Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

quarter-sawn straight staves are planed interiorly and
exteriorly throughout their thickness to achieve a stave of
cylindrical width. After the wooden material has been air-
and/or kiln-dried to approximately 12% moisture content,
the staves and heads are assembled into steel-hoop-bound
barrels. Assembly operations include: setting up staves;
steaming and winching staves to achieve the belly, bilge,
or circumference arch; heating to make wood pliable and
give one last drying after being steam bent; tapping out for
uniform thickness; trussing to tighten stave joints; crozing
interior grooves in each end of the staves where the heads
will be inserted; heading up by inserting heads in the
croze at each end of the staves; hooping up by driving
riveted-steel hoops onto exterior of staves; boring for
testing, lining, and future filling; bunging up the bored
hole; and rolling the barrel out to the marketplace.
Dozens of species of timber from all over the world have
been used to make tight (for liquid) and slack (for non-
liquid) cooperage. Hardwood barrels for spirits and wine
include oak timber from Limousin and Nevers in France;
Alastian and Italian oak; and fork-leafed American white
oak, found principally in the slow-growing forest regions
of the United States of Missouri, Indiana, Tennessee,
Kentucky, and Arkansas. Virtually all of the wooden
barrels made in the United States today [1–2 million
(10
6
)/yr] are 50-gal (189-L)-capacity barrels used by the
bourbon whisky trade. Barrels for bourbon are charred
interiorly about 1/16 in. (1.6 mm) of their 1 in. (25.4 mm)
thickness to bring out the tannin in the wood. Tannin aids
in the coloring and flavoring of spirits and wine.
Wooden barrels have had numerous names, depending
on their size and use. A small sampling of these names
include: pickled-pig’s-feet kit; fish pail; one-quarter, one-
half, and full-beer ponies; hogshead; salmon tierce; tallow
cask; rum puncheon; and port wine pipe. A list of common
international cask sizes is presented in Table 1 (2).
Just as the wooden barrel replaced the crude basketry
used centuries ago, many other types of container have
replaced the wooden barrel: steel and fiber drums, plastic
pails, aluminum and steel cans, fiberglass and cement
tanks, and so on; aluminum and stainless steel replaced
wood for beer barrels (3).
To date, no industrial engineer has come up with a
blueprint to replace the strength of a wooden barrel. In
tests involving high stacking, they can perform better
than steel drums.
BIBLIOGRAPHY
F. J. Sweeney, ‘‘Barrels’’ in The Wiley Encyclopedia of Packaging
Technology, 1st edition, John Wiley & Sons, New York, 1986,
pp. 47–48.
Cited Publications
1. Code of Federal Regulations, Title 49, Sect. 178.155–178.161.
2. F. A. Paine, The Packaging Media, Blackie, Glasgow and
London; John Wiley & Sons, New York, 1977.
3. H. M. Broderick, Beer Packaging, Master Brewers Association
of the Americas, Madison, WI, 1982.
BARRIER AND OVERPRINT COATINGS
MICHAEL W. LEONARD
Sun Chemical—Functional
Coating Research, Kent, United
Kingdom
INTRODUCTION
Barrier coatings provide barriers to certain gases, liquids,
radiation, or other substances. These may include oxygen,
water vapor, aroma, light (including ultraviolet), and, in
the case of food packaging applications, products such as
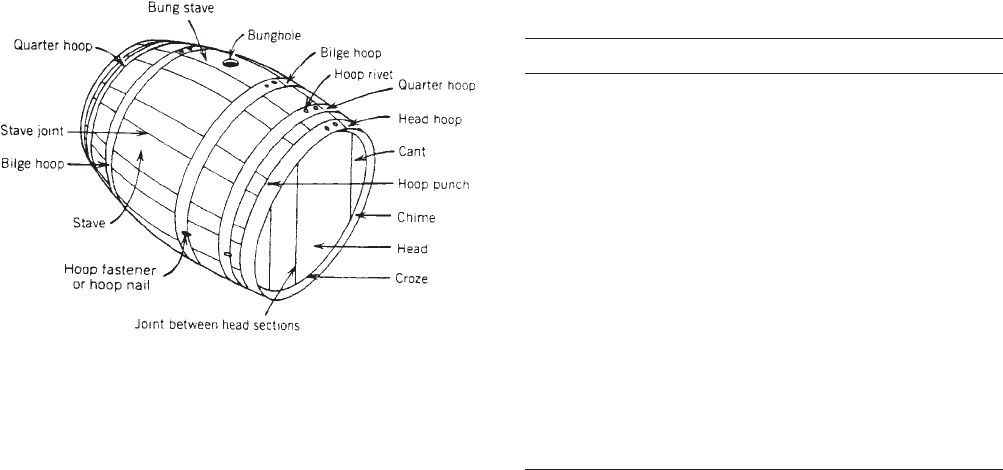
Figure 1. The wooden barrel.
Table 1. Wooden Casks for International Shipments of
Alcoholic Beverages
Beverage Name Gallons
a
Liters
a
Wine
Sherry Butt 137.5/140 500
Hogshead 67.5/69 250
Port Pipe 145/147.5 53
Hogshead 72.5/74 265
Vermouth Hogshead 67.5/70 250
Burgundy Hogshead 57.5/60 215
Spirits
Rum Puncheon 137.5/144 520
Hogshead 67.5/72.5 255
Barrel 50 180
Brandy Hogshead 77.5/79 280
Quarter case 39/40 140
Beer Butt 135 500
Hogshead 67.5 245
Barrel 45 165
Kilderkin 22.5 82
Firkin 11 41
Pin 6 20
a
Gallon (U.S.) and liter sizes are not equivalent.
98 BARRIER AND OVERPRINT COATINGS

oils and fats, and so on. The barrier is usually intended to
prevent both penetration and loss from the package,
which could otherwise cause advanced spoilage of the
packaged product. There is an ongoing industry require-
ment to improve barrier properties in order to extend shelf
life of packaged foods. This section deals only with barrier
coatings, applied to flexible plastic packaging films, for
the purpose of minimizing oxygen and water vapor trans-
mission. In describing the various coating technologies,
comparisons will be drawn with alternative methods of
achieving these barriers, in order to provide a balanced
view of this important property in relation to food
packaging.
The ultimate packaging barriers to oxygen and moist-
ure barrier are tin and glass closely followed by aluminum
foils. These materials provide zero gas and vapor trans-
mission and, in the case of glass, also visibility of the
product, which is often an important point of sale market-
ing consideration. While these materials have served the
food packaging industry very well for many years and are
likely to continue doing so, they have certain drawbacks
which leave the door open for new advances in barrier
technologies. Glass and tin, while infinitely recyclable, are
heavy and therefore expensive to transport, resulting in
less environmentally friendly greenhouse gas (CO
2
) emis-
sion compared to transportation of lightweight plastic
packaging. Plastic packaging laminates including alumi-
num foils, while lighter than tin and glass, render the
packaging difficult (if not impossible) to recycle, resulting
in the need to dispose of it in landfill or by incineration,
both of which are negative options in a world where
sustainability is of increasing importance.
The gradual replacement of tin, glass, and more re-
cently aluminum foils by plastic packaging materials has
been evolving for several decades. The improvements to
polymer film technologies, coupled with inorganic and
organic surface treatments and coatings, continue to close
the gap with tin and glass in terms of barrier performance,
while ensuring that due attention is paid to the environ-
mental impact.
BACKGROUND
There are limited choices for the packaging technologist
who seeks the ultimate gas barrier in single or even
multilayer plastic package structures. This becomes even
more limited when there is a requirement for product
visibility, reducing the options to structures that are
aluminum foil or (Al) metallized film free. The common
polymeric packaging films based upon polyethylene ter-
ephthalate (PET), oriented polypropylene (OPP), cellulose
in many coated guises (e.g., MXXT/W, MS, etc.), and
biaxially oriented nylon (BON/OPA) do not have sufficient
oxygen or moisture vapor barrier properties for the many
food packaging specifications that call for longer shelf
life of foods that spoil if exposed to atmospheres depleted
or rich in these gases. It is usual in many packaging
situations to replace the atmosphere inside a package
with an inert gas substitute. This is known as modified
atmosphere packaging (MAP) or sometimes protective
atmosphere packaging (PAP). Clearly, it is important
that the substitute gas is retained for as long as possible.
Typical MAP gases include nitrogen, oxygen, and carbon
dioxide which as a rule of thumb permeate through
polymeric films in the ratio 1:4:10. Since oxygen is the
most reactive of these three, with respect to food degrada-
tion, it is seen as the most important, while combinations
of nitrogen and carbon dioxide may be selected for reasons
of control of microbial growth, ripening, or cost.
Permeability Data of Several Packaging Films
Low-Density PE
PET
BON
Nitrogen
0.7
0.0108
0.00713
Oxygen
2.2
0.0444
0.0285
Carbon
Dioxide
9.5
0.227
0.066
Ratio
1:3.1:13.5
1:4:21
1:4:9
The manufacturers of such polymeric films have over
the past 60 years developed coatings for their products
which, to varying degrees, improve the gas barrier proper-
ties as well as other specific performance properties such
as heat-seal threshold, product resistance, gloss, and
clarity. Such coatings include organic solvent and aqueous
applied polyvinylidene chloride (PVdC), aqueous or extru-
sion-coated ethylene vinyl alcohol (EVOH), inorganic and
organic sol–gels, and in a few instances aqueous polyvinyl
alcohol (PVOH). Each of these technologies has certain
compromises in performance. Certain polymeric films
have an intrinsic barrier to moisture or oxygen, but rarely
both. Cellulose, being a reasonable barrier to oxygen but
poor moisture barrier, is one example.
Alternatives to aqueous or solvent-based coating appli-
cations have been available for some time now. These
essentially inorganic layers are deposited either as vapor
in a vacuum or by various electron-sputtering or plasma-
enhanced chemical vapor deposition (PECVD) processes.
The most common vacuum-deposited barrier coating is
aluminum, but more recent developments include silicon
oxide (SiOx) and aluminum oxide (AlOx) where the
proportion of oxygen is nonstoichiometric. These coatings
have one distinct advantage of being excellent barriers to
both oxygen and water vapor. The oxide coatings are also
completely transparent and most commonly used on PET
and BON. Although improvements to the gas barrier of
OPP are also achieved, this is less common due to the
expensive nature of the process resulting in mediocre
barrier performance. Since the important substrates are
not heat-sealable, they are invariably used in multilayer
structures, which also serve to protect the oxide coatings
which are particularly susceptible to damage through
physical abuse, which may result in a loss of barrier
properties. Some oxide-coated films are also in-line coated,
with sol–gel technologies, to enhance both barrier and
abuse resistance properties. Post application of protective
coatings is rare due to the risk of damage to the barrier
before coating.
Very recently, there has been a resurgence in the
interest shown by converters to apply barrier coatings
in-line with the printing process for high-barrier
BARRIER AND OVERPRINT COATINGS 99
packaging laminates. New organic polymer technologies
combined with nano-particulate materials applied from
aqueous or solvent-based systems that may not require
extended drying conditions are at an early stage of
commercialization and demonstrating certain advantages
over existing technologies.
BARRIER COATING TECHNOLOGIES
The gas permeation process for any packaging film or
coating is specific to the chemical structure and morphol-
ogy of that material (1). The transmission of gases across a
barrier usually involves several processes. After colliding
with the barrier surface, the gas molecule must be ad-
sorbed and subsequently dissolved in the voids of the
barrier polymer matrix. Diffusion of the gas molecules
then takes over and is governed by the kinetic energy of
the molecules directly influenced by temperature. Diffu-
sion of the gas generally proceeds through a barrier layer
from a more to a less concentrated atmosphere or partial
pressure. In all cases, permeation is controlled by the
solution and diffusion steps, according to Henry’s Law of
Solubility and Fick’s Law of Diffusion, respectively. This is
why metals and the previously mentioned metal oxides
exhibit such high barrier properties in exceptionally thin
films, to both moisture and oxygen. Crystallinity in or-
ganic polymer barriers plays a less important role,
although in coatings based upon PVdC copolymers, where
the drying temperature is critical to imparting crystal-
linity, variation in oxygen and water vapor transmission
has been reported previously by the author (2). An in-
depth consideration of the theories governing gas trans-
mission are dealt with elsewhere in this publication.
PVDC CO POLYMER COATINGS
PVdC was developed by Ralph Wiley and co-workers at the
Dow Chemical Company during the period 1932–1939 (3).
It was initially commercialized under the trade name of
‘‘Saran’’ in 1939. PVdC is still used today by both packaging
film manufacturers and converters. Although it can be
applied from solution, it is far more popular to use the
aqueous dispersion form which enables much higher solids
application at the required viscosities. One reason for the
longevity of this technology is its undoubtedly unique
properties that distinguish it from all other polymer dis-
persions, even today. It is fair to say that PVdC coatings are
still the only organic polymers capable of providing both
moisture and oxygen barrier. The fact that they also
provide heat-sealability, high gloss, transparency, and flex-
ibility are further reasons for its continued use. However,
its use has gradually declined in various regions of the
world as increased importance is placed on environmental
factors. Chlorinated polymers are now excluded in many
packaging structures due to the problems related to incin-
eration and the difficulties associated with handling the
toxic by products including dioxin. Furthermore, as higher
barrier requirements have been introduced, partly driven
by the need for longer shelf life, the need for alternative
polymer technologies and inorganic barrier solutions has
resulted in superior barrier technologies, albeit with their
own set of compromises.
POLY VINYL ALCOHOL AND ETHYLENE VINYL
ALCOHOL-BASED COATINGS
Ethylene vinyl alcohol (EVOH) copolymers are hydrolyzed
copolymers of ethylene and vinyl alcohol. Polyvinyl alcohol
(PVOH), made by the hydrolysis of polyvinyl acetate
(PVAc), provides increasing oxygen barrier with the per-
centage conversion of PVAc, but this is accompanied by
increasing water sensitivity. Consequently, the oxygen
barrier of PVOH, while superior to polymers such as
PVdC at low relative humidity, is drastically compromised
under most ambient conditions that packaging must
resist. Occasionally, PVOH coatings are used where the
coating is protected by an efficient moisture vapor barrier
such as that conferred by polyethylene or polypropylene
films in multi-ply laminates. However, it is not suited,
even in these structures, for high oxygen barrier specifica-
tions where relative humidity is above 50% or the proces-
sing of the packaged product involves pasteurization or
autoclave steam cooking.
EVOH copolymers became commercially available in
1972 in Japan, although their use was very limited until
some 10 years later. By copolymerizing vinyl alcohol with
ethylene, the hydrophobic properties of the latter improve
the resistance to humidity, while preserving much of the
oxygen barrier. Copolymers with the highest oxygen bar-
rier contain in the region of 27–32% ethylene and provide
an environmentally acceptable alternative to PVdC under
similar conditions of use. Again, the oxygen barrier prop-
erties are still sensitive to increasing levels of humidity,
which limits this barrier in certain (e.g., tropical) specifi-
cations. The solubility of EVOH is troublesome; although
some lower ethylene content copolymers are soluble in
alcohol/water solutions, they have a tendency to gel
upon storage, making them less than convenient to use.
In fact, storage of the solutions at elevated temperatures
is the only way to avoid gelation. Therefore, the use of
EVOH in barrier packaging tends to be more commonly
achieved by extrusion coating which is an expensive
converting exercise.
INORGANIC COATINGS
Aluminum-metallized substrates such as PET, OPP, and
cellulose are manufactured by the vacuum metallization
process. These films have been available for around 30
years and provide high levels of barrier to oxygen and
moisture vapor and have undergone an evolutionary
process that has seen improvements to surface stability,
film adhesion, and printing ink adhesion. The presence of
aluminum (foil or metallized film) in food packaging is
gradually being discriminated as environmentally unac-
ceptable and will ultimately decline. The use of other
vacuum metal oxide deposition processes such as
plasma-enhanced chemical vapor deposition (PECVD) to
100 BARRIER AND OVERPRINT COATINGS
lay down ultrathin layers of aluminium oxide or silicon
oxide is also well-advanced, producing transparent films
with equivalent barrier to the more common aluminum
metallization or in some cases even better. The major
disadvantage with these ceramic-type layers is their
brittleness, which makes them prone to damage either
during the converting process or in the final laminate
structure during the packaging operation.
NEW BARRIER COATINGS TECHNOLOGY
Over the last five years, there has been considerable
worldwide investment in nanotechnology. Some of this
investment has been focused on the development of new
barrier coating technology. In an effort to take cost out of
the process whilst achieving the highest possible barrier
properties combined with sustainability (low carbon foot-
print, compostability or recyclability) coatings applied via
the conventional coating processes (gravure, reverse roll,
flexo, blade coat, etc) from water or organic solvent
are ready for commercialization. These coatings exhibit
a balance of properties which makes them suitable for
both film manufacturer and converter applied applica-
tions. Their performance in packaging will now be dis-
cussed with reference to the graphs referenced.
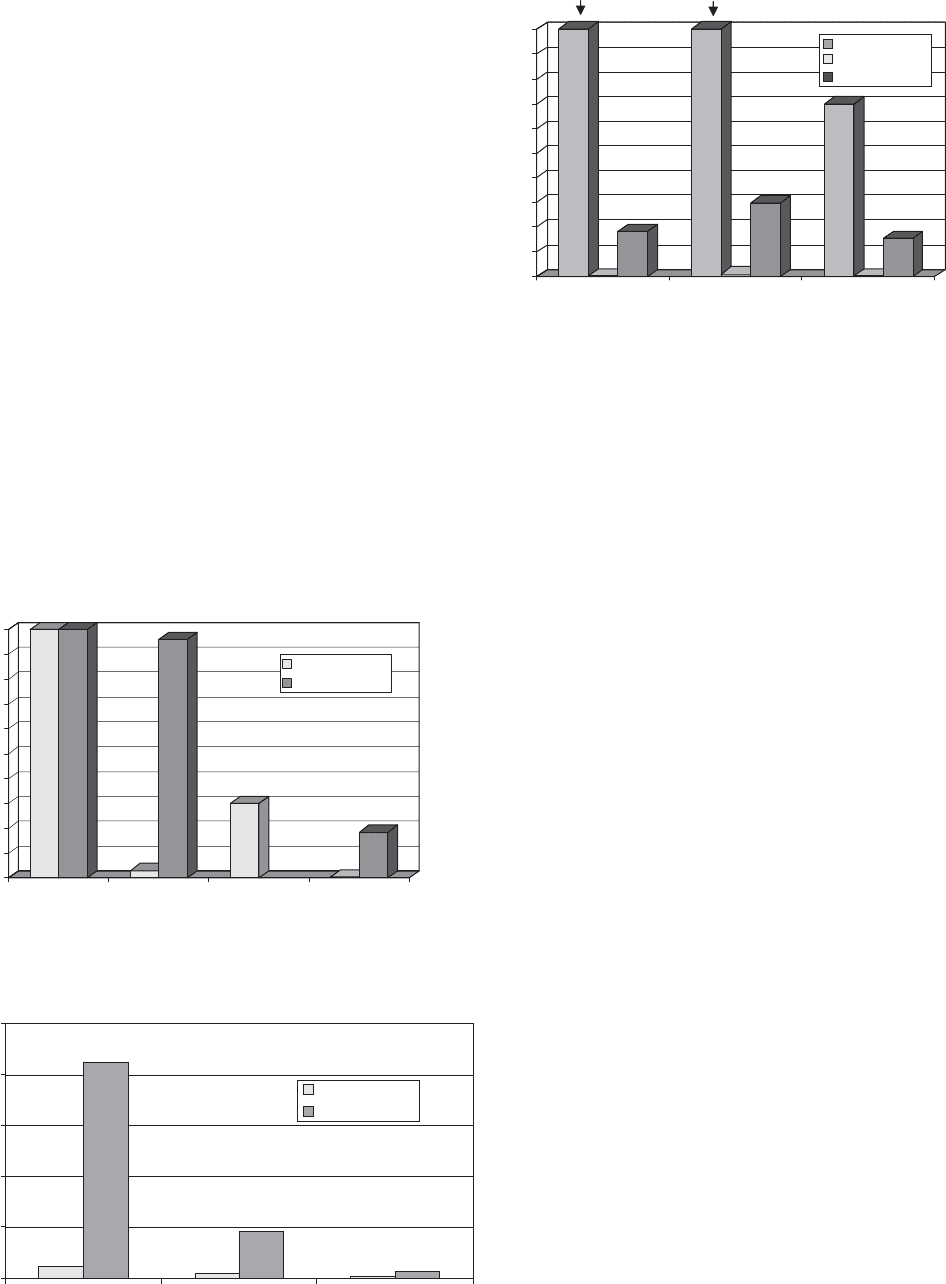
Oxygen Barrier (see Figure 1). Compared to other well-
known organic barrier polymers, this new generation of
barrier coatings offers a superior oxygen barrier that is
less susceptible to high levels of relative humidity. Com-
pared to inorganic barriers the oxygen barrier is very
similar, but inorganics still offer slightly better stability to
high relative humidity. Owing to the combination of inert
particle technologies and ‘‘green’’ polymer selection, the
new-generation coatings are generally more sustainable
and provide better abuse resistance as is demonstrated in
Figure 2. Indeed the latest coating technology can be used
to reinforce the performance of aluminium-metallized and
oxide-coated films, achieving barriers of less than 0.1 cm
3
/
m
2
/day as well as improving the flex resistance of these
substrates. In Figure 3 it can be seen that significant
improvements to the oxygen barrier of common packaging
substrates is possible without the need for complex appli-
cation/vapor deposition processes, opening up the poten-
tial for conversion of high barrier packaging at various
points along the value chain (e.g., film manufacturer or
converter). In many respects these coatings may be seen
as a means to produce cheaper laminate high-barrier
structures (e.g., using barrier-coated OPP) with equiva-
lent barriers to those currently used (e.g., PET).
Compostable and sustainable substrates such as those
derived from cellulose or poly lactic acid (PLA) are being
OTR (cm
3
/m
2
/day @23°C & 50% RH)
0
5
10
15
20
25
30
35
40
45
50
No Coatin
g
EVOH PVDC New Tech
100-110 cm
3
/m
2
/day
23°C/50% RH
23°C/75% RH
Figure 1. Comparison of new-generation oxygen barrier coating
on PET with other barrier polymers.
Al-PET/PE
PET + New Gen/PE Al-PET+ New Gen/PE
No flexes
20 Gelbo flexes
OTR (cm
3
/m
2
/day @23°C & 50% RH)
5
0
10
15
20
25
Figure 2. Oxygen transmission versus flex resistance (in
Gelbo flexes)-new-generation barrier coatings in laminate
structures.
PET OPP OPA
1500cc/m
2
/day
110cc/m
2
/day
OTR (cm
3
/m
2
/day @23°C & 50% RH)
0
5
10
15
20
25
30
35
40
45
50
No Coating
23°C/50% RH
23°C/75% RH
Figure 3. Comparison of new generation oxygen barrier coatings
on common flexible packaging substrates.
BARRIER AND OVERPRINT COATINGS 101
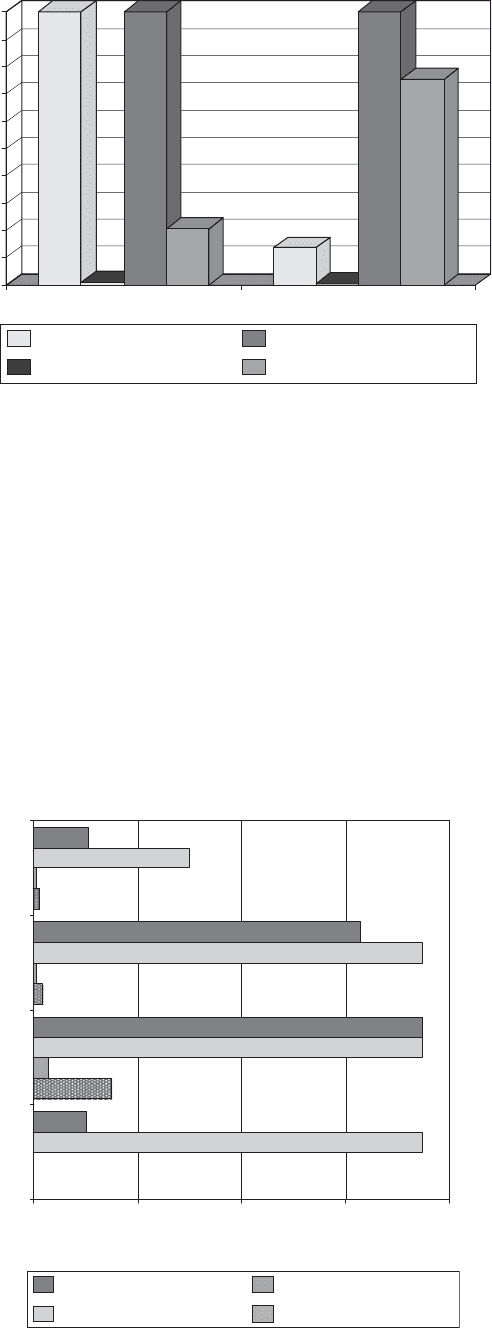
specified more and more by the large retail outlets. These
substrates do not have the appropriate barrier properties
required for many perishable food types. Improvements
through inorganic and new-generation-type coatings are
presently a high priority. In Figure 4 the improvement to
oxygen barrier on PLA is very significant. Cellulose,
however, which has even higher sensitivity to moisture,
shows less improvement.
Moisture Vapor Barrier. Combining oxygen and moist-
ure vapor barrier in organic polymer-based coatings re-
mains a target. Although nano particulate materials are
capable of improving both barrier properties it is still
necessary to apply separate coatings to achieve the high-
est barriers. In Figure 5, improvements to moisture vapor
barrier on PET, PLA, and paper are compared with a
commercial barrier board. With this type of coating tech-
nology, it is possible to confer moisture vapor barrier
performance on substrates such as polylactic acid (PLA)
and cellophane approaching that of polyethylene or OPP.
Present technologies include both water- and solvent-
based products permitting applications from gravure,
flexographic, and lithographic (tower) coaters.
NEAR FUTURE DEVELOPMENTS
The new-generation barrier coating technologies are
set to evolve rapidly in-line with the requirement for a
single coating combining both oxygen and moisture bar-
riers, sustainability and high abuse resistance at low
applied film weights. Heat-sealability and surface print
0
20
40
60
80
100
120
140
160
180
200
PLA Cellophane
OTR (cm
3
/m
2
/day @23°C & 50% RH)
No Coating (23°C/50% RH)
Coating (23°C/50% RH)
No Coating (23°C/75% RH)
Coating (23°C/75% RH)
Figure 4. Indication of oxygen barrier improvements
with new-generation technology on sustainable
substrates.
PET
PLA
Paper
0 20406080
Moisture Vapor Transmission Rate (g/m
2
/24h)
Coated Board
No Coating; 32°C; 80% RH + New Gen’; 32°C; 80% RH
+ New Gen; 25°C; 50% RHNo Coating; 25°C; 50% RH
Figure 5. New-generation barrier coatings: moisture vapor
transmission on sustainable substrates.
102 BARRIER AND OVERPRINT COATINGS

performance will follow. The use of barrier coatings,
especially oxygen barrier types, will also allow considera-
tion for simplifying or lightweighting complex laminates,
resulting in waste reduction and a lower carbon footprint
than is currently the case (4). Furthermore, these highly
transparent and flexible coatings will allow new packa-
ging design innovations where product visibility is an
advantage. Combining these gas barrier coating technol-
ogies with UV light-curing barrier coatings will result in
clear plastic packaging with the potential to supplant
glass, cans, and conventional laminates in a wider variety
of packaged produce.
BIBLIOGRAPHY
1. D. Zagory, in Encyclopedia of Packaging Technology, 2nd
edition, John Wiley & Sons, New York, 1997.
2. M. W. Leonard, ‘‘A Study of the Effects of Increasing Crystal-
linity in PVdC Coated Polypropylene (Prop c) and Cellulose
(MxxT/s),’’ J. Oil Colour Chem. Assoc. 62(12), 461–469 (1979).
3. D. C. Thompson and A. L. Barney, ‘‘Vinylidene Polymers’’ in
Encyclopedia of Chemical Technology, 2nd edition, Vol. 21,
Interscience, New York, 1970.
4. D. R. Illsley, private communication.
BARRIER POLYMERS
BRADLEY FINNIGAN
Precision Fabrics Group,
Newport News, Virginia
Modern synthetic polymers have been used for over 50
years as barriers to mass transport of liquids and gases.
Major applications of barrier polymers include food and
pharmaceutical packaging. The growth in polymer-based
packaging materials at the expense of metal and glass
containers has seen the importance of barrier polymers
continue to rise. Polymeric packaging offers the advan-
tages of weight reduction, formability into useful and
attractive shapes, reduced breakage, transparency, and
cost savings. Recently, emphases on longer-term storage
and convenience factors such as microwavability have
become important. Research remains strong in industry
and academia to offer improved polymer properties and
fabrication methods and to address environmental issues
including recyclability, source reduction, sustainability,
and degradability.
Traditionally, the definition of a barrier polymer
has been strongly attached to the oxygen permeability.
Barrier polymers had oxygen permeabilities less than
about 2.0 nmol/m s GPa ( = 1.0 cc(stp) mil/100 in.
2
day
atm). (See Table 1 for unit conversions.) This definition
unnecessarily limits the range of barrier polymers. Some
polymers with higher oxygen permeabilities are useful
barriers for other molecules.
THE PERMEATION PROCESS
A basic understanding of the permeation process can help
clarify the barrier characteristics of polymers. A permeant
molecule moves through a barrier in a multistep process.
First, the molecule collides with the polymer surface.
Then, it must adsorb and dissolve into the polymer
mass. In the polymer, the permeant ‘‘hops’’ or diffuses
randomly as its own thermal kinetic energy keeps it
moving from vacancy to vacancy as the polymer chains
move. The random diffusion yields a net movement from
the side of the barrier polymer that is in contact with a
high concentration or partial pressure of permeant to the
side that is in contact with a low concentration of per-
meant. After crossing the barrier polymer, the permeant
moves to the polymer surface, desorbs, and moves away.
In virtually every case, the permeation is controlled by
the solution and diffusion steps. The diffusion coefficient,
D, is a measure of the speed of molecules moving in the
polymer. The solubility coefficient, S, is an indication of
the number of permeant molecules that are diffusing.
Together, the diffusion coefficient and the solubility coeffi-
cient describe the permeability coefficient, commonly
called the permeability, P.
P ¼ D S ð1Þ
A low permeability may result from a low diffusion
coefficient or a low solubility coefficient or both.
Table 1. Permeability Units
a
with Conversion Factors
Multiply - to obtain k
nmol
m s GPA
cc mil
100 in:
2
d atm
cc mil
m
2
d atm
cc cm
cm
2
s atm
cc cm
cm
2
s cm Hg
cc 20 mm
m
2
d atm
nmol
m s GPa
1 2 0.129 4.390 10
10
3.336 10
12
0.1016
cc mil
100 in:
2
d atm
0.50 1 6.452 10
2
2.195 10
10
1.668 10
12
5.08 10
2
cc mil
m
2
d atm
7.75 15.50 1 3.402 10
11
2.585 10
13
0.787
cc cm
cm
2
s atm
2.278 10
11
4.557 10
11
2.939 10
12
1 76.00 2.315 10
12
cc cm
cm
2
s cm Hg
2.998 10
13
5.996 10
13
3.860 10
14
1.316 10
2
1 3.046 10
14
cc 20 mm
m
2
d atm
9.84 19.68 1.27 4.32 10
11
3.283 10
13
1
a
Throughout the Encyclopedia,cm
3
(or mL) is used in preference to cc. However, the advantage of using cc here is an obvious visual aid in the complex units
and there are further comments regarding cc versus cm
3
in the text.
BARRIER POLYMERS 103

The permeability for a given polymer–permeant combi-
nation can be used to describe the steady-state transport.
Equation (2) is Fick’s First Law adapted for packaging:
DM
x
Dt
¼
PADp
x
L
ð2Þ
where DM
x
/Dt is the steady-state rate of permeation of
permeant x through a polymer film with area A and
thickness L. P is the permeability, and Dp
x
is the differ-
ence in pressure of the permeant on the two sides of the
film.
Equation (2) shows why reliable tables of permeabil-
ities are important. The packaging engineer has no control
over the Dp
x
since the conditions of the environment and
the contents are fixed. Mechanical, economic, and contain-
ment requirements limit the allowable ranges of area and
thickness. Only P has a wide range of possibilities.
One caveat must be considered before applying equa-
tion (2). The permeation must be at steady state. With
small molecules such as oxygen, steady state is usually
attained in a few hours or less, depending the polymer and
the thickness. However, with larger molecules in barrier
polymers, especially glassy polymers, the time to reach
steady state can be very long, possibly exceeding the
anticipated storage time. The time to reach steady state,
t
ss
, can be estimated with equation (3):
t
ss
ffi L
2
=4D ð3Þ
Equation (2) should only be used when t
ss
is small
compared to the storage time.
Water-vapor transmission is treated differently. The
industry has arrived at a standard condition for reporting
and comparing performance, 37.81C (1001F) and 90% rh
difference. Equation (4) shows how the rate of water-vapor
transmission can be calculated using the value of the
water-vapor transmission rate (WVTR) and the package
geometry:
DM
H
2
O
Dt
¼
WVTR A
L
ð4Þ
When the actual conditions differ from the standard, the
WVTR can be adjusted with great care. Ideally, data have
been reported at the actual conditions; otherwise, adjust-
ments for both the relative humidity difference and tem-
perature must be made. If the polymer is known to be
insensitive to humidity, such as a polyolefin, the humidity
adjustment is merely multiplication by the actual relative
humidity difference on the two sides of the film divided by
90% rh. The temperature effect will be discussed later.
UNITS
The units for permeability are complex, and many correct
combinations are used in the literature. Lamentably,
many incorrect combinations are used too. Table 1 con-
tains conversion factors for several common units for the
permeability. Table 2 contains conversion factors for
several common units for WVTR. In these units the
quantity of permeant is a molar unit, typically a cc(stp).
For the permeability of flavors and aromas, a unit using
the mass of the permeant is useful. A modified unit, the
MZU ( = 10
20
kg m/m
2
s Pa), can be converted to a
molar unit according to equation (5), where MW is the
molecular weight of the permeant in daltons (g/mol).
P in MZU ð10=MWÞ¼P in nmol=m s Gpa ð5Þ
PERMEABILITY DATA
Table 3 contains oxygen, nitrogen, and carbon dioxide
permeability data for several polymers at 201C and 75%
rh. Generally, the permeabilities of N
2
,O
2
, and CO
2
are in
the ratio 1:4:14. The polymers are ranked roughly in order
of increasing permeability. This list contains polymers
that meet the traditional criterion for barrier polymers
and several that do not. The range of permeabilities here
is more than four orders of magnitude. Table 4 contains
diffusion coefficients and solubility coefficients for oxygen
and carbon dioxide in many of the same polymers. Note
that these values are useful for comparison with the
flavor, aroma, and solvent permeation that is presented
later in the chapter.
Barrier polymers are typically used as discrete layers,
as coatings, or in blends. Depending on the packaging
application, oxygen barrier polymers are frequently se-
lected from the group consisting of ethylene–vinyl alcohol
copolymers, polyvinyl alcohol, polyvinylidene chloride,
polyacrylonitrile, polyesters, and various types of polya-
mides. Moisture barrier polymers include polyethylene
(particularly high density polyethylene), polypropylene,
polyvinylidene chloride, cyclic olefin copolymers, and poly-
chlorotrifluoroethylene. OTR and WVTR data for selected
polymers is shown in Table 5. A comprehensive list of
permeability properties of polymers can be found in
reference 4.
The increased demand for transparent packaging has
seen a number of organic, inorganic and ceramic barrier
coatings introduced as alternatives to metallized coatings
and aluminum foil (3). Examples of commercially avail-
able barrier coating materials include polyacrylic acid,
polyvinylidene chloride, polyvinyl alcohol, nitrocellulose,
epoxy-amine and polyamino ether coatings, and nanocom-
posites thereof. In addition to high-barrier, transparency,
and cost-savings drivers, retort and microwave packaging
requirements contributed to the introduction of thin sili-
con oxide (SiO
x
) and aluminum oxide (AlO
x
) coatings.
These coatings are typically applied to bottles and or-
iented polyester films via vapor deposition processes.
Table 2. Water Vapor Transmission Rate Units with
Conversion Factors
Multiply - to obtain k
nmol
m s
g mil
100 in:
2
d
g cm
m
2
d
nmol
m s
1 0.253 6.43
g mil
100 in:
2
d
3.95 1 25.40
g cm
m
2
d
0.155 3.94 10
2
1
104 BARRIER POLYMERS

While SiO
x
and AlO
x
coatings are not polymeric, they offer
an excellent combination of oxygen and moisture barrier
properties to polymer-based packaging.
Multilayer barrier structures are commonly used for
both flexible and rigid applications. These structures can
be the result of coextrusion, lamination, or coating. Typi-
cally, one of the layers provides most of the barrier while
other layers provide inexpensive mechanical integrity,
printability, opacity, sealability, formability, adhesion, or
merely a place to locate reground scrap. The total barrier
performance of a multilayer structure can be estimated
with equation (6):
L
t
P
t
¼
L
1
P
1
þ
L
2
P
2
þ
L
3
P
3
þ ð6Þ
where L
1
, L
2
, and L
3
are the thicknesses of layers and P
1
,
P
2
, and P
3
are the respective permeabilities of the layers.
L
t
is the total thickness, and P
t
is the effective perme-
ability of the multilayer structure. The quantity L
t
/P
t
is
the ‘‘permeance.’’ L
t
and P
t
may be used in equation (2) to
calculate the expected performance. An example of a
seven-layer barrier film is shown in Figure 1.
NANOCOMPOSITE APPROACH TO BARRIER
PERFORMANCE
Polymer nanocomposites containing nanometer-sized
layered silicates (1 nm thick, 100- 500-nm-diameter plate-
lets) have been the focus of considerable research interest
in recent years (5). This is because significant improve-
ments in mechanical, thermal, and barrier properties
have been reported, particularly in nylon systems.
Improved barrier performance in nanocomposites can
result from the high-aspect-ratio platelets providing a
tortuous path for the permeant to travel through, or by
the filler inclusions altering the diffusivity of the host
matrix—for example, by decreasing the free volume of the
polymer—or by changing the polymer crystal type/orien-
tation. Improvements in barrier performance upon filler
addition do not always occur.
A number of companies currently manufacture poly-
olefin and nylon nanocomposites for packaging applica-
tions. However, the use of these materials for barrier
packaging has been limited to niche applications. At
present, research is ongoing to overcome the challenges
that are preventing widespread commercialization. These
Table 3. Permeabilities of Selected Polymers
a
Gas Permeability (nmol/m s GPA)
Polymer Oxygen Nitrogen Carbon Dioxide
Vinylidene chloride copolymers 0.02–0.30 0.005–0.07 0.1–1.5
Ethylene–vinyl alcohol copolymers, dry 0.014–0.095
at 100% rh 2.2–1.1
Nylon-MXD6
b
0.30
Nitrile barrier polymers 1.8–2.0 6–8
Nylon-6 4–6 20–24
Amorphous nylon (Selar
c
PA 3426) 5–6
Polyethylene terephthalate 6–8 1.4–1.9 30–50
Polyvinyl chloride 10–40 40–100
High-density polyethylene 200–400 80–120 1200–1400
Polypropylene 300–500 60–100 1000–1400
Low density polyethylene 500–700 200–400 2000–4000
Polystyrene 500–800 80–120 1400–3000
a
Reference 1; see Table 1 for unit conversion.
b
Trademark of Mitsubishi Gas Chemical Co.
c
Trademark of E. I. du Pont de Nemours & Co., Inc.
Table 4. Diffusion and Solubility Coefficients for Oxygen and Carbon Dioxide in Selected Polymers at 231C, Dry
a
Oxygen Carbon Dioxide
Polymer D,m
2
/s S, nmol/(m
3
GPa)
b
D,m
2
/s S, nmol/(m
3
GPa)
b
Vinylidene chloride copolymer 1.2 10
14
1.01 10
13
1.3 10
14
3.2 10
13
Ethylene–vinyl alcohol copolymer
c
7.2 10
14
2.4 10
12
Acrylonitrile barrier polymer 1.0 10
13
1.0 10
13
9.0 10
14
4.4 10
13
Polyethylene terephthalate 2.7 10
13
2.8 10
13
6.2 10
14
8.1 10
14
Polyvinyl chloride 1.2 10
12
1.2 10
13
8.0 10
13
9.7 10
13
Polypropylene 2.9 10
12
1.1 10
14
3.2 10
12
3.4 10
14
High-density polyethylene 1.6 10
11
7.2 10
12
1.1 10
11
4.3 10
13
Low-density polyethylene 4.5 10
11
2.0 10
13
3.2 10
11
1.2 10
14
a
Reference 1.
b
For unit conversion, see equation (5).
c
42 mol% ethylene.
BARRIER POLYMERS 105

challenges include processing methods and inexpensive
surface modifications to ensure excellent particle disper-
sion and particle alignment, which are required in order to
optimize property performance and reproducibility.
FLAVOR/AROMA/SOLVENT BARRIER
In addition to functioning as gas and moisture barriers,
polymers are also utilized in packaging applications that
require flavor, aroma, or solvent barriers. Table 6 contains
data for the transmission of flavor, aroma, and solvent (F/
A/S) molecules in a few polymers. These data are for 251C
at 0% rh and very low activity (or partial pressure) of the
permeant. This list represents only a small fraction of the
virtually limitless combinations of F/A/S compounds and
polymer films. Reliable data at low activities, as typically
encountered in foods, are difficult to find. However, the
data in Table 6 are consistent with some general rules.
First, polyolefins are not good barriers for F/A/S com-
pounds. Second, vinylidene chloride copolymers and ethy-
lene–vinyl alcohol copolymers are good barriers. A third
rule is not apparent from Table 6. A polymer below its
glass-transition temperature (T
g
)—that is, glassy poly-
mer—is an excellent barrier for F/A/S compounds. Data
are extremely rare; hence, only a few data are given for an
ethylene–vinyl alcohol copolymer. The problem is that the
diffusion coefficients are so low that the experiments take
too long to do with accuracy. The previous author and
associates were unsuccessful in many attempts to test
polystyrene, PET, and nylons.
In these tables of data, different polymers occupy
the top barrier positions. For oxygen, vinylidene chloride
copolymers and ethylene–vinyl alcohol copolymers are the
best barriers. For water vapor, vinylidene chloride copoly-
mers and the polyolefins are the best barriers. For F/A/S
compounds, vinylidene chloride copolymers remain good.
Some glassy polymers that are not given are the best
barriers. Yet of all these polymers, only a few meet the
traditional definition of a barrier polymer.
FACTORS AFFECTING PERMEABILITY
The permeability increases with increasing temperature
for all known cases. A plot of logarithm P versus 1/T in
Table 5. Oxygen and Water-Vapor Transmission Rates of Selected Polymers
Polymer OTR, nmol/(m s)
a
WVTR, nmol/(m s)
b
Polychlorotrifluoroethylene 36
c
0.004–0.009
d
AlOx (Toppan GX-P)
e
0.04 0.006
Vinylidene chloride copolymers 0.02–0.30 0.005–0.05
High-density polyethylene (HDPE) 200–400 0.095
Cyclic olefin copolymers 360 0.02
f
Polypropylene 300–500 0.16
Amorphous nylon (EMS G21) 7.7 0.23
f
Low-density polyethylene (LDPE) 500–700 0.35
Ethylene–vinyl alcohol, 44 mol% ethylene 0.12 0.36
g
Polyethylene terephthalate (PET) 6–8 0.45
Polyvinyl chloride (PVC) 10–40 0.55
Ethylene–vinyl alcohol, 32 mol% ethylene 0.03 0.96
g
Nylon-6,6 7 0.95
Polyamino ether (ICI’s OxyBLOC) 0.2–1.6 1
Nitrile barrier resins 1.8–2.0 1.5
Polystyrene 500–800 1.8
Nylon-6 4–6 2.7
Polycarbonate 520 2.5
Nylon-12 15.9
Polyacrylic acid (Kureha’s Besela) 0.03
h
Polyvinyl alcohol 0.12
i
a
At 231C and 0% rh unless otherwise noted.
b
At 381C and 90% rh unless otherwise noted.
c
Measured at 251C. Reference (4).
d
Measured at 381C and 100% rh. Reference (4).
e
Aluminium oxide coated 48ga polyester film. Units cm
3
or gm/(100 in
2
day atm).
f
Measured at 231C and 85% rh. Reference (4).
g
Measured at 401C and 90% rh. Reference (4).
h
PAA-coated 48ga polyester film. Units cm
3
/(100 in
2
day atm). Measured at 231C and 80% rh after retort.
i
Measured at 241C. Reference (4).
LDPE/LLDPE
tie
Nylon 6
EVOH
Nylon 6
tie
m-LLDPE sealant
7-Layer Barrier Food Packaging Structure
Figure 1. Seven-layer barrier film used in food packaging.
106 BARRIER POLYMERS

kelvin yields a straight line with a slope proportional to the
activation energy for permeation. Usually the slope is
steeper above T
g
than below T
g
. Hence, knowledge of the
permeability at two temperatures allows calculation of the
permeability at a third temperature, provided that T
g
is not
in the range. For many polymers the oxygen permeability
increases about 9% per 1CaboveT
g
and about 5% per 1C
below T
g
. The temperature sensitivity is greater for larger-
permeant molecules. The temperature sensitivity for the
WVTR is theoretically a little more complicated; however, it
is about the same as for the oxygen permeability.
Humidity can affect the permeability of some polymers.
When a polymer equilibrates with a humid environment,
it absorbs water. The water concentration in the polymer
might be very low as in polyolefins or it might be several
weight percent as in ethylene–vinyl alcohol copolymers.
Absorbed water does not affect the permeabilities of some
polymers including vinylidene chloride copolymers, acry-
lonitrile copolymers, and polyolefins. Absorbed water in-
creases the permeabilities in some polymers including
ethylene–vinyl alcohol copolymers and most polyamides.
A few polymers show a slight decrease in the oxygen
permeability with increasing humidity. These include
PET and amorphous nylon. Since humidity is inescapable
in many packaging situations, this effect cannot be over-
looked. The humidity in the environment is often above
50%rh, and the humidity inside a food package can be
nearly 100% rh.
Table 6. Examples of Permeation of Flavor and Aroma Compounds in Selected Polymers at 251C,
a
Dry
b
Flavor/Aroma Compound Permeant Formula P, MZU
c
D,m
2
/s S, kg/(m
3
Pa)
Vinylidene Chloride Copolymer
Ethyl hexanoate C
8
H
16
O
2
570 8.0 10
18
0.71
Ethyl 2-methylbutyrate C
7
H
14
O
2
3.2 1.9 10
17
1.7 10
3
Hexanol C
6
H
14
O 40 5.2 10
17
7.7 10
3
trans-2-Hexenal C
6
H
10
O 240 1.8 10
17
0.14
d-Limonene C
16
H
16
O 32 3.3 10
17
9.7 10
3
3-Octanone C
8
H
16
O 52 1.3 10
18
0.40
Propyl butyrate C
7
H
14
O
2
42 4.4 10
18
9.4 10
2
Dipropyl disulfide C
6
H
14
S
2
270 2.6 10
–18
1.0
Ethylene–Vinyl Alcohol Copolymer
Ethyl hexanoate 0.41 3.2 10
18
1.3 10
3
Ethyl 2-methylbutyrate 0.30 6.7 10
18
4.7 10
4
Hexanol 1.2 2.6 10
17
4.6 10
4
trans-2-Hexenal 110 6.4 10
17
1.8 10
2
d-Limonene 0.5 1.1 10
17
4.5 10
4
3-Octanone 0.2 1.0 10
18
2.0 10
3
Propyl butyrate 1.2 2.7 10
17
4.5 10
4
Low-Density Polyethylene
Ethyl hexanoate 4.1 10
6
5.2 10
13
7.8 10
2
Ethyl 2-methylbutyrate 4.9 10
5
2.4 10
13
2.3 10
2
Hexanol 9.7 10
5
4.6 10
13
2.3 10
2
trans-2-Hexenal 8.1 10
5
d-Limonene 4.3 10
6
3-Octanone 6.8 10
6
5.6 10
13
1.2 10
1
Propyl butyrate 1.5 10
6
5.0 10
13
3.0 10
2
Dipropyl disulfide 6.8 10
6
7.3 10
14
9.3 10
1
High-Density Polyethylene
d-Limonene 3.5 10
6
1.7 10
13
2.5 10
1
Menthone C
10
H
18
O 5.2 10
6
9.1 10
13
4.7 10
1
Methyl salicylate C
8
H
8
O
3
1.1 10
7
8.7 10
14
1.6
Polypropylene
2-Butanone C
4
H
8
O 8.5 10
3
2.1 10
15
4.0 10
2
Ethyl butyrate C
6
H
12
O
2
9.5 10
3
1.8 10
15
5.3 10
2
Ethyl hexanoate 8.7 10
4
3.1 10
15
2.8 10
1
d-Limonene 1.6 10
4
7.4 10
16
2.1 10
1
a
Values for vinylidene chloride copolymer and ethylene–vinyl alcohol are extrapolated from higher temperatures.
b
Permeation in the vinylidene chloride copolymer and the polyolefins is not affected by humidity; the permeability and diffusion coefficient in the ethylene–
vinyl alcohol copolymer can be as much as 1000 times greater with high humidity (1).
c
MZU = (10
20
kg m)/m
2
s Pa); see equation (5) for unit conversions.
BARRIER POLYMERS 107
