of nanotechnology applied to the study
of single molecules, but they have not only provided a picture, but have yielded
quantitative data fully reconciling mechanistic and kinetic studies on ATP
synthesis by this enzyme (283, 290, 291) . The reaction is completely reversible.
Hydrolysis of one ATP causes a 120 ° rotation of γ , and the rotation of γ in the
opposite direction can be driven by proton fl ux coupled to ATP synthesis from
ADP and P
i
. The 120 ° rotation can be decomposed into two sub - steps of 80 °
and 40 ° , where the 80 ° rotation is brought about by the binding of ATP to a
single empty site, and the 40 ° rotation is associated with the release of ADP
from the site. Under physiological conditions the bacterial enzyme is estimated
to rotate at ∼ 100 Hz, and averaged over time all three catalytic sites are occu-
pied, two by ADP and one by ATP. All three sites are involved in driving the
rotation of γ . Pure β subunits do not catalyze hydrolysis at a signifi cant rate,
and even the α
3
β
3
complex shows suboptimal activity and altered affi nities and
kinetics. In the complete structure, “ multisite catalysis ” is now accepted as the
dominant mode of catalysis in vivo , based on the estimated intracellular con-
centrations of substrates and the measured values for the K
m
of the enzyme.
Figure 5.29 Demonstration of the rotation of the shaft ( γ subunit) relative to the F1
complex. (Adapted from reference 287 .)
Actin filament
Streptavidin
His-tag
3
3
α β γ complex
rotation
α
α
β
γ
ATP SYNTHASE (F
1
F
0
-ATPase) 251
252 MITOCHONDRIAL ELECTRON TRANSFER
Multisite catalysis is characterized by an impressive ( ∼ 5 orders of magnitude)
increase in the rate of hydrolysis attributed to the highly cooperative interac-
tion between the subunits and their catalytic sites.
There appears to be a consensus that the binding of ADP and P
i
at one of
the three active sites of the F
1
- ATPase provides the energy for combining the
two substrates to make ATP. In other words, ATP formation is spontaneous
after binding and does not require the input of additional energy. The reaction
is a simple reversal of hydrolysis and has no additional covalent intermediates.
A nearby glutamate participates in acid catalysis. The electrostatic repulsion
between the phosphate groups is overcome by interactions with amino acid
side chains in the active site, and the perfect alignment presumably makes the
formation of a covalent bond and the elimination of water entropically favor-
able. The strong binding energy, however, now makes the release of the ATP
unfavorable and slow. It is the release of ATP which is believed to be the step
driven by the proton translocation and rotation. The release must be triggered
by a change in affi nity for ATP, which can be brought about by a change in
the conformation of the protein subunits. The three orientations of a given β
subunit relative to γ can be shown to correspond to the progression from ATP -
occupied ⇒ ADP - occupied ⇒ empty, the reaction sequence expected for the
hydrolysis reaction.
An authoritative and exhaustive review in 1993 by Boyer (278) has provided
a summary of the biochemical observations related to the ATP synthase mech-
anism and provided critical comments on each. An update by the same author
incorporating information from the crystal structure has appeared subsequently
(294) , and highlights in the development of this penetrating and detailed analy-
sis can be found in the acceptance speeches and publications of the 1997 Nobel
Prize winners in Chemistry. Very signifi cant contributions and refi nements have
also been contributed by Senior and colleagues (see reference 282 for an expert
review). They are testimony to the ingenuity and insights of a large number of
investigators keeping pace with or even staying ahead of the structural studies,
until this amazing molecular engine and its inner workings are now revealed
in exquisite detail. Once more it should be emphasized that biochemical studies
were constantly reviewed and tested for consistency with the structural studies.
The clarifi cation of the subunit stoichiometry was followed by cross - linking
studies under various conditions, demonstrating conformational changes asso-
ciated with the presence of, for example, Mg
2+
- ADP in the catalytic site, con-
trasted to the presence of the nonhydrolyzable ATP analog AMP - PNP. When
the β and γ subunits were cross - linked, enzyme activity was severely inhibited,
a result now fully explainable in terms of the rotary motion associated with
one cycle of activity. Finally, although most of the biophysical studies were
performed with bacterial enzymes, it is a consensus that the other F - and V - type
ATPases operate on the same principles, with perhaps notable differences in
kinetics and mechanisms of regulation.
The noncatalytic nucleotide binding sites have a structural role and are
likely to play a part in regulating or modulating the stability and/or the
structure of this enzyme (266, 278) . Nucleotide exchange at the noncatalytic
sites is slow. One is reminded of the role of one nonhydrolyzable GTP in the
αβ tubulin dimer. Nevertheless, while a physiologically relevant regulatory
role has not yet been demonstrated, abolition of the noncatalytic nucleotide
binding sites by site - directed mutagenesis has shown that they are not dispens-
able. Such a modifi ed enzyme ( α
3
β
3
γ complex) exhibits an initial ATPase
activity, but this activity decays rapidly to zero. This observation has been
interpreted to indicate an entrapment of MgADP in a catalytic site (see refer-
ence 295 for additional references to investigations of the specifi c role of the
noncatalytic sites).
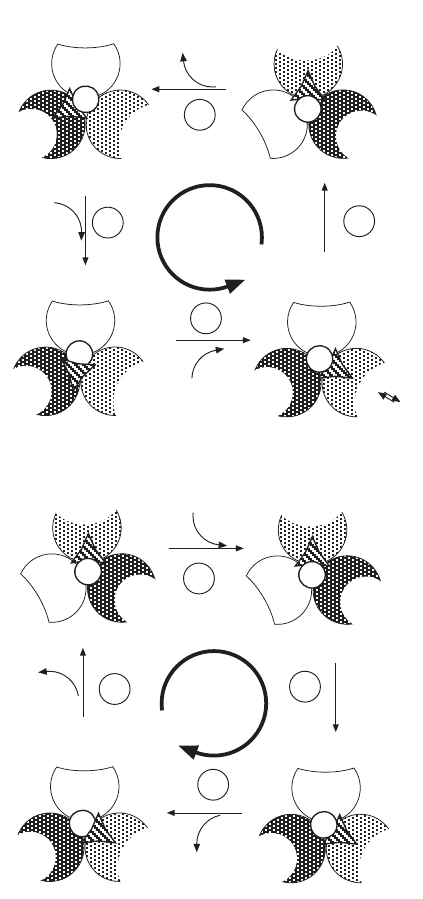
A schematic representation of the three - site model is shown in Figure 5.30
for the synthesis of ATP (Figure 5.30A ) and for the hydrolysis of ATP (Figure
5.30B ). (see 281, 282 , for details).
5.5.4 The F
0
Subcomplex and Proton Flow
Attention now must focus on the stator (b
2
OSCP) in the periphery and on the
F
0
subcomplex (ab
2
c
n
) for which complete high - resolution crystal structures
are not yet available. The F
0
subcomplex provides a path (channel) for the
protons. Proton fl ow through the F
0
channel must be converted into a torque
on the rotor ( γε c
n
) in the center. Thus, two questions can be formulated: (1)
What is the path of the protons through the F
0
subcomplex? (2) How does the
translocation of protons from one side of the membrane to the other produce
a torque? To avoid confusion, the following discussion will refer to the essen-
tial subunits of the bacterial complex, and the reader is referred to Table 5.2
for the corresponding subunits in the mitochondrial complex in various
organisms.
While δ and ε subunits were dispensable in the demonstration of F
1
- ATPase
(α
3
β
3
γ ) as a rotary motor, the ε subunit is likely to be necessary for the coupling
between the F
1
and F
0
subcomplexes. It can be cross - linked via its C - terminal
domain to the α
3
β
3
barrel and via its N - terminal domain to the γ subunit.
Sequence conservation in bacterial and chloroplast ε subunits has focused
attention on an N - terminal domain, and studies employing site - directed muta-
genesis have shown that mutations (e.g., ε H38C) affect coupling between
proton fl ux and ATPase (266) . Cryo - electron microscopy has shown a major
shift of the ε subunit relative to the α and β subunits when viewed down the
threefold symmetry axis upon binding of different nucleotides (266) .
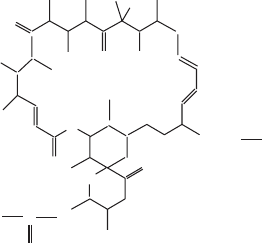
To function as a highly specifi c inhibitor of the ATP synthase in mitochon-
dria, oligomycin (Figure 5.31 ) requires the presence of the OSCP subunit
(oligomycin - sensitivity conferring protein); it was shown to be equivalent to
the bacterial δ subunit. Since it blocks proton fl ow, it had been associated with
the proton translocating mechanism, but it seems clear now that the inhibition
is indirect. The δ subunit is located “ on top ” of the a
3
b
3
barrel, facilitating the
interaction of the tip of the b
2
complex with the α
3
β
3
barrel (see Figure 5.26 ).
A role in the assembly of the ATP synthase is discussed briefl y below.
ATP SYNTHASE (F
1
F
0
-ATPase) 253
254 MITOCHONDRIAL ELECTRON TRANSFER
ADP
ATP
ADP
ATP
ATP
ADP
ATP
ADP - P
i
ADP
ATP
ADP
ATP
P
i
ADP
hydrolysis
1
2
3
4
B
Figure 5.30 (A) Multisite catalysis model for the synthesis of ATP. (B) Multisite
catalysis model for the hydrolysis of ATP. In this model all three catalytic sites are
occupied, except for a very transient period when the product has left and before a
new substrate enters the site. (Adapted from reference 281 .)
ATP
ADP-P
ATP
ATP
ADP-P
ATP
ADP - P
ATP
ATP
ADP
synthesis
1
2
3
4
P
ADP - P
P
ADP - P
ATP
A
The bovine F
0
subcomplex had fi rst been prepared in Racker ’ s laboratory
by a combination of sonication and urea treatment. A more homogeneous
preparation suitable for structural analysis has been devised by the Cambridge
group of J. Walker. The fi nal complex from bovine heart contains nine different
proteins (a – g, F6, A6L) (296) . The stoichiometry for the mammalian mitochon-
drial F
0
complex had also been established independently (e.g., reference 260 ).
All of the subunits have been expressed in bacteria from cloned cDNAs, and
successful re - constitution experiments making various subcomplexes as well
as the entire F
1
F
0
- ATPase have been reported (296) . Complete information
for the yeast mitochondrial F
1
F
0
- ATPase peptides (238) is based primarily on
genetic analyses of respiration - defi cient yeast mutants, followed by biochemi-
cal experiments. Additional gene products were found to be necessary for the
assembly of the complex, but were not present in the purifi ed complex (238,
297) .
From comparisons with the simpler complex in bacteria, one can conclude
that three essential peptides make up the proton - translocating hydrophobic
F
0
core (298) . Subunit a (271 residues in bacteria) has multiple transmembrane
regions, most likely an even number (4 – 8). This places both the N - terminal
and the C - terminal on the same side of the membrane. With the help of fusion
proteins, the ends have been placed on the outside (periplasmic side) of the
bacterial membrane (298) . It is not required for the binding of the F
1
. The b
subunit may be present as a dimer. In bacteria, it has been deduced from the
amino acid sequence that subunit b has a single transmembrane segment near
the N - terminal, with the remaining domain forming a predominantly α - helical,
highly charged (hydrophilic) secondary structure. Early ideas about its interac-
tions with the γ and δ subunits of F
1
(298) have been abandoned in favor of a
model placing the two b subunits on the outside of the α / β complex where it
acts as the stator (see Figure 5.26 ). A high - resolution structure of this stator
has been published recently (271) . The a and b subunits can be cross - linked.
They are attached to the outside of the c ring (see below). A high - resolution
Figure 5.31 Structure of oligomycin.
CH2CCH
3
O
O
O
O
O
O
OH
HO
OH
HO
O
CH
2
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
ATP SYNTHASE (F
1
F
0
-ATPase) 255
256 MITOCHONDRIAL ELECTRON TRANSFER
structure for this interaction is not yet available in spite of the intense interest
focused on this interaction. Protons translocated into the matrix are very likely
taking a path that includes the interface between the a subunit and the c
n
ring.
The b
2
dimer is not believed to be involved in forming the proton channel.
Thus, understanding this specifi c protein – protein interaction promises to
shed much light on the mechanism of torque production by proton
translocation.
The c subunit is present in multiple copies (10 – 14, depending on the species)
compared to the a subunit; 10 – 14 c - subunits are associated to form the c ring.
Within its short sequence (79 residues in bacteria) two transmembrane α -
helices are predicted, fl anking a polar loop region. Such a structure has been
verifi ed by NMR studies of the monomer in chloroform:methanol:water (277) .
Using this structure for molecular modeling structures for the c ring were
proposed. A high - resolution structure for the c ring of a related F - type Na
+
-
ATPase of Ilyobacter tartaricus was fi nally reported in 2005 (275) (Figure 5.28 ),
while Murata et al. (276) published the corresponding structure for the V - type
Na
+
- ATPase of Enterococcus hirae . Some signifi cant differences between these
structures and the mitochondrial c
n
ring can still be expected. First, the number
of monomers per ring may be different; and second, the mammalian ATP
synthase is driven by protons.
Even before detailed crystallographic structures were available, several
revealing structure – function studies were carried out with the bacterial c
subunit modifi ed by site - directed mutagenesis. For example, a Q42E mutant
c - peptide allowed normal assembly of the F
1
F
0
- ATPase, but ATP hydrolysis
was uncoupled from proton translocation (the bacterial enzyme is fully revers-
ible depending on physiological conditions) (298) . The well - known inhibition
of the F
1
F
0
ATPase by DCCD is due to a unique reaction of Asp61 (D61) with
dicyclohexylcarbodiimide (DCCD). Even if only a single c - subunit is deriva-
tized by DCCD, the ATPase activity of the whole complex is abolished. Studies
of the isolated subunit c in a chloroform – methanol – water mixture were
encouraged by experiments suggesting that the isolated protein retains fea-
tures of the native protein, including the reactivity of D61 with DCCD, and
the inhibition of this reaction by the I28T mutation. Experiments of this type
and mutational analysis at this Asp residue have been used to argue that the
carboxyl group of D61 is a participant in proton translocation. At this time it
appears to be the best candidate (277, 298) . Similarly, a mutational analysis of
the bacterial a subunit has identifi ed the R210 residue as an absolutely essen-
tial residue for proton translocation. Thus, the single a subunit and 9 – 12 c
subunits form a path for proton fl ux across the membrane.
Proton fl ow in such a molecular motor must produce a torque. Proton
transfer must be associated with a rotation or displacement of an a - subunit
relative to a c subunit. The model must also include the experimental fact that
several protons and several c subunits are involved in a 120 ° displacement
(more than one proton has to be moved across the membrane for one ATP
to be made). Junge and his colleagues as well as Oster and colleagues have
proposed ingenious solutions in which biochemistry meets engineering (280,
299, 300) ; they await further experimental verifi cation. The reader is referred
to the original literature for details on two major models under consideration
that can be labeled as the “ power stroke model ” and the “ Brownian ratchet
model. ” Evidence from available crystal structures and theoretical consider-
ations favor the ratchet model (299) . In this model the c ring executes stochas-
tic rotational motions. There are two access channels for ions on opposite sides
of the stator (a subunit), but they are not collinear. After an ion has entered
from one side, the c ring has to turn through some angle to allow this ion to
escape on the other side. The unequal concentration and potential of the ions
on either side of the membrane drives a fl ux of ions in one direction, and it
also biases the Brownian rotational fl uctuations of the c ring to produce a net
rotation in one direction. The emerging structural details are clearly of great
importance for defi ning the proton channels, the relevant side chains on the a
and c subunits (already partially determined from mutagenesis experiments),
and the possible conformational changes associated with this mechanism.
Finally, two challenges remain that are closely related in some aspects.
Thermodynamic considerations strongly suggest that on the order of three
protons must be translocated across the membrane for every ATP produced,
or, for every 120 ° turn of the rotor. This had led to predictions at an earlier
stage of exploration that the c ring should have a number of subunits that are
a multiple of three (i.e., 9 or 12). The experimental fi nding of 10 or 14 subunits
in the c
n
ring raises the issue of a “ symmetry mismatch ” : The symmetry of the
c - ring does not match the symmetry of the α
3
β
3
barrel of the F
1
subcomplex.
How many c subunits pass the single a subunit of the stator in a 120 ° turn?
The number must be non - integer, and hence the number of protons passing
from one side to the other on average must also be non - integer. There are
considerations that this symmetry mismatch is an essential characteristic of
the torque - producing mechanism, but the problem will keep biophysicists
occupied in the future.
Upon reaching the end of this chapter, one should expect the reader to be
left with a sense of awe, without, however, being pushed into the camp of those
who believe in “ intelligent design. ” Francois Jacob compared evolution to the
work of a “ tinkerer, ” and it is indeed amazing what several billion years of
tinkering can achieve. It is also truly “ awesome ” to think of the billions of cells
in our body, each containing tens of thousands of little motors going continu-
ously at 50 – 100 rps, thus sustaining a turnover of an amount of ATP/ADP per
day that is equivalent to or greater than our body weight.
5.5.5 Assembly of Complex V and Dimerization
The biogenesis of ATP synthase raises several questions that have already
come up in the discussion of the other complexes of the ETC. One has to
consider (a) the expression of multiple genes and (b) the assembly of multiple
subunits making up the subcomplexes of the mature enzyme. In eukaryotes,
ATP SYNTHASE (F
1
F
0
-ATPase) 257
258 MITOCHONDRIAL ELECTRON TRANSFER
mitochondrial genes as well as nuclear genes contribute to the biogenesis; and
furthermore, the nuclear genes are typically dispersed on different chromo-
somes (in contrast to E. coli , where they form an operon). The control and
coordinate expression of such genes is briefl y discussed elsewhere (Chapter
4 ). In yeast the nuclear genes of ATP synthase appear to be constitutively
expressed, regardless of the carbon source, in contrast to the nuclear genes for
the electron transport complexes. At the same time, there appears to be no
specifi c mechanism to coordinate the expression of nuclear genes and mito-
chondrial genes in yeast. A more detailed discussion can be found in the review
by Ackerman and Tzagoloff (238) .
It seems intuitive that the assembly of a complex of 14 or more subunits
would occur in an orderly fashion, but the challenge has been to elucidate the
assembly pathway. Work in S. cerevisiae has been pioneering by taking advan-
tage of the powerful methodologies of molecular genetics. Thus, it is possible
to investigate gene knockouts and determine how far assembly can proceed
in the absence of a particular subunit. As a fi rst approximation, one can state
that the core subunits of the F
1
subcomplex ( α , β , γ ) and the core subunits of
the F
0
subcomplex (c, a, b) assemble independently, and at some stage they
are joined and combined with other subunits to form the functional complex.
An F
1
subcomplex with ATPase activity can be made in yeast mutants lacking
mtDNA ( ρ
o
mutants). This activity is essential in combination with the adenine
nucleotide transporter to generate a membrane potential for protein import
and for the maintenance of mtDNA in ρ
−
mutants. Alternatively, the assembly
(and hence activity) of ATP synthase has been found to be defective in mutants
even though all known structural genes were normal. Thus, it became apparent
that assembly factors or molecular chaperones are involved (a conclusion
equally applicable to the assembly of the other complexes of the ETC). These
assembly factors are not found in the fi nal complex. Two other properties of
such factors are noteworthy: (1) For example, the factors Atp11p and Atp12p
are specifi cally required only for the assembly of the F
1
subcomplex, in con-
trast to chaperones such as mtHsp70 and Hsp60; (2) because of their restricted
function, their deletion is not necessarily lethal under conditions where gly-
colysis can satisfy the energy requirements of the cells. The most explicit and
detailed description of the ATP synthase assembly in yeast can be found in a
review by the authors who have also contributed much of the experimental
support for the model (238) .
There are two ATP synthase subunits whose signifi cance has been appreci-
ated only recently. The subunits e and g are required for dimerization of
complex V, detected by blue - native gel electrophoresis. In this context, the most
intriguing observation was made that ATP synthase dimers are playing a essen-
tial role in the establishment of the morphology of mitochondrial cristae (37,
38, 301, 302) . Yeast mutants lacking either e or g are devoid of lamellar cristae,
with the inner membrane folded into onion - like structures or highly abnormal
folds. How ATP synthase dimerization can lead to the “ zippering up ” and align-
ment of cristae membranes remains a challenge for the future.
5.6 CONTROL OF RESPIRATION AND
OXIDATIVE PHOSPHORYLATION
5.6.1 General Considerations
It is quite apparent that the energy demands of a given cell or tissue will vary
over time, and the need for regulatory mechanisms becomes obvious. A dis-
tinction to be made at the beginning of this section is to separate mechanisms
involved in long - term control, in the course of development and tissue differ-
entiation, from short - term control mechanisms operating on a time scale of
seconds to hours. A prominent example is muscle, but other tissues and organs
in mammals and other organisms may also exhibit fl uctuations in respiration
and oxidative phosphorylation in response to regulatory signals from hor-
mones, growth factors, or the nervous system. A subtle but very important
aspect of this problem is the balancing between (a) the capture of the free
energy from combustion in the form of ATP and (b) the release of some of
that energy in the form of heat. This is of course the basic mechanism for
maintaining the body temperature in warm - blooded animals, and specifi c
tissues and mechanisms have evolved for this task.
The long - term control in differentiating tissues, or in the adaptation of a
microorganism such as yeast to changing environmental conditions, is likely
to be based on the control of gene expression and the rate of biogenesis of
mitochondria or the components of the inner membrane. In Chapter 3 a dis-
cussion of the morphology of mitochondria and of the density and shape of
cristae has made reference to the likely explanation that the density of cristae
refl ects the capacity for respiration of the particular cells examined. One could
add to this the control of the copy number of mtDNA per cell.
The present discussion will focus on short - term control. A highly readable
and thoughtful review of this subject with many primary references has been
written by Brown (303) . It concludes with the statement that “ there is
no simple answer to the question ‘ what controls respiration? ’ . ” The question
becomes increasingly more complex as one considers an isolated mitochon-
drion, a uniform cell population in the controlled environment of the culture
medium, or an organ. The answer is likely to vary also depending on the origin
of the mitochondria. Even more variations will have to be considered when
one compares mitochondria from different organisms. One may also have to
make a distinction between (a) studies with isolated mitochondria where
extremes of conditions can be explored and (b) in vivo situations where the
parameters cannot be so well - controlled or are not even precisely known. In
the following discussion, therefore, the emphasis will be on the basic principles
that will have to be taken into consideration when the question of the regula-
tion of mitochondrial activity is raised.
A formal analysis of the problem that has led to important insights and
conclusions is based on the theoretical framework now called Metabolic
Control Analysis (304, 305) . The “ Control of Flux ” was a landmark paper by
CONTROL OF RESPIRATION AND OXIDATIVE PHOSPHORYLATION 259
260 MITOCHONDRIAL ELECTRON TRANSFER
Kacser and Burns fi rst published in 1973 and then released as a recent updated
and annotated reprint (304) . It begins with the emphatic statement that “ fl ux
is a systemic property and questions of its control cannot be answered by
looking at one step in isolation — or even each step in isolation ” , and it ulti-
mately challenges our familiar and simplifying concept of a “ rate - controlling
enzyme ” or of a “ bottleneck in the pathway. ” In its explicit formulation it
constitutes a highly mathematical treatment seeking to express how sensitive
a fl ux in a pathway is to an enzyme ’ s concentration or activity. A fl ux control
coeffi cient is introduced as a quantitative measure of the system ’ s sensitivity
to modulations of one of its components (enzyme activity/concentration), and
it is a measure of the importance of this enzyme for control of fl ux through
the pathway, regardless of whether and how this enzyme is controllable.
A coeffi cient of 1.0 would indicate that an enzyme is controlling the pathway
and that in practice a 1% increase in enzyme concentration/activity would
lead to a 1% increase in fl ux. Such an enzyme would constitute the typical
“ bottleneck ” in a pathway. Conversely, a coeffi cient of 0 would indicate
that the enzyme is in excess and that changing its concentration fractionally
would not lead to a signifi cant change in the fl ux. It appears that the control
coeffi cient for many enzymes is small, hence heterozygous individuals are
normal and mutations inactivating one allele are classifi ed as recessive. Post -
transcriptional mechanisms may infl uence such simple considerations based
on gene dose. The formal treatment also introduces elasticity coeffi cients for
each enzyme, formerly known as the controllability coeffi cients, which roughly
defi ne the sensitivity of an enzyme to all the metabolites and effectors that
interact with it. The reader is referred to the original literature for a formal
discussion of the terminology and mathematical treatment.
While the treatment is highly theoretical and lends itself to explicit computer
simulations, it is also highly suitable for the interpretation of experimental data
from well - known pathways such as glycolysis and oxidative phosphorylation.
Input substrate concentrations can be varied, specifi c inhibitors can be used to
modulate the activity of individual enzymes in the pathway, and more recently
it has become possible to manipulate individual enzyme levels by genetic meth-
odologies (e.g., in yeast). On the other hand, the best computer model is useless
if it is too simple to refl ect the reality inside the cell.
Applications of metabolic control analysis to oxidative phosphorylation
have been numerous over the years, and experimental systems have included
isolated mitochondria, intact cells, perfused tissues, and whole living organisms.
Recent examples by Brand and his colleagues describe (a) the metabolic
control analysis of the effects of cadmium on oxidative phosphorylation in
mitochondria isolated from potato tubers (306) and (b) the control of oxida-
tive phosphorylation in liver mitochondria, hepatocytes, and other tissues
(307 – 312) . Before discussing the formalism used by these groups and the con-
clusions derived from this “ top - down approach ” (i.e., “ starting from the top
and analysing the whole system ” ), a more conventional set of considerations
will be presented.
