Scheffler Immo E. Mitochondria
Подождите немного. Документ загружается.

THE DYNAMICS OF MITOCHONDRIAL MORPHOLOGY 41
membranes will cause mitochondria to grow, and eventually there must be a
signal for such a mitochondrion to divide, although insertion of lipids and
proteins into the inner membrane alone may simply increase the number of
cristae. Mitochondrial fi ssion appears a priori to be an essential event in pro-
liferating cells, since mitochondria have to multiply and ultimately be divided
between daughter cells. A surprising fi nding is that mitochondria and peroxi-
somes share a signifi cant number of components of their division machinery
(60) . Some spectacular electron micrographs (24) showing bilaminar septa
during progressive stages of mitochondrial divisions leave one with little doubt
about the overall mechanism, but they raise new questions about the control
of this process which remain largely unanswered. Since an inner and an outer
membrane are involved, the process is not a simple pinching - in - half of a mem-
brane vesicle, and it may be more analogous to the division of a bacterium,
where the plasma membrane and the cell wall represent separate structures
surrounding the cytosol. Microscopic observations suggest that fi ssion is pre-
ceded by stretching, but constrictions may appear at one site transiently and
disappear without the initiation of fi ssion (34) .
There is an alternative view of the division of mitochondria, based on
observations with a select number of organisms. In the unicellular red alga
Cyanidioschyzon merolae a structure referred to as an MD ring (mitochon-
drion dividing apparatus) was observed. This ring - like structure was found on
the outside of mitochondria, and it was interpreted to function in a manner
analogous to the contractile ring in cytokinesis (61) . The authors claimed
that cytochalasin B inhibited “ mitochondriokinesis, ” and they deduced the
presence of actin fi laments in the ring. However, these fi laments could not be
stained with anti - actin antibodies or with phalloidin. Because such rings have
not been observed in any other organism, the generality and signifi cance of
these observations must remain in doubt.
The fi ssion mechanism in yeast requires a complex of at least three major
proteins that is localized on the outer mitochondrial membrane, with major
domains exposed to the cytosol. Fis1p (hFis1p in humans) is an integral mem-
brane protein with a single transmembrane domain. Its C - terminus is anchored
in the outer membrane, and its N - terminal domain in the cytosol contains an
array of six - helices forming a tetratricopeptide - like fold with a binding pocket
for interactions with either of two closely related components, Mdv1p and
Caf4p. Structural studies suggest that these can form homo - oligomeric struc-
tures. Fis1p is distributed uniformly on mitochondria and required for the
association of Mdv1p (or Caf4p) on the mitochondrial surface. Together, Fis1p -
Mdv1p (in yeast) assist in the incorporation of the dynamin - related GTPase,
Dnm1p, in a fi ssion complex that can be visualized by punctate staining on
mitochondria. The assembly pathway and its regulation remain to be eluci-
dated in detail. A signifi cant amount of information has, however, been accu-
mulated on dynamin and dynamin - related proteins (DRP1 or DLP1 in humans)
(62 – 64) . Dnp1 behaves like a soluble protein when purifi ed, and it contains a
GTPase domain at the N - terminal and a C - terminal GTPase effector domain.

42 STRUCTURE AND MORPHOLOGY. INTEGRATION INTO THE CELL
As fi rst recognized in the study of endocytosis, the protein assembles at the
location of the constriction where a membrane vesicle buds from its parent
membrane. A plausible and favored model suggests that the dynamin or
dynamin - like proteins aggregate in a ring - like structure at the site of the mem-
brane constriction and ultimately pinch off the vesicle. A tubular structure like
a mitochondrion could be pinched into two pieces at such a site. GTP hydro-
lysis represents a likely driving force, but a detailed mechanism is still elusive.
For example, it is not clear whether constriction is achieved by a contraction
of a dynamin ring due to conformational changes, or whether the ring con-
tracts with the concomitant loss of dynamin monomers (62) . Further specula-
tions in the case of mitochondria include the formation of an initial, local
constriction with the help of the inner - membrane protein Mdm33p, or a
stretching of mitochondria by molecular motors interacting with cytoskeletal
elements (59) . Clearly, the mechanism must include the outer and inner mem-
branes in a concerted fashion, and there must be signals for when and where
to assemble the fi ssion apparatus.
3.3.1.2 Fusion Collisions brought about by the movement of the organ-
elles may lead to fusions, especially when at least one tip makes contact with
TABLE 3.1 Genes/Proteins Involved in Mitochondrial Fusion and Fission (59)
Yeast
Gene/Protein
Human
Homologue
Localization in
Mitochondria Function
Fzo1 Mnf1/2 Outer membrane (IMP) Fusion
Mgm1 OPA1 IMS or inner membrane
(IMP?)
Fusion
Ugo1 Outer membrane (IMP) Fusion
Mdm30 Cytosol/outer membrane Fzo1 degradation
Pcp1 hPARL Inner membrane (IMP) Mgm1p processing
Dnm1 Drp1/DLP1 Cytosol/outer membrane
(peripheral)
Fission
Fis1 hFis1 Outer membrane (IMP) Fission
Mdv1 Cytosol/outer membrane
(peripheral)
Fission
Caf1 Cytosol/outer membrane
(peripheral)
Fission
Mmm1 Outer membrane - inner
membrane spanning
Tubulation
Mdm10 Outer membrane (IMP) Tubulation
Mdm12 Outer membrane (IMP) Tubulation
Mdm31 Inner membrane (IMP) Tubulation
Mdm32 Inner membrane (IMP) Tubulation
Mdm33 Inner membrane (IMP) Inner membrane fi ssion?
Gem1 Miro - 1/2 Outer membrane (IMP) Ca
2+
signaling?
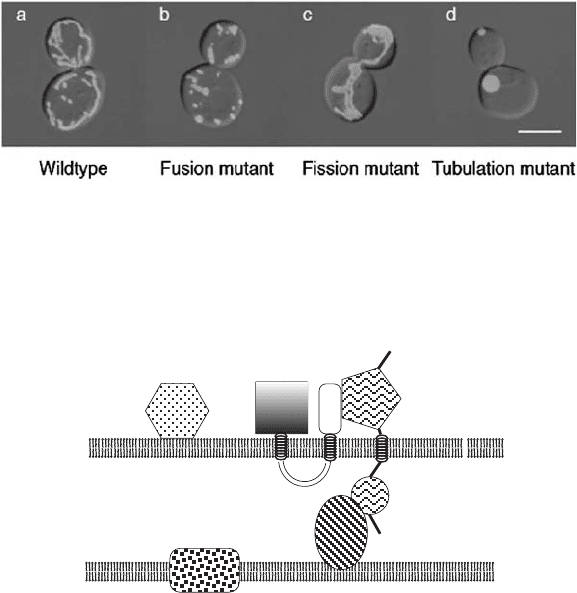
THE DYNAMICS OF MITOCHONDRIAL MORPHOLOGY 43
Figure 3.11 GFP targeted to the matrix in wild - type yeast cells and in various mutants:
fusion mutant ( fzo1 Δ ), fi ssion mutant ( dnm1 Δ ), tabulation mutant ( mmm1 Δ ); GFP
fl uorescence images were superimposed on images from a differential interference
contrast microscope. (From the review of Okamoto and Shaw (59) .) See color plates.
Figure 3.12 Schematic representation of fusion proteins in the outer (OM) and inner
(IM) membrane. (From the review of Okomoto and Shaw (59) .)
another, or with the side. Contact sites and the creation of contact zones are
inferred from observations with the electron microscope, but unfortunately
such images represent a static situation that cannot be further resolved in time.
In contrast to the secretory pathway, the membrane fusions occur between
identical organelles, although some asymmetry between docking vesicle and
target membrane may be transiently created when a mitochondrial tip encoun-
ters a side. Needless to say, fusion involves the coordinate but presumably
sequential fusion of biochemically distinct outer and inner membranes. It is a
unique process that excludes all other intracellular membranes. The same
mechanisms responsible for the dynamic nature of cristae will operate subse-
quently to rearrange the cristae in the fused organelle.
Mitochondrial fusion is a developmentally programmed mechanism in sper-
matogenesis in fruit fl y males. The analysis of a mutation causing male sterility
led to the discovery of a gene, fuzzy onion (Fzo), named after the appearance
Mdm30p
Fzo1p
N
C
N
C
Ugo1p
Mgm1p
Pcp1p
IMS
MATRIX
O M
I M
44 STRUCTURE AND MORPHOLOGY. INTEGRATION INTO THE CELL
of a large structure formed when mitochondria fail to fuse normally during
spermatogenesis. Advances in genomics greatly aided in the search for homol-
ogous genes in yeast (Fzo) and humans (Mfn1 and Mfn2 — mitofusin). When
the gene was found in yeast, the characterization of the Fzo protein and its
function led to some rapid advances and greatly stimulated the search for
additional genes required for the fusion pathway. The Fzo1 protein is an
integral outer - membrane protein, with an evolutionarily conserved GTPase
domain on the cytoplasmic side. Two transmembrane segments are connected
by a short linker exposed to the intermembrane space. The presence of a
temperature - sensitive allele in yeast prevents mitochondrial fusions at the
nonpermissive temperature, and the mitochondria become increasingly frag-
mented. A second GTPase essential for fusion was discovered unexpectedly
by studies of a gene required for mitochondrial genome maintenance in yeast.
The Mgm1p is a GTPase related to the family of dynamins; the corresponding
mammalian gene is OPA1, initially named for a mutation in OPA1 that causes
autosomal dominant optic atrophy (65) . It is an integral inner - membrane
protein with its functional domain localized in the intermembrane space.
Another dynamin - like GTPase has already been introduced in the context of
fi ssion (see above). A third protein, Ugo1p, so far identifi ed only in fungi, spans
the outer membrane and has functional domains on the inside to interact with
Mgm1p, and on the outside to interact with Fzo1p. Fzo1p, Ugo1p, and Mgm1p
thus form a fusion complex connecting the inner and outer membranes (53,
59) . A recent development identifi es two functional isoforms of Mgm1p pro-
duced by alternative topogenesis. The sorting is controlled by a novel protein,
Ups1p (in yeast) and PRELI (in humans) (66) .
The most dramatic progress in the study of mitochondrial fusion was
achieved recently when Meeusen et al. (67) developed an in vitro mitochon-
drial fusion assay. Effectively, two populations of mitochondria were labeled
with two different chromophores in the matrix, mixed, concentrated, and
brought into contact by centrifugation, and fusion could be observed by the
mixing of the chromophores in the fused mitochondria. For the fi rst time,
conditions in this in vitro system could be chosen to restrict the fusion only to
the outer membranes. Such a step required functional Fzo1 proteins on both
membranes, suggesting that cytoplasmic domains of the Fzo1p bind to each
other in a homotypic interaction. In support, X - ray crystallographic data reveal
that the C - terminal domains of Mfn1 (the human homologue) form a dimeric,
antiparallel coiled - coil structure (68) . Endogenous levels of GTP are suffi cient,
and the GTPase activity of Fzo1 is required, since fusion did not take place
when temperature - sensitive Fzo1 proteins were present. Most intriguingly,
outer - membrane fusion requires the proton gradient component of the inner
membrane ( Δ pH), but not the electrochemical component ( ΔΨ ). Thus, when
mitochondria are brought close together physically, the Fzo1 proteins act as
tethers, similar to the SNARE proteins in the secretory pathway. In vivo this
initial interaction is likely brought about by an interaction with cytoskeletal
elements and ATP - dependent molecular motors. The fi nal steps in fusion and
THE DYNAMICS OF MITOCHONDRIAL MORPHOLOGY 45
the use of the proton gradient are still mysterious. To achieve inner - membrane
fusion, two additional requirements must be met: A suffi ciently large electro-
chemical gradient ( ΔΨ ) must be present across the inner membrane, and ele-
vated levels of GTP must be available for hydrolysis. This observation clearly
implicates the second GTPase (Mgm1p/OPA1) localized in the IMS. It is also
consistent with a role of Mgm1/Opa1 in maintenance of cristae morphology
and ATP synthase assembly (69, 70) .
A major challenge for the future will be to understand the control of fi ssion
and fusion. In wild - type cells these processes must be balanced, except when
the number of mitochondria increases in preparation for cell division. At one
level, one expects control over the synthesis and turnover of the major proteins
introduced above. Undoubtedly, other participating proteins will be added to
the picture, some of them universal and some species - specifi c. Their assembly
into fusion and fi ssion complexes is also likely to be under some control mecha-
nism. For example, Mgm1p exists in two forms, a “ precursor ” and a mature
form that are both required for fusion. After import and cleavage of the target-
ing signal, the precursor is processed by the inner - membrane rhomboid prote-
ase Pep1. The exposure of the cleavage site in turn is dependent on matrix ATP
levels. As usual, our understanding is more complete in yeast compared to
mammalian cells, where it has been suggested that different variants of OPA1
are created by variation in splicing instead of proteolytic processing (53, 71) .
Jeyaraju et al. (72) have investigated the human orthologue called PARL
(presenilin - associated rhomboid - like protein) in animal cells. PARL has an
unrelated, vertebrate - specifi c N - terminal domain. The protein is an integral
inner - membrane protein with the N - terminal on the matrix side. After import
and processing, a second cleavage by PARL activity (in trans) occurs. The
highly conserved sequence of this domain, along with the presence of potential
Ser/Thr phosphorylation sites, suggested to these authors to test alanine sub-
stitutions at these residues. To summarize a much more detailed analysis, the
authors provided evidence for the involvement of the PARL protease in the
control of mitochondrial morphology. The non - phosphorylated, cleavable form
of the enzyme was able to induce mitochondrial fragmentation when tran-
siently overexpressed in HEK 293 cells. In contrast, aspartate substitutions at
these positions to mimic phosphorylation prevented this process.
In another recent study it was shown that the F - box protein Mdm30 binds
to Fzo1 and mediates its proteolysis by a proteasome - independent mechanism
(73) . This represents a novel mechanism for Fzo1 degradation in yeast in addi-
tion to the mechanism involving ubiquitinylation and proteasome - dependent
turnover.
Fusion events involving mitochondria that are developmentally pro-
grammed have been characterized in many systems (74) . A later section will
detail some salient insights into this phenomenon in the context of cell
differentiation.
One should also consider the behavior of the mitochondria and the mito-
chondrial genomes when two cells fuse, as in mating of haploid yeast cells. This
46 STRUCTURE AND MORPHOLOGY. INTEGRATION INTO THE CELL
aspect will be considered in more detail below. The observation of constant
fusions and fi ssions of mitochondria in a mammalian somatic cell has implica-
tions for the geneticist. In the absence of additional information (see below)
the dynamic behavior leads one to conclude that mitochondrial genomes in a
heteroplasmic cell are continuously mixed and redistributed, with the result
that a given mitochondrion contains both types of genomes at any time. It
may serve as a mechanism to “ homogenize ” mitochondrial genomes among
the many mitochondria in a cell. The implications for our understanding of
human mitochondrial diseases will be discussed in a later chapter. In some
organisms it may also serve to promote recombination between mitochondrial
genomes.
3.3.2 Distribution During Cell Division
When a cell contains a large number of mitochondria more or less evenly dis-
tributed throughout the cytoplasm, and doubling in number and volume during
the cell cycle, no signifi cant conceptual problem exists for distributing these
mitochondria evenly into the daughter cells during cytokinesis. Furthermore,
if mitochondria become attached evenly to the newly formed microtubules of
the mitotic half spindles, their segregation is easy to visualize. The problem
during the cell cycle, therefore, is to assure a doubling of mitochondrial mass
and number and to maintain a constant ratio of nuclear to mitochondrial
genes. Cell differentiation during development, along with mitochondrial
proliferation and turnover in terminally differentiated cells like muscle and
neurons, poses a different problem.
A special problem arises when cell division occurs not by the classical
process of cytokinesis in which a larger cell is partitioned into two cells either
by a contractile ring, as in animal cells, or by the assembly of a phragmoplast
in plant cells. In the budding yeast Saccharomyces cerevisiae a bud is started
and enlarged during the cell cycle, and all the constituents for a new viable
cell have to be transported through a narrow connection between the bud and
the mother cell. How this is achieved for the nuclear chromosomes has been
a subject of study for some time, but the problem of equipping the bud with
mitochondria has been illuminated by pioneering studies starting in the labo-
ratory of M. Yaffe (see reference 50 for an expert review). Taking a genetic
approach, the Yaffe laboratory has identifi ed yeast ts mutants that are defec-
tive in the transfer of mitochondria and nuclei into growing daughter buds at
the nonpermissive temperature. Others have contributed over the years, and
the subject has started to merge with the general topic of mitochondrial mor-
phology and dynamics in yeast and other eukaryotes (52, 59) . A pathway that
may be unique to fungi has been referred to as the tabulation pathway.
The original mdm1 - 1 allele, along with additional mdm1 alleles isolated
later, defi nes a gene encoding a protein (Mdm1p) with several intriguing
properties. The protein is essential for viability. With some mutant proteins the
problem of mitochondrial distribution can be separated from nuclear distribu-

THE DYNAMICS OF MITOCHONDRIAL MORPHOLOGY 47
tion. The protein sequence has some similarity to intermediate fi lament pro-
teins of vertebrates, and it forms 10 - nm fi laments in vitro . Filament formation
fails at the nonpermissive temperature in vitro with the protein encoded by
the original mdm1 - 1 (ts) allele. On the other hand, indirect immunofl uores-
cence microscopy with anti - Mdm1p antibodies reveals punctate staining
distributed throughout the cytoplasm of wild - type cells. With mutant alleles
affecting mitochondrial inheritance, the mitochondrial morphology in the
mother cells is also abnormal. Instead of the usual reticulated, tubular wild -
type structure, one fi nds small, round mitochondria in clusters. These observa-
tions invite speculation that a cytoplasmic fi lamentous network or scaffold
exists which is responsible for the maintenance of the mitochondrial morphol-
ogy of yeast discussed above, and that it also plays a major role in guiding
presumably threadlike mitochondrial structures through the connection into
the bud. It should be mentioned that fungi do not have the typical intermedi-
ate fi lament proteins of higher eukaryotes. The yeast fi laments made from
Mdm1p may represent an alternate solution to a problem that reached an
evolutionary dead end, and they were replaced by proper intermediate fi la-
ments in more advanced organisms. The mdm1 mutation does not appear to
affect actin - and tubulin - based cytoskeletal functions.
Mdm1p may not be the only protein involved in this peculiar yeast cyto-
skeletal structure. Additional mutants isolated in the Yaffe lab have led to the
characterization of other MDM genes ( m itochondrial d istribution and m or-
phology), of which MDM14 encodes a 35 - kDa protein that appears to interact
with the Mdm1p and is distributed in the cytoplasm in a similar fashion. Not
unexpectedly, on second thought, some mdm mutants are affected in proteins
in the outer mitochondrial membrane ( mdm10, mdm12 ) (50, 75) . (See Figure
3.13 .) Clearly, association with a cytoskeletal element implies the existence of
a protein domain on the mitochondrial surface that must at least bind to the
scaffold, and perhaps even have motor - like properties. A related gene product
in the outer membrane is the Mmm1p ( m aintenance of m itochondrial m or-
phology) identifi ed by Burgess et al. (76) . Depletion of these proteins has
pronounced effects on mitochondrial morphology, and null mutations yield a
temperature - sensitive phenotype. As pointed out by Berger and Yaffe (50) ,
the fact that these null mutants grow at the permissive temperature on glucose
(albeit slowly) suggests that a bypass or backup pathway must exist to assure
mitochondrial transmission to daughter buds. Nevertheless, mdm10 and mdm12
mutants lose mitochondrial DNA and generate respiration - defi cient cells at
higher - than - normal rates. New members have been added to the MMM and
MDM groups of genes (52, 59, 77, 78) . The corresponding proteins have been
localized in the inner membrane (Mdm31p and Mdm32p), or in (or on) the
outer membrane (Mdm10p, Mdm12p, Mmm1p, Mmm1p). The Mmm1p is most
unusual in spanning both the inner and outer membrane, with its N - terminus
in the matrix and its C - terminus on the cytoplasmic side. It acts like a rivet for
these two membranes, and at the same time it forms a complex with the
Mdm10p and Mdm12p (the MMM complex) and transiently interacts with
48 STRUCTURE AND MORPHOLOGY. INTEGRATION INTO THE CELL
Mmm2p. Based on phenotypes exhibited by mutants lacking the MMM
complex, the complex is implicated in at least three signifi cant processes:
attachment of mitochondria to actin, formation of tubular mitochondria, and
the the anchoring of the mtDNA nucleoids to the inner membrane. The fi rst
is likely to be relevant for moving mitochondria around in the cytosol and into
the bud during cell division. The second could also involve attachment to a
cytoskeletal element (still undefi ned; Mdm1p?), or it could include the con-
struction of an external scaffold that constrains mitochondria to a tubular
shape. In its absence the mitochondria turn into spheres. Finally, the N -
terminal domain of Mmm1p in the matrix has been colocalized, together with
other proteins (e.g., Mgm101p) and mtDNA, in a large structure referred to
as the mtDNA nucleod. It is the site of both mtDNA replication and transcrip-
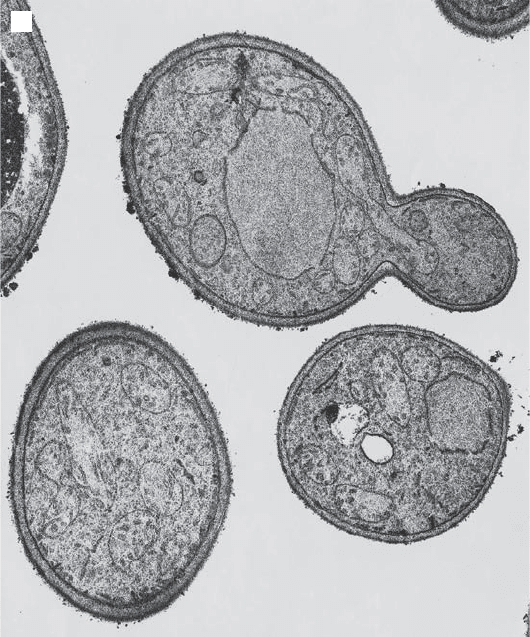
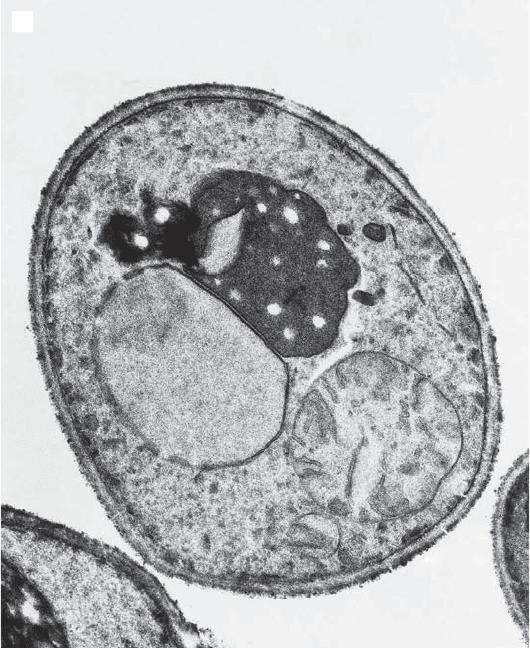
Figure 3.13 (A) Mitochondria in a budding, wild type yeast cell. Note the movement
of one elongated mitochondrion into the bud. (B) Abnormal, giant spherical mito-
chondria in yeast cells with the mdm10 mutation (50, 75) . (Photographs provided by
M. Yaffe, University of California, San Diego.)
A
THE DYNAMICS OF MITOCHONDRIAL MORPHOLOGY 49
tion, to be discussed in a later chapter. In the present context it is important
to recognize that daughter cells must inherit mitochondria with mtDNA.
Therefore, dysfunction of the MMM complex results not only in abnormal
mitochondrial shape and inheritance, but also in severe mtDNA instability.
The overall potential complexity of the processes under consideration is
illustrated by the failure of mitochondrial inheritance in the mdm2 mutant. In
this mutant the OLE1 gene is affected, encoding a fatty acid desaturase (79) .
Mitochondrial membrane fl uidity evidently affects mitochondrial transmis-
sion, but it may be too simple - minded to think of a mitochondrion squeezing
through a tiny pore connecting mother cell and bud. A much more exhaustive
screen of 768 yeast mutants ( ∼ 2/3 of all essential genes) has uncovered
119 genes required for maintenance of mitochondrial morphology (56) .
They encode proteins involved in ergosterol biosynthesis, mitochondrial
protein import, actin - dependent transport processes, vesicular traffi cking, and
B
Figure 3.13 (Continued)
50 STRUCTURE AND MORPHOLOGY. INTEGRATION INTO THE CELL
ubiquitin/26S proteasome - dependent protein degradation. It becomes quite
apparent that the physical as well as functional integration of mitochondria
into a cell are intimately linked, and many details still elude us.
Meiosis and sporulation in yeast represent a special kind of cell division in
which most of the emphasis is usually placed on the behavior of the chromo-
somes. What about mitochondria? One study has defi ned morphological tran-
sitions in the early stages followed by a dramatic fragmentation of a single
large mitochondrion into small spherical structures that were distributed to
the spores (80) . Mechanisms for such events remain largely speculative at this
time. If, as discussed above, fusion and fi ssion occur normally at equal rates,
one may simply have to inhibit fusions to promote the breakup of a large
mitochondrion into smaller units. The phosphorylation or dephosphorylation
of an essential docking protein may be all that is required, but identifying it
is another problem.
Before meiosis can occur in yeast cells, one must have a diploid cell formed
from the fusion of two haploid cells. When respiration - defi cient yeast mutants
were fi rst investigated by means of mating experiments, and observations were
made on the stability of phenotypes during mitotic divisions of the zygote, a
puzzling observation was made. Zygotes containing heteroplasmic mtDNA
populations generated homoplasmic progeny within approximately 20 genera-
tions, much faster than expected on the basis of stochastic models assuming
random segregation (see references 81 and 82 for a recent review). This obser-
vation is even more puzzling in light of the discussion about the dynamic
nature of the large mitochondrial structure and the implied constant mixing
of mtDNAs. A study by Nunnari et al. (35) has reinvestigated this problem
with a most elegant and ingenious approach. Mitochondria of one haploid cell
were labeled with GFP; mitochondrial proteins of the other mating type were
labeled with the vital dye TMR - CH
2
Cl, which not only localized in mitochon-
dria because of the membrane potential, but eventually becomes covalently
attached to mitochondrial proteins. When such cells were allowed to mate, the
fusion and complete mixing of the two mitochondrial protein populations
could be directly observed by wide - fi eld fl uorescence microscopy: These obser-
vations were fully consistent with observations described above on the dynamic
behavior of yeast mitochondria. The more provocative result was obtained
when mtDNA from one mating partner was labeled selectively with bromo-
deoxyuridine (by a not totally trivial procedure). Its distribution in the zygote
could subsequently be determined immunochemically after fi xation and per-
meabilization of the cells. The results showed unequivocally that while mito-
chondrial proteins from the two mitochondrial populations mixed readily, the
corresponding mtDNA populations remained segregated in the two lobes of
the zygote derived from the haploid cells. This result was a visual confi rmation
of previous genetic experiments which had inferred the unequal distribution
of mtDNA in the zygote from an analysis of mtDNA in zygotic progeny. In
such progeny the type of mtDNA (genetically marked) depended on the posi-
