Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


1 Electrophoretic Techniques20
The ionic strength should be as low as possible to keep Joule heat
development to a minimum. And, the higher the ionic strength of
the buffer, the lower will be the mobilities of the sample ions.
In isoelectric focusing a different buffer concept is applied: either
amphoteric buffering components migrate to an endpoint in the sys-
tem and become decharged, or the buffering groups are fixed to the
static separation medium.
Another special case is free flow electrophoresis (FFE).
1.1.1
Free Flow Electrophoretic Methods
In the free-flow approach, originally developed by Hannig (1982), a
continuous stream of buffer flows in a 0.5–1.0 mm wide layer inside
a cooled glass cuvette. The buffer is supplied to the cuvette over the
entire width. At one end the sample is injected at a defined spot, and
at the other end the fractions are collected in an array of tubes. The
electrical field is applied perpendicular to the buffer flow.
The varying electrophoretic mobilities perpendicular to the flow
lead to differently strong but constant deviations of the components,
so that they reach the end of the separation chamber at different
though stable positions (see Figure 1.1).
Fig. 1.1: Schematic drawing of
continuous free flow electrophor-
esis, according to Wagner et al.
(1989).
Isoelectric focusing will be
described later in detail.
In contrast to all other methods
FFE is a continuous system.
Hannig K. Electrophoresis. 3
(1982) 235–243.
Wagner H, Kuhn R, Hofstetter
S. In: Wagner H, Blasius E. Ed.
Praxis der elektrophoretischen
Trennmethoden. Springer-
Verlag, Heidelberg (1989)
223–261.

1.1 The Principle of Electrophoresis and Some Methodological Background 21
Different separation methods can be applied: zone electrophoresis in
homogeneous and discontinuous buffer systems, and isoelectric
focusing (see below).
Discontinuous field electrophoresis is a specialty of free flow electro-
phoresis with a zone concentration effect: The sample solution is sup-
plied in a wide zone through the central openings, the buffer solu-
tions on the right and on the left hand side have a twenty times
higher conductivity than the sample solution. The sample ions are
more or less strongly deflected towards the anode or the cathode,
depending on their charges. When they reach the borderline between
sample and buffer stream, their electrophoretic mobility is consider-
ably reduced, resulting in a concentration of the fraction at the bor-
derline.
Free flow electrophoresis is the method of choice for separating
large particles like sub cellular components, as shown in the paper by
Zischka et al. (2003).
1.1.2
Gels for Electrophoretic Techniques
Most electrophoretic separations in proteomics are performed in gel
matrices. There are basically two types of gels: granulated and com-
pact gels. Granulated gels, like dextranes, are almost exclusively used
for chromatography, compact gels for electrophoretic techniques.
There is just one exception: dextrane gels are well suited for prepara-
tive applications and pre-fractionation according to charge, as shown
below. A detailed description for working with dextrane gels for pre-
parative IEF can be found elsewhere (Westermeier, 2004).
The authors are not aware of any proteomics applications in agar-
ose gels; therefore we describe only polyacrylamide gels.
1.1.3
Electroendosmosis Effects
This phenomenon occurs, when fixed charges belonging to the gel
matrix or to glass surfaces are present in an electric field. Those are
for instance carboxylic groups in gels and silicium oxide groups on
the glass surface. These groups become ionized in basic and neutral
buffers: in the electric field they will be attracted by the anode. As
they are fixed in the matrix, they cannot migrate. In order to maintain
the physico-chemical balance, this will be compensated by the coun-
terflow of H
3
O
+
ions towards the cathode. This effect is called electro-
endosmosis. In practice, this phenomenon is observed as a water
flow towards the cathode, which carries proteins along with it and
causes blurred zones and streaking. As described further below, it
All these separations are
performed in a continuous flow.
This one of the rare methods
where a high buffer concentra-
tion is employed for trapping
sample components in a
certain zone.
Zischka H, Weber G, Weber
PJA, Posch A, Braun RJ,
Bhringer D, Schneider U,
Nissum M, Meitinger T,
Ueffing M, Eckerskorn C.
Proteomics 3 (2003) 906–916.
Westermeier R: In Cutler P. Ed.
Protein Purification Protocols.
Second edition. Methods in
Molecular Biology, Volume
244, Humana Press, Totowa,
NJ (2004) 225–232.

1 Electrophoretic Techniques22
can also cause difficulties during the transfer of proteins between
IPG strips and the second-dimension gel.
1.2
Polyacrylamide Gel Electrophoresis
1.2.1
The Polyacrylamide Gel
Polyacrylamide gels are polymerized from acrylamide monomers and
a cross-linking reagent – usually N,N'-methylenebisacrylamide. The
reaction is started with ammonium persulfate as catalyst; TEMED
provides the tertiary amino groups to release the radicals. The pore
size can be exactly and reproducibly controlled by the total acrylamide
concentration T and the degree of cross-linking C:
T ¼
ða þ bÞ · 100
V
½%; C ¼
b · 100
a þ b
½%:
a is the mass of acrylamide (g),
b the mass of methylenebisacrylamide (g),
V the volume (mL).
When C remains constant and T increases, the pore size decreases.
When T remains constant and C increases, the pore size follows a
parabolic function: at high and low values of C the pores are large.
For T = 10% the minimum lies at C = 4%; with higher C values the
gels become are brittle and relatively hydrophobic. For T = 16% the
minimum lies at C = 6%; these gels are used for separation of small
peptides
For protein and peptide separations T values between 4% and 16%
T are used. It has been observed that higher T values can cause pro-
tein degradation. In practice the applied C values are 2.5–3.0% for
zone electrophoresis and 3% for isoelectric focusing.
Polyacrylamide gels must be polymerized in closed cassettes to
exclude oxygen, which would interrupt the polymer chain formation.
Often the monomer solutions are degassed with the help of a vacuum
pump for optimal polymerization effectiveness. The polymerization
efficiency is additionally influenced by the monomer concentration,
the quality of the reagents, temperature, and pH value. The polymer-
ization is exothermic. Particularly, when a highly concentrated gel
solution polymerizes, a substantial amount of heat is generated. The
higher the temperature, the faster and more efficient is the polymeri-
zation.
When the monomer concentra-
tion is very low and the pH
value very high, the gel will not
polymerize. For instance, it
would be very difficult to poly-
merize a 4%T gel with a pH of
8.8.

1.2 Polyacrylamide Gel Electrophoresis 23
The standard catalyst system with TEMED and ammonium persul-
fate works only for gels containing neutral and basic buffers. Acidic
gels are polymerized with alternative reagents, like ascorbic acid and
ferrous sulfate together with hydrogen peroxide. Preparation of acidic
gels is not trivial, because polymerization occurs very quickly and is
difficult to control.
1.2.1.1 Silent Polymerization
It should be noted that after the crude gel has formed, there is a silent
polymerization following, which takes several hours and completes
the formation of the final matrix. Therefore the gels should not be
used immediately after preparation, and they should not be placed
into a refrigerator or a cold room. The gels need to be left at room
temperature for a couple of hours or overnight. After this they can be
used or stored in the cold. Gels containing an alkaline buffer have a
limited shelf life, because at high pH values the matrix begins to
hydrolyze after a few weeks.
& Practical tip: In order to get reproducible gels,
the concentrations of the catalysts have to be
balanced in favor of a higher TEMED and a
lower ammonium persulfate concentration. At
basic pH, ammonium persulfate can react with
the Tris; this effect is minimized by adding more
TEMED, and by reducing the ammonium
persulfate content.
1.2.1.2 Vertical and Horizontal Gels
In a classic setup polyacrylamide gels are run in vertical chambers
while they are still in the casting cassettes. The samples are applied
into wells formed with a comb during polymerization (see Figure
1.2). As an alternative, the gels can be run with an open surface on a
horizontal flatbed system. Here the sample wells are positioned
within the stacking gel area (see Figure 1.4).
Laboratory-made horizontal gels are usually run with liquid buf-
fers, connected to the gel with paper wicks. The electrodes are then
placed into the buffer tanks. The Laemmli buffers are used. Ready-
made horizontal gels contain a different buffer (see below). Horizon-
tal gels can also be run without the buffer tanks: either thick filter
cardboard, soaked with a concentrated buffer, or ready-made poly-
acrylamide buffer strips, which contain the necessary buffer ions for
one run, are placed on the edges of the gel (see also page 108). In this
case electrode wires are placed on top of these electrode strips. The
If a gel is not perfectly polymer-
ized it will become particularly
obvious during mass spectro-
metry analysis of tryptic digests
of cut-out protein bands or
spots. Incompletely polymer-
ized gels cause high back-
ground in the mass
spectrogram.
A comprehensive guide how to
prepare and run horizontal
polyacrylamide gels can be
found in “Electrophoresis in
Practice” (WILEY VCH).
Kleine B, Lçffler G, Kaufmann
H, Scheipers P, Schickle HP,
Westermeier R, Bessler WG.
Electrophoresis 13 (1992) 73–
75.
1 Electrophoretic Techniques24
buffer strip concept reduces chemical and radioactive liquid waste
considerably (Kleine et al. 1992).
1.2.1.3 Discontinuous Gels
In order to prevent aggregations of proteins or other sample mole-
cules during entering the gel matrix, usually a stacking gel is poly-
merized on top of the resolving gel. The stacking gel contains a differ-
ent buffer composition and has larger pore sizes than the resolving
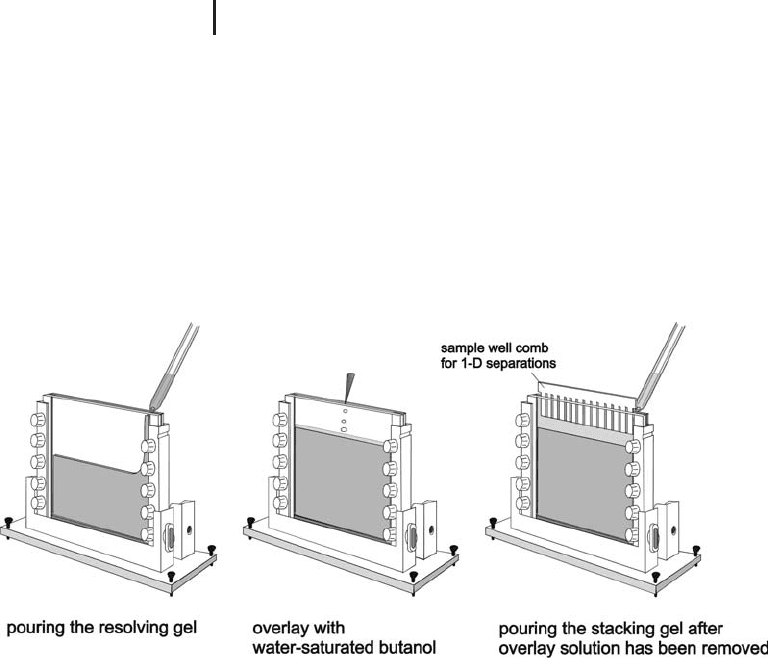
gel. Figure 1.2 shows how such discontinuous gels are prepared. The
stacking gel is poured about one hour before electrophoresis.
Fig. 1.2: Preparation of a discontinuous polyacrylamide gel. The resolving
gel is polymerized for at least 5 hours or overnight at room temperature.
The stacking gel is poured about one hour before electrophoresis.
Stacking gels are used for one-dimensional separation, when the
applied sample molecules are in liquid phase, like in 1-D SDS electro-
phoresis, cationic detergent acidic electrophoresis, and Blue native
electrophoresis.
1.2.1.4 Gradient Gels
Porosity gradients are prepared by continuously changing the acryla-
mide concentration in the polymerization solution while pouring the
gel, so that the concentration in the casting cassette decreases from
bottom to top. The density of the highly concentrated solution is sup-
plemented with glycerol so that the layers in the cassette will not mix.
In principle a concentration gradient is poured. For casting linear gra-
dients the law of the communicating vessels is followed: when the
level in one of the tubes decreases, liquid will flow through the con-
The mode of functioning of the
stacking gel is in detail
described in “Electrophoresis in
Practice” fourth edition 2004,
page 34ff.
For 2-D electrophoresis in
vertical systems the stacking gel
is not necessary.
The easiest is to use gravitation
to create the liquid flow; the
velocity can be controlled by
placing the gradient maker on
a certain level and opening the
clamp only partly. In some
laboratories the support of a
peristaltic pump is preferred.
1.2 Polyacrylamide Gel Electrophoresis 25
necting channel until the level is equal. The light solution will be
immediately mixed with the heavier solution in the mixing chamber.
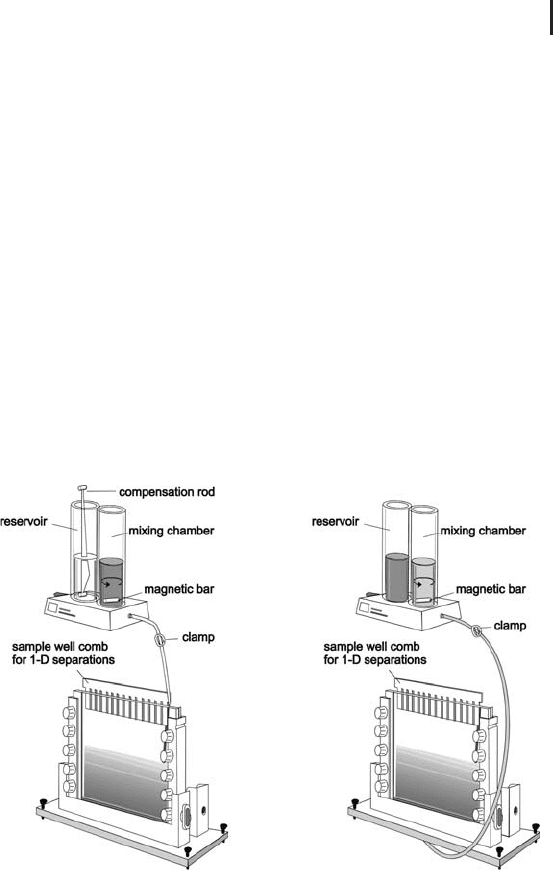
Figure 1.3 shows casting of a single gel in two different ways: on the
left hand side the solution is poured from the top of the cassette. In
this case the mixing chamber contains the dense solution, the reser-
voir the light solution. A compensation rod is placed into the reser-
voir for volume and density compensation. Alternatively the mono-
mer solution can be introduced through the bottom, then the solu-
tions are swapped, the compensation bar is not inserted (see right
hand side of Figure 1.3). This option is also preferred for casting mul-
tiple gels.
Exponential gradient gels are prepared by placing a stopper into
the mixing chamber, thus keeping the volume of the mixing chamber
fixed. Exponential gradients are not often used.
In the field of proteomics gradient gels are used as immobilized
pH gradients for isoelectric focusing, or as porosity gradients in Blue
Native Electrophoresis, and sometimes for SDS gel electrophoresis.
Fig. 1.3: Preparing a linear acrylamide gradient.
Left side: pouring from the top. In the beginning the mixing
chamber contains only dense solution, when the liquid level
decreases; light solution from the reservoir flows in and is mixed
with the liquid in the mixing chamber.
Right side: pouring from the bottom. The solutions are swapped;
the gel solution is introduced through a hole in the bottom of
the casting stand. The magnetic bar is rotated with a magnetic
stirrer (not shown).
This procedure is described in
“Electrophoresis in Practice”
fourth edition 2004, page 37.

1 Electrophoretic Techniques26
1.2.1.5 Gel Sizes
Proteomics requires large gels, because highly complex protein mix-
tures have to be separated as efficiently as possible. Large gels also
offer more space for highly abundant proteins; they have a higher
dynamic range. Some expert laboratories work with gel sizes of
4030 cm (Klose and Kobalz, 1995). But in general, the standard size
for a large 2-D gel is 2520 cm, because there are upper limits for
practically handling the gels. Also, the prices for scanners increase
exponentially, when a size of letter format or A4 is exceeded. For verti-
cal systems mostly the gels are 1 mm thick. Some laboratories prefer
1.5 mm thick gels because those have a higher mechanical stability.
However, staining of these thicker gels takes longer and is in some
cases less sensitive. Horizontal gels are usually 0.5 mm thin.
There are two other standard gel sizes for vertical systems:
1616 cm and 88 cm (minigels); these gels are much easier to han-
dle, however for 2-D electrophoresis they suffer from limited resolu-
tion. Their major application area in proteomics is 1-D SDS electro-
phoresis and blotting. For 1-D separations in horizontal flatbed sys-
tems 25 cm wide and 11 cm long gels are used for running multiple
samples.
1.2.1.6 Mechanically Supported Polyacrylamide Gels
Although polyacrylamide gels have a much better mechanical stabi-
lity than agarose gels, they can swell, shrink, and become fragile after
they have been removed from the cassettes. Particularly during stain-
ing gels can easily break into many pieces. Some laboratories use
therefore gel strengtheners, which are either acrylamide derivatives
or supplements to acrylamide monomer solutions.
As an alternative, gels can be bound to one of the cassettes glass
plates, which has been pretreated with Bind-Silane. This procedure is
particularly useful, when selected bands or spots have to be cut out
with an automated spot picker. In this case the spot picker cuts the
spots from the gel while using the information on the spot position
coordinates from the image analysis results. This concept would not
work, when the gel would shrink or swell between scanning and spot
picking.
Instead of binding a gel to a glass plate, it can be cast on a film sup-
port. This has become a standard for gels, which are run on a hori-
zontal flatbed apparatus, but is also applied for vertical gels. Lately
new supporting films have been developed, which do not exhibit
fluorescent background anymore. Furthermore these films are trea-
ted with a novel binding chemistry, which does not increase the gel
pore sizes near the film surface.
Klose J, Kobalz U. Electrophor-
esis 16 (1995) 1034–1059.
Nevertheless many laboratories
are satisfied with the resolution
of 2-D separations in smaller
gels.
Gels with improved mechanical
strength and elasticity are
obtained with these reagents,
however on cost of resolution
and pattern quality.
If the gel has to be stained, the
reagents and dyes can diffuse
into the gel only from the open
side, which can lead to longer
staining time and to lower
sensitivity, for instance for silver
staining.
Gels on film supports are easier
to ship, handle and store than
glass plate supported gels.

1.2 Polyacrylamide Gel Electrophoresis 27
1.2.1.7 Reversible Polyacrylamide Gels
Practical experiences in mass spectrometry analysis of peptide mix-
tures show that better signals are obtained from samples digested in
liquid phase rather than in gel plugs. Besides methylenebisacryla-
mide a number of other cross-linking reagents exist, listed and com-
pared by Righetti (1983). Some of them possess a cleavage site, which
allows solubilization of the gel matrix after electrophoresis (see
Table 1.1).
Tab. 1.1: Alternative crosslinkers for polyacrylamide gels (examples).
Substance Cleavage site Cleavage agent Comment
N,N¢-methylenebisacrylamide
(Bis)
None Not possible Standard crosslinker
N,N¢-(1,2-Dihydroxyethylene)bisacrylamide
(DHEBA)
1,2-diol Periodic acid
N,N¢-Diallyl-ditartardiamine
(DATD)
Ester bonds Hydrolysis with
a base
Problems with inefficient
polymerization
N,N¢-Bisacryloylcystamine
(BAC)
Disulfide bond Thiol reagents DTT in the sample must
be completely scavenged
by iodoacetamide
Furthermore, reversible polyacrylamide gels can become very inter-
esting as a sample preparation method for the “top-down” approach
in proteomics, where intact proteins are directly analyzed with very
high resolution mass spectrometry like FT-ICR and orbitraps (see
page 232 f).
1.2.2
SDS Polyacrylamide Gel Electrophoresis
In general this is the mostly applied electrophoretic method for pro-
tein analysis. In proteomics SDS PAGE of proteins is employed for
many different purposes:
.
Second dimension in high-resolution 2-D elec-
trophoresis;
.
1-D protein separation prior to tryptic digestion
and LC-MS of peptides;
.
Second dimension for “dual detergent” electro-
phoresis (after separation with 16-BAC or
CTAB);
.
Second dimension for Blue native electrophor-
esis;
.
Third dimension in IPAS.
Righetti PG.: Isoelectric
focusing: theory, methodology
and applications. Elsevier
Biomedical Press, Amsterdam
(1983).

1 Electrophoretic Techniques28
1.2.2.1 Theoretical Background
Sodium dodecyl sulfate (SDS) is a very strong anion detergent, and
solubilizes all proteins including the very hydrophobic ones. It dena-
tures proteins by dissolving hydrogen bonds and unfolds the tertiary
and secondary structures. SDS and proteins form complexes with a
necklace-like structure composed of protein-decorated micelles con-
nected by short flexible polypeptide segments (Ibel et al. 1990). As a
result of the necklace structure large amounts of SDS are incorpo-
rated in the SDS–protein complex in a ratio of approximately 1.4 g
SDS/g protein. SDS masks the charge of the proteins themselves and
the formed anionic complexes have a reasonably constant net nega-
tive charge per unit mass. Usually a reducing agent such as DTT is
added to the sample to cleave the disulfide bridges between cysteines.
Then the polypeptides become completely unfolded, all quaternary
structures are dissolved.
With SDS PAGE (SDS–polyacrylamide gel electrophoresis) the
polypeptides are separated according to their molecular weights (M
r
).
All proteins – also those with basic pIs – will migrate towards the
anode. The electrophoretic mobility of proteins treated with SDS and
DTT depends largely on the molecular weight of the protein. At a cer-
tain polyacrylamide percentage there is an approximately linear rela-
tionship between the logarithm of the molecular weight and the rela-
tive migration distance of the SDS–polypeptide complexes of a cer-
tain molecular weight range. The molecular weights of the sample
proteins can be estimated with the help of co-migrated standards
with known molecular weights.
& Note: SDS PAGE cannot deliver the exact
molecular mass of a protein, it allows only
estimation.
1.2.2.2 Sample Preparation
In the standard sample preparation procedure for 1-D SDS electro-
phoresis the proteins are boiled for 3 minutes in the sample buffer
containing 2% SDS, 50 mmol/L Tris-Cl pH 8.8, 0.01% Bromophenol
blue, and 1% (w/v) DTT. For vertical gels the sample buffer must con-
tain 25% glycerol to prevent mixing of the sample with the upper buf-
fer. When the sample has cooled down, back folding and aggregating
of subunits must be prevented: either by adding the same amount of
reductant again or by alkylating the cysteines with 2.5% (w/v) iodo-
acetamide. Alkylation has several advantages: higher stability of the
samples, prevention of artifactual lines across the gel and for down-
Ibel K, May RP, Kirschner K,
Szadkowski H, Mascher E,
Lundahl P. Eur J Biochem 190
(1990) 311–318.
For complex protein mixtures
mostly gels with T values of 12
to 13 are used for the optimal
separation in the range
between 10 kDa and 100 kDa.
Exact masses can only be deter-
mined with mass spectrometry.
It should be noted that during
electrophoresis the polypeptides
become partly alkylating by
acrylamide. For database
searches after mass spectro-
metry it is very important that
either none or all cysteines are
alkylated.

1.2 Polyacrylamide Gel Electrophoresis 29
stream analysis with mass spectrometry ensuring alkylation of all
cysteines.
For some clinical applications, or for the detection of antibodies by
blotting, sometimes non-reduced samples are applied, in order to
maintain the quaternary structure of the immunoglobulins. In this
case the molecular weights cannot be determined, because the disul-
fide bridges are still intact, many of the polypeptides are still folded.
Non-reduced samples must not be boiled; this would cause fragmen-
tation of some polypeptides.
1.2.2.3 Buffers and Gels
Tris-chloride / Tris-glycine
The standard buffer system for SDS
PAGE is based on the discontinuous Tris-chloride / Tris-glycine sys-
tem described by Laemmli (1970). With this buffer system, reproduci-
ble and well resolved patterns are obtained even with high protein
loads. It should be noted that different laboratories prepare the buf-
fers in slightly different ways, this can cause differences of the run-
ning conditions. When the gel buffer is titrated with a pH meter, the
measuring electrode should be well calibrated. Some laboratories
measure the added hydrochloric acid by the volume.
The running buffer and the gels contain 0.1% SDS. Sometimes the
gels are cast without SDS, because the SDS migrating into the gel
from the cathodal buffer is sufficient. During electrophoresis the
negatively charged chloride, SDS and glycine ions migrate towards
the anode, the positively charged Tris ions migrate towards the cath-
ode. The buffer reservoirs of the electrophoresis chamber must be
large enough to prevent depletion of the buffer ions.
& Note: Use only Tris base for making the buffers,
not “Tris buffer”. The gel buffers are made with
Tris and HCl, they contain only chloride. The
cathodal running buffer must not contain any
chloride; only SDS, Tris and glycine. Do not
titrate the upper running buffer to adjust the
pH!
Because of the very alkaline pH 8.8 in the gel of the Laemmli system,
the polyacrylamide matrix becomes hydrolyzed during storage. After
two months the sieving property of the matrix is almost completely
gone. In the standard laboratory practice this is not a big issue,
because the gels are consumed within one week after preparation.
The situation is different, when ready-made gels are used.
Some people call this “native”
SDS electrophoresis.
Lmmli UK. Nature 227
(1970) 680–685.
Adding HCl to the upper
running buffer eliminates the
stacking effect and causes
extended running time.
The problem is not only the
limitation in shelf life, but also
the lack of reproducibility
because of the ongoing hydro-
lysis of the matrix.
