Westermeier R., Naven T., H?pker H.-R. Proteomics in Practice: A Guide to Successful Experimental Design
Подождите немного. Документ загружается.

1 Electrophoretic Techniques40
1.4.1.1 Titration Curve Analysis
The net charge curves are also called “titration curves”. The titration
curves of proteins can be displayed by a simple method (Rosengren
et al. 1976): A square gel – containing carrier ampholytes, but no sam-
ples – is submitted to an electric field until the carrier ampholytes
form a pH gradient from 3 to 10. Then the gel is rotated by 90
degrees, the sample proteins are pipetted into a groove in the gel
across the pH gradient. Now an electric field is applied perpendicular
to the pH gradient: the carrier ampholytes are uncharged at their pIs
and will not move. But, as shown in Figure 1.10, the proteins will
migrate towards the cathode or the anode according to their charge
sign and mobility, and will thus form titration curves (net charge
curves).
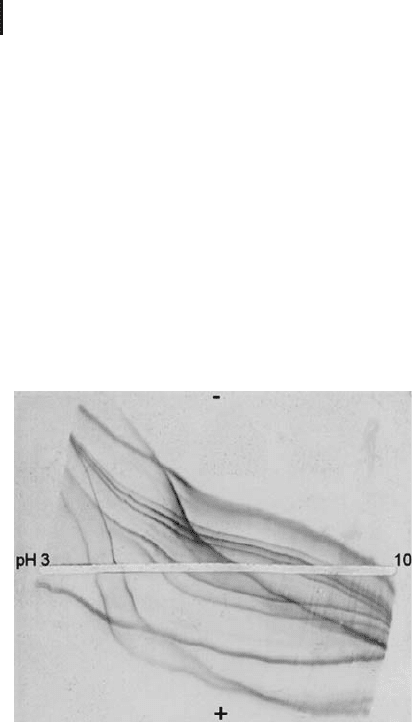
Fig. 1.10: Titration curves of a mixture of pI marker proteins
under native conditions. The steeper the curve, the faster will be
the migration of a protein in IEF. Note that some proteins have
a flatter curve above their pI.
The proteins possess very individually shaped curves also under dena-
turing conditions. Practical experience has shown that more proteins
have a steeper curve below their pI. This is the reason, why mostly
sample application close to the anode results in better pattern.
1.4.1.2 pH Gradients
In practice there are two ways to establish a pH gradient in a gel:
.
pH gradients which are formed in the electric
field by amphoteric buffers, the carrier ampho-
lytes;
Rosengren A, Bjellqvist B,
Gasparic V. In: Radola BJ,
Graesslin D. Ed. Electro-
focusing and isotachophoresis.
W. de Gruyter, Berlin (1977)
165–171.

1.4 Isoelectric Focusing 41
.
Immobilized pH gradients in which the buffer-
ing groups are part of the gel medium.
Carrier ampholytes generated pH gradients This was the first devel-
oped technique for isoelectric focusing. Svensson (1961) has designed
the theoretical basis for preparing “natural” pH gradients, while the
practical realization is the work of Vesterberg (1969): the synthesis of
a heterogeneous mixture of isomers of aliphatic oligoamino-oligocar-
boxylic acids. The result is a spectrum of low molecular weight
ampholytes with closely neighboring isoelectric points, and with high
buffering power at their pI.
The general chemical formula is the following:
–CH
2
–N–(CH
2
)
x
–N–CH
2
–
II
(CH
2
)
x
(CH
2
)
x
II
NR
2
COOH
R = H or –(CH
2
)
x
–COOH, x = 2 or 3
These carrier ampholytes possess the following properties:
.
High buffering capacity and solubility at the pI;
.
Good and regular electric conductivity at the pI;
.
Absence of biological effects;
.
Low molecular weight.
Naturally occurring ampholytes such as amino acids and peptides
have only low or no buffering capacity at their isoelectric point. They
can therefore not be used.
The pH gradient is established by the electric field. At the begin-
ning, the gel with carrier ampholytes has a uniform mean pH value.
Almost all the carrier ampholytes are charged: those with the higher
pI positively, those with the lower pI negatively.
When an electric field is applied, the negatively charged carrier
ampholytes migrate towards the anode, the positively charged ones to
the cathode, with velocities depending on their net charges. The car-
rier ampholytes align themselves in between the two electrodes
according to their pI and will determine the pH of their environment.
A (relatively) stable and gradually increasing pH gradient results; for
instance from 3 to 10. The carrier ampholytes lose a great part of their
charge, so the conductivity of the gel decreases substantially.
Because these are mixtures of buffers, the gradient profile will
never be perfectly linear. By adding narrow range gradient mixtures
to wide gradient mixtures the final shape of the gradient can be mod-
ified according to special resolution requirements.
Svensson H. Acta Chem Scand
15 (1961) 325–341.
Vesterberg, O. Acta Chem.
Scand 23 (1969) 2653–2666.
Alternative synthesis chemis-
tries have been developed later.
The chemical structures of
other amphoteric buffers are
different. Their function is the
same, but the properties of the
gradients can be different.
By controlling the synthesis and
the use of a suitable mixture
the composition can be moni-
tored for the preparation of
regular and linear gradient.
The anodal end becomes acidic
and the cathodal side basic.

1 Electrophoretic Techniques42
To maintain a gradient as stable as possible, electrode solutions are
applied between the gel and the electrodes, an acid at the anode and a
base at the cathode. Should, for example, an acidic carrier ampholyte
reach the anode, its basic buffering group would acquire a positive
charge from the medium and it would be attracted back by the elec-
tric field.
Isoelectric focusing in carrier ampholytes generated pH gradients
can be performed in various media:
.
In free liquid phase in capillaries, free flow elec-
trophoresis apparatus and rotating tubing;
.
In gels made from agarose with very low electro-
endosmosis;
.
In granulated dextrane gels (Sephadex
);
.
In polyacrylamide slab gels or individual gel
rods in glass or plastic tubes.
Carrier ampholytes as solvents for proteins
Carrier ampholytes have
another very important function: they help to solubilize proteins,
which stay in solution only in presence of buffering compounds.
Therefore they are also needed for the immobilized pH gradient tech-
nique.
Limitations of carrier ampholyte pH gradients
Isoelectric focusing is a
relatively slow separation method, because proteins have only very
low net charges when they come close to their isoelectric points, and
therefore low mobility. High spatial resolution requires long separa-
tion distance, which causes a long migration time under a high elec-
tric field strength.
Because the carrier ampholytes are in free solution, the gradient
will become instable during long IEF time: the gradient drifts. The
pattern becomes time-dependent and most of the basic proteins are
drifting out of the gel together with the basic part of the gradient.
Another problem: proteins of the sample behave like additional car-
rier ampholytes and modify the profile of the gradient. Thus the gra-
dient shape becomes also sample-dependent.
Immobilized pH gradients These problems are solved with the appli-
cation of immobilized pH gradients, generated by buffering acryla-
mide derivatives copolymerized with the gel matrix (Bjellqvist et al.
1982). The acrylamide derivatives containing the buffering groups are
called Immobilines
. An Immobiline is a weak acid or weak base
defined by its pK value.
Particularly under denaturing
conditions the matrix is highly
viscous leading to slow migra-
tion. Additionally, unfolded
polypeptides migrate slower in
the gel than native proteins.
Carrier ampholyte generated
gradients are also influenced by
the sample load and salt
contents in the sample.
Bjellqvist B, Ek K, Righetti PG,
Gianazza E, Gçrg A, Wester-
meier R, Postel W. J Biochem
Biophys Methods 6 (1982)
317-339.
1.4 Isoelectric Focusing 43
The general structure of an Immobiline is the following:
CH
2
=CH–C–N–R
II I
OH
R contains either a carboxylic or an amino group.
The acidic substances have dissociation constants in the range
from pK 0.8 to pK 4.6, the basic from pK 6.2 to pK 12. In order to
buffer at precise pH values, at least two different Immobilines are
necessary, an acid and a base. Figure 1.11 shows a diagram of a poly-
acrylamide gel with polymerized Immobilines, the pH value is
defined by the ratio of the Immobilines in the mixture.
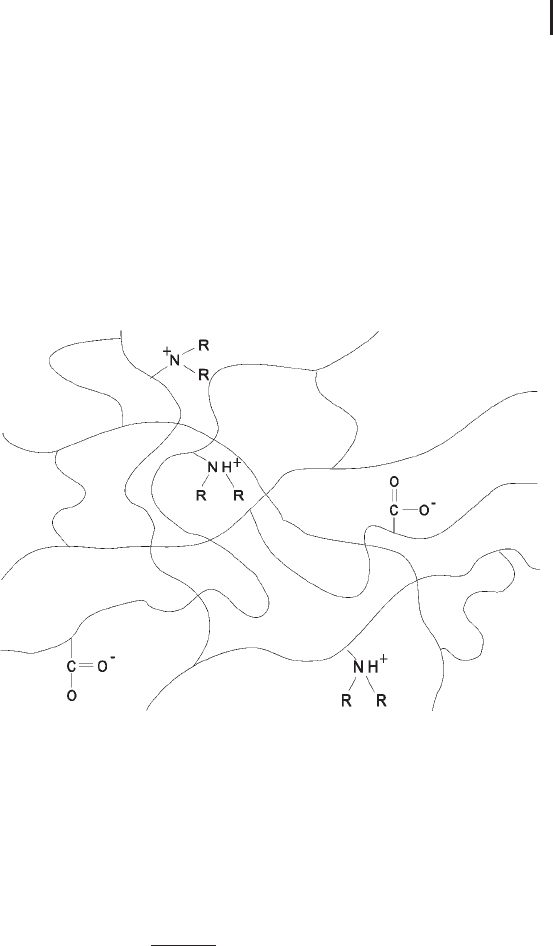
Fig. 1.11: Immobilized pH buffer. Schematic drawing of a
polyacrylamide network with co-polymerized buffering groups.
A pH gradient is obtained by continuously varying the ratio of
Immobilines. The principle is that of an acid/base titration, and the
pH value at each stage is defined by the Henderson–Hasselbalch
equation:
pH ¼ pK
B
þ log
C
B
C
A
C
A
when the buffering Immobiline is a base.
C
A
and C
B
are the molar concentrations of the acid and basic
Immobiline, respectively.
The wider the pH gradient, the
more different Immobiline
homologues are needed.
This concept generates an abso-
lutely continuous pH gradient.

1 Electrophoretic Techniques44
If the buffering Immobiline is an acid, the equation becomes:
pH ¼ pK
A
þ log
C
B
C
A
C
B
Celentano et al. (1986) and Altland (1990) developed PC programs
for the calculation and simulation of immobilized pH gradients for
the optimization of buffering power and ionic strength distribution.
Practice shows that a pre-calculated pH gradient recipe does not auto-
matically result in a usable IEF gel. Every recipe has to be tested and
optimized with practical experiments.
Further reading There are two books by P.G. Righetti which deal
exclusively with isoelectric focusing in theory and applications.
1.4.2
Preparation of IEF Gels
Carrier ampholyte gels
Casting of such homogeneous polyacryl-
amide gels is very simple: the monomer solution containing the car-
rier ampholytes is mixed with the catalysts and pipetted into a poly-
merization cassette, or into glass or plastic tubes. The carrier ampho-
lyte concentration is mostly 2% (w/v), the gel concentration 4% T and
3% C. To exclude oxygen the solution is carefully overlaid with dis-
tilled water.
The preparation of granulated gels is more intricate, because it is
very important to achieve the optimum liquid content (Westermeier,
2004).
Immobilized pH gradients Immobiline stock solutions with concen-
trations of 0.2 mol/L are used. Two solutions with acrylamide mono-
mers, crosslinker, and catalysts are required to prepare an immobi-
lized pH gradient: one contains the Immobiline cocktail for the acidic
end, the other the cocktail for the basic end. The gel forming mono-
mers are diluted to 4% T and 3% C. The acidic solution is made den-
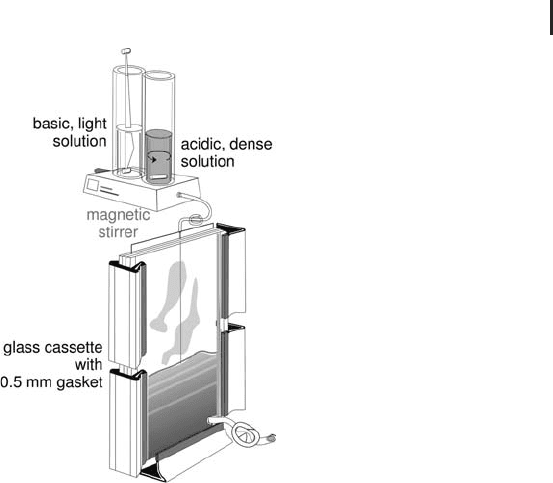
ser by adding glycerol. The gel is prepared by linear mixing of the two
different monomer solutions with a gradient maker (see Figure 1.12),
like for pore gradients. In principle a concentration gradient is
poured. 0.5 mm thick Immobiline gels, polymerized on a support
film have proved most convenient. During polymerization, the buffer-
ing carboxylic and amino groups covalently bind to the gel matrix.
Because of the low electric conductivity of immobilized pH gradients,
all contaminating compounds, like acrylamide monomers and the
catalysts, have to be removed by washing the gels several times with
deionized water.
Celentano F, Gianazza E, Dossi
G, Righetti PG. Chemometr
Intel Lab Systems 1 (1987)
349–358.
Altland K. Electrophoresis 11
(1990) 140–147.
Righetti PG. In: Work TS, Ed.
Burdon RH. Elsevier Biome-
dical Press, Amsterdam (1983).
Righetti PG. In: Burdon RH,
van Knippenberg PH. Ed. Else-
vier, Amsterdam (1990).
Westermeier R. In Cutler P. Ed.
Protein Purification Protocols.
Second edition. Methods in
Molecular Biology, Volume
244, Humana Press, Totowa,
NJ (2004) 225–232.
1.4 Isoelectric Focusing 45
Fig. 1.12: Preparing an immobilized pH gradient.
Pouring a gradient into a gel cassette.
1.4.3
Isoelectric Focusing in Proteomics
1.4.3.1 IEF Based on Carrier Ampholyte Gradients
Free flow IEF
The free flow electrophoresis apparatus described
above can also be used for isoelectric focusing. Either carrier ampho-
lytes or multi-components buffers, which are mixtures of amphoteric
and non-amphoteric buffers, are added. The latter mixture cannot
develop linear pH gradients, but they reduce the costs. With IEF a
higher number of fractions can be obtained than with the discontinu-
ous field electrophoresis.
Free flow electrophoresis and isoelectric focusing has successfully
been applied for fractionation and proteins in proteomics workflows
(Hoffmann et al. 2001; Obermaier et al. 2005). Lately it has also been
used for the separation of peptide mixtures as a replacement of
strong cation exchange chromatography (Xie et al. 2005). Free flow
IEF is a very reliable and reproducible technique, however good
operator skills and highly sophisticated instrumentation is required.
Liquid phase isoelectric focusing systems Several types of IEF sys-
tems in liquid phase are on the market, which employ membranes
for the separation of the fraction compartments. Temperature differ-
Hoffmann P, Ji H, Moritz RL,
Connolly LM, Frecklington DF,
Layton MJ, Eddes JS, Simpson
RJ. Proteomics 1 (2001) 807–
818.
Obermaier, C, Jankowski V,
Schmutzler C, Bauer J, Wild-
gruber R, Infanger M, Kohrle J,
Krause E, Weber G, Grimm D.
Electrophoresis 26 (2005)
2109–2116.
Xie H, Bandhakavi S, Griffin
TJ. Anal Chem 77 (2005)
3198–3207.

1 Electrophoretic Techniques46
ences and sticking of proteins are tried to be avoided either by con-
tinuously streaming fluids or rotation. The instrumental efforts are
less than with free flow apparatus, but there is always the danger that
hydrophobic proteins stick to membranes.
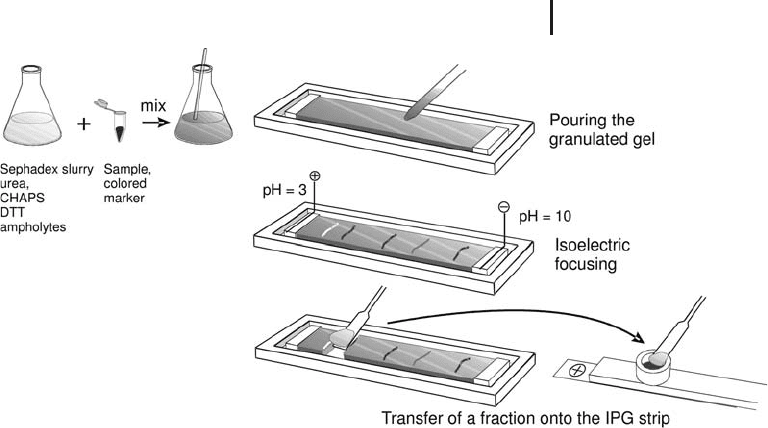
IEF in a granulated gel bed Gçrg et al. (2002) have developed a very
quick, reproducible and efficient pre-fractionation method: pre-
separation of complex protein mixtures by carrier ampholyte IEF in a
granulated gel bed. The procedure is performed on standard flatbed
electrophoresis equipment at an exactly adjusted temperature of
20 C. The technique is simple: the sample is mixed with low electro-
endosmosis Sephadex, which has been reswollen with rehydration
solution containing urea, CHAPS, DTT and carrier ampholytes. For
the visualization of pH value zones after IEF a mixture of non-protein
amphoteric dyes is added. The slurry is pipetted into a trough and
covered with a thin layer of silicone oil. After 4 hours the proteins are
focused at their isoelectric points. The pH gradient can be deter-
mined by interpolation between the colored marker zones. As shown
in Figure 1.13, the pH fractions are removed with a spatula and
directly applied onto rehydrated IPG strips. In the electric field the
proteins migrate out from the granulated gel into the polyacrylamide
gel of the IPG strip. The fractions can also be stored in a freezer at
–70 C.
This technique has several advantages:
.
Standard laboratory equipment is used.
.
The technique is easy to carry out.
.
The separation is quick.
.
Denaturing sample pre-fractionation according
to pI in presence of urea, detergents, etc.
.
No protein losses.
.
It can be done with high and low protein
amounts
.
It is also desalting the sample – no microdialysis
is required.
Subsequent separations of the pH fractions in the respective narrow
pH interval IPG strips show a considerable increase in spot number.
Many more of the lower expressed proteins can be detected in this
way.
Gçrg A, Boguth G, Kçpf A, Reil
G, Parlar H, Weiss W. Proteo-
mics 2 (2002) 1652–1657.
1.4 Isoelectric Focusing 47
Fig. 1.13: Pre-fractionation of a complex protein mixture with
isoelectric focusing in Sephadex.
Isoelectric focusing in gel rods for 2-D electrophoresis In the original
high resolution 2-D electrophoresis method the first dimension is car-
ried out in thin gel rods in glass or plastic tubes. The history of this
technique goes back to a paper in German language by Stegemann
(1970), combining isoelectric focusing (IEF) and SDS polyacrylamide
gel electrophoresis. The resolution of 2-D electrophoresis was consid-
erably increased by the introduction of denaturing conditions during
sample preparation and isoelectric focusing by O’Farrell in the year
1975. With this modification the method gained a wide acceptance.
The gel rods contain urea, detergent, reductant and carrier ampho-
lytes to form the pH gradient in the electric field. Usually the sample
is loaded onto the cathodal side of the gel rod, which becomes the
basic end of the gradient in the electric field. This “O’Farrell” techni-
que has been used for about two decades – and is still used by some
laboratories – without major modifications. The potential of the
method for a systematic approach to create a protein database had
been recognized very soon. Anderson and Anderson (1978) have
designed instruments and operation procedures to prepare and run
multiple 2-D gels under the most reproducible conditions, in order to
develop a “Human protein index” (Anderson and Anderson, 1982).
Gels with about 2020 cm have become standard for an adequate
spatial resolution. If one assumes that up to 100 bands can be
resolved in a 20 cm long one-dimensional gel, a theoretical separation
space of 10,000 proteins could be reached in such a gel. In practice
several thousands of proteins can be detected.
Stegemann H. Angew Chem
82 (1970) 640.
O’Farrell PH. J Biol Chem 250
(1975) 4007–4021.
Anderson NG, Anderson NL.
Anal Biochem 85 (1978)
331–354.
Anderson NG, Anderson NL.
Clin Chem 28 (1982)
739–748.

1 Electrophoretic Techniques48
Due to the high concentration of urea the viscosity in the gel is very
high. The denatured proteins are unfolded and thus highly retarded
by the gel matrix. Therefore long migration times are needed, which
lead to a destabilization of the gradient. The soft, thin, and long gel
rods demand high experimental skill. Between pH 6.7 and 7 there is
very often a lack of buffering power, leading to mechanical instability
of the thin gel rods, and an empty area in the spot pattern. The hand-
ling of the technique was rather cumbersome; in many cases the pat-
terns were not reproducible enough. Besides the influence of the
sample composition also batch to batch variations of the carrier
ampholyte mixtures were the reasons for differences in the profiles of
the pH gradients. Gradient drift with prolonged isoelectric focusing
time lead to losses of almost all basic and some of the acidic proteins.
A remedy had soon been found for the display of basic proteins:
O’Farrell et al. (1977) have introduced a modification of the first
dimension run: NEPHGE (non-equilibrium pH gradient electrophor-
esis). Here the sample is loaded onto the acidic end of the gel and the
proteins are separated while the gradient drifts towards the cathode.
The run is stopped after a defined time period, which is approxi-
mately one third of the regular focusing time. However, due to the
time factor it is hard to achieve a good reproducibility. The proteins
are not focused like in IEF; the resolution is limited by the number of
different carrier ampholyte homologues.
Meanwhile most laboratories prefer to use the technique of IEF in
individual IPG strips according to Gçrg et al. (2000), which will below
be described in detail. In these gels all proteins are strongly focused
at their isoelectric points.
There are indications that it is not always an advantage, when pro-
teins are tightly focused at their isoelectric points. High molecular
weight and hydrophobic proteins aggregate with themselves when
they are too tightly focused, and cannot migrate out from the first
dimension gel anymore. This can explain, why sometimes proteins
are missing in an IPG strip 2-D gel, but they are present after a 2-D
electrophoresis run with the traditional NEPHGE procedure.
Some laboratories employ isoelectric focusing in gel rods and
immobilized pH gradient for 2-D electrophoresis side by side as com-
plementary methods.
1.4.3.2 IEF Based on Immobilized pH Gradients
Immobilized pH gradient strips
Generally it is very useful to run dif-
ferent samples in completely separated individual gels. Immobilized
pH gradient gels are the only electrophoresis slab gels, which do not
show any edge effects during the run, when they are cut into strips.
The gels are cut after they have been dried down on the film. Instruc-
Because of the technical limita-
tions, mainly acidic proteins
could be studied in the past.
2-D electrophoresis had some-
times been seen as a technique,
which produces operator-
dependent results.
O’Farrell PZ, Goodman HM,
O’Farrell PH. Cell 12 (1977)
1133–1142.
The proteins are not focused,
but stacked between the
different carrier ampho lyte
homologues.
Gçrg A, Obermaier C, Boguth
G, Harder A, Scheibe B, Wild-
gruber R, Weiss W. Electrophor-
esis 21 (2000) 1037–1053.
This observation has been
made particularly with
membrane proteins.
Gçrg A, Weiss W. In Rabilloud
T, Ed. Proteome research: Two-
dimensional gel electrophoresis
and identification methods.
Springer, Berlin Heidelberg
New York (2000) 107–126.

1.4 Isoelectric Focusing 49
tions and pH gradient recipes for preparing immobilized pH gradient
strips in the laboratory can be found in several book chapters: Gçrg
and Weiss (2000 and 2005) and Westermeier (2004). However, this is
a multistep procedure, which needs some expertise and skill.
Much more reproducible results are obtained, when ready-made
IPG strips are used. Commercially produced strips are prepared
according to GMP industry standards, and they are quality controlled.
A wide choice of different gradients and strip lengths is available on
the market. Additional gradients are being developed for achieving
higher resolution.
Gel sizes The commercial strips are 3 mm wide and have to be
reconstituted to the original thickness of 0.5 mm with rehydration
solution prior to IEF. Thicker and wider strips would have a higher
protein loading capacity. This would, however, not be an advantage,
because the SDS polyacrylamide gel of the second dimension would
show overloading effects of proteins and a high detergent back-
ground. In theory this could be compensated by using SDS gels
thicker than the conventional 1 to 1.5 mm. However, staining of such
thick gel slabs would be very time consuming and not allow a high-
throughput approaches.
Strips are available in several different lengths from 7 cm to 24 cm.
In proteomics usually 18 and 24 cm strips are used, because the high-
est possible resolution is required for proteome analysis. Miniformat
strips with 7 cm length are ideal for optimization of the sample pre-
paration method.
When spatial resolution does not have the highest priority, like for
protein identification with western blotting, small gel formats from 7
to 13 cm are sufficient.
pH gradient types One of the advantages of immobilized pH gradi-
ents is the possibility to reproducibly produce linear gradients from
pH 3 to pH 10. However, there are samples with an uneven distribu-
tion of the proteins over the pH gradient from 3 to 10. The non-linear
gradient pH 3–10 is flat in the acidic range to accommodate more dif-
ferent and higher concentrations of acidic proteins. So far the widest
gradient was designed and applied by Gçrg et al. (1999): pH 3–12. For
increased resolution and higher protein loads acidic (pH 4–7, pH
3–7) and basic (pH 6–11, pH 6–9, pH 7–11) gradients are employed.
Narrow intervals with only one or two pH units allow very high pro-
tein loads and excellent spatial resolution. The narrowest intervals, so
called “ultra zoom gels” with 0.5 pH units, have been applied to 2-D
electrophoresis by Hoving et al. (2000).
Gçrg A, Weiss W. In: Cell
Biology. A Laboratory Manual,
Vol. IV, Ch. 23, 3rd edition. (JE
Celis, Ed.), Elsevier Science &
Academic Press (2005) pp.
175–188.
Westermeier R. Electrophoresis
in Practice. WILEY-VCH, Wein-
heim (2004).
It is very annoying, when the
valuable sample is not sepa-
rated well, just because of a
little mistake occurring in the
laboratory.
With miniformat gels results
are obtained after a few hours.
Gçrg A, Obermaier C, Boguth
G, Weiss W. Electrophoresis 20
(1999) 712–717.
Hoving S, Voshol H, van
Oostrum J. Electrophoresis 21
(2000) 2617–2621.
