Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.


1 Electrophoretic Techniques50
Basic IPG gradients As already mentioned above, polyacrylamide
gels are sensitive to high pH values. The higher the pH value, the fas-
ter they become hydrolyzed. There is no problem during storage of
IPG gels, because the pH is buffered with a small amount of acidic
compounds, and at the freezing temperatures of storage and delivery
of ready-made gels this process does not occur. But during isoelectric
focusing these buffering anions will be electrophoretically removed.
At 20 C running temperature the very basic pH value will cause
hydrolysis of the matrix, and the basic buffering groups will be partly
cut off the gel. This results in a flattening out of the gradient at the
basic end with time: what was pH 11 will come closer to a real pH
value of 9. Basic IPGs should therefore be run at a minimum of time;
narrow basic IPG gels were restricted to 18 cm length because of the
restricted running time.
A new development allows using IPG gels with a gradient 7–11
with 24 cm length. By replacing acrylamide monomers with a more
stable reagent, these IPG gels can be prevented from hydrolysis.
Advantages of immobilized pH gradient strips in 2-D electrophoresis
As already mentioned above, isoelectric focusing in pre-manufac-
tured IPG strips is a highly reproducible method compared to gels
with carrier ampholytes generated pH gradients. There are several
reasons for this fact:
.
Industrial production reduces variations caused
by human interference.
.
The chemistry of the buffering acrylamide deri-
vatives is better controllable.
.
The film-supported gel strips are easy to handle.
.
The fixed gradients are not modified by the sam-
ple composition, and they do not drift with IEF
time.
.
Stable basic pH gradients allow reproducible
separation and display of basic proteins.
The concept of immobilized pH gradients offers a number of addi-
tional beneficial features:
.
Different ways of sample applications are feasi-
ble; the dried strips can be directly rehydrated
with the sample solutions.
.
Higher protein loads are achievable.
The consequence: The very
basic proteins are lost, the
wrong isoelectric point values
are determined for basic
proteins.
The recipe for these IPGs and
the reagent are proprietary to
GE Healthcare Life Sciences.
Readymade IPG strips are
usually produced according to
GMP routines.
In contrast to carrier ampho-
lytes, which are mixtures of
several hundred homologues.
Thin gel rods require high
experimental skill.
Very important for reproducible
results.
In contrast to NEPHGE, where
the basic proteins are not
focused at their pI.
This procedure prevents
proteins from precipitating at
the application point.
This allows also the detection of
low expressed proteins.
1.4 Isoelectric Focusing 51
.
Various additives, like detergents and reductants,
can be added to the rehydration solution.
.
Less proteins are lost during equilibration in
SDS buffer, because the fixed charged groups of
the gradient hold the proteins back like a weak
ion exchanger (Righetti and Gelfi, 1984).
.
Probably the most powerful feature is the possi-
bility to reach almost unlimited spatial resolu-
tion with very narrow pH intervals. This is
needed for protein identification and characteri-
zation.
& Dried gel strips can be stored at –20 C to –80 C
from months to years before reconstitution.
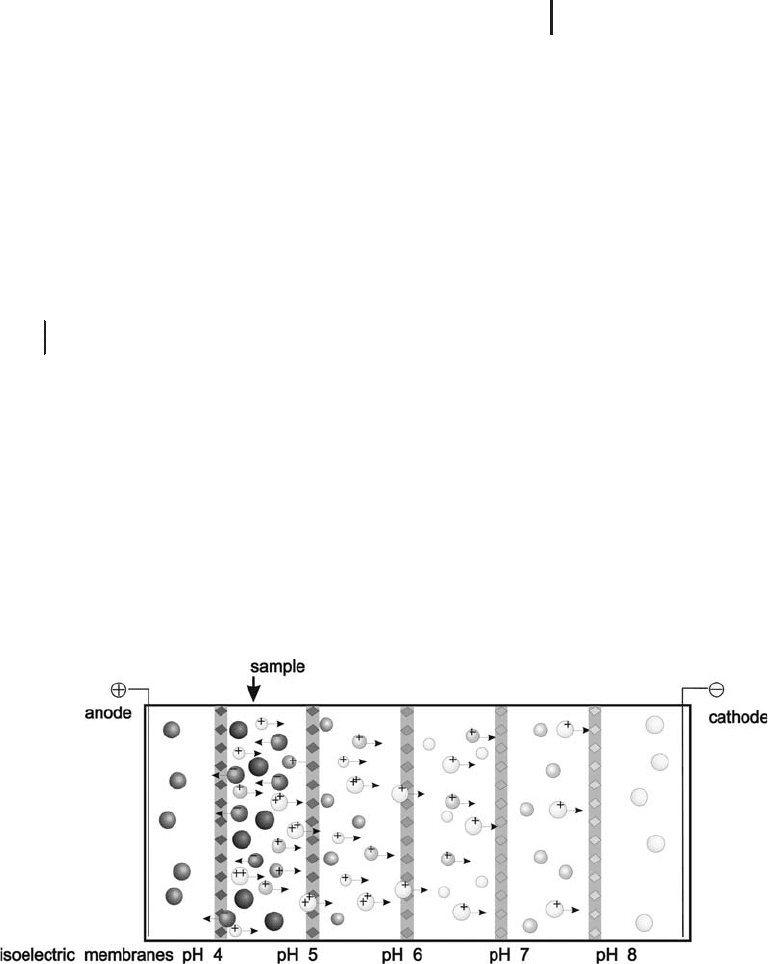
Fractionation between buffering isoelectric membranes
The principle
of this technique is based on the concept of immobilized pH gradi-
ents. The separation occurs in the liquid phase in a multi-compart-
ment apparatus, which is divided by buffering isoelectric membranes
with defined pIs (Wenger et al. 1987). The electrodes are located in
the two outer segments. The membranes are prepared by polymeriz-
ing a polyacrylamide gel layer with basic and acidic Immobilines
around a micro fiber filter or polyester grid. The amounts of Immobi-
lines needed for a certain pH value are precalculated in the same way
like for the immobilized pH gradients. Figure 1.14 shows the princi-
ple of fractionation with isoelectric membranes.
Fig. 1.14: Pre-fractionation of a protein mixture in liquid phase
with isoelectric membranes.
Some of them would inhibit gel
polymerization.
Righetti PG, Gelfi C. J Biochem
Biophys Methods. 9 (1984)
103–119.
Gradients can be engineered
according to special needs; they
are absolutely continuous.
Wenger P, de Zuanni M, Javet
P, Righetti PG. J Biochem
Biophys Methods 14 (1987)
29–43.

1 Electrophoretic Techniques52
In a mixture the sample proteins are charged. In the electric field,
a protein with a high pI is protonated and migrates through the com-
partments towards the cathode until it reaches a membrane with a
higher pI. It cannot pass through this membrane, because it will
become deprotonated. If it will get a negative net charge there, it will
migrate back towards the anode until it reaches the previous mem-
brane with a lower pI. There it will become protonated again, and so
on. It is thus trapped in the compartment.
Herbert and Righetti (2000) as well as Speicher and Zuo (2000)
could show that sample prefractionation via multicompartment elec-
trolyzers with isoelectric membranes greatly enhances the loadability,
resolution and detection sensitivity of 2-D maps in proteome analysis.
In contrast, it has been reported that sometimes sticking of hydro-
phobic proteins to the membranes are causing electroendosmotic
effects and clogging of the membrane pores.
Off-Gel isoelectric focusing in a multicompartment device The pro-
teins or peptides are separated according to their isoelectric point in a
multiwell device, which is connected to immobilized pH gradient
strips (Michel et al. 2003). The separation occurs in liquid phase, not
in the gel. The protein fractions are directly recovered in solution for
further analysis.
Isoelectric focusing followed directly by mass spectrometry Analo-
gous to the 1-D SDS electrophoresis LC-MS approach the proteins
can be digested directly after isoelectric focusing in IPG strips with
subsequent LC-MS (Giorgianni et al. 2003).
Isoelectric focusing of peptides Essader et al. (2005) have found that
the use of narrow range immobilized pH gradients pH 3.5–4.5 for
peptide separation in the first dimension yielded 13% more protein
identifications than the optimized off-line strong-cation-exchange
chromatography as a first dimension in shotgun proteomics. In this
work greater than 11,500 peptides and 3,700 proteins were identified
with high statistical confidence. The approach has a second advan-
tage: Accurate pI prediction can then be employed using currently
available algorithms to very effectively filter data for peptide/protein
identification, and thus lowering the false-positive rate for standard
peptide identification algorithms.
Herbert B, Righetti PG. Elec-
trophoresis 21 (2000)
3639–3648.
Speicher DW, Zuo X. Anal
Biochem. 284 (2000)
266–278.
Michel PE , Reymond F,
Arnaud IL, Josserand J, Girault
HH, Rossier JS. Electrophoresis
24 (2003) 3–11.
Giorgianni F, Desiderio DM,
Beranova-Giorgianni S. Electro-
phoresis 24 (2003) 253–259.
Essader AS, Cargile BJ, Bundy
JL, Stephenson JL. Proteomics
5 (2005) 24–34.

1.5 Two-dimensional Electrophoresis 53
1.5
Two-dimensional Electrophoresis
As already mentioned above, high-resolution two-dimensional elec-
trophoresis is still the mainly applied separation technique in proteo-
mics. The separation according to two completely independent phy-
sico-chemical parameters of proteins, isoelectric point and size, offers
the highest resolution. Several thousand proteins can be separated,
displayed and stored in one gel without degradation.
It is often tried to compare a 2-D gel result of a certain sample with
a 2-D pattern displayed on a website or in a scientific paper. That
does not work, because there are too many variables. There are stan-
dardization initiatives on the way, which try to design recommended
procedures for sample preparation and running conditions. The con-
ditions for these procedures must be highly stringent; otherwise pat-
tern comparisons across different laboratories will never be possible.
However, a protein can never securely be identified by its spot posi-
tion in a 2-D gel. The spot has to be excized from the gel, the protein
has to be digested with a proteolytic enzyme, and the peptides have to
be analyzed with mass spectrometry.
1.5.1
Sample Preparation
The sample treatment is the key to adequate results. The protein com-
position of the cell lysate or tissue must be reflected in the pattern of
the 2-D electrophoresis gel without any losses or modifications. Of
course, the sample must not be contaminated with proteins and pep-
tides not belonging to the sample. “Co-analytical modifications”
(CAM) of proteins need to be avoided. This is not trivial, because var-
ious protein–protein interactions may happen in such a complex mix-
ture, and pre-purification of the sample can lead to uncontrolled
losses of some of the proteins.
The key points for appropriate sample acquisition and preparation
for 2-D electrophoresis in proteomics are:
.
Avoid uncontrolled loss of proteins;
.
Disrupt all aggregates and complexes
.
Prevent enzyme activities;
.
Remove disturbing compounds without remov-
ing proteins.
With higher protein loads
sample preparation becomes
even more critical, because also
the contaminants concentra-
tions are increased.
The lost protein might be the
most important ones
Because they are too big to
enter the gel
Proteases and phosphatases are
also active under denaturing
conditions
Such as salts, lipids, polysac-
charides, nucleic acids, polyphe-
nols

1 Electrophoretic Techniques54
1.5.1.1 Sample Acquisition
It should be mentioned that the way, how samples are collected and
stored, is very important. It is relatively easy for body fluids and
lysates from cultured cells, because the proteins are evenly distribu-
ted. Strict rules have been designed and are followed for collecting
clinical samples to prevent degradation. Some samples are particu-
larly critical. It has, for instance, been reported that the protein com-
position of brain cells starts to change dramatically immediately after
death. Skçld et al. (2002), for instance, keep the time between removal
of a total rat brain and having it completely frozen at –80 C below
3 minutes.
When patient tissue material is studied, the cells to be analyzed
must be well defined. A highly selective procedure for tissue analysis
is needle dissection or laser capture micro dissection under the
microscope (Banks et al. 1999), where diseased cells and control cells
are separately collected to gain enough material for a 2-D electrophor-
esis.
As a rule of thumb about 10
7
to 10
8
cells – or about 100 mg protein
– are required for a silver- or fluorescent dye-stained 2-D gel. If the
sample material has to be collected with laser capture or needle micro
dissection of tissue cells under the microscope, it can take several
days to a week to obtain enough protein sample for a 2-D electrophor-
esis.
1.5.1.2 Sample Treatment
To avoid protein losses, the treatment of the sample must be kept to a
minimum; to avoid protein modification, the sample should be kept
as cold as possible; to avoid losses and modifications, the time should
be kept as short as possible.
Too much salt in the sample disturbs isoelectric focusing and leads
to streaky patterns. Amphoteric buffers in cell cultures, like HEPES,
overbuffer the gradient in the areas of their pIs, which results in ver-
tical narrow areas without protein spots. The chemicals used must be
of the highest purity.
The proteins have to be extracted from cells or tissue material.
Liquid samples have to be denatured to prevent the formation of poly-
peptide oligomers, aggregates and interactions. Some material con-
tains proteolytic enzymes, which are still active under denaturing
conditions. Phosphatases have to be inhibited to maintain the post-
translational modifications to be determined.
Skçld K, Svensson M, Kaplan
A, Bjçrkesten L, strçm J,
Andren PE. Proteomics 2
(2002) 447–454.
Banks R. Dunn MJ, Forbes MA,
Stanly A, Pappin DJ, Naven T,
Gough M, Harnden P, Selby
PJ. Electrophoresis 20 (1999)
689–700.
Pre-labeling of cysteines with
CyDyes increases the sensitivity
of detection substantially, and
therefore reduces the amount
of required cells considerably.

1.5 Two-dimensional Electrophoresis 55
1.5.1.3 Denaturing Conditions
A frequently expressed wish of proteomics researchers is the possibi-
lity to separate the protein mixtures under native conditions, in order
to conserve the three-dimensional structures of the proteins. There
are a number of reasons, why high molar urea, a reductant, and non-
ionic or zwitterionic detergents must be present in a sample for high-
resolution 2-D electrophoresis:
.
Aggregates and complexes between proteins and
proteins, polysaccharides, and lipids would be
too big to enter a gel matrix. They are usually
disintegrated by the addition of high urea con-
centrations and reduction of the sample.
.
Under native conditions, a great part of the pro-
teins exists in several different conformations.
This would lead to even more complex 2-D pat-
tern, which could not be evaluated. The proteins
have to be denatured in order to display them in
single conformations.
.
Different oxidation steps must be prevented by
the addition of a reducing agent.
.
All proteins, also the hydrophobic ones, have to
be brought into solution. Because buffers and
salt ions would disturb isoelectric focusing, non-
charged chaotropes, like urea and thiourea, and
detergents have to be used for the solubility of
all proteins.
.
Protein–protein interactions are avoided by
applying denaturing conditions.
.
For the analysis of the proteome, it should be
possible to match the theoretically calculated iso-
electric point of a polypeptide with pI position in
the 2-D map. This is only possible, when the
three-dimensional structure is cancelled and all
buffering groups are exposed to the medium.
Bjellqvist et al. have shown that this is possible
with immobilized pH gradients.
& Composition of the standard lysis solution:
9 mol/L urea, 4% (w/v) CHAPS, 1% (w/v) DTT,
2% (v/v) carrier ampholytes, 0.002% (w/v)
Bromophenol blue.
Unfortunately it is impossible
to separate complex protein
mixtures under native condi-
tions.
Sometimes the sample needs to
be treated by precipitation to
dissolve strongly coherent
complexes.
In presence of more than 7
mol/L urea most polypeptides
exist only in one single config-
uration.
Also the addition of carrier
ampholytes supports the solubi-
lity of hydrophobic proteins.
In the three-dimensional config-
uration some buffering groups
are hidden, which causes a shift
of the pI.
Bjellqvist B, Hughes GJ,
Pasquali C, Paquet N, Ravier
F, Sanchez J-C, Frutiger S,
Hochstrasser D. Electrophoresis
14 (1993) 1023–1031.

1 Electrophoretic Techniques56
Urea The high urea concentration is needed to convert proteins into
single conformations by canceling the secondary and tertiary struc-
tures, to get and keep hydrophobic proteins in solution, and to avoid
protein–protein interactions. Only in special cases a second, stronger
denaturing chaotrope has to be added to urea in order to increase the
solubility of very hydrophobic proteins, like membrane proteins:
thiourea. The purity of urea is very critical: Isocyanate impurities and
heating must be avoided, because these would cause carbamylation
of the proteins, resulting in artifactual spots.
Detergent CHAPS is a zwitterionic detergent, and preferred to non-
ionic polyol mixtures like Triton X-100 and Nonidet NP-40, because
of its higher purity. It increases the solubility of hydrophobic pro-
teins.
Reducing agents The reductants DTT or DTE prevent different oxi-
dation steps of the proteins. Both are interchangeable. 2-mercap-
toethanol should not be used, because of its buffering effect above
pH 8 (Righetti et al. 1982). This would cause horizontal streaks in the
area between pH 8 and 9. Also, keratin contaminations, seen with
mass spectrometry, have been traced back to contaminated 2-mercap-
toethanol (Parness and Paul-Pletzer, 2001).
Unfortunately DTT as well as DTE can become ionized above their
pK of 8 and migrate towards the anode during IEF in basic pH gradi-
ents after some time. The cysteines are no longer protected, which
leads to back folding and aggregation of some of the polypeptides,
resulting in modifications of their pIs and thus horizontal streaks.
Additionally, non-specific reactions of cysteines with urea cause arti-
factual spots.
Tributylphosphine (TBP), introduced into 2-D electrophoresis by
Herbert et al. (1998), is generally not recommended as a replacement,
because of its poor stability in the electric field. Also its replacement
by Tris(2-carboxyethyl) phosphine (TCEP) does not improve the
separation. As a remedy Herbert et al. (2001) have then proposed alky-
lation of proteins prior to IEF, particularly for separations in basic pH
gradients. Also this procedure cannot be recommended, because
complete alkylation of all proteins in a complex mixture is not easy to
control. Therefore additional artifactual spots are observed, resulting
from incomplete alkylation. Furthermore, the pIs of the basic pro-
teins become modified, which is another disadvantage.
The phenomenon of streaking and appearance of artifactual spots
in basic regions is abolished, when the first dimension IEF is run
with an excess of hydroxyethyldisulfide (HED, “DeStreak”) according
to Olsson et al. (2002). For rehydration of the IPG strip the reductant
is replaced by 100 mmol/L HED (“DeStreak”). The proteins are
Urea is not stable in solution,
repeated freeze – thawing must
be avoided.
Mass spectrometry is particu-
larly sensitive to contamina-
tions coming from detergents
like Triton X-100.
Righetti PG, Tudor G,
Gianazza E. J Biochem Biophys
Methods 6 (1982) 219–22.
Parness J, Paul-Pletzer K. Anal
Biochem 289 (2001) 98–99.
Herbert BR, Molloy MP,
Gooley AA, Walsh BJ, Bryson
WG, Williams KL Electrophor-
esis 19 (1998) 845–851.
Herbert B, Galvani M,
Hamdan M, Olivieri E,
MacCarthy J, Pedersen S,
Righetti PG. Electrophoresis 22
(2001) 2046–2057.
Olsson I, Larsson K, Palmgren
R, Bjellqvist B. Proteomics 2
(2002) 1630–1632.

1.5 Two-dimensional Electrophoresis 57
extracted in the conventional way with a reductant like DTT or DTE;
also alternative reductants like 2-mercaptoethanol, TBP, or TCEP can
be used. The lysis solution must not contain DeStreak. When the
reduced proteins electrophoretically enter the IPG strip, the HED
immediately oxidizes the protein cysteinyl groups to mixed disulfides.
In this way all thiol groups are converted into one defined form,
which will not react anymore. Back folding and aggregation of subu-
nits are thus prevented. Non-specific reactions of cysteines with urea
cannot happen anymore. The resulting pattern shows round spots,
the number of spots is reduced compared to separations in presence
of reductant, and there is a light shift of the basic pIs towards basic
pH because of the missing negative charges of the thiols.
When DeStreak (HED) is used, it is highly recommended to use
cup- or paper bridge-loading at the anodal end of the IPG strip
instead of rehydration loading. More information can be found on
pages 79 and 85 where rehydration and sample loading is explained.
If rehydration loading is needed, the DTT content of the final rehy-
dration solution (sample diluted with rehydration solution) must not
contain more than 1 mmol/L DTT.
An alternative way to adequate and reproducible 2-D pattern in
basic gradients has been suggested by Hoving et al. (2001): the addi-
tion of higher amounts of DTT to the gel, adding more DTT to a cath-
odal paper strip, and a few more measures described later in the iso-
electric focusing chapter.
Carrier ampholytes Carrier ampholytes, which had been designed
for generating pH gradients, improve the solubility of proteins con-
siderably by substituting ionic buffers. In a mixture they are charged.
They do not disturb IEF like buffer addition, because they migrate to
their pIs, where they become uncharged. Dedicated pH intervals, pre-
pared for the addition to immobilized pH gradients, are called IPG
buffers.
Various IPG buffer mixtures are designed for the respective pH gra-
dients. Sometimes in practice, the use of carrier ampholyte mixtures
for wide gradients, like Pharmalytes pH 3–10, instead of dedicated
IPG buffers, has shown better results for narrow gradients.
Dyes The anionic dye Bromophenol blue is very useful as a control
for the start and running conditions. The low amounts used do not
disturb the analysis.
If DeStreak is mixed with a
reductant, it will become
reduced to 2-mercaptoethanol.
As described above, 2-mercap-
toethanol causes horizontal
streaking.
Hoving S, Gerrits B, Voshol H,
Mller D, Roberts RC, van
Oostrum J. Proteomics 2
(2002) 127–134.
Note: The composition of
carrier ampholytes or IPG
buffers used will influence the
result.
This should be checked in a
practical optimization experi-
ment for a different sample
type.
Bromophenol blue can be inter-
changed with Orange G.

1 Electrophoretic Techniques58
1.5.1.4 Alternative Lysis Solutions
Thiourea / urea solution
For the extraction and solubilization of
highly hydrophobic proteins like membrane proteins a combination
of 7 mol/L urea plus 2 mol/L thiourea lysis solution can be very help-
ful to get more proteins into solution (Rabilloud, 1998).
& Composition of the alternative lysis solution:
7 mol/L urea, 2 mol/L thiourea, 4% (w/v)
CHAPS, 1% (w/v) DTT, 2% (v/v) carrier
ampholytes, 0.002% (w/v) Bromophenol blue.
Generally, extraction with urea and thiourea combined increases the
number of spots considerably. It can be observed in the literature that
many researchers have started to use this combination of chaotropes
for all types of samples. However artifacts can be observed as well.
Figure 1.15 shows the results of 2-D electrophoresis of rat liver
extracts: the lysis buffer differed only in the chaotropes used. The
thiourea-containing sample shows many more spots, but also a phe-
nomenon typical for thiourea gels: a vertical streak in the acidic area
and blurred spots in the pH range below the streak. This effect
becomes more pronounced with increased focusing times. So far a
remedy for this effect has not been found.
Fig. 1.15: 2-D electrophoresis of rat liver extracts.
A: Lysis solution contained 8 mol/L urea.
B: Lysis solution contained 7 mol/L urea and 2 mol/L thiourea.
Gels: IEF in 18 cm IPG 3–10 non-linear, SDS PAGE in 2020 cm
gels of 12.5% T, 1 mm thick. Silver staining.
SDS procedure In some cases the anionic detergent SDS can be
employed for sample preparation: up to 2% SDS has been used.
Before the sample is applied to denaturing isoelectric focusing the
SDS sample has to be at least 20-fold diluted with urea and a non- or
Rabilloud T. Electrophoresis 19
(1998) 758–760.
This streak and the blurred
spots are not sample-specific. It
seems that thiourea starts to
focus itself.
The precipitation clean-up
procedure described below can
also remove the SDS from the
proteins.

1.5 Two-dimensional Electrophoresis 59
zwitterionic detergent containing solution. In the electric field the
SDS will separate from the proteins and migrate into the anode. The
major reasons for using SDS are:
.
In human serum and plasma the formation of
oligomers are only prevented by boiling the sam-
ple in SDS.
.
Organisms with tough cell walls sometimes
require extraction with 1% SDS before they are
diluted with lysis buffer.
.
Some very hydrophobic proteins may require
extraction with high percentage of SDS.
.
It is also applied for plant protein extraction (see
below).
Unfortunately SDS does not always completely separate from the pro-
teins, even under high field strength. This can result in the shift of
some isoelectric points to a more acidic value.
New zwitterionic detergents and sulfobetains A series of novel zwit-
terionic detergents (Chevallet et al. 1998) and non-detergent sulfobe-
tains (Vuillard et al. 1995) have been tried. Some hydrophobic mem-
brane proteins could be solubilized, which otherwise would have
been lost. Molloy (2000) has reported that membrane proteins go bet-
ter in solution with 7 mol/L urea plus 2 mol/L thiourea and alterna-
tive zwitterionic detergents like ASB 14 or sulfobetain in the lysis
solution. Nevertheless, up to now none of these additives has per-
formed so well in general that it has replaced CHAPS in the standard
cocktail. A perfect solution has not yet been found.
1.5.1.5 Protease Inactivation
Some proteases are also active in presence of urea and detergents.
Protease inhibitors can inactivate most of the proteolytic activities,
however in some cell lysates not completely. PMSF is frequently used
(8 mmol/L), but it is a toxic compound. It has to be added to the sam-
ple prior to the reductant, because thiols deactivate PMSF. Pefabloc
(AEBSF) applied as 5 to 10 mmol/L is less toxic, but might lead to
charge modifications of some proteins (Dunn, 1993). This can also
happen with application of protease “cocktails”. Some proteases are
inhibited by the denaturing conditions, some by basic pH. Therefore
Tris base – below 40 mmol/L – is sometimes added to the lysis solu-
tion.
Proteases can be inactivated by boiling the sample in SDS buffer
for a few seconds prior to the addition of urea-containing lysis solu-
tion. Completely irreversible protease inactivation is only obtained
Hughes GJ, Frutiger S, Paquet
N, Ravier F, Pasquali C,
Sanchez JC, James R, Tissot JD,
Bjellqvist B, Hochstrasser DF.
Electrophoresis 13 (1992) 707–
714.
Gçrg A, Drews O, Weiss W. In
Simpson RJ, Ed. Purifying
proteins for proteomics: A
laboratory manual. Cold Spring
Harbor Laboratory Press, Cold
Spring Harbor, New York
(2003) 407.
Chevallet M, Santoni V, Poinas
A, Rouquie D, Fuchs A, Kieffer
S, Rossignol M, Lunardi J,
Gerin J, Rabilloud T. Electro-
phoresis 19 (1998)1901–1909.
Vuillard L, Marret N, Rabilloud
T. Electrophoresis 16 (1995)
295–297.
Molloy MP. Anal Biochem 280
(2000)1–10.
There are no protease inhibi-
tors, which can stop all
protease activities completely.
Dunn MJ. Gel electrophoresis
of proteins. Bios Scientific
Publishers Alden Press, Oxford
(1993).
In practice the sample is
extracted with a protease inhi-
bitor cocktail, and then the
sample is precipitated.
