Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.


standard Gibbs free-energy change would be
A þ ATP , B þ ADP þ P
i
G
¼ 4:0 7:3 ¼3:3 kcal= mol ð5:15Þ
The equilibrium constant for reaction (5.15) is
K
0
A=B
¼
½B
eq
½A
eq
½ADP
eq
½P
i
eq
½ATP
eq
¼ exp þ
3:3
RT
¼ 216:0 ð5:16Þ
However, ATP in cells is never in equilibrium with ADP and P
i
. Under physiological
conditions the relationship is approximated by
½ATP
½ADP½P
i
’ 500 ð5:17Þ
Now we can see that the equilibrium ratio of B to A in the coupled reaction is
K
A=B
¼
½B
eq
½A
eq
¼ K
0
A=B
½ATP
½ADP½P
i
¼ð216Þð500Þ¼1:1 10
5
ð5:18Þ
Thus, the equilibrium ratio of B to A would be increased by a factor of more than 10
7
by coupling the reaction with the conversion of ATP to ADP. Note that for this coupling to
work, there must be some act ual connection between the two reactions. In this example, A
may first react with ATP, transferring a phosphate to A. The phosphorylated A might then
be an unstable compound that converts into phosphorylated B. The final stop might then
be the dissociation of the phosphate from B, leaving the free product. Thus, there is no
step where A is converted directly to B. If all the steps just described each can proceed to
a reas onable extent, this mechanism would circumvent the activation energy barrier. The
net effect of this coupling is that as long as ATP is present, the equilibrium between A and
B will be displaced toward formation of the product. This kind of coupling is exploited in
numerous biochemical reactions.
5.2 ELEMENTARY KINETICS
One last preliminary subject is needed before we look at specific metabolic reaction sys-
tems. If the thermodynamics is favorable, the reaction may proceed. However, thermody-
namics tells us nothing about how fast the reaction will go. As noted above, a solution of
sugar in water in contact with air is thermodynamically unstable. However, the reaction
proceeds immeasurably slowly. How fast the reaction occurs is part of the study of che-
mical kinetics. We start with some basic kinetics, since the same approach is useful to
model other kinds of biological changes, including microbial growth, growth of plant or
animal populations, and transport of pollutants in the environment or within organisms.
Recall reaction (5.4). In a const ant-volume batch system the specific reaction rate, r,
is defined in terms of any of the reaction participants as
r ¼
1
a
d½A
dt
¼
1
b
d½B
dt
¼
1
c
d½C
dt
¼
1
d
d½D
dt
ð5:19Þ
88 ENERGY AN D METABOLISM

The specific reaction rate, r, depend s on the concentrations of the reacting species as well
as factors such as temperature, pressure, and ionic strength. We hold those last three con-
stant and examine the dependence on reactant concentration alone. We will also examine
only the forward reaction, r
f
, the elimination of A and B by conversion into products,
neglecting any formation of A and B from the reverse reaction of C and D. The equation
relating the reaction rate to the concentrations of reactants is called the rate law.
At this point we invoke an assumption that the reaction proceeds exactly as written,
without the format ion of intermediate compounds. Such a reaction is called an elemen-
tary reaction. Although not strictly applicable, elementary kinetics are a good approxi-
mation for many complex biochemical reactions, such as biodegradation of organics by
microorganisms.
The rate of an elementary reaction is proportional to the product of the reactants raised
to their stoichiometric coefficients. This is called the law of mass action. Referring to the
reaction rate for A ðr
A
¼ arÞ instead of the specific reaction rate, the law of mass action
gives the following rate law:
r
A
¼
d½A
dt
¼ k½A
a
½B
b
ð5:20Þ
Thus, if we know the stoichiometry of the reaction and can assume it to be an elementary
reaction, we can write down the rate law. The order of the reaction is defined as the sum
of the exponents of the concentrations in the rate law. In this case, the order is a þb. The
coefficient k is called the rate coefficient.
For example, the air pollutant nitrogen pentoxide (N
2
O
5
) decomposes spontaneously in
air according to the following rate law:
d½N
2
O
5
dt
¼k½N
2
O
5
ð5:21Þ
This is an example of a first-order rate law. The rate coefficient for this reaction at 45
C
has a value of 0.0299 min
1
. First- order rate laws are used very commonly. For example,
the bacterial biodegradation of organic compounds in lakes and rivers is usually approxi-
mated as a first-order decay process, even though it is a complex metabolic process.
Equation (5.21) is easily integrated to yield concentration, c, as a function of time for a
closed system (such as a reaction in a beaker) given an initial concentration c
0
:
c
c
0
¼ expðktÞð5:22Þ
Equation (5.22) shows that for first-order reactions the concentration changes by fixed
ratios in fixed time periods. One such ratio leads to the concept of half-life , t
1=2
. Setting
c=c
0
¼ 0:5, and rearranging gives the relationship between the half-life and the rate
coefficient:
t
1=2
¼
ln 0:5
k
¼
0:693
k
ð5:23Þ
Bear in mind that this relationship, and indeed the very idea of a half-life, is valid only for
first-order reactions. In the nitrogen pentoxide example, the half-life is 23.2 min. Thus a
ELEMENTARY KINETICS 89

vessel containing 100 ppmv initially would have 50 ppmv after 23.2 min and 25 ppmv
after 46.4 min.
Example 5.3 The oxidation of ferrous iron(II) to ferric(III) in water follows first-order
kinetics if oxygen is not limiting. A solution of 20 mg/L ferrous iron at pH 8.0 is aerated
for 60 min. The ferric iron that is formed precipitates and can be removed by filtration.
The remaining iron concentration is 4.0 mg/L. What are the rate coefficient and the half-
life?
Answer Rearrange equation (5.22) gives k ¼lnðc=c
0
Þ=t ¼ 0:027 min
1
. Then use
equation (5.23), we have t
1=2
¼ 0:693=0:027 ¼ 25:8 min.
Sometimes a higher-order reaction will behave as if it were first order. For example, if
the reaction is A þ B ) C, the rate law is d½A=dt ¼k½A½B. Now suppose that B is
present far in excess of A. Then as A is depleted, B will decrease only a small amount
proportionally. For a specific example, suppose that the initial concentrations ½A
0
¼ 2:0
and ½B
0
¼ 100: 0. Now as A becomes depleted by 50%, B will only decrease by 1%.
Thus, B can be assumed to have a constant concentration. The concentration of B can
be combined to form a new rate coefficient, k
0
¼ k½B , and the new rate law becomes
first order: d½ A=dt ¼k
0
½A. This assumption is called a pseudo-first-order reaction.
This effect is one reason why a great many reactions can be treated as being first order
even if the underlying reaction mechanism is very complicated.
5.3 ENZYME KINETICS
Enzymes function not only to catalyze reactions but to enable control of those reactions
via sensitivity to environmental conditions and to the presence of cofactors, which can
increase or decrease rates. A detailed treatment of the kinetics of enzyme reactions will
be beneficial for two reasons. First, it will lead to a clearer understanding of the action of
enzymes. Second, the kinetic equations themselves can be useful in modeling biochemical
processes in environmental systems. This can be true even when the process involved is
not a simple enzyme reaction. For example, the first system we describe will be Michaelis–
Menten kinetics. The resulting model is derived rigorously. However, an empirical
equation of the same form, the Monod equation, is used effectively to model substrate
utilization by systems consisting not only of many different enzymes and of substrates,
but of mixed populations of microbial organisms.
Recall from general chemistry that a catalyst is a substance that affects the rate of a
reaction but is not changed by it, and that it acts by lowering the activation energy, E
a
.
The activation energy refers to the height of a barrier between reactants and products of a
reaction. The molecules in a given system have a distribution of energies. If the activation
energy is high, fewer molecules possess enough to surmount the barrier. Lowering the
height of the barrier means that a higher proportion of the molecules has enough energy
to get over it, and the rate at which molecules pass is increased. The effect of activation
energy and temperature on the rate constant of a first-order reaction is given by the
Arrhenius equation:
ln k ¼ B
E
a
RT
ð5:24Þ
90 ENERGY AN D METABOLISM
where k is the rate constant for the reaction, B is an arbitrary constant, R is the gas law
constant, and T is the temperature in kelvin. As an example, the hydrolysis of sucrose is
catalyzed by hydrogen ions, with an activation energy of 25,600 cal/mol. The yeast
enzyme invertase lowers the activation energy barrier to about 8000 cal/mol. By writing
equation (5.24) first for the hydrogen catalysis, then for the enzyme, assuming that B is the
same for both reactions, and subtracting the two, one gets the following expression for the
ratio of the two rate constants:
log
k
enzyme
k
hydrogen
¼
25;600 8000
2:3 RT
¼ 12:4 ð5:25Þ
at the human body temperature of 310 K. Thus, by reducing the activation energy 69%,
the enzyme enhances the rate of this reaction by a factor of 2:5 10
12
!
5.3.1 Single-Substrat e Kinetics
The simplest enzyme system we can study is one with a single substrate, S, and single
product, P: that is, the reaction
S þ E ! P þ E ð5:26Þ
The reaction rate, r, is the mass of product produced or substrate consumed per unit time
per unit volume. This is expressed as
r ¼
d½S
dt
¼
d½P
dt
ð5:27Þ
Experiments with such systems have been conducted at varying concentrations of total
enzyme ½E
t
and substrate ½S . Two observations are commonly made based on experimen-
tal observations:
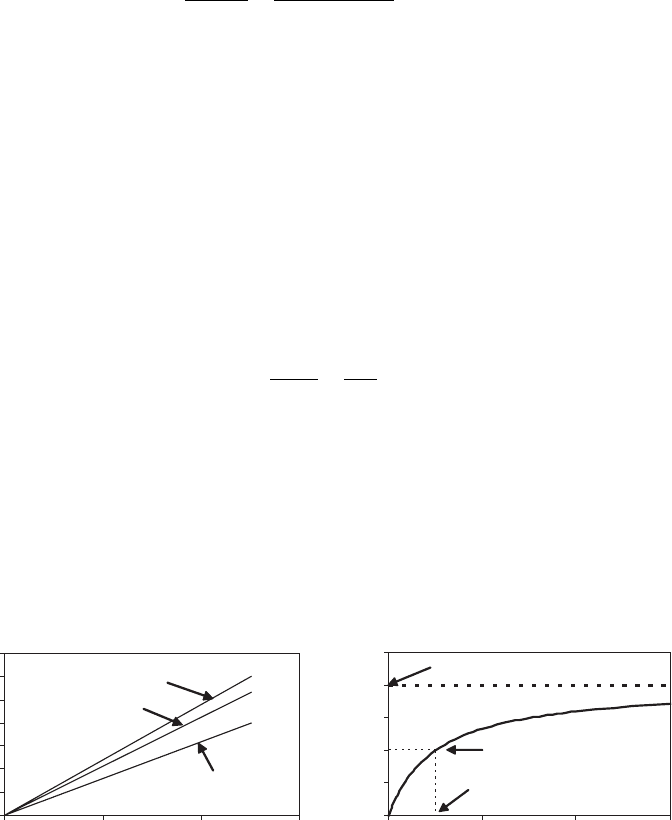
The rate of the reaction at any particular substrate concentration is proportional to
the total concentration of enzyme if the substrate concentration is held constant
(Figure 5.1a).
0.00
0.05
0.10
0.15
0.20
0.25
0.0 1.0 2.0 3.0
S (mmol/L)
r (mmol/L·s)
r
max
= k*E
t
r = r
max
/2
S = K
m
0.00
0.05
0.10
0.15
0.20
0.25
0.30
0.35
0.0 0.2 0.4 0.6
E
t
(mmol/L)
r (mmol/L·s)
S = 0.5 mmol/L
S = 1.0
S = 1.5
(a)(b)
Figure 5.1 Typical behavior of single substrate–enzyme reactions: ðaÞ constant S; ðbÞ constant E
t
.
ENZYME KINETICS 91

If the total enzyme concentration, E
t
, is held constant, the rate of reaction shows
a saturation effect of substrate concentration. At low concentrations of substrate, the
rate is proportional to substrate concentration. However as substrate concentration
becomes large, the rate levels off, until it is independent of concentration
(Figure 5.1b).
An empirical expression was proposed in 1902 by Henri that satisfied these observa-
tions. Michaelis and Menten developed a theoretical derivation, later improved by Briggs
and Haldane, resulting in what is now called the Michaelis–Menten equation. The deriva-
tion starts by assuming the following reaction mechanism:
S þ E ,
k
1
k
1
ES ð5:28Þ
ES !
k
2
P þ E ð5:29Þ
The species ES represents a com bined enzyme–substrate complex in which the sub-
strate is bound to the active site of the enzyme. The first reaction, in which the complex
is formed, is assumed to be reversible and in equilibrium. The second part, decomposition
of the complex into product with recovery of the free enzyme, is assumed to be irrever-
sible. The total enzyme concentration is given by
½E
t
¼½Eþ½ESð5:30Þ
Experimentally, ½E
t
is known since it is the amount of enzyme present initially. The
species E and ES usually cannot be measured directly. Now the rate equations for the
reaction rate and the rate of change of [ES] can be written assuming elementary reaction
kinetics:
d½S
dt
¼k
1
½S½Eþk
1
½ESð5:31Þ
d½ES
dt
¼ k
1
½E½Sðk
1
þ k
2
Þ½ESð5:32Þ
At this point another key assumption is introduced that simplifies the result. Equations
(5.30) to (5.32) do not have an analytical solution but can be solved numerically. Exam-
ination of numerical solutions shows that after a short startup period, the concentration of
the enzyme–substrate complex is fairly constant. Thus, equatio n (5.32) can be set to zero.
This is the quasisteady-state assumption. With this assumption, and using equation
(5.30) to eliminate [E], equation (5.32) can be rearranged into
½Sð½E
t
½ESÞ
½ES
¼
k
1
þ k
2
k
1
¼ K
m
ð5:33Þ
where K
m
is known as the Michaelis constant. Solving equation (5.33) for [ES] gives
½ES¼
½E
t
½S
K
m
þ½S
ð5:34Þ
92 ENERGY AN D METABOLISM

Finally, it is noted the rate of the reaction in equation (5.29) must be equal to the overall
rate of reaction, r (with the units ½M t
1
. Therefore, from ele mentary reaction kinetics
we can say:
r ¼ k
2
½ESð5:35Þ
Using equation (5.35) to eliminate [ES] from equation (5.34), we obtain
r ¼
k½E
t
½S
K
m
þ½S
ð5:36Þ
This is the final form of the Michaelis–Menten equation, which satisfies the observations
made above. Note that k ¼ k
2
. We have dropped the subscript 2 from the rate constant for
simplicity. The two coefficients of equation (5.36) are determined by fitting to experimen-
tal data. Usually, the equation is linearly transformed so that linear regression methods
can be used. However, modern statistical software makes nonlinear regression easy and
eliminates accuracy problems with the linear transformation methods.
The Michaelis–Menton equation approaches its maximum value of k½E
t
as the sub-
strate concentration increases without limit. When the substrate concentration is equal
to K
m
, the reaction rate will be one-half its maximum value at that same enzyme concen-
tration.
Example 5.4 The reaction rate for an enzyme reaction is 0.1 mol/Lmin a substrate con-
centration 0.01 M. Doubling the substrate concentration increases the rate by 50%. What
is K
m
for this reaction? What is the maximum rate for this reaction?
Answer Taking equation (5.36) and writing the first rate r
1
at concentration S
1
, and
the second r
2
at S
2
, we can cancel out k½E
t
by taking the ratio r
1
=r
2
and then solve for K
m
:
r
1
r
2
¼
K
m
þ S
2
K
m
þ S
1
S
1
S
2
K
m
¼
S
1
S
2
ðr
2
r
1
Þ
r
1
S
2
r
2
S
1
¼
ð0:01Þð0:02Þð0:15 0:10Þ
ð0:10Þð0:02Þð0:15Þð0:01Þ
¼ 0:02 M
The maximum rate is equal to k½E
t
, which from equation (5.36) is
k½E
t
¼ r
1
K
m
þ S
1
S
1
¼ð0:10Þ
0:02 þ 0:01
0:01
¼ 0:30 mol=L min
The Michaelis–Menten equation is well known and often used. However, it is instruc-
tive to consider conditions under which the equation is not accurate. One is if the ratio of
½E
0
=½S is large (see Problem 5.2). Also, the pseudosteady-state assumption does not
apply at the beginning of an experiment, when enzymes are just mixed with substrate
and no complex has been formed. Of course, there are many situations in which the
basic mechanism may be more complicated, such as in the case of multiple substrates
or the use of cofactors.
What is the effect of assumi ng that reaction (5.29) is irreversible? If it is assumed
reversible, with an equilibrium constant equal to K
P
, and renaming K
m
to K
S
, and
ENZYME KINETICS 93

again invoking the quasisteady-state assumption, we obtain
r ¼½E
t
ðk
2
=K
S
Þ½Sðk
1
=K
P
Þ½P
1 þ½S=K
S
þ½P=K
P
ð5:37Þ
One important thing to note about this case is that the rate depends on the product con-
centration as well as substrate concentration, and that the rate decreases with increasing
product concentration.
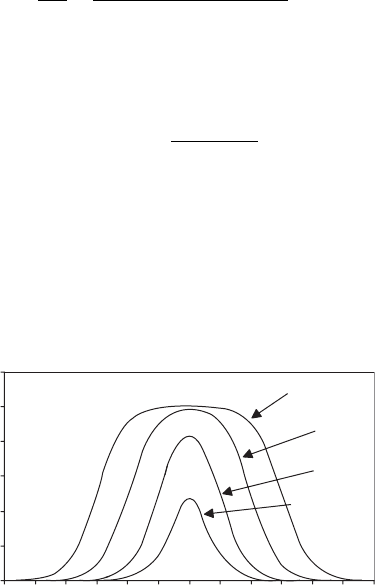
Two other single-substrate concentration effects are worth noting. Substrate activa-
tion describes the situatio n observed in Figure 5.2, in which the effect of substrate con-
centration on reaction rate shows a sigmoidal shape. It can be modeled by adding a
reaction to (5.28) and (5.29) in which the free enzyme is in equilibrium with an inactive
form. Activation is different from enzyme induction (discussed in Section 6.2.2). In induc-
tion the presence of substrate actually stimulates the organism to produce the enzyme; that
is, the enzyme is absent when not needed.
Substrate inhibition is the case when adding substrate beyond an optimum amount
causes a reduction in the reaction rate (Figure 5.2). It can be modeled by assuming that
a second substrate molecule complexes reversibly with the enzyme but that this new com-
plex does not produce product directly. The resulting rate equation is
r ¼
k ½E
t
1 þ K
m
=½Sþ½S=K
I
ð5:38Þ
where K
m
and K
I
are constants. This expression is known as the Haldane equation, and is
equivalent in form to the Andrews equation (Section 11.7.7), often used to model micro-
bial biodegradation of toxic substances. Equation (5.38) has a maximum at the substrate
concentration:
½S
max
¼
ffiffiffiffiffiffiffiffiffiffiffi
K
m
K
I
p
ð5:39Þ
Many industrial organic chem icals are both biodegradable and toxic to microorganisms,
and thus their biodegradation may be modeled by the Haldane equation. Examples of such
compounds include benzene and phenol.
0.00
0.02
0.04
0.06
0.08
0.10
0.12
0.0 1.0 2.0 3.0 4.0
S (mmol/L)
r (mmol/L
·
s)
Activation
Inhibition
Figure 5.2 Substrate activation and inhibition.
94
ENERGY AND METABOLISM

Inhibition can be important in the normal control of metabolism. For example, the
amino acid isoleucine inhibits one of the enzymes involved in its formation, preventing
an oversupply from being produced. This is an example of feedback inhibition.
5.3.2 Multiple Substrates
In the case where two substrates, S
1
and S
2
, bind reversibly to a enzyme to make a single
product, we may assume the following reaction mechanism:
S
1
þ E ,
k
1
k
1
ES
1
S
2
þ E ,
k
2
k
2
ES
2
S
2
þ ES
1
,
k
12
k
12
ES
1
S
2
S
1
þ ES
2
,
k
21
k
21
ES
1
S
2
ES
1
S
2
!
k
P þ E
ð5:40Þ
where the first four reactions are in equilibrium with dissociation constants of K
1
, K
2
, K
12
,
and K
21
, respectively. The resulting equation for the rate is
r ¼ k½E
t
½S
1
K
1
þ½S
1
½S
2
K
2
þ½S
2
ð5:41Þ
where
K
1
¼
K
21
½S
2
þK
1
K
12
½S
2
þ K
12
ð5:42Þ
It is interesting that an empirical equation of the same form as (5.41) is sometimes used to
model dual substrate microbial kinetics, except that K
1
is treated as a constant. For exam-
ple, such a model has been used where the two substrates are chemical oxygen demand
(COD) and oxygen, or COD and nitrate.
Besides the substrate inhibition described above, inhibition can come from compounds
other than the substrate. Some inhibitors will react irreversibly with the enzyme, removing
it from availability permanently. These are poisons. An example is cyanide, which deac-
tivates an enzyme in the respiration process. Others act by competing with substrate in
reversible reactions with the enzyme. Their effect can be categorized in terms of their
effect on the coefficients of the Michaelis–Menten equation and in terms of how they
bind to the enzymes.
Competitive inhibitors bind to the same active site as the substrate. They do not
change the maximum reaction rate, but do change K
m
. Their effect can be overcome
by increasing substrate concentration. For example, TCE is cometabolized with substrates
such as methanol. The presence of TCE reduces the biodegradation rate of the methanol
but not its maximum biodegradation rate. Noncompetitive inhibitors bind at a different
site than the substrate. They act by changing the shape of the enzyme, and therefore its
ENZYME KINETICS 95
activity. The maximum rate is changed but not K
m
. Uncompetitive inhibitors bind to the
enzyme–substrate complex, preventing the formation of products. Both maximum rate
and K
m
are reduced.
5.3.3 Effect of pH
Like all proteins, enzymes have a variety of acidic and basic groups, each with its own
ionization constant. For the enzyme to have optimum activity, it must exist in a particular
ionization state. If the pH changes enough to change the ionization significantly on any
one group, the enzyme’s activity will decrease. This can be modeled by assuming that the
enzyme is in equilibrium with two versions of itself, one containing one additional proton
over the optimum, E
þ
, and another with one proton less than optimum, E
, with equili-
brium constants associated with the conversion to each of those states:
E
þ
,
K
1
E ,
K
2
E
ð5:43Þ
With this mechanism it is possible to derive the following equation for the fraction of the
total enzyme in the optimum state as a function of the hydrogen ion conce ntration:
½E
½E
t
¼
1
1 þ½H
þ
=K
1
þ K
2
=½H
þ
ð5:44Þ
Notice that this has the same form as the Haldane equation for substrate inhibition as in
equation (5.38). In a similar fashion, the optimum pH is found to be
pH
optimum
¼
pK
1
þ pK
2
2
ð5:45Þ
Figure 5.3 shows the effect of pH on enzyme activity according to equation (5.44). All
curves have an optim um pH of 7.0. The outer curve has pK
1
and pK
2
values of 4 and 10,
respectively; inner curves are 5 and 9, 6 and 8, and 6.75 and 7.25, respectively. Note that
the farther apart the pK
a
values, the broader the optimum. Furthermore, if the pK
a
values
are close to each other, the maximum activity falls substantially below 100%. This means
0.0
0.2
0.4
0.6
0.8
1.0
1.2
1 2 3 4 5 6 7 8 9 10 11 12 13
pH
[E] / [E]
t
pK: 4 - 10
5 - 9
6 - 8
6.75 - 7.25
Figure 5.3 Effect of pH on enzyme activity for various ranges of pK
a
.
96
ENERGY AND METABOLISM
that even at the optimum pH, much of the enzyme is in an inactive form because one or
the other of the acid–base groups is in the wrong state of association.
5.3.4 Effect of Temperature
At first it would seem that the Arrhenius equation (5.24) would be all that is needed to
describe the temperature effect. In fact, it does hold, but only in the lower range of tem-
peratures associated with life. At higher temperat ures another reaction occurs: the dena-
turation of the enzyme. One way to handle this is to treat the denaturation reaction as a
simultaneous equilibrium. The dissociation equilibrium constant, K
d
, is related to tem-
perature by
K
d
¼ exp
G
d
RT
¼ exp
H
d
RT
exp
S
d
R
ð5:46Þ
where G
d
, H
d
, and S
d
are Gibbs free energy, enthalpy, and entropy of deactivation,
respectively. For example, the enthalpy and entropy of dissociation for trypsin are 68 kcal/
mol and 213 cal/molK, respectively, and the Gibbs free energy for the reaction is
1.97 kcal/mol. With this and a similar relationship for the rate constant, k, in equation
(5.36) the following expression for maximum rate in the Michaelis–Menten equation
can be derived:
r
max
¼ k½E
t
¼
bT expðE=RTÞ
1 þ expðS
d
=RÞexpðH
d
=RTÞ
ð5:47Þ
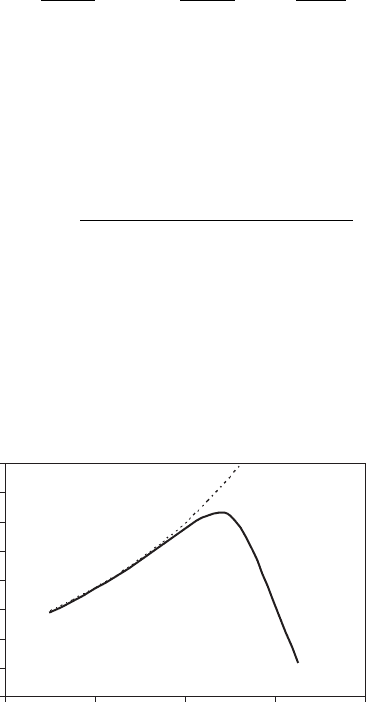
where b is a kinetic rate coefficient. This is curve (a) in Figure 5.4.
Figure 5.4 shows this relationship. Note that the side of this curve below the tempera-
ture optimum is slightly concave upward. This portion can be approximated empirically
as a simple exponential, as shown in Figure 5.4b:
r ¼ r
20
y
T20
ð5:48Þ
0
50
100
150
200
250
300
350
400
020406080
r (mm
3
/min)
T (°C)
(a)
(b)
Figure 5.4 Effect of temperature on hydrogen peroxide decomposition by catalase; ðaÞ is equation
(5.47) with E ¼ 3:5 kcal=mol, H
d
¼ 55:5 kcal=mol, S
d
¼ 168 kcal=mol K, b ¼ 258 mm
3
=min;
(b) is equation (5.48) fitted to equation (5.47) at 20
C and 25
C; r
20
¼ 185:8mm
3
=min, y ¼ 1 :024.
(Based on Bailey and Ollis, 1986.)
ENZYME KINETICS 97
