Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

BIBLIOGRAPHY
‘‘Ampuls and Vials,’’ R. P. Abendroth and J. E. Lisi, in The Wiley
Encyclopedia of Packaging Technology, 1st ed., Wiley, New
York, Owens-Illinois, Inc, 1986, pp. 16–20.
1. The United States Pharmacopeia XXII, and the National
Formulary, The United States Pharmacopeial Convention,
Inc., Rockville, MD, 1990.
ANALYTICAL METHODS FOR FOOD
PACKAGING AND SHELF LIFE STUDIES
CRISTINA NERIN
Aragon Institute of Engineering
Research (I3A), CPS;
University of Zaragoza, Dept.
Analytical Chemistry, Zaragoza,
Spain
INTRODUCTION
According to the Encyclopedia of Packaging Technology,
shelf life is the time after production and packaging that a
product remains acceptable under defined environmental
Figure 6. Tube converting for vial manufacture.
Figure 5. Vial contour-forming sequence.
38 ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES
conditions. To establish this period of time is nowadays
required for every food product, not only for commercial
reasons but also for safety reasons, as all foods deteriorate
with time. The main causes for these deteriorative pro-
cesses are as follows:
1. Oxidation processes during which the color fades
and may give way to a brownish appearance,
rancid tastes, and odors and the food becomes
unacceptable.
2. Microbiological, which is the consequence of the
micro-organisms growth, either molds, bacteria or
yeasts, some of them pathogens and dangerous for
human beings. Often together with the micro-organ-
isms, new compounds appear that are responsible
for the bad odors or off flavors.
3. Water losses, which is the consequence of the aging
that affect the texture, hardness, appearance, and
the taste.
However, although the causes of the limitation of the
shelf life were known, the analysis of the macroscopic
effects usually is not enough to establish the limits, and
more and more other parameters are required to establish
the end of the shelf life.
Besides, the evaluation of the deadline cannot be sub-
jective, just as a global measurement, and analytical
procedures are needed to standardize the parameters
used for establishing the deadline limits.
The 21st century is characterized by globalization. In
the food area, this means to have the food packaged,
because in these conditions, the transport, storage, logis-
tic, and distribution chain can reach distant markets, and
a global commercialization can take place. Foodstuffs
themselves cannot stand for long time, and new technol-
ogies such as vacuum packaging and modified atmosphere
packaging, combined with the use of high barrier materi-
als and active packaging, can extend the shelf life of
the food inside the package. Emerging technologies like
intelligent packaging are then proposed to show that the
packaged food is still good and safe.
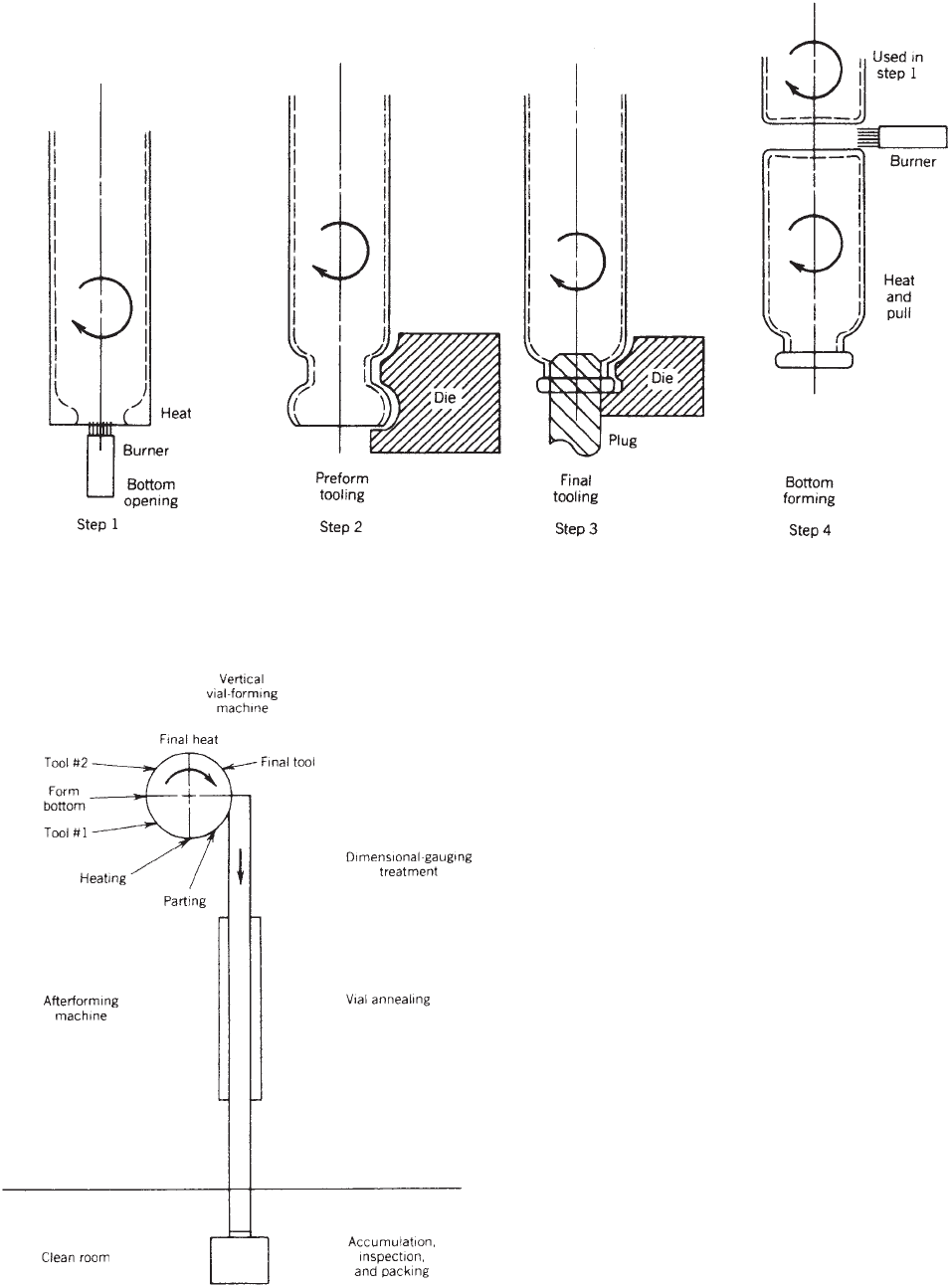
But the challenge is how to measure the shelf life
of food. When developing the packaging material or
the packaging system, there are different materials,
parameters, and variables to modify and to measure.
Figure 1 shows a scheme in which the material, the
food, and the internal atmosphere play an important
role in the shelf-life definition and testing. Interactions
between them as well as absolute measurements have to
be carried out to study the shelf life. According to this
process and depending on the type of samples that
we have, different parameters and different analytical
methods will be used. This chapter deals with the main
analytical methods, which can be useful for shelf-life
evaluation, taking into account that the main purpose in
this task is to have an objective and independent series of
methods. Avoiding the subjective and personal skills is the
main way to measure the key parameters. There is not
an exhaustive list of analytical methods, and only those
considered as the most appropriate or most common in
this type of studies are referred to.
As Figure 1 shows, the food, internal atmosphere, and
packaging material should be tested to establish the shelf
life. The food qualities have to remain constant and be as
good as possible and the most similar to the fresh food or
just-made food. These qualities involve color, taste, hard-
ness, texture, volatile compounds, odor, water content,
chemical contaminants, absence of microbiological con-
tamination, and freshness, among others.
The internal atmosphere means the analysis of gases
such as oxygen, carbon dioxide, and other permanent
gases and vapors, such as water, volatile compounds
transferred by the packaging material, or those released
by the food.
Finally, the packaging material, which involves the
testing of the permeability properties, the analysis of
likely contaminants as potential migrants, the migration
tests to ensure that the material is safe for being in
Figure 1. Scheme of the food packaging to measure
the shelf life.
ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES 39
contact with the food, and the sorption properties of the
material to guarantee that the material does not affect
the quality of food, by scalping aromas or by trapping the
compounds responsible for the color, flavor, or other food
attributes.
In all cases, the first key point is the selection of the
parameters to be measured in a quantitative manner for
the shelf-life studies, and the second point is the selection
of the analytical technique and the procedure to measure
the changes, if any, in the whole system.
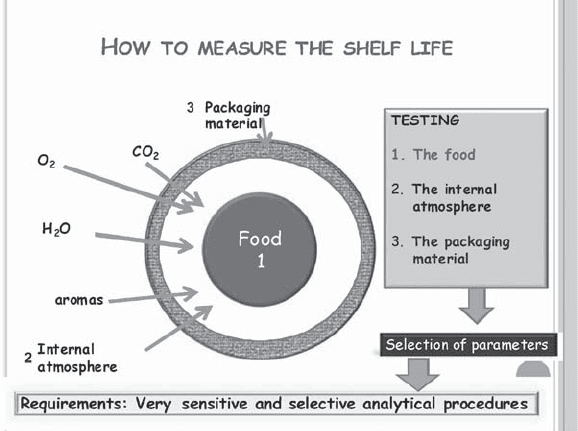
THE ANALYTICAL PROCESS
Figure 2 shows a scheme of the analytical process. Three
main steps can be distinguished: the ‘‘sampling,’’ the
‘‘sample treatment,’’ and the ‘‘final analysis.’’ A short
comment of the main procedures involved in each step
will be described below.
THE SAMPLING STEP
Usually, this is the first step, as the sample has to be taken
for analysis. Some critical requirements should be fol-
lowed to be sure that the final analysis represents a real
diagnostic of the sample. The three points mentioned in
the Figure 2 have to be decided by the analyst, and such a
decision should be based on scientific parameters. The
first step, the sample size, seems to be obvious, as if low
concentrations are expected for the analytes, higher
amount of sample should be taken. However, the sample
size will condition the second step of the analytical
procedure, which is the sample treatment, and it will
affect the handling, time, and price of the analysis. How-
ever, if the sample size is small the representativeness of
the sample is in danger. Then, to decide about this
parameter, it is recommended to think about the detection
limit of the final analytical technique in which the vari-
able under study will be measured as well as the expected
concentration of the analyte after the test. Sometimes,
only differences between the concentration before and
after the shelf-life studies are going to be measured
instead of absolute values. In this case, the sensitivity is
higher, as the absolute value measured is higher than the
difference itself. When absolute values have to be mea-
sured, for example, the concentration of an analyte formed
during the shelf life study, the expected value can be
extremely low, and thus higher amount of sample is
needed.
Representativeness
This point is critical and affects any kind of analysis. It is
close-related with the homogeneity of the sample. With
food samples, the lack of homogeneity can let us take
discriminant portions of food for the analysis, and ob-
viously in this case the diagnostic is wrong. To avoid
this problem, a previous homogenization is recommended.
Sample size is also conditioned here, as with small sample
size there is a high risk of discrimination, which ruins the
analysis. All parts of the food should be taken and mixed
together to reproduce the real situation as much as
possible. Interactions between the different components
in the food sample is also very important, what requires
that all the components were represented in the sample
taken for analysis.
Homogeneity
The problem of homogeneity is more common in food
analysis, where the samples can be macroscopically homo-
geneous but nonhomogeneous at microscopic level. Differ-
ent devices are nowadays available to homogenize the
samples. Mills, blenders, mixers, and similar machines
Figure 2. The analytical process.
40 ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES
are common in any laboratory. Liquid and solid portions
are mixed together if they are present in the food sample.
Then, an homogeneous sample is produced, usually
smashed and fluid enough to be sure that any portion of
it has the same composition.
SAMPLE TREATMENT
The sample treatment involves the sample dry, the ex-
traction and the clean-up steps. To dry the sample is not
always required before the analysis, although the values
should be always referred to the dry sample for compar-
ison. Sometimes, an aqueous extraction is going to be
applied to the sample, and then there is no point to remove
the water. Anyway, it is important to measure the water
content in a different aliquot from that used for the sample
analysis.
However, the extraction and the clean up steps are
usually the bottleneck of the analytical process (1). It is in
fact the area in which more analytical development has
taken place in the last decade, and there are several
reasons for that. Let us go to explain more in detail the
newest extraction techniques that can be applied for shelf-
life studies.
It is true that the shelf-life studies require sensitive
analytical methods for measuring low concentrations of
compounds that are formed either during the shelf life of a
foodstuff or disappear as long as the time goes on. Alter-
natively, the packaging has to be measured, and then low
concentrations of compounds are also analyzed either in
the packaging material or in the atmosphere inside the
packaging. In any case, the analytes have to be isolated
from the matrix, the interferences have to be removed as
efficiently as possible, and simultaneously the concentra-
tion of the analyte in the final solution that will be
analyzed has to be as high as possible. This way, the
detection and quantification limits will be surpassed, and
no limitations concerning the sensitivity will be present.
The classic extraction procedures involve the liquid–
liquid extraction, in which the liquid sample to be ex-
tracted is shaken with a small volume of a nonmiscible
liquid-extracting phase, nonsoluble with the sample, in
which the analytes will be efficiently dissolved and then
extracted from the matrix. It is obvious that for an
aqueous matrix, the extracting agent will be an organic,
and vice versa. To gain sensitivity using this extraction
procedure, the volume of extracting agent can be reduced
as much as possible, but in any case, handling low
volumes of liquids and putting them in intimate contact
with a much higher volume of the liquid sample is
difficult, and the risk of losing efficiency in the process is
high. Another disadvantage of this classic extraction is the
use of high volumes of organic solvents, which are toxic,
dangereous, and not environmental friendly.
Also included in the classic extraction panorama are
the solid–liquid extraction techniques or better known as
‘‘leakage,’’ which are usually from a solid matrix that can
be either a foodstuff or a packaging material. Among
them, Soxhlet extraction is the most common. Its high
efficiency is based on the fact that it is a continuous
extraction in which new solvent is continuously put in
contact with the sample while maintaining the total
volume of solvent. As the partition constant is reached
each time, the efficiency can be high without increasing
the total volume of solvent but increasing the total mass of
the analyte extracted. Although it is a good and well-
recognized method, it takes time, usually from 6 to 20 h,
and it requires a high amount of solvent.
An improvement of the classic extraction is the accel-
erated solvent extraction (ASE), which consists of carrying
out the extraction at high temperature and high pressure.
The increase of temperature increases the extraction
coefficient and then the extraction efficiency, but when
increasing the temperature, the solvents used as extract-
ing agents are evaporated and they would be in vapor
phase. To avoid the evaporation and maintain the solvent
in liquid phase, a high pressure is applied, and thus the
extraction is accelerated both in time and in efficiency.
When the analytes are dissolved in a liquid solvent
either in aqueous solution or organic solvent, the solid-
phase extraction (SPE) is the most common technique to
isolate and concentrate the analytes and to remove the
interferences from the matrix. SPE is also useful in the
cleanup step, which is usually applied to the sample after
any extraction to remove the interferences. The solid
phase usually is inside a cartridge, ready to use, and
commercially available, and the nature of this solid phase
can vary from the current stationary phases based on
polymeric sorbents to modified silica such as C18, pure
adsorbents such as alumina or active charcoal to ionic
exchange, which are both cationic and anionic phases. Its
versatility and ease of use make SPE one of the most
common techniques in any analytical procedure in which
the isolation of analytes or concentration steps and
cleanup are required.
But without a doubt, the trends in extraction systems
drive us to the microextraction techniques in which a few
drops of solvent, if any, are employed as extracting agents
while maintaining the efficiency and having short periods
of extraction.
For these reasons, new microextraction techniques
have been developed to avoid the mentioned problems.
The most important ones will be described in this chapter.
Solid-Phase Microextraction (SPME)
SPME was proposed for the first time by Arthur and
Pawliszyn in early 1990s (2), and it has been used more
and more widely in sample preparation. SPME is based on
the
partitioning of analytes
between a coated fiber and a
sample. The coated fiber consists of a small fused silica rod
covered with a thin layer of a sorbing material, which acts
as stationary phase. When exposed to the vapor phase
above a solution (head space sampling) or by direct
immersion in the solution, this fiber enters a mass-trans-
fer process driven by the second law of thermodynamics,
according to which the chemical potential of each com-
pound should be equal throughout the system. The che-
mical species will cross the interface until their
concentrations are such that their corresponding partial
molar free energies are the same in all parts of the system
ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES 41
formed by the fiber and the sample. After the coated fiber
is exposed to the sample for a given period, it is inserted
into the injection port of a chromatograph to release the
analyte. In gas chromatography (GC), it is carried out by
thermal desorption, whereas in high-performance liquid
chromatography (HPLC) it is accomplished by dissolution
and injection with the elution solvent (3, 4). In both
approaches, all the substances eventually reach the ana-
lytical instrument detector to produce a trace in which
there is no large solvent peak. Although a small amount of
analyte is transferred, it is sufficient to produce a sig-
nificant analytical signal in modern detectors. The SPME
uses a capillary fiber on which the stationary phase is
coated or chemically bonded; therefore, it is a solventless
technique. Usually, only the analytes and related mole-
cules are trapped on the fiber, and for this reason most of
the interferences remain in the matrix and do not affect
the final analysis (5). This technique is useful in both
modes, headspace, and total immersion, and its high
sensitivity, based on the high concentration rates that
the analytes have on the capillary fiber are without a
doubt their main advantages. There is no limitation for
either of the nature of samples, as both packaging materi-
als and foodstuffs can be analyzed (6–16).
Liquid Microextraction Techniques
Using liquid phases for extraction from liquid matrices,
several microextraction techniques have been recently
developed and applied to food packaging and shelf-life
studies. Among them we can point out the single drop
microextraction (SDME), the liquid–liquid–phase micro-
extraction (LLPME), and the hollow fiber liquid phase
microextraction (HFLPME).
SDME. SDME was first introduced by Liu and Das-
gupta (17), but it is felt that the works by Jeannot and
Cantwell (18, 19), who studied its kinetics and mass
transfer model, and by He and Lee (20), who gave the
technique for much of today’s aspect, provided the basics
needed to consider it as an independent technique. Many
applications have been introduced since then (21) and are
summarized in many devoted reviews (22–25). It consists
of the partition of the analytes between two nonmiscible
solvents, but in this case only one drop of the extracting
solvent hanging from the tip of a syringe is used. Once the
extraction is finished, the drop is withdrawn into the
syringe and directly injected into the gas chromatograph
for the final analysis. It can be considered as solventless
technique as well, as the amount of solvent is negligible.
Also, most of the matrix interferences are avoided in both
exposure modes, as only the compounds soluble in the
drop of solvent, either in headspace mode or in total
immersion in the sample solution are extracted. There is
no doubt that this technique is useful in many applica-
tions and allows the analysis at high sensitivity level. The
drawback of this technique is the fall of the single drop
during the sampling, which requires the procedure to
start again with a new sample, as the presence of the
lost drop of solvent in the same vial affect the extraction
greatly. In such a case, the analytes are simultaneously
extracted in both drops, although only one is analyzed,
which is why a new sampling in a different vial is
required.
Liquid–Liquid Microextraction. Several approaches have
been recently proposed to increase the extraction effi-
ciency using only a few drops of organic solvent. Among
them, those that employ a few microliters of solvent
directly added to the aqueous liquid sample are useful
when using liquid samples, and no particulate matter or
slurries are involved. The supernatant solvent is recov-
ered after the extraction and analyzed. For quantitative
purposes, the use of an internal standard is compulsory to
guarantee the quanitative results, as the handling of such
a low volumes of solvents is always difficult.
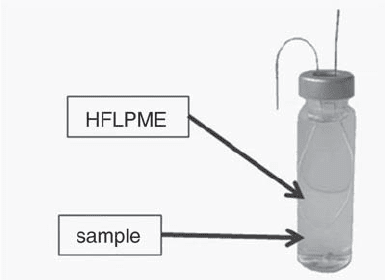
HFLPME. Another improvement of the SDME that
eliminates its main disadvantage is the use of polymeric
membranes as support of the small volumes of solvent.
Few microlitres can be swallowed inside the pores of a
semipermeable polypropylene capillary tube, and then
this hollow capillary can be used for extraction, which
allows high concentration factors (26, 27). When organic
compounds in aqueous solution have to be analyzed,
HFLPME is the appropriate technique for sampling and
extraction, using for instance, an organic solvent as
extracting agent inside the hollow fiber, being the sample
outside (see Figure 3). This technique has been recently
automatized and proposed for migration studies from
packaging materials (27).
Several studies have been carried out in the last 3 years
dealing with different approaches of this technique, and
there are several options in which up to three liquid
phases can be involved to extract and concentrate ionic
analytes from complex matrices (28–30).
TOTAL DISSOLUTION
When the analyte to be measured cannot be directly
extracted, the total dissolution of the sample is required.
This problem often happens when dealing with plastic
samples, in which the analytes are inside the matrix or
even linked to it. For example, if the shelf life of a new
Figure 3. Scheme of the HFLPME extraction from a liquid
sample.
42 ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES
active packaging material is under study, then the remain-
ing active agents in the material should be analyzed. If it is
a plastic material, then it may be difficult to apply an
efficient extraction, as the recoveries are commonly low.
Thus, the total dissolution is preferred. The solvent and
conditions used for the total extraction, have to be opti-
mized. If a synthetic polymer is dissolved, the polymer
itself has to be removed from the solution, as the com-
pounds of interest are the small molecules, no the large
polymeric chains. The addition of methanol, or in general
a solvent in which the polymer is insoluble, is usually
enough to precipitate the polymer, and another filtration
removes the polymer from the solution (31). Obviously, the
compounds of interest have to be soluble in the solvents
used in the precipitation step and they have to be free
from the polymer, which means that no chemical bonds
exist between the polymer and the compounds of interest.
Large volumes of organic solvents are obtained, and
additional concentration and cleanup steps are usually
required to cope with the final analysis. Anyway, only a
few times the total dissolution is required in shelf-life
studies but often is necessary for the packaging analysis.
Final Analysis
The final analysis means the identification and quantifi-
cation of the compounds of interest. The analytical tech-
nique to be used depends on several variables as follows:
. The chemical structure and properties of the
analytes
. The concentration level in which the analytes are
expected to be in the sample
. The information required
For volatile compounds, the most appropriate techni-
que is GC. Depending on the information required, differ-
ent detectors can be used, such as follows: (a) flame
ionization detector (FID), which is ideal for organic com-
pounds with carbon atoms, as the response is based on the
carbon atoms number; (b) the electron capture detector
(ECD), which is very sensitive for electronegative atoms
such as halogens and oxygen; (c) the nitrogen-phosphor-
ous detector (NPD) with a high sensitivity for the com-
pounds that contain these atoms; and (d) the thermal
conductivity detector (TCD), which is only sensitive for
those compounds that have a thermal conductivity very
different from that of the carrier used as background or
reference, usually the hydrogen. That means that this
detector is mainly used for permanent gases, such as
nitrogen, carbon dioxide, and so on. and it is no sensitive
for other compounds. None of the mentioned detectors can
be used for identification purposes if the nature of the
analyte is unknown, as only using the standards, what
means the pure compounds analyzed exactly in the same
conditions as the sample, the compounds could be identi-
fied. For identification purposes, the mass spectrometry
(MS) coupled to GC is the right technique. The molecule is
broken by electronic impact that produces positive ions,
which are the characteristic mass fragments of each
molecule that constitue the mass spectrum. Then, this
mass spectrum of each compound is obtained and com-
pared with the mass spectra contained in the MS library.
The software of this hyphenated technique (GC-MS) give
as the list of compounds with the most similar spectra and
is the analyst who decides which one can be present in the
sample, according to the matching parameters provided
by the software.
Two different systems for MS detectors can be chose:
the quadruple and the ion trap. For quantitative purposes,
it is generally recommended the quadrupole, whereas for
qualitative objectives the ion trap is more appropriate, as
it pemits to have the MS
n
fragmentation. This means that
once the molecule is fragmented and the characteristic
masses are known, the analyst can select some of these
characteristic masses and apply a new fragmentation.
This operation can be repeated several times. Even the
isomers or quiral compounds, which are very similar
between them, have differences in some of the fragmenta-
tion profile; these differences can be used for unequivocal
identification. Recent advances on the MS detectors have
launched in the market new instruments with novel
systems that increase the sensitivity of the MS detectors.
Combinations of MS-MS, triple quadrupole (TQ) or time of
flight (TOF) techniques are now available. These analy-
tical tools allow the identification of unknowns and permit
as well the quantitation of the compounds at low level of
concentration. The selection of the right technique de-
pends on the sample, the analyte, and the information
required from the sample.
There are also several options for injecting the com-
pounds into the GC. The head space (HS) technique is
useful for the analysis of volatile compounds. It consists of
injecting the vapor in equilibrium with the sample at a
fixed temperature. The sample is thermostatized, and only
a fraction of the vapor in equilibrium is taken for the
analysis. This technique can be used in either manual or
automatic modes, but the reproducibility is much higher
in the latter. As only vapor is introduced into the GC, most
of the interferences from the matrix remain in the sample,
either in liquid or solid state, and the sample treatment is
considerably simplified. For this reason, this technique is
applied to a wide series of samples and volatile analytes.
The scarce handling and time consuming required make it
appropriate for the analysis of volatile compounds.
An improvement of this technique is the dynamic HS,
also called purge and trap (P&T), which is coupled to the
GC. The advantage is that in the dynamic system, the
analytes are continuously purged from the sample using
an inert gas and trapped on a solid adsorbent, which is
thermally desorbed to introduce the analytes directly into
the GC. This technique is the most sensitive for volatile
compounds, as the total mass of the analytes present in
the sample is introduced into the analytical system and
arrives at the detector (31, 32). To have an idea of the high
sensitivity of P&T-GC-MS, we can compare the real mass
of analytes at the detector in this case with that of liquid
injection in GC-MS. For example, 10 mL of sample con-
taining 1 ng/mL of a volatile analyte are analyzed by both
direct injection into GC-MS and HS-GC-MS. If 1 mLis
injected in the former case, 1 pg arrives at the detector.
However, in P&T-GC-MS, the total mass of the same
ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES 43
volatile compound contained in the 10 mL of sample
arrives at the detector, which is 10 ng of analyte. This
amount is 10,000 times higher in the detector. To have the
equivalent mass at the detector in both cases, the concen-
tration of the analyte in the sample should be 0.1 pg/mL,
that is 0.1 ppt. However, the main drawback of this
technique is also its high sensitivity, as it is very difficult
to have a blank sample, and often interfering compounds
appear and overlap the peaks of interest. Another problem
is the calibration plot for quantitative purposes, because
in this system the equilibrium is not reached, as the
volatile compounds that would be in equilibrium in the
vapor phase are continuously removed from the vapor and
trapped on the solid trap. Then, the calibration plot should
be prepared exactly in the same conditions as those used
for the sample.
Another interesting approach for injecting the volatile
or semivolatile compounds into the GC column, apart from
the injection of liquid solutions, is the SPME injection.
Nowadays, there are commercially available automatic
injectors for SPME, HS, P&T, and of course for liquid
injections into the GC.
The analysis of non/volatile compounds is usually
carried out by liquid chromatography (LC), in which the
compounds present in the sample are separated. Several
detectors can be used; the most common ones are ultra-
violet-visible spectrometry (UV-VIS), molecular fluores-
cence spectrometry (Fl), refraction index (RI), and MS.
UV-VIS is the most general and common one coupled to
HPLC, whereas Fl is more selective as only fluorescent
compounds can be detected. RI is usually applied to
analyze sugars, wheras MS can be applied to any com-
pound with a molecular mass higher than 50 units of mass
and able to be ionized in the ionization step of the LC-MS.
As in GC, the separation takes place in the chromato-
graphic column where the stationary phase exerts an
interaction with the compounds. Depending on the nature
of this stationary phase, either partition, ionic exchange,
or size exclusion can be the main process between the
compounds (the analytes) and the column. The analyst
has to take an important decision and choose the right LC
column, according to the analyte and the sample. Also
other conditions such as the size of the column and the
mobile phase in each analytical procedure have to be
optimized in each case. Recent developments to increase
the resolution in LC launched in the market new instru-
ments in which a higher pressure, more narrow LC
columns, and lower particle size in the LC columns occurs.
This is the case of the new systems of ultrahigh perfor-
mance, such as ultrahigh performance liquid chromato-
graphy (UPLC), which increase the resolution and
considerably reduce the time of analysis compared with
the normal HPLC. The UPLC-UV analysis is not in real
time but it can last only a few minutes to have the whole
chromatogram with more than 20 compounds (33).
When using MS as detector, one of the main decisions to
take is the ionization step. This is a critical step in which
the analytes are transformed into ions, either positive or
negative ions, which can be driven to the MS detector in
which they are separated according to their mass and then
counted (abundance). The ionization step in LC is
commonly a soft ionization that mainly produces the
molecular ion. The interface between the ionization step
and the MS analyzer has been the most difficult part of the
development of this hyphenated technique of LC-MS. This
is because the ionization step is applied to the liquid
sample injected into the LC and the nonvolatile solvent,
usually water, methanol-water, or acetonitrile, which has
to be removed efficiently to get the high vaccum required
for MS, to avoid the saturation of the MS detector, and to
remove the interferences associated to the solvents. Two
different ionization devices and techniques are the most
common in current LC-MS, and they are the Electrospray
(EI) and the atmospheric pressure chemical ionization
(ApCI), although more and more the EI is gaining impor-
tance, as most problems are solved using the EI.
During the last 10 years, new approaches dealing with
LC-MS have been launched to the market to increase the
sensitivity and reproducibility, and to widen the type of
analytes that can be analyzed. Also, the instrumental
equipments are nowadays more friendly for the users
and easier to handle.
Also, the advanced MS techniques such as the TOF
supply new tools for the identification of unknowns, using
the exact mass values. Without a doubt the identification
of unknown compounds is the most challenging area, and
new and more sophisticated techniques are required for
this purpose. Artifacts formed during the analysis, degra-
dation compounds caused by the interaction between the
components in the packaging materials or in the food, or
degradation compounds coming from the additives used in
the food or from the packaging materials in contact with
the food, which include the presence of compounds non
intentionally added, pose new analytical problems and
make the analyst face a real challenge in food packaging
and shelf-life studies. To help with the identification, MS-
MS can be used. As was mentioned above, this consists of
identifying the characteristic ion (MS) and then applying
the fragmentation to this ion to break this fragment. The
breakage of each fragment is specific and is of great help to
identify the compounds. Triple quadrupole instruments in
which the first fragments (parent ions) obtained in the
first quadrupole goes through the second quadrupole in
which a gas, usually argon, enters and crash into the
fragments, causing the second fragmentation (daughters)
of each ion, which are analyzed in the third quadrupole.
These systems are also available in LC-MS and are useful
for identification purposes, although the sensitivity using
the triple quadrupole is lower than that obtained when
using only the first quadrupole.
Recently double-dimension chromatography has been
developed.
This is a
new hyphenated technique in which
two different chromatographic separations using two col-
umns of different polarity and different size each, con-
nected in series, are applied to the same sample. Usually,
the first column is of normal size, for example 30 m in GC,
and the second one is short, about 1.5 m. The final detec-
tion can be FID or even better the MS or its different MS
options. This double dimension can be also used in liquid
chromatography. In this case, the use of for example a
size exclusion column as the first one to eliminate the
polymers, proteins, or in general the macromolecular
44 ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES

compounds, and a reverse phase (C18 or similar) as second
dimension can be a good alternative for analyzing in one
single run the compounds of interest without the inter-
ferences from the matrix (34).
A different approach from chromatography is electro-
phoresis, where the compounds are separated based on
the application of an electric field. Then, the ionic com-
pounds move through the liquid solvent according to their
mass and their charge. Although gel electrophoresis is old
and novery sensitive, the new capillary electrophoresis
(CE) has been shown as a good technique for ionic organic
molecules, such as drugs, proteins, amino acids, and so on,
as these compounds can be easily transformed into ionic
ones just controlling the pH. This technique can be also
used in shelf-life studies, but its frequency is not as high
as the chromatographic techniques. The new hyphenated
technique CE-MS can be one alternative in future studies.
OTHER ANALYTICAL PROCEDURES
Figures 4, 5, and 6 summarize the most common techni-
ques for shelf-life studies. Besides the analytical techni-
ques mentioned above, the optical techniques, such as UV-
Vis spectrophotometry or infrarred spectrometry (IR) are
useful. The former is used to measure the color and color
changes (VIS) occurred in the food or in the packaging
materials either in absorption or in reflexion modes. The
latter (IR) is used to identify the packaging materials or
organic components. The most recent advances in near
infrared (NIR) allow also to quantify some components in
not complex samples, mainly in packaging materials more
than in food.
Also, the combination of different techniques provide
an useful an important information in the shelf-life stu-
dies (35) and can be observed as a powerful tool for having
as much information as possible about the state of the
packaging material, the food inside the packaging, and the
internal atmosphere between them.
BIBLIOGRAPHY
1. C. Nerin, Analy. Bioanalyt. Chemi. 388, 1001 (2007).
2. C. L. Arthur and J. Pawliszyn, Anayti. Chem. 62, 2145 (1990).
3. R. Batlle, C. Sanchez, and C. Nerin, J. AOAC Int. 84, 431
(2001).
4. C. Nerin, M. R. Philo, J. Salafranca, and L. Castle,J.
Chromatogr. A 963, 375 (2002).
5. C. Nerı
´
n, E. Contı
´
n, and E. Asensio, Analyt. Bioanalyt. Chem.
387, 2283–2288, (2007).
6. L. Pillonel, J. O. Bossett, and R. Tabacchi, Lebens. Wiss.
Technol. 35, 1 (2002).
7. W. Wardencki, M. Michulec, and J. Curylo, Int. J. Food Sci.
Technol. 39, 703 (2004).
8. A. K. Malik, V. Kaur, and N. Verma, Talanta 68, 842 (2006).
9. J. B. Quintana and I. Rodriguez, Analyt. Bioanalyt. Chem.
384, 1447 (2006).
10. F. Pragst, Analyt. Bioanalyt. Chem. 388, 1393 (2007).
• Colour
• Microbiology
• Compounds released
Spectrophotometry
Reflexion spectrometry
Colorimetry (CIE-LAB)
In vitro tests
In vivo tests
Cell culture
PCR
SEM
Volatile compounds
Degradation compounds
Food artifacts
Aromas
Figure 4. Testing the food.
• Permeability studies
• Diffussion of compounds throughout
the packaging material
• Monitoring the concentration of
active a
g
ents in the packa
g
in
g
• Classical systems
(automatic
instruments)
• New system, cheap
and home-made
Figure 6. Testing the packaging.
• O
2
• Radicals
• Aromas
Automatic systems
Oxysense
Mocon…
HS-SPME
SDME
HFLPME
Figure 5. Testing the internal atmosphere.
ANALYTICAL METHODS FOR FOOD PACKAGING AND SHELF LIFE STUDIES 45
11. G. Ouyang and J. Pawliszyn, Analyt. Bioanalyt. Chem. 386,
1059 (2006).
12. J. S. Aulakh, A. K. Malik, V. Kaur, and P. Schmitt-Kopplin,
Crit. Revi. Analyt. Chem. 35, 71 (2005).
13. C. Bicchi, C. Cordero, E. Liberto, B. Sgorbini, and P. Rubiolo,
J. Chromatogr. A 1184, 220 (2008).
14. H. Kataoka, H. L. Lord, and J. Pawliszyn, Encyclopedia of
Separation Science, Academic Press, London, UK, 2000.
15. J. Pawliszyn, Solid Phase Microextraction: Theory and Prac-
tice, Wiley VCH, New York, 1997.
16. J. Pawliszyn, Applications of Solid Phase Microextraction,
Royal Society of Analytical Chemistry, Cambridge, UK, 1999.
17. H. H. Liu and P. K. Dasgupta, Analyt. Chem. 68, 1817 (1996).
18. M. A. Jeannot and F. F. Cantwell, Analyt. Chem. 68, 2236
(1996).
19. M. A. Jeannot and F. F. Cantwell, Analyt. Chem. 69, 2935
(1997).
20. Y. He H. K. Lee, Analyt. Chemi. 69 4634 (1997).
21. R. Batlle and C. Nerı
´
n. J. Chromatog. A 1045, 29–35 (2004).
22. D. A. Lambropoulou and T. A. Albanis, J. Biochem. Biophys.
Met. 70, (2007) 195.
23. D. A. Lambropoulou, I. K. Konstantinou, and T. A. Albanis, J.
Chromatog. A 1152, 70 (2007).
24. E. Psillakis and N. Kalogerakis, Trends and Analyt.l Chem.
21, 53 (2002).
25. C. Nerı
´
n, J. Salafranca, M. Aznar, and R. Batlle, Analyt.
Bioanalyt. Chem. 2008.
26. J. Romero, P. Lopez, C. R. R. Batlle, and C. Nerin, J.
Chromatog. A 1166, 24 (2007).
27. D. Pezo, J. Salafranca, and C. Nerin, J. Chromatog. A 1174,85
(2007).
28. S. Pedersen-Bjergaard and K. E. Rasmussen, J. Chromatog. B
817, 3 (2005).
29. S. Pedersen-Bjergaard, K. E. Rasmussen, and G. A. Mills,
Trends Analyt. Chem. 26, 843 (2007).
30. S. Pedersen-Bjergaard and K. E. Rasmussen, J. Chromatog. A
1184, 132 (2008).
31. C. Nerı
´
n, C. Rubio, J. Cacho, and J. Salafranca. Chromato-
graphia 41(3/4), 216–220 (1995).
32. C. Nerı
´
n,
C. Rubio
, J. Cacho, and J. Salafranca. Food Addi-
tives Contami. 15(3), 346–354 (1998).
33. K. Bentayeb, R. Batlle, J. Romero, and C. Nerı
´
n, Analyt.
Bioanalyt. Chem. 388, 1031–1038 (2007).
34. C. Nerı
´
n, J. Salafranca, J. Cacho, and C. Rubio, J. Chromatog.
A 690, 230–236 (1994).
35. R. Becerril, R. Go
´
mez-Lus, P. Gon˜i, P. Lo
´
pez, and C. Nerı
´
n,
Analyt. Bioanalyt. Chem, 388, 1003–1011 (2007).
ANTI-COUNTERFEITING PACKAGING
STEVE WERBLOW
Steve Werblow Communications
Ashland, OR
Counterfeiting is a huge business, threatening not only
sales revenue, but also the health and safety of consumers
who may end up with adulterated food or medicine, unsafe
engine parts, or other dangerous fake goods. Liability for
those counterfeit products, as well as the negative pub-
licity that can befall a company whose brand has been
compromised by piracy, can have devastating impacts on
business.
In the United States, the Brand Protection Alliance
asserts that counterfeiting and piracy cost the U.S. econ-
omy $200 to $250 billion per year. The Organization for
Economic Co-Operation and Development in Europe cal-
culated that international trade alone in counterfeit goods
represented an additional $200 billion in 2005; the Inter-
national Chamber of Commerce (ICC) estimates the
total counterfeit economy at more than three times that
amount. In fact, in the middle of the first decade of the
new millenium, ICC figured the counterfeiting industry
represented about 10% of world trade, equivalent to the
12th largest economy in the world.
Counterfeit products range from cheap bootleg videos
to phony bottles of wine sold for thousands of dollars to
fake aircraft parts.
Fortunately, label and packaging technology can help
deter or thwart counterfeiters. Printing techniques, ma-
terials such as substrates and inks, and high-tech sensors
can all play a role in protecting brand integrity.
OVERT AND COVERT
The first decision to make in an effort to protect a brand
from counterfeiting is to determine whether the effort
should be visible or invisible to the counterfeiter.
A highly visible, or overt, tactic can warn off potential
counterfeiters, much as a flashing alarm light does in an
expensive car. An adept thief could break into the vehicle
and disable the alarm, but is more likely to consider
saving the time and hassle by moving on to an unprotected
vehicle.
Overt measures can also communicate to consumers. It
may put them on guard against counterfeit products. It
may reinforce a sales message that says, in essence, ‘‘this
product is so desirable, people want to counterfeit it.’’ And
it may also reassure customers that the product they are
buying is well protected.
By contrast, covert measures tend to be secret and
unobtrusive, generally revealed only through close obser-
vation or with special readers or detectors. The theory
behind covert tactics is often that if counterfeiters do not
know what the authentication features are, they are less
likely to fake them.
Many anti-counterfeiting efforts rely on a combination
of overt and covert methods. It’s like protecting your home
with a good lock, a loud dog, and a burglar alarm.
TACTICS AND MATERIALS
The U.S. $20 bill introduced in 1998 illustrates the use of a
variety of anti-counterfeiting features, all of which may be
employed in the manufacture of labels and packaging.
Rare paper, embedded fibers and strips, watermarks,
46 ANTI-COUNTERFEITING PACKAGING
sophisticated printing, special inks—each feature makes
the bill difficult to reproduce and easy to authenticate.
Special Paper
The U.S. currency anti-counterfeiting effort begins with
cotton/linen paper manufactured specifically for the U.S.
Bureau of Engraving and Printing for the production of
currency. A swipe with an iodine pen is an easy authenti-
cation measure: Unlike wood-based paper, which contain
starches that turn black in the presence of iodine, the
linen/cotton blend will not show a dark line where the pen
left its mark.
In addition to its unique composition, the banknote
paper includes embedded red and blue fibers, so average
paper stocks cannot be substituted for the real thing.
Papers with embedded fibers or planchettes—in-
clusions that look like confetti or tiny dots—are widely
available as label stocks. Even the use of expensive or
unusual substrates, such as textured stock or parchment,
may serve as a mild deterrent to counterfeiters.
Watermarks
Watermarks are a classic anti-counterfeiting feature found
in the U.S. notes as well as currency and other important
documents around the world. A watermark is created
during the manufacture of the paper: While the paper is
still wet, it is pressed with a cylinder roll bearing an image
in relief. The raised areas on the roll compress fibers in the
paper, making the paper thinner in those areas. Thinner
paper transmits more light, allowing the watermark to
‘‘appear’’ when light is shined through the note.
Embossing
Embossing a document, label, or package—creating a
raised figure or pattern on the surface using a die, heat,
or varnish—can be used to deter counterfeiters. Emboss-
ing and texture are increasingly important as scanners
and printers improve. Though new imaging technologies
are better than ever at reproducing color and print quality,
they cannot reproduce the texture of an embossed surface.
Reactive Inks
Inks formulated to react to stimuli by changing color
challenge counterfeiters and can be relatively easy for
consumers to recognize. The $20 bill features an optically
variable ink (OVI) that appears to shift from green to
black as light hits it from different angles. Thermochro-
matic inks, which change color in response to changes in
temperature, can be activated when a shopper puts her
thumb on a label. Other inks may fluoresce in the presence
of ultraviolet (UV) light or change colors when exposed to
specific chemicals.
Inks with highly specific spectral signatures can be
authenticated with photospectrometers, even when they
appear no different from counterfeiters’ inks to the naked
eye. Inks provide a good illustration of the difference
between overt and covert technology. Thermochromatic
inks are overt—the authenticating feature can easily be
seen by the public. Measuring the photospectral signature
of an ink requires a specialized reader and the knowledge
that the feature is included in the package, making it a
covert technology.
Taggants
Inks may also be impregnated with taggants, microscopic
markers that can be detected with various decoders. For
instance, a microscope can reveal cell-sized particles that
feature a company’s logo. A hand-held reader wand can
detect specific reflection, refraction or electromagnetic
properties of taggants in ink. One Australian wine produ-
cer even blended DNA from a grape vine into its label ink
as a very unique taggant.
Printing Techniques
High-quality printing is a classic anti-counterfeiting mea-
sure. Microprinting, or the creation of extremely tiny
characters such as the words ‘‘United States of America’’
around Andrew Jackson’s portrait on the U.S. $20 bill,
cannot be readily achieved by most small printers or
replicated on inkjets in a home counterfeiting operation.
Similarly, printing elaborate scrollwork and other ultra-
fine geometric patterns—called guilloche printing—can
challenge counterfeiters. Some anti-counterfeiting compa-
nies offer sophisticated guilloche patterns that reveal a
signal word, such as ‘‘VOID,’’ if a protected document is
photocopied. Digital ‘‘watermarks’’ can be embedded in
graphics and detected by scanners.
Holograms
A technology that got its start in the 1940s, holograms
have long been used to authenticate valuable items such
as credit cards, electronic parts, software, and even
clothing.
There are two types of holograms. Surface-relief, or
rainbow transmission, holograms are the familiar rain-
bow-colored images that are common in packaging be-
cause of their low cost and minimal thickness. Surface-
relief holograms are embossed onto thermoplastic film
backed with aluminum. Lippmann holograms—created
by capturing wave interference patterns on special photo-
graphic materials—offer the illusion of greater depth and
clarity, are more difficult to produce, and can include
multiple frames, microprinting, and extremely fine details
that can aid in authentication.
Holograms are interesting and attractive enough to
capture the eye of consumers, which offers some overt
brand protection, but the relative ease of counterfeiting
surface-relief holograms themselves mean that the most
effective security features on a good hologram are more
covert.
A variant on holograms, latent image technology, uses a
polarizer to reveal a very sharp, difficult-to-counterfeit
image.
Electronic
Tags
Integrated circuit (IC)
and radio-frequency identification
(RFID) technologies are catapulting anti-counterfeiting
ANTI-COUNTERFEITING PACKAGING 47
