Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

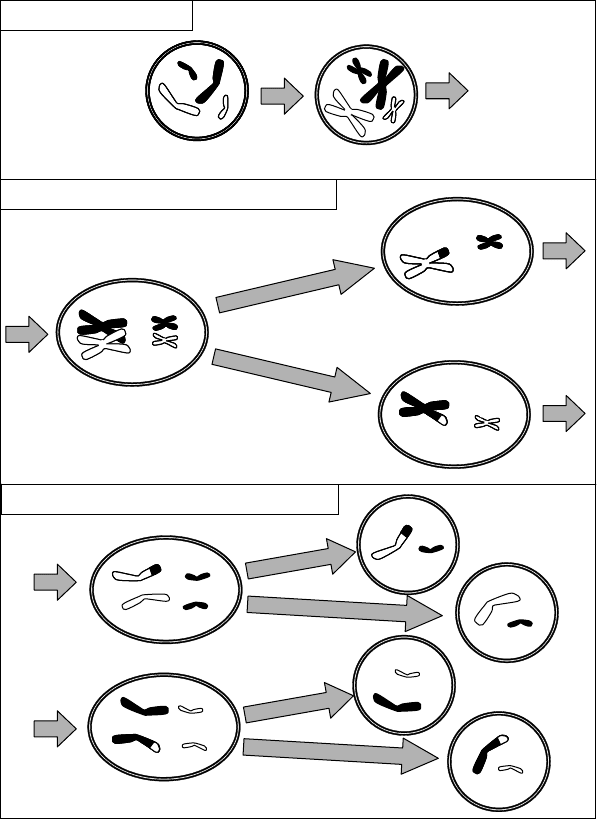
chromosomes, more combinations would be possible. In all, there would be 2
N
, where N is
the number of chromosome pairs. For humans with 23 pairs, this results in
2
23
¼ 8:4 10
6
possible combinations. If crossing over did not occur, this is the number
of genetically unique offspring that a single human couple could possible produce. The
combinations occur randomly, depending on how the chromosomes align before being
pulled apart by the spindle. The random distribution of chromosomes to the daughter
cells in the first stage of meiosis is called independent assortment.
When random assortmen t is combined with crossing over, the numb er of possible
daughter cells that can be formed is extremely large. This is an important source of the
random variation that drives evolution. Even without mutations, crossing over can create
new genes by combining pieces of two old ones. The great variation in offspring that
Interphase: replication
Meiosis I: crossing over and first division
Meiosis II: alignment and second division
One diploid cell
Four haploid cells
Figure 4.7 Meiosis: crossing over and independent assortment of chromosomes.
78
THE CELL: THE COMMON DENOMINATOR OF LIVING THINGS
results can help a species adapt rapidly to changing environmental conditions. This is one
of the great evolutionary advantages of sexual reproduction.
The meiotic process just described is actually only the first step, called meiosis I. After
that step, two daughter cells are produced that although already haploid, contain two of
each type of chromosome that originated in the replication process. In meiosis II, these
are separated in another division process, resulting in a total of four haploid cells, each of
which probably has a unique genetic complement.
PROBLEMS
4.1 Name the kinds of physicochemical or chemical effects the following types of
pollutants might have on plasma cell membranes: (a) dissolved hydrophobic solvents
such as benzene or TCE; (b) surfactants such as detergents; (c) salts such as chloride
or sulfate; (d) strong oxidizers such as chlorine or ozone.
4.2 Why do animal cells not need cell walls to maintain their structure?
4.3 Consider a solution of 500 mg of NaCl per liter as an analog for tap water. What is the
particle concentration in mmol/L? What will be its osmotic pressure?
4.4 A fruit fly has four pairs of chromosomes. How many possible combinations can be
produced in its gametes by random assortment (with no crossing over)? List the
possibilities.
REFERENCES
Atlas, R. M., and Bartha Richard, 1981. Microbial Ecology: Fundamentals and Applications,
Addison-Wesley, Reading, MA.
Bailey, J. E., and D. F. Ollis, 1986. Biochemical Engineering Fundamentals, McGraw-Hill, New York.
Fried, G. H., 1990. Schaum’s Outline: Theory and Problems of Biology, McGraw-Hill, New York.
Gaudy, A. F., and E. T. Gaudy, 1988. Elements of Bioenvironmental Engineering, Engineering Press,
San Jose, CA.
Grady, C. P., Jr., G. T. Daigger, and H. C. Lim, 1999. Biological Wastewater Treatment, Marcel
Dekker, New York.
Henze, et al., 1986. Activated Sludge Model No. 1, International Association on Water, London.
Holland, J. H., 1992. Genetic algorithms, Scientific American, Vol. 267, No. 1, pp. 66–72.
Lim, Daniel, 1998. Microbiology, 2nd ed., WCB McGraw-Hill, New York.
Postlethwait, J. H., and J. L. Hopson, 1995. The Nature of Life, McGraw-Hill, New York.
Smith, E. L., R. L. Hill, I. R., Lehman, R. J. Lefkowitz, P. Handler, and White, 1983. Principles
of Biochemistry: General Aspects, McGraw-Hill, New York.
Stryer, L., 1995. Biochemistry, 4th ed., W.H. Freeman, New York.
Van de Graaff, K. M., and R. W. Rhees, 1997. Schaum’s Outline: Theory and Problems of Human
Anatomy and Physiology, McGraw-Hill, New York.
Watson, J. D., 1968, The Double Helix, Atheneum, New York.
White, A., P. Handler, and E. L. Smith, 1973. Principles of Biochemistry, McGraw-Hill, New York.
REFERENCES 79

5
ENERGY AND METABOLISM
All the biochemical reactions in a living organism, taken together, are the organism’s
metabolism. The metabo lic processes of life can ultimately be described as a complex
set of chemical reactions distributed in space and time, and coupled by shared intermedi-
ate compounds and transport mechanisms. In this chapter we focus on the chemical reac-
tions themselves, but the reader should also pay attention to the where and when of the
chemical choreography of the cell.
Our first concern will be whether or not a particular biochemical reaction can proceed.
This question is answered by thermodynamics. In general, a reaction is feasible if the pro-
ducts are collectively more stable (in a lower-energy state) than the reactants. Thermody-
namics also describes how energy is produced or consumed by a reaction. Although a
reaction may be therm odynamically feasible, there may be an energy barrier between pro-
ducts and reactants that limits the rate at which it occurs. We describe the rate at which
reactions occur using chemical kinetics. Finally, we examine several of the most impor-
tant metabolic reactions found in living things.
5.1 BIOENERGETICS
Thermodynamic relationships govern whether a reaction can occur, although not whether
it will. A reaction may be thermodynamically feasible but may occur too slowly for prac-
tical consideration. We will also be very concerned with whether a reaction provides
energy for doing biochemical work or requires such energy from other sources.
5.1.1 Some Basic Thermodynamics
The second law of thermodynamics states that processes in an isolated system, including
chemical reactions, tend to proceed from a less likely to a more likely state: that is, to
Environmental Biology for Engineers and Scientists, by David A. Vaccari, Peter F. Strom, and James E. Alleman
Copyright # 2006 John Wiley & Sons, Inc.
80
increase entropy (S), or randomness. However, organisms are not isolated systems. They
are better approximated as a closed system, in which energy, but not matter, can cross
their boundaries. In particular, organisms approximate closed systems at constant tem-
perature and pressure. The appropriate measure for energy added to a closed system at
constant pressure is the change in enthalpy, H,
H ¼ U þ P V ð5:1Þ
where U is the resulting change in the internal energy of the system, P is its total pres-
sure, and V is the change in volume of the system.
In closed systems a decrease in entropy can be compensated for by adding energy from
outside the system. This causes the entropy outside the system to increase enough so that
the total entropy change is positive. For a closed system at constant temper ature and pres-
sure, the relevant quantity to determine if a process is feasible is the change in the Gibbs
free energy, G , which includes the effects of both entropy and enthalpy:
G ¼ H T S ð5:2Þ
To summarize, in isolated systems changes in energy are described in terms of the
internal energy, U, and the system will always change in a way that increases its entropy,
S. However, for closed systems at constant pressure, it is more convenient to describe both
of these processes in terms of enthalpy, H, and Gibbs free energy, G, respectively.
Thus, a reaction is feasible in closed systems only if it causes a decrease in Gibbs free
energy. For an individual chemical, the Gibbs free energy increases with concentration
logarithmically. For example, the Gibbs free energy of compound A would be
G
A
¼ G
A
þ RT ln½Að5:3Þ
where G
A
is the standard Gibbs free energy of A, corresponding to the Gibbs free
energy at a molar concentration o f A, ½A, equal to 1.0 M in solution or 1.0 atm partial
pressure for gases. The standard Gibbs free energies of formation for several biochemical
compounds are give in Table 5.1. (N ote that chemical activity should be used instead of
concentration, to be strict. However, we will use concentration, which approximates activ-
ity, in order to make the development easier to follow.)
Consider the reaction
aA þ bB , c C þ d D ð5:4Þ
in whi ch a moles of A and b moles of B react to form c moles of C plus d moles of D. As a
result, the Gibbs free energy of the mixture will be decreased by aG
A
þ bG
B
and
increased by cG
C
þ dG
D
, producing a net change in Gibbs free energy for the reaction,
G:
G ¼ cG
C
þ dG
D
aG
A
bG
B
¼ cðG
C
þ RT ln½CÞ þ dðG
D
þ RT ln½DÞ
aðG
A
þ RT ln½AÞ bðG
B
þ RT ln½BÞ ð5:5Þ
BIOENERGETICS 81

Some rearrangement gives the following expression for the change in Gibbs free energy of
the reaction at any concentration of reactants:
G ¼ G
þ RT ln
½C
c
½D
d
½A
a
½B
b
ð5:6Þ
where
G
¼ cG
C
þ dG
D
aG
A
bG
B
ð5:7Þ
is the standard Gibbs free-energy change for the reaction.
If one starts with a mixture of A and B, they would begin to convert to C and D because
if the latter have a low enough concentration, their Gibbs free energy will be less than that
of the reactants. As the reaction proceeds, the combined Gibbs free energy of A and B will
decrease, and the contribution from C and D will increase. Eventually, a point is reached
where any change in the Gibbs free energy of the products exactly balances changes in the
Gibbs free energy of the reactants. Any further reaction would cause an increas e in the
total Gibbs free energy. The total Gibbs free energy is then at a minimum, and if the reac-
tion proceeded infinitesimally, the change in Gibbs free energy given by equation (5.6)
would be zero. Thus, substituting equilibrium concentrations into (5.6) and setting G
TABLE 5.1 Standard Gibbs Free Energies of Formation for
Several Biochemical Compounds
a
Compound Formula G
(kcal/mol)
Oxalic acid (1) (COOH)
2
166.8
Proprionic acid (liq) (2) C
3
H
5
O
6
91.65
Pyruvic acid (liq) (2) C
3
H
4
O
3
110.75
Acetic acid* C
2
H
4
O
2
93.8
Lactic acid (cr) (2) C
3
H
6
O
3
124.98
Glucose (3) C
6
H
12
O
6
216.22
Glycerol (3) C
3
H
8
O
3
116.76
Ethanol (3) C
2
H
6
O 43.39
Ethylene (gas) (1) C
2
H
4
16.282
Methanol (liq) (1) CH
4
O 57.02
Methane (gas) (1) CH
4
12.14
Alanine (3) C
3
H
6
O
2
N 88.75
Glycine (2) C
2
H
5
O
2
N 88.62
Urea (aq) (1) CH
4
ON
2
48.72
Ammonia (gas) (1) NH
3
4.0
Water (liq) (3) H
2
O 56.69
CO
2
(gas) (3) CO
2
94.45
Nitric acid (1) HNO
3
19.1
a
Values for 1 M aqueous solutions at pH 7.0 and 25
C, except as noted; cr,
crystal form.
Source: (1) CRC Handbook of Chemistry and Physics, 60th ed.; (2) Lange’s
Handbook of Chemistry; (3) Lehninger, Biochemistry.
82 ENERGY AN D METABOLISM

to zero, we have
G
¼RT ln
½C
c
eq
½D
d
eq
½A
a
eq
½B
b
eq
¼RT ln K
eq
ð5:8Þ
where K
eq
is defined as the equilibrium coefficient:
½C
c
eq
½D
d
eq
½A
a
eq
½B
b
eq
¼ K
eq
ð5:9Þ
From equation (5.8) we also have that
K
eq
¼ exp
G
RT
ð5:10Þ
The equilibrium constant is a unique thermodynamic constant for each reaction. We
have already met one example of an equilibrium constant in the K
a
for acid–base reactions
as defined in equation (3.2).
By convention, when biochemists compute K
eq
and G
for reactions that involve
water or hydrogen ions, they assume that their concentrations are held constant at 1.0
and 10
7
M (pH 7.0), respectively.
Example 5.1 Compute G
and K
eq
for the complete oxidation of oxalic acid.
Answer First write the balanced equation for the oxidation:
C
2
H
2
O
4
þ
1
2
O
2
! 2CO
2
þ H
2
O
Then use Table 5.1 and equation (5.7):
G
¼ cG
C
þ dG
D
aG
A
bG
B
¼ 2ð94:45Þþ1ð56:69Þð166:8Þ0:0 ¼78 :79 kcal=mol
Note that G
for elements (oxygen, in this case) equals zero. Next, use equation (5.10):
K
eq
¼ exp
G
RT
¼ exp
78:79
ð1:987Þð298:15Þ
¼ 1:14
Keep in mind that equation (5.8) holds only at equilibrium , whereas equation (5.6) is
valid at any combination of reactant and product concentrations. If equation (5.6) evalu-
ates to a negative value, the reaction will tend to proceed in the forward direction as writ-
ten. If it is positive, the reverse reaction will tend to occur. This also means that the actual
amount of Gibbs free energy released by a biochemical reaction will depend on the con-
centrations in the cell. As reaction products build up in relation to reactants, the Gibbs free
energy yield decreases. If product concentrations are held low enough, any reaction can be
made to produce a large amount of Gibbs free energy.
BIOENERGETICS 83
Put another way, it is important to distinguish between G and G
. The latter is the
change in Gibbs free energy only when the reactants are all at concentrations of 1 M (if a
reactant is a gas, its partial pressure is used instead of concentration, and the partial pres-
sure is assumed to be 1 atm). A positive value for G
does not mean that the reaction
cannot proceed. It only means that the equilibrium is tilted toward the reactants. The reac-
tion can be made to proceed by keeping the concentration of one or more of the produc ts
low. On the other hand, G depends on the actual concentration of reactants and pro-
ducts. When the numerator of the quotient on the right-hand side of equation (5.6) is
less than the denominator, the logarithm will b e negative. If it is negative enough so
that its absolute value exceeds G
, G will be negative and the reaction will tend to
proceed spontaneously toward equilibrium. If, on the other hand, the numerator is more
than the denominator, G will be positive, and the reaction will tend to proceed in the
reverse direction.
As an analogy, G
is like the amount of potential energy that would be released
by a fluid falling through a standard elevation change, say, 1 meter. This is a property
of the fluid (a function of its specific gravity). On the other hand, G is like the
potential energy released by an actual change in elevation. This can be made arbitrarily
large, or reversed in sign, by changing the beginning and ending elevations for the
flow.
Equation (5.8) embodies what is known as Le Cha
ˆ
telier’s principle. This states that if
a reaction at equilibrium is distur bed, such as by adding one of the reacta nts or products to
the solution, the reaction will proceed in a direction so as to partially eliminate the dis-
turbance. For example, cells convert glucose-6-phosphate to fructose-6-phosphate as part
of glycolysis. If the fructose-6-phosphate were not subsequently consumed, allowing it to
accumulate, the reaction could stop or even reverse itsel f, so as to maintain the proper
ratio between product and reactant. This is an important mec hanism for the control of
biochemical reactions.
Also remember that the thermodynamic relationships say nothing about how fast a
reaction will proceed. The sucrose crystals in a sugar bowl are unstable in contact with
air in terms of equation (5.6), but do not react at a measurable rate. However, in the pre-
sence of microbial enzymes and other requirements of microbes, such as moisture and
nutrients, the sugar and oxygen are soon converted to carbon dioxide and water.
The enthalpy, H, is the actual amount of energy released by a reaction in a closed
system under constant pressure and temperature. So why do we use G, which is the
enthalpy reduc ed by a factor involving the change in entropy T S [equation (5.2)]
when we discuss the energy provided by a reaction? The answer is that the Gibbs free
energy is the energy available to do work. This work may include the driving of other
reactions ‘‘uphill’’ in a thermodynamic sense. For example, the Gibbs free energy
released in oxidizing sucrose can be used to synthesize amino acids. The H value for
the oxidation of glucose to carbon dioxide to water is 680 kcal/mol, whereas the G
value for this reaction is 686 kcal/mol, which gives K
eq
¼ 3:046.
The Calorie counts for fats, proteins, and carbohydrates represent values for H for
the complete oxidation (except for proteins, because the nitrogen is excreted mostly as
urea, not as nitrate). However, in humans the processes of digestion and absorption
also require energy, amounting to about 6%, 4%, and 30% of the energy in fats, carbohy-
drates, and proteins, respectively. This energy is wasted as heat. This accounts for why
people often want to eat les s in hot weather. It also suggests that they would be less
uncomfortable if they substituted pasta for meat on warm days.
84 ENERGY AN D METABOLISM

5.1.2 Oxidation–Reduction
Among the energy-intensive reactions are oxidation–reduction, or redox, reactions.
Oxidation is the loss of an electron, reduction is the gain. Con sider how easy it is for
some molecules to lose a proton. The acids readily give up a proton (hydrogen ion) in
aqueous solution, but the hydrogen’s electron is left behind. Subsequent removal of the
electron constitutes oxidation and liberates energy. As a result, removal of a hydrogen
atom (with its electron) often is the same as oxidation in biochemical systems. This is
referred to as dehydrogenation. For example, methanol can lose two hydrogens in a
reaction with oxygen to make formaldehyde:
2HC
H
H
OH + O
2
⇔ 2C
O + H
2
O
H
H
Sometimes it is less obvious if a reaction is an oxidation. Reactions of covalently
bonded carbon atoms rarely involve com plete transfer of electrons. Whether an organic
compound was oxidized or reduced can be determined if there is an increase or decrease
in the oxidation state or oxidation number of its atoms, particularly of its carbon atoms.
The oxidation state is determined for each carbon in the molecu le by assuming that all of
the electrons that participate in bonding with the carbon are assigned to the more electro-
negative of the bonded pair. The oxidation state will then be the electrical charge that
remains on the carbon.
For example, because carbon is more electronegative than hydrogen, a 1 is added to
its oxidation state for each hydrogen bonded to it. Since oxygen is more electronegative
than carbon and a pair of electrons are shared in a carbonyl bond (C
O), the carbonyl
bond contributes þ2 to the carbon’s oxidation state. An OH connected to a carbon con-
tributes þ1. Although the oxidation state really refers to a single atom, the oxidation state
of an organic compound can be considered to be the sum of the oxidation state of all its
carbon atoms.
Thus, the carbon in methane has an ox idation state of 4, and carbon dioxide is þ4.
The carbons in sugars have an oxidation state of 0, whereas the alcohol methanol is 2.
Reaction by addition of a water molecule (hydrolysis) does not change the oxidation state
of the carbons in an organic compound.
The oxidation state of different compounds can be compared using the mean oxida-
tion state of carbon (MOC). The MOC is the sum of the oxidation states of all the carbon
atoms in a molecule, divided by the number of carbon atoms. The concept can be
extended to complex mixtures including suspensions such as wastewaters and sludges,
leading to the MOC. The MOC can be estimated using the total organic carbon
(TOC) and the theoretical oxygen demand (ThOD):
MOC ¼ 4 1:5
ThOD
TOC
ð5:11Þ
The TOC can be computed from the chemical formula as the mass of the carbon per
mole of the compound divided by its molar mass. ThOD is the mass of oxygen required
stoichiometrically to oxidize a material completely. The ThOD can be computed from the
balanced equation for the oxidation. The calculation of TOC, ThOD, and MOC is shown
in Example 5.2. ThOD can also be estimated in the laboratory by calculating the chemical
oxygen demand (COD). A sample is oxidized using potassium dichromate with strong
BIOENERGETICS 85

acid and a catalyst under boiling conditions and the amount of oxygen equivalent to the
dichromate is computed.
Example 5.2 Compute the oxidation number of oxalic acid by counting electrons shared
across bonds using equation (5.11).
Answer Each carbon in the oxalate molecule is bonded to two oxygens, one by a dou-
ble bond. Since oxygen is more electronegative than carbon, one electron from the single
bond and two from the double bond are assigned to the oxygens, leaving each carbon with
a charge of þ3. The carbon–carbon bond does not redistribute an electron. Therefore, the
average oxidation number of the two compounds is 3.0.
According to the balanced equation for the oxidation of oxalic acid in Example 5.1,
1
2
mol of O
2
is required to oxidize each mole of oxalic acid. The molar mass of oxygen
is 32 g/mol, and for oxalate it is 90 g/mol. Therefore, the ThoD is ð0:5Þð32=90Þ¼0:178 g
O
2
=g oxalate. The molecular formula of oxalate is C
2
H
2
O
4
, so the TOC ¼ð2Þ
ð12=90Þ¼0:267 g C=g oxalate. Using these values with equation (5.11) yields
MOC ¼ 3:0, which agrees with the value computed directly.
Equation (5.11) assumes that there are not significant amounts of nitro, azo, or halo-
genated compounds present. These can result in MOC values that are too low. It is also
assumed that inorganic interferences with the COD measurement are absent. These can
include cyanide, sulfide, cyanate, thiocyanate, sulfite, nitrite, thiosulfate, Fe
2þ
,Mn
2þ
,or
H
2
O
2
. The MOC can be used as an indication of the extent to which an organic waste has
been treated by oxidation processes.
In oxidation, ele ctrons are never released directly, but are taken up by other com-
pounds, called electron acceptors, which thus become reduced. Hydrogens (and their
electrons) are usually picked up in pairs. Thus, oxygen accepts a pair of electrons and
becomes H
2
O, and nitrate becomes ammoni a. Organic molecules can also accept elec-
trons, as in fermentation reactions, for example. The oxidized forms of some metals
[e.g., Fe(III) and Mn(IV)], can also act as electron acceptors.
A cell will often need to oxidize or reduce compounds. Since every oxidation must be
accompanied by a reduction (one compound loses an electron, another gains), the cell
could do this by coupling the oxidation of one compound to another that needs to be
reduced. However, the great variety of individual oxidations and reductions that may
be needed would result in a huge variety of combined reactions, and it would be necessary
to have an enzyme syst em for each. A simpler appro ach is to have a smaller numb er of
intermediary compounds that accept electrons in oxidations and then give them up in
separate reduction reactions. This is called reaction coupling.
Several of the important compounds involved in coupling redox reactions are nucleotides
that have been mentioned previously, including NAD, NADP, and FAD. In their reduced
forms they are referred to as NADH
2
, NADPH
2
, and FADH
2
(often wr itten without the
subscript ‘‘2’’), respectively. NAD serves as an oxidizing agent in many biochemical reac-
tions, extracting a hydrogen with its electron. Conversely, NADH
2
is a reducing agent.
These acronyms are a shortened form. NAD is actually NAD
þ
, and NADH
2
is actually
NADH þ H
þ
, and similarly for NADP and FAD. In this book we use the simpler notation.
5.1.3 Phosphate Compounds and ATP
Another form of reaction coupling involves the liberation of energy in reactions, because
the energy liberate d by one reaction could be used to drive another. As in redox coupling,
86 ENERGY AN D METABOLISM
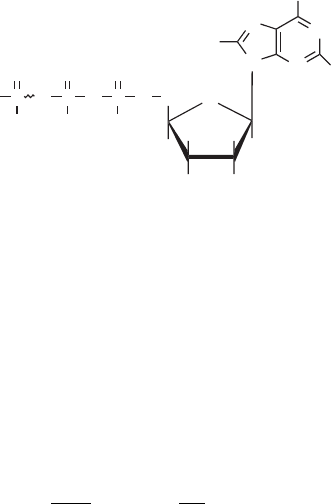
however, there is a need for an intermediary to serve as a sort of broker in the transfer of
energy from one set of reactions to another. This function is served mostly by a single
compound, adenosine triphosphate (ATP).
Energy is stored in a high-energy bond between phosphate and organic com pounds.
Such bonds have standar d Gibbs free energies of hydrolysis in the range 2to
13 kcal/mol. The reaction that bonds phosphate to an organic is called phosphorylation.
The reverse reaction is hydrolysis. Phosphorylation of an organic is often a step in its
oxidation. For example, the first step in oxidation of glucose is conversion to glucose-6-
phosphate.
A number of compounds have energies in the higher end of the range given above,
including adenosine triphosphate (ATP). ATP is used to provide energy to almost all bio-
chemical reactions that require energy. The sta ndard Gibbs free energy for the hydrolysis
of ATP to adenoside diphosphate (ADP) is 7.3 kcal/mol:
ATP , ADP þ P
i
G
¼7:3 kcal=mol ð5:12Þ
However, since other reactions remove phosphate continually, this reaction is not at
equilibrium in the cell. The actual G can be as negative as 11 or 12 kcal/mol or
more. ATP is not stored in significant quantities by cells. If production stopped, a muscle
cell would deplete its supply in a few minutes. However, the turnover rate is quite high.
Humans produce and use about 40 kg in a day.
N
N
NH
2
N
N
H
H
O
HH
OHOH
H
CH
2
H
OP
OH
O
OPOPHO
OO
OH OH
adenosine triphosphate (ATP)
5.1.4 Reaction Coupling
To show how reaction coupling works, consider the following hypothetical reaction:
A , B G
¼þ4:0 kcal=mol ð5:13Þ
The equilibrium ratio for this reaction will have the following value at 310 K (based on
equation (5.7):
K
A=B
¼
½B
eq
½A
eq
¼ exp
4:0
RT
¼ 1:5 10
3
ð5:14Þ
Thus, at equilibrium only a small portion of A will be converted to B. However, if by some
mechanism, reaction (5.13) could be combined with reaction (5.12), the net reaction and
BIOENERGETICS 87
