Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.

1 Electrophoretic Techniques30
Long shelf life SDS gels For long shelf life, the pH value of the gel
buffer has to be reduced to or below pH 7. This requires a modifica-
tion of the buffer composition.
Tris-acetate / Tris-tricine Tris-acetate buffer with a pH of 6.7 has pro-
ven to have a good storage stability and separation capacity for flatbed
gels. Glycine has to be replaced by tricine as the terminating ion.
Since tricine is more expensive, it is only used in the cathodal buffer.
The anodal buffer contains Tris-acetate. The running buffer can be
applied as polyacrylamide electrode strips, instead of connecting the
gels to liquid buffers in tanks, see also page 108. The molecular weight
distribution of proteins obtained with this buffer system is slightly
different from that achieved with the Laemmli system. This buffer
system is also applied by rehydratization of pre-polymerized, washed
film-supported gels for SDS in horizontal flatbed chambers (Clean-
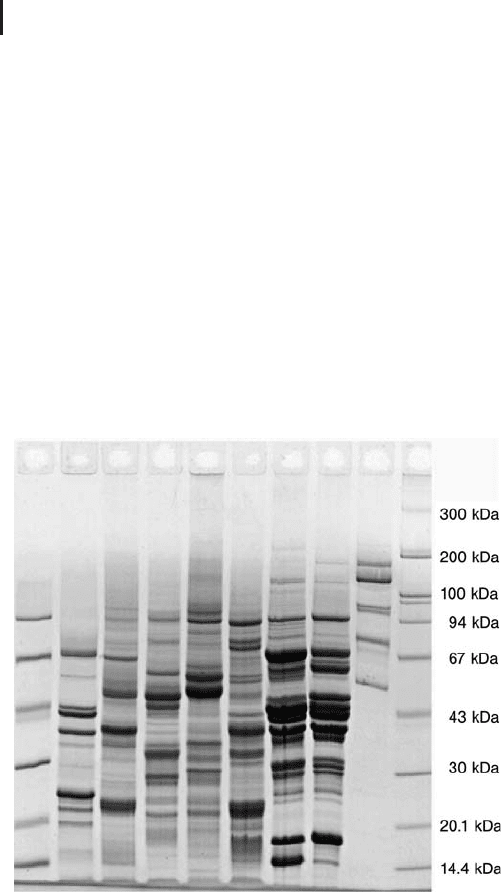
Gel). Figure 1.4 shows a SDS electrophoresis result in a 10% T gel.
Fig. 1.4: SDS electrophoresis in a CleanGel 10% T.
Samples: plant seed proteins and M
r
standards.
Hot Coomassie brilliant blue staining.
Bis-Tris / Bis-Tris-tricine This is similar to the above system. Tris is
replaced by Bis-Tris to obtain improved buffer capacity at neutral pH
(Wiltfang et al. 1991). Also with this buffer system the spot patterns
are different from Laemmli buffer patterns.
Because of the tricine in the
cathodal buffer, these gels show
a very good separation of small
peptides, when a gradient gel or
a 15% T gel is used.
Wiltfang J, Arold N, Neuhoff V.
Electrophoresis 12 (1991)
352–366.

1.2 Polyacrylamide Gel Electrophoresis 31
PPA-chloride / Tris-glycine Here the Tris in the gel is replaced by
PPA (piperidino propionamide) and titrated with hydrochloric acid to
pH 7. This gel buffer can be combined with the standard running
buffer Tris-glycine in the cathodal buffer. For the anodal buffer PPA
would be required instead of Tris. Because this compound is rather
expensive, the large volume anodal buffer usually contains a cheaper
compound: diethanolamine-acetate (DEA).
Tris-borate for improved separation of glycoproteins Glycoproteins
migrate slower than pure polypeptides, since the sugar moiety does
not bind SDS. When a Tris-borate-EDTA buffer is used, borate sticks
to the sugars, the sugar moieties become also negatively charged:
therefore the speed of migration will increase (Poduslo, 1981). The
glycine in the running buffer is simply replaced by boric acid. The
use of gradient gels is also beneficial for better MW estimations. See
also cationic detergent electrophoresis on page 34 f.
Homogeneous gels Usually a homogeneous gel with 12.5% T and
3% C is used; the proteins of major interest in the size range from 10
kDa to 100 kDa are optimally resolved. Only in special cases the
matrix concentration is modified to increase the resolution in certain
molecular size ranges.
The rule of thumb is:
.
Lower T value: better resolution for high molecu-
lar weight proteins.
.
Higher T value: better resolution for low molecu-
lar weight proteins.
Gradient gels Gradient gels offer a broader separation interval than
homogeneous gels, also the linear relation interval between the loga-
rithm of M
r
and the migration distance is wider than for homoge-
neous gels. And they exhibit a zone sharpening effect.
Peptide gels The resolution of peptides below 10 kDa is not suffi-
cient in conventional Tris-chloride / Tris-glycine systems. The pep-
tides smaller than 10 kDa co-migrate with the SDS front. A number
of modifications of the standard gel and buffer system have been pro-
posed, for instance the addition of 8 mol/L urea to the gel by Hashi-
moto et al. (1983), and, additionally, increasing the Tris concentration
in the gel to 1 mol/L by Anderson et al. (1983). The most efficient
technique has been developed by Schgger and von Jagow (1987):
The resolving gel has a composition of 16% T and 6% C; the gel buf-
fer concentration is increased to 1 mol/L Tris-chloride; the pH is low-
ered to 8.4; and tricine is used as terminating ion instead of glycine.
In this way the destacking of peptides and SDS is much more effi-
The M
r
distribution is very
similar to Laemmli gels.
Poduslo JF. Anal Biochem 114
(1981) 131–139.
Hashimoto F, Horigome T,
Kanbayashi M, Yoshida K,
Sugano H. Anal Biochem 129
(1983) 192–199.
Anderson BL, Berry RW, Telser
A. Anal Biochem 132 (1983)
365–275.
Schgger H, von Jagow G. Anal
Biochem 166 (1987) 368–379.

1 Electrophoretic Techniques32
cient than in the standard Tris-glycine system. This method yields lin-
ear resolution from 100 kDa to 1 kDa.
The Schgger system requires long running time because of the
high buffer concentration in the gel, which would otherwise become
overheated with high electric power. By adding 30% v/v ethylenegly-
col to the monomer solution, the buffer concentration can be reduced
to 0.7 mol/L (Westermeier, 2004). The resolution is still very good
and cracking of glass plates and smiling effects are avoided.
1.2.3
Blue Native Electrophoresis
Blue Native polyacrylamide electrophoresis has been developed by
Schgger and von Jagow (1991) for the separation of membrane pro-
teins and hydrophobic protein complexes in enzymatically active
form. Membrane proteins and complexes are extracted with the help
of a mild non-ionic detergent like Triton X-100, dodecyl-b-D-malto-
side, or digitonin. The gel buffer is composed of e-amino caproic acid
and Bis-Tris and titrated to the physiological pH 7.4. The anode buffer
is Bis-Tris titrated to pH 7.0 with HCl, the cathodal buffer tricine, Bis-
Tris and Coomassie blue dye. The anionic dye Coomassie brilliant
blue G-250 is added to the sample and the cathodal buffer. The dye
binds to the hydrophobic proteins and complexes without disturbing
protein–protein interactions and provides negative charges to them
independently of their original net charge. Thus under the mild con-
dition of pH 7.4 all sample components migrate into the anodal direc-
tion, and they are visible during the separation. In fact the dye–pro-
tein complexes are soluble in the detergent-free buffer medium.
Because all protein and complex surfaces are negatively charged, they
repel each other, and they will not aggregate. In the middle of the
run, the cathodal buffer is exchanged with a non-dye-containing buf-
fer, in order to achieve a clear background. The gels do not need to be
stained, because the Coomassie dye is still bound to hydrophobic pro-
teins and the complexes. Valuable hints for performing Blue native
PAGE including a trouble shooting guide can be found in a recently
published review by Wittig et al. (2006).
Porosity gradient gels from 4.5% T to 16% T are used, to allow large
super-complexes to enter and to prevent smaller molecules and com-
plexes to leave the gel on the end. Very sharp zones can be observed,
because the complexes migrate until the gel network gets too tight
for further migration. The working range is between 10 kDa and
10,000 kDa. A stacking gel with the same buffer, but with a slightly
lower concentration than the end of the gradient of 4% T is used for
optimum sample entry.
Westermeier R. Electrophoresis
in Practice. WILEY-VCH, Wein-
heim (2004) 242–245.
Schgger H, von Jagow G. Anal
Biochem 199 (1991) 223–231.
Wittig I, Braun H-P, Schgger
H. Nat Protocols 1 (2006)
419–428.
Blue Native electrophoresis is
often run in minigels or
medium gel format. In the
proteomics environment large
gel systems are preferred with
2520 cm gels in the second
dimension.
1.2 Polyacrylamide Gel Electrophoresis 33
In order to determine the complex partners the lanes are cut out,
these polyacrylamide strips are equilibrated in SDS buffer and placed
on top of a SDS gel. Thereby the complexes become dissociated into
the single proteins, which are separated in a second dimension.
Because in SDS electrophoresis the Coomassie dye will separate from
the proteins and migrate in the front, the gels need to be stained
afterwards, which is mostly performed with Coomassie blue (Gran-
vogl et al. 2006).
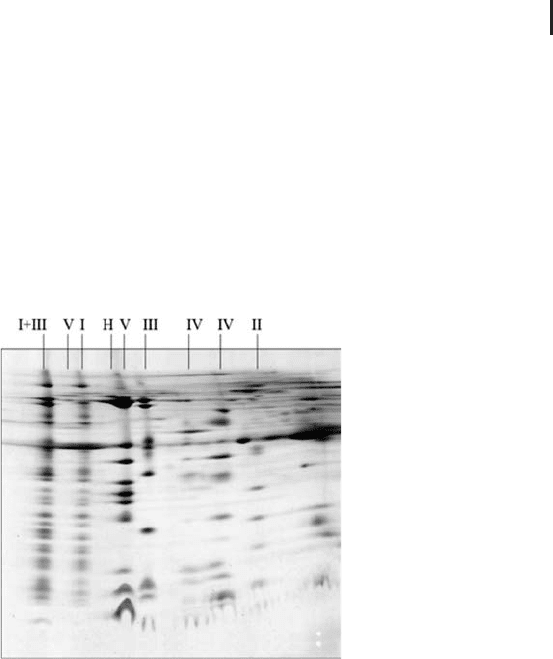
Figure 1.5 shows an example of two-dimensional electrophoresis
employing Blue Native electrophoresis in the first dimension (from
Eubel et al. 2003) and SDS electrophoresis in the second.
Fig. 1.5: Blue Native PAGE / SDS PAGE electrophoresis of mitochondrial
complexes of Arabidopsis thaliana (from Eubel et al. 2003). The complexes
were solubilized with digitonin and they are annotated with Roman numerals.
I+III is a super complex composed of complexes I and III. Coomassie brilliant
blue post-staining of the SDS gel.
The technique is applied in two major areas of proteomics:
.
Analysis of very hydrophobic proteins, like mem-
brane proteins.
.
Functional proteomics: analysis of protein com-
plexes and super-complexes.
A comprehensive review on applications of Blue native PAGE can be
found in the paper by Krause (2006).
For differential analysis of complexes it is very useful to apply the
DIGE concept. The complexes can easily be labeled with the CyDyes
after solubilization, prior to adding the Coomassie dye (Perales et al.
2005).
Granvogl B, Reisinger V,
Eichacker LA. Proteomics 6
(2006) 3681–3695.
Eubel H, Jnsch L Braun H.
Plant Physiol 133 (2003)
274–286.
Krause F. Electrophoresis 27
(2006) 2759–2781.
Perales M, Eubel H, Heine-
meyer J, Colaneri A, Zabaleta
E, Braun H-P. J Mol Biol 350
(2005) 263–277.

1 Electrophoretic Techniques34
When digitonin is used for extraction, even the big super-com-
plexes are kept together and intact during the separation. After cut-
ting out the lanes and equilibration in a less mild detergent, namely
dodecyl maltoside containing buffer, the single complexes can be
separated in a second-dimension Blue Native gel. In the resulting pat-
tern the composition on the super-complexes is displayed. With activ-
ity staining, a zymogram technique, its function can be assigned to
the particular spot. All this is nicely demonstrated in a paper by Sun-
derhaus et al. (2006). In this paper these complexes are also directly
visualized by single particle electron microscopy.
Three-dimensional electrophoresis The analysis of smaller subsets of
protein mixtures frequently provides improved pattern and allows to
detect proteins which otherwise do not enter the gel. A possible pro-
cedure is the three-dimensional electrophoresis procedure introduced
by Werhan and Braun (2002). The first dimension is the separation of
protein complexes by blue native polyacrylamide electrophoresis. The
visible bands are eluted electrophoretically from the gel, destained,
and further analyzed by standard denaturing 2-D electrophoresis:
IEF/SDS PAGE. Interestingly, this method reveals also proteins with
high hydrophobicity; most probably due to the considerable reduction
of complexity of the protein mixture.
Stegemann et al. (2005) have introduced a three-dimensional gel
block for performing blue native electrophoresis and SDS electro-
phoresis in one go.
1.2.4
Cationic Detergent Electrophoresis
Another possibility for getting poorly soluble proteins separated and
analyzed is the combination of two electrophoretic separations in pre-
sence of two different ionic detergents. Usually the extraction and
separation with a cationic detergent is selected as the first dimension,
because cationic detergents are less denaturing than SDS. The pro-
tein–detergent micelles migrate towards the cathode. The separation
pattern is different from that obtained with SDS electrophoresis. Pro-
teins are either solubilized with cetyltrimethylammonium bromide
(CTAB) or benzyldimethyl-n-hexadecylammonium chloride (16-BAC)
and applied on an acidic polyacrylamide gel containing the respective
detergent. A sodium or potassium phosphate buffer pH 2.1 with a
stacking gel pH 4.1 is used. It has been observed that membrane gly-
coproteins are separated much better in acidic CTAB electrophoresis
than in basic SDS electrophoresis (Buxbaum, 2003).
Sunderhaus S, Dudkina N,
Jnsch L, Klodmann J, Heine-
meyer J, Perales M, Zabaleta
E, Boekema E, Braun H-P.
J Biol Chem 281 (2006)
6482–6488.
This procedure can also be cate-
gorized into pre-fractionation
with Blue native electrophoresis
followed by 2-D electrophoresis.
Werhahn W, Braun H-P. Elec-
trophoresis 23 (2002) 640–
646.
Stegemann J, Ventzki R,
Schrodel A, de Marco A.
Proteomics 5 (2005)
2002–2009.
Buxbaum E. Cationic electro-
phoresis and electro transfer of
membrane glycoproteins. Anal
Biochem 314 (2003) 70–76.
1.3 Blotting 35
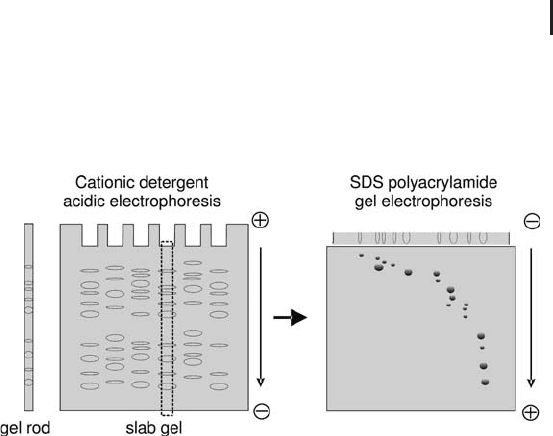
For 2-D electrophoresis the first dimension is either run in indivi-
dual gel rods in thin glass tubes or carried out in slab gels, which are
cut into strips for the second dimension (see Figure 1.6). After the
first dimension the strip is equilibrated in SDS sample buffer and
applied on the second-dimension gel (MacFarlane, 1989).
Fig. 1.6: Schematic representation of 2-D electrophoresis with
cation detergent / SDS PAGE. Here the first dimension is performed
in a vertical slab gel.
Hartinger et al. (1996) were the first to use the technique to success-
fully separate integral membrane proteins. Usually the patterns
achieved with this technique are not easy to compare. However, when
the samples are prelabeled with different CyDyes, mixed and applied
together on the gel, the same proteins of the different samples will
co-migrate, and the patterns can be analyzed with a spot co-detection
algorithm. It was shown by Helling et al. (2006) that very scarce sam-
ples can be analyzed in this way by pre-labeling the cysteines of the
proteins with saturation dyes. In this approach the first dimension
was performed with CTAB in narrow gel rods instead of slab gels.
1.3
Blotting
Blotting is the transfer of macromolecules on immobilizing mem-
branes for specific and sensitive detection. The electrophoretic trans-
fer of electrophoretically separated proteins onto a membrane with
subsequent immuno detection was introduced by Towbin et al.
(1979). This method is frequently called “Western blotting”. For pro-
tein blotting either nitrocellulose or PVDF is used as membrane
material. PVDF membranes have a higher binding capacity for pro-
teins than nitrocellulose, but nitrocellulose binds small proteins
better.
Large gel formats are preferred
for this technique, because the
spots are crowded along the
diagonal axis and require as
high resolution as possible.
MacFarlane DE. Anal Biochem
132 (1983) 231–235.
Hartinger J, Stenius K, Hoge-
mann D, Jahn R. Anal
Biochem 240 (1996) 126–133.
Helling S, Schmitt E, Joppich
C, Schulenborg T, Mllner S,
Felske-Mller S, Wiebringhaus
T, Becker G, Linsenmann G,
Sitek B, Lutter P, Meyer HE,
Marcus K. Proteomics 6 (2006)
4506–4513.
Towbin H, Staehlin T, Gordon
J. Proc Natl Acad Sci USA. 76
(1979) 4350–4354.
The term Western Blotting is
derived from the name of
Edwin Southern, who intro-
duced the first blotting tech-
nique: the transfer of DNA
onto nitrocellulose by capillary
forces for subsequent hybridiza-
tion (Southern EM. J Mol Biol.
98 (1975) 503–517).
1 Electrophoretic Techniques36
Usually SDS polyacrylamide electrophoresis is employed for the
separation, because all proteins have been solubilized and migrate in
the same direction, and the epitopes are easier accessible due to the
denaturing effect of SDS. Proteins are too big to elute quantitatively
from a SDS polyacrylamide gel by diffusion or capillary forces, there-
fore they are transferred electrophoretically.
1.3.1
Electrophoretic Transfer
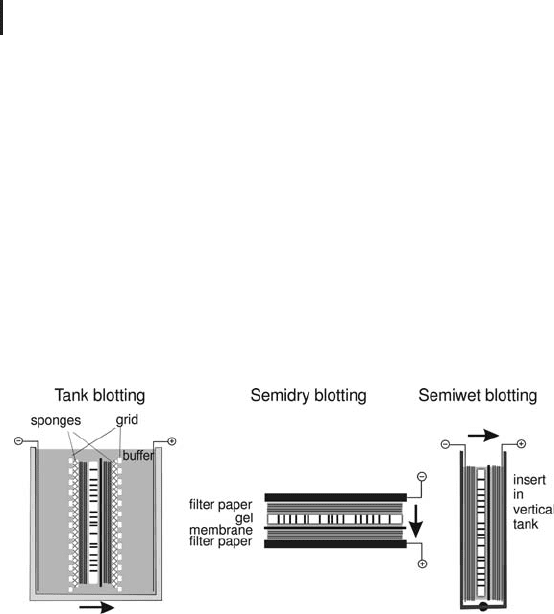
As shown in Figure 1.7 there are several ways to perform the electro-
phoretic transfer: tank blotting, semi-dry blotting and semi-wet blot-
ting. All three have in common that the gel and the membrane form
a sandwich with a stack of filter papers on both sides.
Fig. 1.7: Blotting transfer methods.
If isoelectric focusing is used for the separation of the proteins, it is
more efficient to transfer the proteins by diffusion with pressure blot-
ting (Towbin et al. 2001).
1.3.2
Protein Detection on the Membrane
After the transfer, which can take about an hour (semidry blotting) to
overnight (tank blotting), the free binding sites of the membrane is
blocked with a protein mixture, which will not interfere with the sub-
sequent probing with an antibody. After some washes this “primary
antibody” is detected with a secondary antibody, which recognizes
this particular antibody. This secondary antibody is conjugated with a
set of specific molecules, which can be easily detected with a subse-
quent development procedure with high sensitivity. The most sensi-
tive detection methods are using enhanced chemiluminescence
(ECL): the antibody–horseradish conjugate recognizes the primary
antibody; the substrate reaction is coupled with a secondary reaction
For blotting small or medium
sized gels are preferred, in this
way the volume of expensive
antibody solutions is kept to a
minimum.
Towbin H, zbey , Zingel O.
Electrophoresis. 22 (2001)
1887–1893.
More detection techniques are
described in “Electrophoresis in
Practice”.
1.3 Blotting 37
which causes chemiluminescent light emission for a certain time per-
iod. This light signal is accumulated by exposing the membrane on
an X-ray film, or by placing it into an absolutely dark cabinet where
the signal is recorded with a sensitive CCD camera. With a special
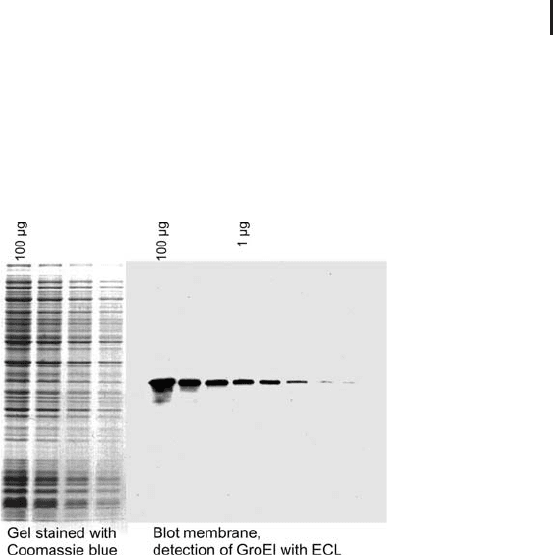
variant of ECL down to 1 pg of a protein band is detectable. Figure 1.8
shows an example for detection of a single protein with ECL versus a
Coomassie blue-stained gel showing all proteins.
Fig. 1.8: Comparison of a gel with total proteins Coomassie
blue-stained versus a blot membrane with one protein detected
with ECL. Note the high sensitivity of blotting detection.
The proteins on a blotting membrane can be reprobed for several
times after stripping the antibodies off and washing the membrane.
For this case it is recommended to use PVDF membranes or sup-
ported nitrocellulose.
Multiplex detection on blots As an alternative to enzyme–substrate
detection methods fluorescent dyes, like CyDye, can be conjugated to
the secondary antibody. In this case the fluorescent signal is directly
recorded with a fluorescent scanner. This procedure offers almost the
same sensitivity like enhanced chemiluminescence, and with differ-
ent fluorescence-conjugated secondary antibodies several different
antigens can be probed in one procedure without the need for strip-
ping antibodies off from the membrane. Besides multiplex detection
this allows also a relative quantification of changes of expression
levels of an antigen, when the second primary antibody is directed
against a protein with stable expression level.
Blotting from 2-D electrophoresis gels There are many papers pub-
lished where immunoblotting was successfully performed after 2-D
For this method low fluorescent
membranes must be used.
Blotting from 2-D electrophor-
esis gels had been the standard
for protein identification, until
mass spectrometry became so
sensitive that it detected too
many constituents of the blot-
ting membranes.

1 Electrophoretic Techniques38
electrophoresis. However, it is frequently observed that an antibody
does not recognize a protein anymore, when the sample had been
separated with 2-D electrophoresis; but the same protein can easily
be detected on a blot after 1-D SDS electrophoresis. It is still not
known, what causes this effect. There is a hypothesis that focusing a
protein at its isoelectric point and/or the presence of high molar urea
in the first dimension causes a different and stronger denaturation
for some proteins than SDS treatment alone.
Non-immunological detection procedures There are a number of
general protein staining techniques for blot membranes, which are
reversible or will not interfere with the subsequent specific detection
methods. The most practical are the fluorescent dyes, which are intro-
duced below in the context of staining of 2-D gels on page 119 ff.
Specific proteins can be detected with other ligands than antibo-
dies. For instance, glycoproteins are detected with high specificity
using lectins coupled to very sensitive avidin–biotin detection proce-
dures. This method is the most reliable specific detection of glycosy-
lated proteins, as the glyco-specific staining methods often produce
some wrong positive signals. For the detection of protein–protein
ligands there is a method called “Far Western Blotting”: The probing
protein is tagged with a labeled antibody for detection (Burgess et al.
2000).
In proteomics blotting is mostly employed for the verification of a
biomarker or drug target. When the new approach of antibody proteo-
mics has succeeded with delivering antibodies against each human
protein, blotting will gain increasing importance for the identification
of very low abundant proteins.
1.4
Isoelectric Focusing
In proteomics isoelectric focusing is mainly applied for the following
purposes:
.
First dimension in high-resolution 2-D electro-
phoresis of complex protein mixtures.
.
Pre-fractionation of complex protein mixtures
according to charge.
.
For the separation of very heterogeneous mix-
tures of tryptic peptides instead of strong cation
exchange chromatography in MDLC-MS.
Burgess R, Arthur TM, Pietz
BC. Methods Enzymol. 328
(2000) 141–157.
The application of blotting for
N-terminal sequencing or mass
spectrometry analysis has
almost been completely aban-
doned.
1.4 Isoelectric Focusing 39
1.4.1
Theoretical Background
Isoelectric focusing is performed in a pH gradient. Proteins are
amphoteric molecules with acidic and basic buffering groups. Those
become protonated or deprotonated depending on the pH environ-
ment. In basic environment the acidic groups become negatively
charged, in acidic environment the basic groups become positively
charged. The net charge of a protein is the sum of all negative or posi-
tive charges of the amino acid side chains. When net charges of pro-
teins are plotted over the pH scale, each protein has an individual net
charge curve. The intersection of the net charge curve with the x axis
is the isoelectric point – the pH value where the net charge is zero.
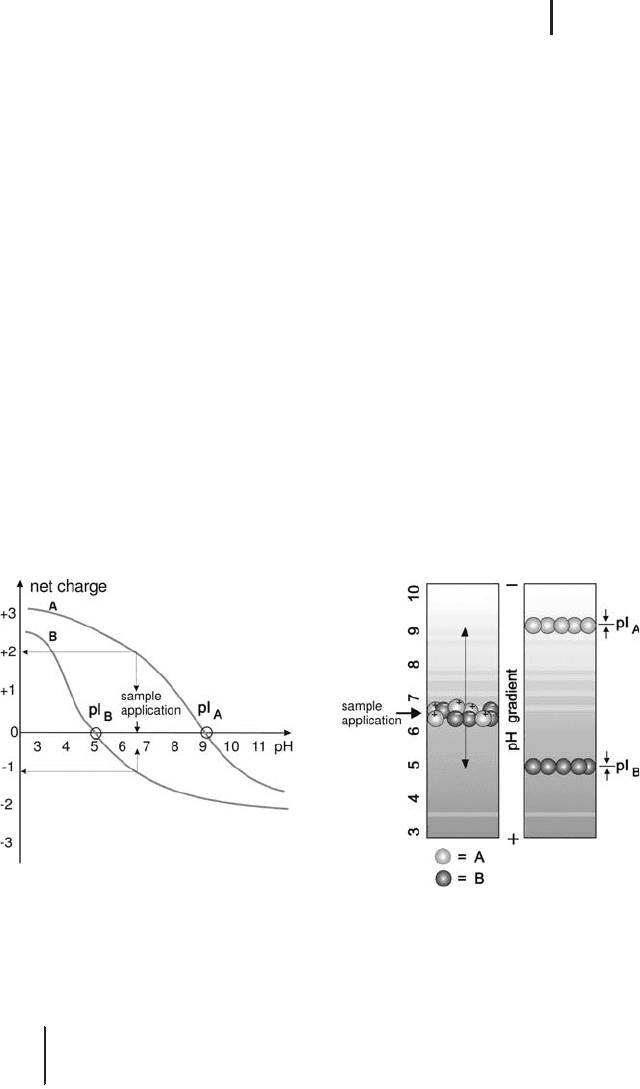
This is shown for two model proteins in Figure 1.9.
When a protein is applied on a certain pH value of the gradient,
and an electric field is applied, it will start to migrate towards the elec-
trode of the opposite sign of its charge. Because it migrates inside a
gradient, it will arrive at a pH value of its isoelectric point (see Figure
1.9) after some time. At its pI it has no net charge anymore and stops
migrating. Should it diffuse away above or below its pI it will become
charged again and migrate back to its pI. This is called the “focusing
effect”, which results in very high resolution.
Fig. 1.9: The principle of isoelectric focusing.
Left: Net charge curves of two model proteins A and B. At the point of
application, A will have two positive, B will have one negative charge(s).
Right: Migration of A and B to their pIs in the pH gradient of an isoelectric
focusing gel.
& In principle: Isoelectric focusing is a very high
resolution separation method, and the pI of a
protein can be measured.
