Vaccari D.A., Strom P.F., Alleman J.E. Environmental Biology for Engineers and Scientists
Подождите немного. Документ загружается.

where T is the temperature in degrees Celsius, r
20
the reaction rate at 2 0
C, and y an
empirical coefficient. This expression is commonly used to describe the effect of tempera-
ture on biological growth rates in biological waste treatment processes. In the example of
Figure 5.4, the denominator of equation (5.47) is very close to 1.0 up to a temperature of
40
C. Thus, the numerator carries most of the effect of temperature and the exponential
form of (5.48) holds.
5.3.5 Other Considerations
Keep in mind that the deactivation due to temperature described above is reversible. Irre-
versible denaturation can be modeled in a number of ways, depending on the mechanism.
In the simplest case, first-order decay may suffice. Irreversible denaturation by heat may
be one of the principal mechanisms of heat sterilization.
It is also important to know that many enzymes function when associa ted with mem-
branes or other biological structures and may not function at all when extracted into solu-
tion. Some, such as pancreatic lipase, function only when absorbed at the interface
between a lipid droplet and an aqueous solution.
5.4 BIOCHEMICAL PATHWAYS
With the preceding discussion of the types of compounds that form living things (Chapter 4)
and of the enzymes that facilitate and control their chemical transformations and of ther-
modynamic relationships governing individual reactions, we can now examine some of
the most important of these transformations. We are usually interested not in individual
reactions but in a sequence of reactions, called a pathway, leading from initial reactants to
final products. For example, the oxidation of the carbon in glucose to carbon dioxide
involves some 21 reactions, usually divided into two pathways: glycolysis, which forms
an intermediate called pyruvate, and the Krebs cycle, which produces the CO
2
.
The detailed reactions of each pathway are not given here. Instead, we examine the
overall reactions for several major pathways, with a few details about what occurs within
the pathway. The rate of an overall reaction pathway is often governed by a single limiting
step in the sequence of reactions. In the case of biodegradation of xenobiotic compounds,
it is often the first step that limits the overall rate. Biochemical pathways can be classified
in several ways. One of the simpl er ways is to divide them into the following: Catabolic
pathways are those that break down organic compounds, usually to provide energy;
anabolic pathways are those that synthesize complex organics, such as to form new
cell material, from simpler precursors. However, many metabolic pathways do not fit
easily into these two categories.
5.4.1 Glycolysis
First we consider ways of extracting energy from organics without external oxidizers such
as oxygen. Essentially, this means producing ATP from ADP (phosphorylation) for
short-term use (on the order of seconds or minutes). Phosphor ylation of ADP by reaction
with organic molecules, and without an electron acceptor such as oxygen, is called sub-
strate-level phosphorylation. For longer-term energy supply, cells store glucose (as
starch in plants or as glycogen in animals) or lipids. Thus, our starting point is glycolysis,
98 ENERGY AN D METABOLISM
which provides a ready source of ATP by partial oxidation of glucose. It incorporates sub-
strate-level phosphorylation. Its products feed into respiration pathways, for more energy
extraction, or to fermentation pathways for waste elimination.
Glycolysis is the conversion of glucose to pyruvic acid, an important metabolic inter-
mediate. It is the first of several pathways for the production of ATP from glucose
(Figure 5.5). Glycolysis consists of nine reactions involving nine intermediate com-
pounds. The six-carbon glucose first enters into several reactions involving ATP, forming
a fructose with two phosphate groups. Since this consumes ATP, it represents an invest-
ment by the cell to get the reaction going, forming an activated intermediate compound.
This splits into two three-carbon sugars, each with one phosphate, which is oxidized by an
NAD to form NADH
2
. Both three-carbon sugars then go through a series of steps that
generate two ATPs each, for a total of four. Thus, the net reaction of glycolysis is
glucose þ 2ADP þ 2P
i
þ 2NAD ) 2 pyruvic acid þ 2ATP þ 2NADH
2
ð5:49Þ
Since each ATP has a standard Gibbs free energy of 7.3 kcal/mol and the complete
oxidation of glucose has 686 kcal/mol, this reaction has an efficiency of only 2.1%. Addi-
tional energy is contained in the pyruvic acid and the NADH
2
, but these are unavailable
without an oxidizing agent. This is why anaerobic bacteria yield less cell mass than aero-
bes do, since they do not harvest as much energy for synthesis. In the aerobic process of
respiration, described below, both the pyruvate and the NADH
2
can be made to yield more
ATP.
Pyruvate is a link to numerous other biochemical pathways, besides feeding into the
respiration pathway. It can be made into alanine, leading to synthesis of amino acids.
Pyruvate is also the starting point for many fermentation products described below.
Some of the other glycolysis intermediates are also used to synthesize other sugars or
amino acids. There also are other less important pathways for glucose catabolism. The
cell shifts to them to control the level of ATP in the cell or to form other compounds.
5.4.2 Fermentation
Besides ATP and pyruvate, glycolysis produces the reducing agent NADH
2
. Unless this
were reoxidized to NAD, the cell’s supply of NAD would be depleted. Also, pyruvate is
retained by the cell. A buildup of pyruvate would decrease the rate of the glycolysis by the
law of mass action. Many cells use the pyruvate and NADH
2
in respiration with oxygen
(described in the next section) to produce more ATP. In the absence of oxygen, cells use a
different pathway, in which the NADH
2
is used to reduce the pyruvate to various products
that can leak out of the cell as waste products. This process, which forms such partially
oxidized by-products to regenerate NAD, is called fermentation. More generally, fermen-
tation is an anaerobic biochemical process in which organic compounds serve as both
electron donors and acceptors. Thus, some organics become reduced (e.g., NAD) and
others are oxidized (e.g., ethanol to acetic acid), and no inorganic electron acceptor
(such as oxygen) is needed.
In animals, the rapid generation of ATP needed for muscle power can only be gener-
ated by glycolysis. Animal cells eliminate p yruvate and regenerate NAD by a single-step
reaction that forms lactic acid. However, lactic acid is a ‘‘blind alley’’ in animals and can-
not be used further. But unlike pyruvate, it can diffuse out of the cell, where the blood
transports it to the liver. There it is converted back to pyruvate to enter other pathways.
BIOCHEMICAL PATHWAYS 99
The accumulation of lactic acid in the muscles causes the pain that results from vigorous
exercise. Rest allows time for the elimination and conversion of the lactic acid.
Bacteria and yeast can produce other products. The most important is ethanol, which is
formed by yeast in two steps, with acetaldehyde as an intermediate. Specific organism and
cultivation conditions yield specific end products. Yeast can produce glycerol in addition
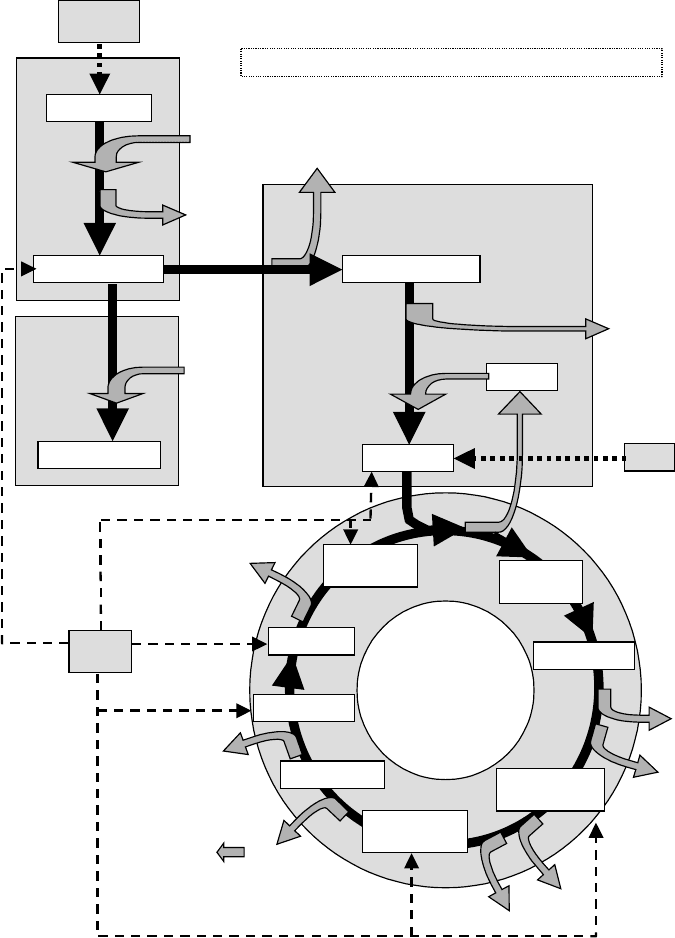
Glucose (C-6)
Pyruvate x 2 (C-3)
2 ATP
4 ATP
2 NADH
2
Glycolysis
Lactic Acid (C-3)
NADH
2
Fermentation
Acetaldehyde (C-2)
CO
2
Each pyruvate
Acetyl-CoA
Oxaloacetic
Acid (C-4)
Citric
Acid (C-6)
CoA-SH
Transition to Krebs Cycle
NADH
2
KREBS CYCLE
Isocitric acid
CO
2
NADH
2
α-Ketoglutaric
acid (C-5)
Succinyl-CoA
(C-4)
GTP
ATP
CO
2
NADH
2
Malic Acid
Fumaric acid
Succinic acid
FADH
2
NADH
2
Each
pyruvate
Glycogen
or starch
Fats
Amino
Acids
All NADH
2
and FADH
2
produced go to the CYTOCHROME SYSTEM
(Nine
steps)
Figure 5.5 Glycolysis, fermentation, and the Krebs cycle. Inputs and outputs from each cycle are
shown, as well as connections with the metabolism of other biochemical compounds.
100
ENERGY AND METABOLISM
to ethanol, although it is derived from intermediates of glycolysis and not from pyruvate.
Clostridium produces acetone, isopropanol, butyrate, and butanol. Proprionic acid bac-
teria produce proprionate. Coliforms produce formic acid, acetic acid, hydrogen, and
CO
2
. Enterobacter produces ethanol, 2,3-butanediol, formic acid, and lactic acid.
Fermentation produces other compounds of commercial importance, some of which
may originate with pyruvate, such as penicillin. However, although they are produced dur-
ing fermentation, they are not end products used by the cell. Thus, thes e are properly
referred to as secondary metabolites, not fermentation products.
5.4.3 Respiration
Respiration is a process in which organic compounds or reduced inorganics (such as
hydrogen or ferrous iron) are oxidized by inorganic electron acceptors for the produc tion
of energy. Eukaryotes can only use oxygen as a final electron acceptor, in what is called
aerobic respiration. Microorganisms can also use nitrate, sulfate, some metals, and even
carbon dioxide, in what is generally called anaerobic respiration. In environmental
applications the term anoxic respiration is generally used for nitrate reduction, and anae-
robic respiration is limited to the other forms.
Strictly speaking, glycolysis is not a part of respiration, although it is the first step lead-
ing to respiration for glucose. Nevertheless, when discussing respiration, many scientists
and engineers are referring to the overall conversion of glucose or other carbohydrates to
carbon dioxide and water:
C
6
H
12
O
6
þ 6O
2
) 6CO
2
þ 6H
2
O ð5:50Þ
Respiration occurs in two phases. The first is the Krebs cycle [also called the citric
acid cycle or tricarboxylic acid (TCA) cycle]. The Krebs cycle completes the job of oxi-
dizing the carbon that originated with glucose, forming CO
2
and ATP. However, much of
the energy is left in the reducing power of NADH
2
or FADH
2
. These are converted to ATP
in the second phase, called the electron transport system or cytochrome system. The
cytochrome system is also responsible for reducing oxygen to water, the other product
of the overall reaction for the oxidation of glucose. Both the Krebs cycle and the cyto-
chrome system are cyclic because intermediates involved in each reaction are regenerated
by other reactions.
Both of these process are mediated by enzymes bound to membranes and require the
presence of the membranes in order to function. In eukaryotes this occurs within the mito-
chondria. Pyruvate must diffuse into the mitochondria to enter the process. Prokaryotes do
not have internal membrane structures. Their respiratory enzymes are bound to their cell
membrane. The discussion here focuses on eukaryotes, but the process is similar in pro-
karyotes. Mitochondria consist of an inner and an outer membrane, forming an inner and
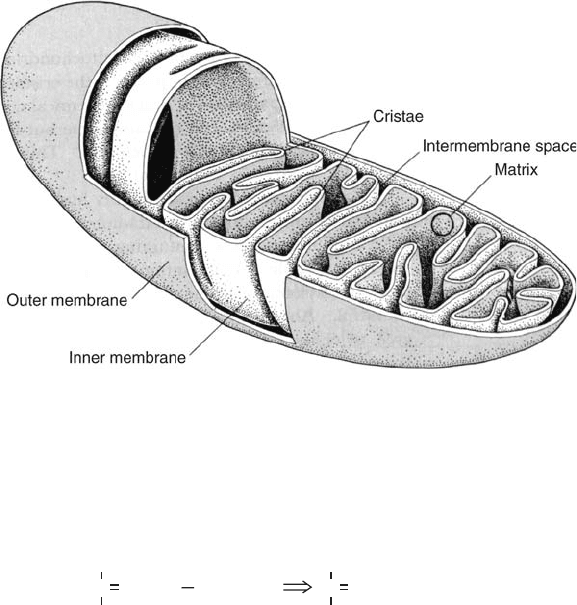
an outer compartment. The inner membrane is folded extensively (Figure 5.6). The Krebs
cycle occurs within the inner compartment. The cytochrome system is integral to the inner
membrane and involves reactants in both compartments.
Glycolysis forms two three-carbon pyruvate molecules for each glucose. When oxygen
or another suitable electron acceptor is available, the pyruvate enters into respiration
instead of fermentation. Respiration begins when the pyruvate diffuses into the inner com-
partment of the mitochondria. There, each pyruvate becomes covalently bonded through a
sulfhydryl bond with a coenzyme, called coenzyme A CoA, which itself is a derivative of
BIOCHEMICAL PATHWAYS 101
ADP. The process is an oxidation and requires an NAD. It resu lts in a compound called
acetyl-CoA:
CH
3
C
COOH
O + CoA
SH + NAD
CH
3
C
S-CoA
O + CO
2
+ NADH
2
pyruvate
acetyl-CoA
ð5:51Þ
The removal of one CO
2
for each pyruvate accounts for two of the six carbons originating
in the glucose. The NADH
2
carries muc h of the energy of oxidation and will be used to
generate ATP in the electron transport system.
Acetyl-CoA is another important metabo lic intermediate. It is a point of connection
between a number of pathways, including the breakdown of lipids and amino acids for
energy. But for purposes of the present discussion, a pair of acetyl-CoAs serve the purpose
of carrying four carbons from the original glucose molecule into the Krebs cycle.
Krebs Cycle The Krebs cycle oxidizes the two carbons remaining from each pyruvate to
CO
2
. One ATP is formed as a result, and the remaining four pairs of hydrogens (with their
energetic electrons) reduce the cofactors NAD or FAD. The overall reaction is
acetyl-CoA þ 3NAD þ FAD þ ADP
) 2CO
2
þ CoA þ 3NADH
2
þ FADH
2
þ ATP
ð5:52Þ
Since two such reactions occur for each glucose, all of the original carbon atoms are
accounted for. Only two additional ATPs are formed, for a total of four when combined
with glycolysis. Thus, the efficiency so far, in terms of standard Gibbs free energy, is four
times 7.3 kcal/mol for the ATPs, divided by 686 kcal/mol glucose, or 4.3%. However,
Figure 5.6 Mitochondrion structure, showing membrane structure and division into inner and
outer compartments. (From Smith et al., 1983.)
102
ENERGY AND METABOLISM
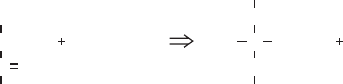
there are a total of 10 pairs of energetic hydrogen electrons that have been captured by
cofactors.
Some details of the cyclical reactions of the Krebs cycle may help clarify the picture.
The cycle starts when the C
4
oxaloacetate combines with two carbons of the acetyl-CoA
that originated with pyruvate. The products of this first reaction regenerate the CoA and
form the C
6
tricarboxylic acid citric acid:
COOH
CH
2
C
COOH
O
HO C COOH
CH
2
CH
2
COOH
COOH
acetyl-CoA
oxaloacetate citric acid
CoA
ð5:53Þ
A series of eight reactions complete the cycle, in which the two carbons added by the
first step are effectively removed, along with the four pairs of hydrogen. (Some of the
hydrogen originates in H
2
O, which is incorporated at several steps.) Some of the inter-
mediates may be used to synthesize other compounds needed by the cell. The reducing
power of the NADH
2
may be used in synthesis reactions of other pathways. Both of these
reduce the energy yield of the Krebs cycle; however, this is not necessarily inefficient
since useful products are made. However, the cell must maintain a supply of oxaloacetate
so that the cycle can continue. If intermediates are drawn off, oxaloacetate can be formed
from pyruvate and CO
2
.
The Krebs cycle is normally controlled by feedback inhibition by the products of the
cycle. High levels of the ratios NADH
2
/NAD, ATP/ADP, or acetyl-CoA/CoA indicate that
the cell has ample energy, and slow the cycle.
The Cytochrome System The six carbons from the glucose have been disposed of, but
the substantial reducing power in the form of NADH
2
and FADH
2
has yet to be converted
to energy in a useful form: namely, as ATP. In eukaryotes this conversion occurs with a
series of enzymes and other compounds bound to the inner mitochondrial membrane,
which comprise the cytochrome system (Figure 5.7). NADH
2
reduces the first of these
compounds, flavin mononucleotide (FMN), which spans the membrane from one side to
the other. FMN takes the two hydrogens, passes their electrons to the next membrane
compound (at a lower energy), and releases the protons to the outside of the membrane.
It may be useful to think of the electrons as dropping to a lower voltage, to use an elec-
tronic analogy, each time they pass from one electron carrier to the next. Thus, the net
effect is to use some of the energy from the electrons to move a pair of protons to the
outer compartment of the mitochondrion.
The compound that receives the electrons does a similar thing; it passes them on at a
yet lower energy, which may or may not result in moving protons to the outer compart-
ment. In all, six protons are transported. Consider the result: Energy is used to transport
protons, creating a pH gradient. This pumping of protons across a membrane into an area
of increasing proton concentration stores energy, just as to pumping air into a tank pro-
duces a pressure difference that also stores energy. But there isn’t only a pH gradient.
Since the protons are transported without their electrons or complementary anions, an
electric charge gradient, an actual voltage, is also accumulated. The combined chemical
and electrical potential across the inner membrane of the mitochondrion is called the
chemiosmotic potential.
BIOCHEMICAL PATHWAYS 103
The energy of the chemiosmotic potential is used when the protons flow back across
the membrane to the inne r compartment through a complex of transmembrane proteins
called ATP synthase, producing ATP from ADP. Think of air that has been pumped into
a pressure vessel then flowing back out through a turbine to produce electricity.
Altogether, 3 mol of ATP is produced for each mole of NADH
2
.TheFADH
2
acts similarly,
except that its electrons have a lower energy to start with, so it enters the chain farther
along. It provides only enough energy to produce 2 mol of ATP per mole of FADH
2
.
Finally, what becomes of the energy-depleted electrons in the transport chain? The
final components of the transport chain are a series of membrane-bound compounds called
cytochromes. The final cytochrome performs one last reduction, that of oxygen. Each
1
2
mol of O
2
receives 2 mol of electrons and 2 mol of H
þ
, forming H
2
O and completing
the process of respiration. Because of the use of an electron acceptor, ATP production in
this system is called oxidative phosphorylation.
Assuming that none of the intermediate compounds or NADH
2
have been shunted off
for other cellular purpos es, the final tally for ATP is a maximum of 36 mol per mole of
glucose oxidized. Now the efficiency based on standard Gibbs free energy is 38%, a big
improvement over glycolysis or the Krebs cycle. Since organisms will use every oppor-
tunity for growth, a more typical number of ATPs actually produced by glycolysis and
respiration is 20 to 25.
In prokaryotes, the electron transport chain is located in the cell membrane. Ba cteria
perform the function of the electron transport system without mitochondria by pumping
protons outside the cell, depleting it within to create the chemiosmotic potential .
Alternative Electron Acceptors Some bacteria can switch to other electron acceptors,
such as nitrate, when oxygen is absent. The nitrate becomes reduced ultimately to
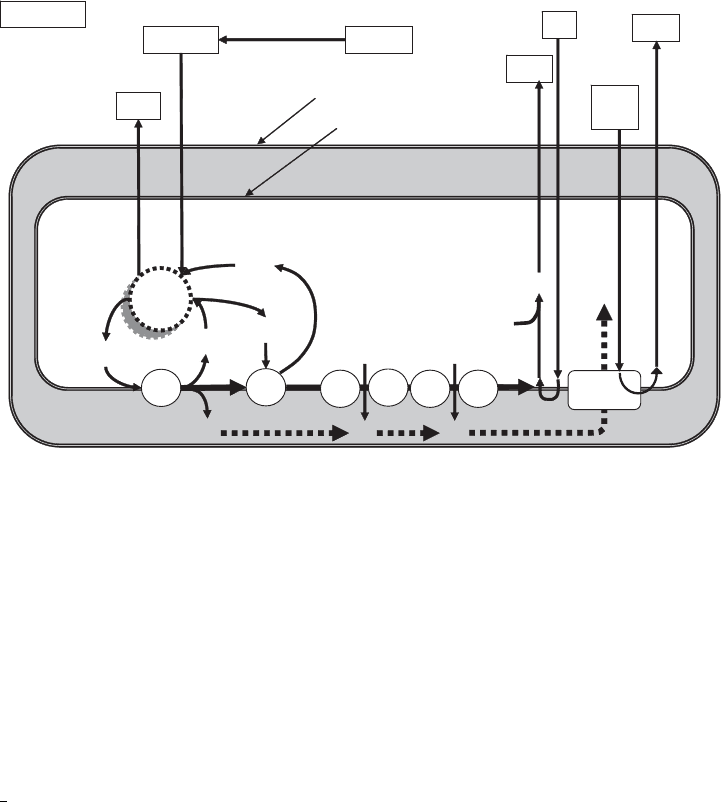
Outer mitochondrial membrane
Inner mitochondrial membrane
GlucosePyruvate
Glycolysis
Krebs
Cycle
Krebs
Cycle
CO
2
FMN
CoQ
NADH
2
NAD
2H
+
2e
-
FADH
2
FAD
Cyt
b
Cyt
c
Cyt
a
Cyt
a
3
2H
+
2H
+
2H
+
2H
+
O
2
2H
+
H
2
O
ATP
synthase
H
2
O
ADP
+P
i
ATP
2e
-
2e
-
H
+
Cytoplasm
Outer compartment
Inner compartment
Figure 5.7 Cytochrome electron transport system, with a relationship to glycolysis and the Krebs
cycle.
104
ENERGY AND METABOLISM
nitrogen by the process of denitrification. It is thought that this occurs in a series of steps
as follows:
NO
3
) NO
2
) NO ) N
2
O ) N
2
However, the nitrate requires a higher-energy electron for its reduction. It gets it at an
earlier point in the electron transport system, so that only 2 mol of ATP is formed per
mole of NADH
2
. This is why organisms that can will always use oxygen when it is pre-
sent and switch to nitrate only in the absence of oxygen.
Sulfate and carbon dioxide can also function as electron acceptors for certain micro-
organisms. However, the organisms that do so cannot also use oxygen. When sulfate is
reduced, the product is hydrogen sulfide, a poisonous gas. Carbon dioxide is reduced to
methane by molecular hydrogen by a special group of organisms wi thin the archaea. Oxi-
dized metal ions such as ferric (III) iron can also be an electron acceptor. This occurs, for
instance, in the sediments of wetlands, where oxygen is limiting. The iron becomes
reduced to ferrous (II) iron. If an environment contains oxygen, nitrate, sulfate, and car-
bon dioxide, the oxygen will tend to be used up first, producing H
2
O. Then the nitrate,
then sulfate, and then CO
2
will be used. In homogeneous environments, one electron
acceptor will not be utilized until the energetically more favorable one is depleted. How-
ever, in some situations microenvironments may form, allowing several of these reactions
to proceed simultaneously. For example, in biologi cal slime layers in aquatic systems,
microorganisms near the slime layer surface may have access to oxygen while organisms
below the surface may be depleted of oxygen and will use nitrate.
5.4.4 Oxidation of Fats and Amino Acids
Obviously, glucose is not the only fuel used by living things. Our foods contain other
sugars, such as lactose in milk and fructose in the disaccharide sucrose (table sugar).
The success of dieters hinges on the body’s ability to use fat as fuel; of course, this is
why the body stores fat in the first place. Under starvation conditions, the body obtains
its energy for basic cell function by cannibalizing itself, by oxidizing its proteins.
The sugars are converted fairly easily into either glucose or another intermediate in the
glycolysis pathway. In the case of sucrose and glycogen, these polys accharides are split
into simple sugars by phosphorolysis (splitting by phosphate) instead of hydrolysis. This
results in glucose-6-phosphate, the intermediate in glycolysis that just follows the point
where an ATP is reacted with glucose to get things going. Thus, the extra ATP is not
needed, and glycolysis yields one more ATP than for glucose itself.
The human body maintains only enough stored glycogen to last about a day. Then it
must switch to fats as a fuel. Most fats are stored in the body as triglycerides. Because
they are not soluble in water, they must be transported in the blood as lipoproteins. After a
fatty meal the concentration in the blood may be sufficient to give it a milky opalescence.
Lipase enzymes in the blood or in fatty tissues hydrolyze the lipoproteins to glycerol and
to fatty acids bound to blood proteins. The glycerol enters glycolysis after a few steps
involving ATP. The fatty acids enter the cell and react with CoA similar to the way pyr-
uvate does at the start of the Krebs cycle. The fatty acid-CoA compounds are then trans-
ported into the inner compartment of the mitochondria, where a series of reactions split
off the last two carbons of the fatty acid, with the CoA, forming acetyl-CoA and a shorter
fatty acid-CoA. The process, called beta oxidation, is repeated until the fatty aci d has
BIOCHEMICAL PATHWAYS 105
been consumed. The acetyl-CoAs produced then enter the Krebs cycle, where they are
further oxidized. In addition, for every acetyl-CoA formed, one NADH
2
and one
FADH
2
are formed, feeding the electron transport system production of ATP. Th is
accounts for the high-energy yield of fats in comparison to carbohydrates.
Proteins are first hydrolyzed into their component amino acids, followed by deamina-
tion, removal of the amino group. Finally, each of the 20 amino acids is converted to
either pyruvate, acetyl-CoA, or one of the other intermediates in the Krebs cycle, for
further oxidation. Free amino acids are not stored in the body. Excess proteins in the
diet thus must be eliminated by the mechanism just described. Deamination releases
ammonia to the blood, whi ch can be toxic and must be rapidl y removed. This can be
accomplished by incorporation into new amino acids or by excretion either directly as
ammonia (fish), uric acid (bi rds and reptiles), or urea (mam mals).
5.4.5 Photosynthesi s
Virtually all the organic carbon in our environment, from the carbon in a person’s finger-
nails to the carbon in a plastic pen, was formed by plants from CO
2
in the air. The energy
for this conversion comes entirely from sunlight. Among the few known exceptions in
nature are ecosystems found on the ocean floor and hot springs that obtain their energy
from oxidation of reduced inorganic compounds issuing from deep below the ocean floor
in hot-water vents. Most human-made sources of energy, such as fossil fuels and weather-
driven electric plants (wind and hydroelectric), ultimately come from the sun. Only
nuclear, geothermal, and tidal electrical facilities, plus a portion of wind energy driven
by Earth’s rotation, do not derive their energy from the sun.
Use of the sun’s energy to synthesize carbohydrates is called photosynthesis. Only cer-
tain bacteria, algal protists, and green plants are capable of photosynthesis. Organisms
that can synthesize their own carbohydrates from inorganic precursors are called auto-
trophs. Those that use sunlight to provide the energy for this are called photoautotrophs.
Some bacteria can use inorganic energy sources, such as H
2
,H
2
S, NH
3
, or reduced metal-
lic salts such as manganese or ferrous iron, to form carbohydrates from CO
2
. These are
called chemoautotrophs or lithoautotrophs. All other organisms, including all animals
and fungi, and most bacteria, depend ultimately on the autotrophs for organic carbon and
energy. Organisms such as animals, fungi, and many bacteria that must obtain their
organic carbon ultimately from autotrophs are called heterotrophs.
Photosynthesis is somewhat more complicated to describe than respiration. However,
once we have understood respiration, there are enough similarities to respiration in reverse
to describe it in those terms. Recall that in respiration, there is a separation between the
oxidation of organic carbon to CO
2
(glycolysis and the Krebs cycle), which produces
reducing power as NADH
2
, and the electron transport system, which consumes the redu-
cing power and reduces oxygen to water.
In photosynthesis, the CO
2
gets reduced, produc ing glucose. The reducing power
comes from photons of light. The overall net reaction of photosynthesis is
6CO
2
þ 6H
2
O ) C
6
H
12
O
6
þ 6O
2
ð5:54Þ
The two major parts of photosynthesis are the light reactions, which are analogous to
electron transport in respiration, and the dark reactions, which can be compared to the
reverse of the Krebs cycle and glyc olysis. Several basic experimental facts support the
106 ENERGY AND METABOLISM
division of photosynthesis into light and dark reactions: (1) Plants give off oxygen only in
the light; and (2) if a suspension of algae is illuminated for some time in the absence of
CO
2
, then placed in the dark with CO
2
, the CO
2
is incorporated into carbohydrate for a
brief time. Additional work with tracer elements further supports the idea.
In the light reactions, energy from light is used to oxidize H
2
OtoO
2
and produce ATP
and/or NADPH
2
. As in respiration, electron transport is involved and even uses cyto-
chromes. In fact, one type of organism, the purple nonsulfur bacteria, use the same elec-
tron transport chain for both respiration and photo synthesis. In algae and plants there are
actually two electron transport systems just for photosynthesis, which act in concert. The
light reactions require a membrane structure, the chloroplast (except in cyanobacter),
which is similar to the mitochondrion. Furthermore, ATPs are formed as a result of the
generation of a chemiosmotic potential by the electron transport system. In the dark reac-
tions, the ATP and NADPH
2
are used in a cyclic reaction to form sugars from CO
2
.Some
of the reactions involved are the reverse of parts of glycolysis.
Chloroplasts are the cel lular organelles in plants and alga e where all of the light reac-
tions and some of the dark reactions are located. Bacteria perform the function of chlor-
oplasts using their cytoplasmic membrane, as they do for the respiratory electron transport
system. Like mitochondria, they contain an internal membrane structure that divides the
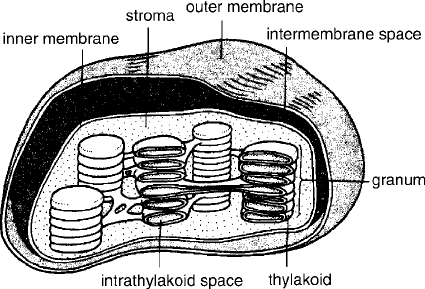
interior into stacks of flattened hollow disks, thylakoids (Figure 5.8). The inner compart-
ment of the thylakoid is called the lumen, the outer compartment is called the stroma.
The membranes of the thylakoids are studded with three groups of proteins and other
compounds (Figure 5.9). Two of these groups are photosystem I and photosystem II,
which perform the principal task of capturing the light energy and transporting the elec-
trons. Each photosystem consists of pigments, proteins, and electron transport com-
pounds, such as cytochromes. The third group is actually a single complex, called the
CF1 particle. Like the ATP synthase particle in the mitochondrion, The CF1 particle
uses the energy stored in the chemiosmotic potential created by the electron transport sys-
tems (in this case of the photosystems) to generate ATP.
The photosystems consist of an association of membrane-bound particles, one of which
is called the antenna. The antenna is a complex of numerous chlorophyll molecules, most
bound to proteins. Chlorophyll is the green pigment of plants, which absorbs much of the
light energy for photosynthesis. The antenna then transfers the energy to a ‘‘special pair’’
Figure 5.8 Chloroplast structure. (From Fried, 1990. # The McGraw-Hill Companies, Inc. Used
with permission.)
BIOCHEMICAL PATHWAYS 107
