Yam, Kit L. (ed.). The Wiley encyclopedia of packaging technology
Подождите немного. Документ загружается.

efforts to a new level. Tiny printed circuits can be affixed
to labels and packages to carry significant amounts of
information. Active RFID chips draw power from built-in
batteries to send a signal many meters; as battery tech-
nology becomes increasingly miniaturized and inexpen-
sive, active RFID will find many new uses in packaging.
Passive RFID chips do not require a power source—a radio
query from an electronic reader provides enough energy to
allow the chip to report its data—so they can be extremely
small, thin and inexpensive. Users may write new infor-
mation to IC tags, adding flexibility.
TAMPER EVIDENT FEATURES
Rather than try to reproduce a package, some counter-
feiters use a brand’s genuine packaging and refill it or
adulterate the ingredients. Any tampering with a package
represents a significant threat to both the brand and the
consumer, so anti-tampering features can be extremely
important elements of a brand protection campaign.
From the days of sealing wax, signet rings and signa-
tures across the folds of an envelope, tamper-evident
features have been employed. In many ways, the princi-
ples remain the same as they have been for centuries.
Induction Sealing
The familiar foil seals under the lids of products from pain
reliever to peanut butter utilize a technology called in-
duction sealing. A laminate consisting of a foil and poly-
mer on a paper backing is placed across the top of a plastic
or glass container. Passing the container and lidding
material under an electrical induction coil causes the foil
layer to heat up in response to the electromagnetic energy
supplied by the coil. The foil is released from the backing,
and the polymer (heated by the aluminum) fuses with the
lip of the container. The result is a tight seal. Any attempt
to access the contents of the container is immediately
apparent in the form of a torn or missing seal. Many
induction seals are designed to leave a residue on the lip of
the container, showing that the laminate has been
removed.
Tamper-Evident Films
Many polymer materials are employed as heat-sealed
wraps or capsules over lids to provide evidence of tamper-
ing. The materials are typically quite durable, but are
prone to ragged tearing or shattering when someone
applies force to them or attempts to slip them off of the
package. Many tamper-evident films also include holo-
grams or other anti-counterfeiting features to establish
authenticity and to ensure that they are not replaced with
a simpler shrink film after the package has been opened.
Tamper-Evident Labels
Some pressure-sensitive labels include a layer that re-
mains firmly adhered to the product, leaving behind
adhesive residue or displaying a message such as ‘‘void’’
if the label has been removed. Other labels are constructed
with perforations or built-in weak points to prevent their
intact removal. (This is often seen with price tags, which
are difficult to remove intact and move to a more expen-
sive product. The same benefits apply when preventing
the movement of wine or pharmaceutical labels to un-
authorized packages.)
RFID Chips
Radio-frequency identification (RFID) chips are playing
an increasing role in tamper protection. Active RFID
chips, which include a power supply, can sound a radio-
frequency distress call, including specific product or pack-
age information, if they have been tampered with. Passive
RFID chips, which respond to electronic queries from
readers, may provide evidence of tampering when they
are deployed on destructible labels—tampering destroys
the antenna, so the chip’s non-performance can serve as a
warning that tampering has taken place.
BIBLIOGRAPHY
1. The Economic Impact of Counterfeiting and Piracy, Organiza-
tion for Economic Co-Operation and Development, Paris
(2008).
2. B. Jones, ‘‘First Line of Defense,’’ Manufacturing Today Europe
(April–May 2007).
3. M. Noach and E. Feder, The Tadbik Group, personal commu-
nication (2008).
4. U.S. Department of the Treasury, ‘‘U.S. Treasury and Federal
Reserve Issue a New $20 Bill’’ (May 20, 1998).
5. Web site www.UsingRFID.com.
6. Web site of the BP Council (www.bpcouncil.org).
ANTI-FOG COATING
CRAIG D. BOYD
Sun Chemical
One of the main purposes of a package is to facilitate
consumers to repeatedly purchase the product that
it contains. Inks and coatings are continually expected
to have a higher contribution to the overall performance
and/or aesthetics of the package. There is possibly no
coating that offers a more significant effect on the
consumers’ acceptance of a package than the anti-fog
coating.
CONDENSATION PROBLEMS DEFINED
Whenever a sealed high-humidity environment is placed
through a temperature gradient, moisture condensation
will occur. This condensation will materialize as small
beads of water droplets forming on the inside of the
48 ANTI-FOG COATING
package. The droplets will begin to vaporize and then form
a fog on the inside of the package and transform a clear
package into an opaque one and thus conceal the contents
from the consumer. Most manufactures want the consu-
mer to see their product. This is normally achieved either
by packaging the product in a clear film or by printing a
pictorial of the product on the outside of the package.
Therefore, it is obvious that when a manufacturer
packages a product in a clear package, they expect that
it will remain clear. The produce manufacturer wants the
consumers to see the multicolor mixture of the green
lettuce, orange carrots, red cabbage, and radishes.
CONVENIENT, HEALTHY EATING
In order to attract more consumers, produce manufactures
are marketing the convenience of prewashed produce.
These packages are an attractive way of quickly preparing
a healthy meal. Today, there are literally dozens of differ-
ent salad and vegetable blends sold in the produce depart-
ment in the grocery store. These packages are sealed in a
plastic bag and the produce contains a relatively large
amount of moisture.
REFRIGERATION UNIT TYPES
There are two different classification of refrigeration units
used in the produce section of the grocery store. They are
either vertical or horizontal. Because cold air is denser
than warm air, the horizontal refrigeration units allow for
the entire package to maintain a consistent temperature
and therefore will not produce a great amount of conden-
sation. The main drawback of the horizontal unit is that it
conceals the packages from consumers as they navigate
through the produce section. Because of this, the horizon-
tal unit is not used very often any more. The vertical unit,
when positioned properly in the store, can allow consu-
mers to see products from virtually across the produce
section of the store. The largest problem with the vertical
unit is that it allows the refrigerated package to be in
direct contact with the warm ambient air in the store. This
configuration is a perfect design to allow the outside of the
package, the side facing the consumer, to be exposed to a
rather drastic temperature gradient. Thus vertical units
create a perfect environment for the generation of a great
amount of moisture condensation. In the vertical refrig-
eration unit, only the most outside package will experi-
ence condensation. Therefore, the greater the popularity
of the product, the less time any one package will set on
the outside of the unit. Ironically, the less popular the
product, the more the package will require an anti-fog
coating.
PRODUCE PACKAGE TYPES
Produce packages fall into two different categories. First is
the ‘‘pillow’’ pouch. These pouches are made from soft,
subtle films that will contain loose packaged produce. This
is the most popular package for produce. Lettuce and cole
slaw blends as well as baby carrots and small pieces of
vegetables such as broccoli flowerets are usually packaged
in the pillow pouch. These pouches have very little rigidity
and thus the package will contain many wrinkles to allow
for the water droplets to collect. The second type of
produce packages are vertical stand-up packages. These
vertical packages are made from very rigid films and will
not allow any wrinkles to form. These packages usually
contain more rigid produce like romaine lettuce or celery
stalks. In order to work properly, these packages will
utilize different types of anti-fog coatings. The rigid
packages will be printed with an anti-fog that will cause
the condensation to bead up and then gravity will draw
the water beads to the bottom the pouch where they will
do no harm. The pillow pouch will use an anti-fog that will
allow the water vapor to easily wet out the wrapper. This
way the moisture will not gather and will not create any
fogging. This type of anti-fog is required for the pillow
pouch because the wrinkles will cause the water beads to
gather and will eventually cause fogging. The coatings for
rigid pouches are designed to produce a dried coating that
will have a very low surface energy, while the coatings for
the pillow pouches are designed to have a very high
surface energy.
NOT JUST PRODUCE PACKAGING
Anti-fog coatings are used not only in produce packaging,
but often also in raw and prepared meat and poultry
packaging. For example, meat may be packaged in rigid
trays using modified atmosphere packaging (MAP), where
the trays are flushed with an optimum composition of
O
2
/N
2
and then sealed with a shrink film. These pack-
ages allow a greatly increased shelf life when compared to
the old-fashioned meat wrappers. However, these
packages can easily experience condensation and will
often need an anti-fog coating on the inside of the shrink
film. Preprepared meats have become very popular at
grocery stores. Rotisserie chickens are available at most
grocery stores. These chickens are placed in an opaque
rigid tray and then covered with a clear rigid bubble. The
inside of the clear bubble will often contain an anti-fog
coating.
NOT ALL REFRIGERATED PACKAGES REQUIRE ANTI-FOG
COATINGS
Any package where the film is in direct contact with
the product does not require an anti-fog coating no
matter how much moisture the package contains. When
the film is in direct contact with the product, no matter
how the structure of the refrigeration unit, there is no
area of air space that will allow the formation of conden-
sation. For example, an unwashed head of lettuce with a
film that is tightly wrapped around the lettuce will never
experience condensation and therefore will not require the
use of an anti-fog coating. Another example of a package
that will not contain an anti-fog coatings are meat
ANTI-FOG COATING 49
wrappers that are in direct contact with the meat. For
example, the old-fashioned styrofoam meat trays that are
tightly wrapped with a flexible film do not require an anti-
fog coating.
ANTI-FOG COATING APPLICATIONS AND
FORMULATIONS
Since anti-fog coatings are designed to deter condensation
formation on the inside of the package, these coatings are
always applied to the inside of the wrapper. Therefore, all
anti-fogs at a minimum have two things in common. They
must be FDA compliant for direct food contact and must be
designed to print and adhere to the untreated side of the
film. Since the inks are applied to outside of the film, the
treated side is always on the outside of the package. These
coatings are normally applied with at a very low coating
weight, resulting in a very thin film of o1mm. Anti-fog
coatings are tested for smudge resistance, block resis-
tance, and offset (transferring to the back side of the
film) resistance.
Some anti-fog coatings will affect packaging seal bond
strengths, while other will not. The coatings that do
interfere with seal bonds must be patterned out of the
heat seal areas. Anti-fog coatings can be difficult to reach
correct register because they are absolutely clear. There-
fore, the coatings that do not interfere with heat seal
bonds are more popular. The problem with these coatings
is that they are much more prone to offsetting, so achiev-
ing the correct film weight is essential with either coating.
Excessive coating applications can greatly interfere
with heat seals, while a lack of coating will not allow the
coating to reach its anti-fog potential.
Anti-fog coatings for flexible films are normally applied
using the flexographic process; coatings for rigid dome lids
for the precooked poultry are normally spray applied.
CONCLUSION AND REVIEW
Some flexible substrates are manufactured with anti-fog
agents added to them. These films will only provide
marginal anti-fog properties because the active agents
are diffused throughout the entire thickness of the film,
while the coatings are concentrated on the surface. There-
fore, the use of anti-fog coatings are typically required for
high-performance applications.
There is far more to anti-fog coatings than immediately
meets the eye. Whether a package receives an anti-fog
coating is always the decision of the manufacturer. This
decision is based on the type of the product, the type of
refrigeration unit, and the turnaround time of the pro-
duct. The requirements for the correct type of anti-fog are
mostly dependent on the type of product and package. The
purpose of anti-fog coating is to allow the clear film to
remain clear so that the consumer can easily see the
colorful and wholesome product that the package con-
tains. The main use of anti-fog coatings is a matter of
manufacturers wanting to increase market share and
product differentiation.
ANTIMICROBIAL PACKAGING
S. Y. CHO
Institute of Life Sciences and
Biotechnology, Korea
University, Seoul, Korea
D. S. LEE
Department of Food Science
and Biotechnology, Kyungnam
University, Masan, Korea
J. H. HAN
PepsiCo Fruit and Vegetable
Research Center, Frito-Lay Inc.,
Plano, Texas
INTRODUCTION
In the past, the functions of packaging had been limited to
the roles of containment, protection of its content from
environmental effects, consumer convenience, and com-
munication of the product information. While the conven-
tional functions of packaging was considered as passive,
the new paradigm of packaging—called active packa-
ging—has been emerged. The active packaging may be
defined as a packaging system in which the product, the
packaging, and the environment interact in a positive way
to extend shelf-life or to achieve some characteristics that
cannot be obtained otherwise (1). Antimicrobial packaging
is a form of active packaging that can control the growth of
microorganisms on the surfaces of foods and packaging
materials and eventually reduce cell counts in the
products.
Antimicrobial function of packaging materials can be
achieved by providing unfavorable environmental condi-
tions to microorganisms by eliminating growth require-
ments, rendering direct contact of microorganisms to the
immobilized antimicrobials on the packaging material
surface, or transferring antimicrobial agents originally
incorporated into the packaging materials. With the above
actions, antimicrobial packaging extends the shelf life of
food and secure consumer safety by controlling spoilage
and pathogenic microorganisms in foods.
According to the Flexible Packaging Association (2), the
market size of flexible packaging in the United States in
2007 is about $23,500 million, garnering 18% $130,000
million packaging market. The largest market for flexible
packaging is food industry (retail and institutional) ac-
counting for over 57% of shipment. Active role of food
packaging has been empathized for sustainable develop-
ment of food packaging industry. Antimicrobial packaging
would make the food packaging industry possible to
achieve an innovative development with the aid of reg-
ulatory requirements such as food product liability and
HACCP. Therefore, research and development trends of
antimicrobial packaging systems may progress to achiev-
ing more effective antimicrobial activity with broader
spectrum, utilizing natural extracts and creating new
applications.
50 ANTIMICROBIAL PACKAGING
A BRIEF HISTORY OF ANTIMICROBIAL PACKAGING
Early Works on Antimicrobial Packaging
As an early stage application of antimicrobial packag-
ing, shelf-life extension of perishable product had been
achieved by applying modified atmosphere packaging
(MAP) technologies that provided adverse environments
of microbial growth. MAP eliminates oxygen level and
elevates the concentration of carbon dioxide to inhibit the
growth of aerobic microorganisms.
The antimicrobial polymeric materials were first intro-
duced to protect biomedical devices from microbial con-
tamination in Japan (1). A considerable progress in
antimicrobial packaging technology has been made in
1990s, and various antimicrobial products was developed
and commercialized in household goods, textiles, surgical
implants, biomedical devices, and food packaging materi-
als. The progress could be attributed to the development
of inherently antimicrobial polymers and also to the
development of antimicrobial agent that was incorporated
into polymer matrix. For example, chitosan-based poly-
mers and copolymerized acrylic with protonated amine
co-monomer have been developed as antimicrobial packa-
ging materials (3). Preservatives with antimicrobial
activity play an important role in preventing microbial
contamination. Many of these agents such as silver sub-
stituted zeolite, potassium sorbates, sodium benzoate,
propionic acid, and acetic acid have been successfully
incorporated directly into packaging materials to control
the microbial contamination (4, 5).
Current Works on Antimicrobial Packaging
The use of chemical preservatives as antimicrobial agents
caused public concerns on the potential risks of chemical
preservatives that might migrate into food products.
These agents are categorized as food additives and con-
trolled by legislation.
Although the chemical preservatives in packaging ma-
terials had been carefully regulated by domestic autho-
rities, the increased consumer demand for preservative
free foods has rendered the development of antimicrobial
packaging with natural antimicrobial agents. The natural
antimicrobials and GRAS antimicrobials may include
bacteriocins, enzymes, plant extracts, and natural essen-
tial oils (5, 6). They hold a great potential and represent
excellent activities for controlling microbial contamina-
tion. With the effort of finding safe antimicrobial agents,
considerable studies have been exerted on developing
antimicrobial biopolymer matrix as a carrier for natural
antimicrobial agents (6).
ANTIMICROBIAL PACKAGING SYSTEM
Antimicrobial packaging is a system that is designed
to control the growth of microorganisms by extending
the microbial lag phase and by reducing the growth rate,
thereby extending the shelf life of perishable products
and enhance the safety of packaged products (4). Anti-
microbial packaging can be constructed using antimicro-
bial packaging materials and/or antimicrobial agents.
Antimicrobial packaging systems can be classified into
three types according to the mode of antimicrobial agent’s
action: absorption, release, and immobilization (5). The
first type is the packaging materials containing antimi-
crobial agents that eliminate oxygen and moisture in the
packaging system by absorption. This type of packaging
controls the growth of microorganisms by providing
unfavorable conditions to cell growth. The second is the
packaging materials containing antimicrobial agents that
migrate to the surface of food materials. The antimicrobial
action is achieved by the release of the antimicrobial
agents from the packaging material. The third type is
those containing antimicrobial agents without migration.
This type of packaging needs direct contacts between
packaging materials and food product since the antimi-
crobial agents in the packaging material does not release
into foods.
The antimicrobial agents may be coated, incorporated,
immobilized, or surface modified onto the packaging mate-
rials according to the characteristics of the antimicrobials
and to the antimicrobial mechanisms how the agents are
working (6). Thermal polymer processing methods such as
extrusion and injection molding are used for heat-stable
antimicrobial agents like silver substituted zeolites. Mean-
while, solvent compounding methods may be a more sui-
table method to combine the antimicrobial agents and the
packaging materials for heat-sensitive antimicrobials such
as enzymes and volatile compounds (7).
Antimicrobial Packaging Containing Oxygen Absorbing
Agent
Modified atmosphere packaging (MAP) with the absence
of oxygen along with elevated concentration of carbon
dioxide has been used for controlling the aerobic micro-
organisms that are responsible for food spoilage. Gener-
ally, oxygen concentrations of 0.1% v/v or less in package
headspace are required to inhibit the growth of aerobes
(1). However, MAP does not guarantee this anaerobic
condition in the packaging system throughout the shelf
life since the flexible packaging materials show consider-
able gas permeabilities. Introduction of oxygen absorbing
agent in the packaging system rendered active control of
oxygen level in the package headspace suitable for anti-
microbial purpose by removing oxygen molecules.
In general, oxygen scavenging technologies are con-
structed by the oxidation of oxygen absorbing agents: iron
powder, ascorbic acid, photosensitive dyes, enzymes (e.g.,
glucose oxidase and alcohol oxidase), unsaturated fatty
acids (e.g., oleic acid or linoleic acid), and immobilized
yeast on a solid substrate (8). Iron-based material is
known to the most effective oxygen scavenging system
among the above substances. It has been reported that 1 g
of iron will react with 300 ml of oxygen (8). The oxygen-
absorbing antimicrobial packaging has been successfully
commercialized in bakery, pasta, and meat products by
applying the agents enclosed in sachets or incorporated in
polymer matrix. Recently, diversified matrices for oxygen
absorbents are available in film, tray, label, and closure
liner as well as sachet (8). In spite of the strong anti-
microbial activity of the oxygen scavenging system
ANTIMICROBIAL PACKAGING 51
against aerobic microorganisms, this system does not
possess the effective activity against anaerobic micro-
organisms.
Antimicrobial Packaging Containing Antimicrobial Agent
that Migrate into Food
The packaging materials may act as a carrier for anti-
microbial agents to perform their active role to control
microorganisms. Some of the antimicrobial agents may
be coated or directly incorporated into the packaging
materials and subsequently migrate to the food system.
The antimicrobial action is achieved by release of the
antimicrobial agents from the packaging material. The
released antimicrobial agents control the growth of micro-
organisms by altering cell membrane properties or by
inhibiting essential metabolic pathways of the microor-
ganisms (4–6).
Most spoilage incidents occur primarily at food surface
by the contamination of aerobic microorganisms. The
concentration of the antimicrobial agents above their
minimal inhibitory concentration (MIC) is required to
impart antimicrobial function. Without the antimicrobial
packaging concept, the excess amount of preservatives
such as benzoates and sorbates should be included in foods
to control the spoilage microorganisms. Thus, releasing
antimicrobial additives to the food surface conveniently
increases the additives concentration in the food surface
above the MIC while maintains the preservative concen-
tration inside the food at sufficiently low level (4). Con-
sidering that the use of preservatives for shelf-life
extension has been strictly controlled by food safety
authorities, antimicrobial packaging is advantageous in
reducing potential risks of consuming excess amount of
food preservatives.
An additional advantage of antimicrobial packaging is
the sustainable antimicrobial activity. The antimicrobial
agents initially included in food ingredients might be
inactivated by interacting with other food components.
For example, bacteriocins and enzymatic antimicrobial
agent applied in the foods or onto the food surface may
interact with proteolytic enzymes in food and may cause
the loss of antimicrobial activities. On the contrary, in-
corporation of the above substances in packaging films did
not cause the loss of antimicrobial activity, controlled the
release of the antimicrobial agents, and maintained anti-
microbial activity for fairly long periods.
Nonvolatile Migration. The incorporated antimicrobials
are migrated to the food surface in either solute or gas
states. The migrating solutes are nonvolatile materials
such as organic acids and their salts, enzymes, bacterio-
cins, fungicides and some of natural extracts, while the
gases are volatiles such as alcohols, small phenolics,
aldehydes, and others (4–6, 8). They moved from the
film matrix to the food surface and diffused or dissolved
into the food. Diffusion is a primary mechanism of non-
volatile solute transfer in the film matrix, in which relates
to the release rate. The migration kinetics of nonvolatile
solute followed the Fick’s second law of diffusion, where
diffusion coefficient depends on the type of film materials,
microstructural voids in film matrix, and environmental
temperature (9, 10). The migration of the antimicrobial
agents in the film to the food surface requires directly
contact. The contact between the film matrix and the food
surface throughout the shelf life should be assured for
their migration and, consequently, their antimicrobial
action. For this, the food should be a continuous matrix
without any factors that interfere with the diffusional
migration. This food matrix could be a liquid solution,
a semisolid paste, or a smooth solid matrix without
significant pores, holes, or heterogeneous particles.
The antimicrobial agents in the food surface will move
into the center of the food by diffusion or dissolution. The
solubility and diffusion coefficients of the agent in the food
are very important factors that govern the rate of agent
removal in the food surface. The antimicrobial concentra-
tion in the food surface could be maintained above the
MIC for their effectiveness in controlling the microbial
growth when the agent transfer in the food and films are
balanced (5).
Volatile Migration. The migrating antimicrobials could
be gaseous compounds released from a gas emitting
material. The gaseous antimicrobials can be migrated
into the food surface and its packaging headspace or into
the air gap between the package and the solid food (4).
The migrating volatile gases include SO
2
, ethanol,
essential oils, and a component of natural herb and spice
extracts. The advantage of using volatile antimicrobial
agents is that the packaging film does not need to contact
directly to the food surface. Therefore, food matrix in this
system could be highly porous foods, powdered foods,
particulate foods, or shredded and irregularly shaped
foods. Sometimes, however, the use of volatile antimicro-
bial is limited by the incompatibility of the agent with the
packaging materials, or by the loss of volatile antimicro-
bials during incorporation into packaging by extrusion or
coating (5).
The volatile antimicrobial agent initially incorporated
in the packaging film or sachet material is transferred to
the film surface and subsequently evaporated to the
packaging headspace. The vaporized agent in the head-
space is then absorbed to the food surface. Eventually, the
agent concentration in the food surface is equilibrated
with the volatilized antimicrobials in the headspace (4).
For the antimicrobial efficacy of the agent, the agent
concentration in the food surface should be maintained
above the MIC of target microorganisms. The concentra-
tion of migrated agent in the food surface is determined by
the release rate of the volatile agent from the packaging
material and by the absorption rate of headspace volatile
agents into the food. The release rate of the agent from the
packaging material to the headspace depends on the
volatility of the agent which represents the partitioning
behavior between packaging material and headspace. The
absorption rate of the agent into food depends on the
solubility of the agent into the food ingredient. Therefore,
the composition of the food is a very important factor of the
agent’s solubility to the food materials. In general, the
volatile agents are more soluble in lipid foods than in
hydrophilic food materials.
52 ANTIMICROBIAL PACKAGING
The desired antimicrobial concentration of a food sur-
face can be attained by controlling the release rate of
antimicrobial agent from the packaging material surface.
When we use an appropriately selected film layer that has
a specific permeability of the volatile substance, the anti-
microbial’s release rate could be controlled efficiently. The
microencapsulation of the volatile substance with appro-
priate wall materials may control the release of antimi-
crobials. This encapsulation method can also provide a
solution against the loss of volatile antimicrobials during
packaging material fabrication processes.
Antimicrobial Packaging without Antimicrobial Agent
Migration
Antimicrobial packaging can also use agents that are not
migrated into the food. The nonmigrating antimicrobial
packaging system can be achieved using inherently bioac-
tive polymers or developing novel packaging materials
where the antimicrobials are attached to the nonactive
films by either covalent bond or ionic immobilizations (4).
In this antimicrobial packaging system, many antimicro-
bials are enzymes or chemicals that are directly and
indirectly participated in the microbial inactivation func-
tion. They participate in the biological reactions that lead
to the microbial inactivation or produce biologically active
radicals, ions, and reactive singlet oxygen that may induce
antimicrobial effects.
Since the biologically active compounds are not mobile,
the incorporation of the antimicrobials in the matrix
during film fabrication processes is not considered neces-
sary. Although the incorporated antimicrobials uniformly
distributed in the film matrix, their activity is limited to
the film surface only. Therefore, imparting a biologically
active ingredient to the film surface would be sufficient to
exhibit the antimicrobial efficacy of the nonmigrating
antimicrobial packaging film. Therefore, assigning an
antimicrobial activity without migration of antimicrobials
can be performed by immobilizing nonmigrating antimi-
crobials on the polymer film surface, by coating a very thin
layer of active matrix on to the packaging film, or by
attaching antimicrobial compounds covalently on the film
surface with the aid of multifunctional ligands (11).
The active materials in the film surface may catalyze
antimicrobial action or provide sufficient energies to form
ions and radicals on the food surface which are capable to
control microbial growth. Therefore, this type of antimi-
crobial film is particularly effective for the packaging with
liquid food and is advantageous for regulatory com-
pliances. Since antimicrobial agents do not migrate into
the food system, the antimicrobial agents that are not
permitted as food ingredient and food additives may be
used for this purpose as food contact substances.
Inherently Antimicrobial Packaging Materials. Some
polymers are inherently antimicrobial, and they have
been utilized in many fields currently such as biomedical
instruments, filters, membranes, and packaging materi-
als. Cationic polymers such as chitosan, poly
L-lysine,
lysozyme, and numerous synthetic polymers that have
quaternary ammonium, phosphonium, and biguanide
compounds have exhibited to be effective antimicrobial
polymers.
It has been well known that chitosan (a natural poly-
saccharide), and its derivatives have an antimicrobial
effect due to the presence of a positively charged ammo-
nium group. Besides the natural antimicrobial polymers,
antimicrobial synthetic polymers have been produced by
polymerizing biologically active monomers. The widely
used biologically active materials for synthetic polymers
are the cationic compounds including quaternary ammo-
nium compounds (QACs), biguanide groups, quaternary
pyridinium compounds, phosphonium compounds, sulfo-
nium compounds, and 2-(4u-thiazolyl) benzimidazol (TBZ)
(Table 1). The functional groups are linked to reactive
groups of monomers and, in turn, have been polymerized
to form synthetic antimicrobial polymers (Figure 1).
Therefore, most synthetic antimicrobial polymers can be
understood as polymerized biocides, and the polymer
backbone has the multiplied activity of the antimicrobial
functions of attached biocides. Many of these polymers are
amphiphilic and positively charged.
The antimicrobial action of polycationic compound has
been considered as the disruption of cytoplasmic mem-
brane of bacterial cell (13). Polycationic antimicrobial
polymers are absorbed onto the negatively charged bacter-
ial cell surface at physiological pH by electrostatic inter-
action. The absorbed polycations are bound to cytoplasmic
membrane, and disrupt it. The consequent leakage of
potassium ions and other cytoplasmic constituents lead
to cell death. In particular, the mode of antimicrobial
action of QACs is the damage of cytoplasmic membrane
of bacteria through surfactant-like interaction resulting in
the loss of permeability properties of the membrane (14).
This means that the functional groups of the antimicrobial
polymer must diffuse through the microbial cell wall, be
water soluble, and have no interaction with the microbial
cell wall. For this, spacer molecules that link the biologi-
cally active agents to the polymer backbone are required to
allow the agent to have sufficient freedom of motion when
the polymer backbone does not act as the spacer (11).
Physical modification of polymers often produces biolo-
gically active packaging films. Ultraviolet or electron beam
irradiation on polyamide films increases positively charged
amine concentration on the film’s surface, resulting in
enhanced cell adhesion potential. However, this type of
film has limited microcidal effect, and microbial adsorption
on the film surface diminishes antimicrobial activity. In-
corporation of antimicrobial agents into this film matrix
may overcome these drawbacks successfully (7).
Immobilized Antimicrobial Agents in the Packaging Film
Matrix. The immobilized antimicrobial agents are not
removed from polymer surfaces. Various immobilization
techniques have been developed to overcome the contact
problems of antimicrobial residues to food surfaces.
The antimicrobial materials used for immobilization
include the hydrolysis product of a quaternary amine-
containing organosilicon salt, hexachlorophene, acrifla-
vine, antibiotics such as streptomycins and gentamycins,
and antimicrobial enzymes including lactoferrin, sulfhy-
dril oxidase, and bile-salt- stimulated lipase. The changes
ANTIMICROBIAL PACKAGING 53

in conformation and denaturation of proteins and peptides
may result in reduced antimicrobial activities of antimi-
crobial enzymes. The active site should be protected
during the film formation.
INGREDIENT OF ANTIMICROBIAL PACKAGING
Film Matrix
The antimicrobial packaging system is comprised of film
matrix and antimicrobial agents. In considering the
biologically active role of the antimicrobial agent in the
film, the relationships between the film matrix and the
antimicrobial agent would be critical, since thermal de-
gradation of the antimicrobial agent during film fabrica-
tion and its chemical compatibility with the film material
may limit the antimicrobial activity. Synthetic polymeric
packaging materials are produced by thermal processing
method such as extrusion. Thermally stable antimicrobial
agents should be incorporated. In general, synthetic flex-
ible films consist of many layers of different films. Low-
density polyethylene (LDPE) has been used for the film
layer that is in direct contact with the food. Therefore,
LDPE has been most widely used packaging materials
among the synthetic polymers for antimicrobial agent
carriers. For further modification of the contact film layer,
poly(ethylene-co-methacrylic acid) (PEMA) has also been
explored as a film matrix (15).
Thermally unstable antimicrobial agents cannot be
used with extruded synthetic polymer films, but they
can be applied to the biopolymeric film matrix instead.
In general, biopolymer films are formed by removing the
solvent from the film solution in the cast plate. Therefore,
they do not require high temperature for polymer melting.
Examples of biopolymers are hydrocolloids including poly-
saccharide and protein, lipid, and the composite of hydro-
colloid and lipid. For antimicrobial purposes, the use of
biopolymeric films such as chitosan, soy protein, whey
protein, corn zein, methyl cellulose (MC), and hydroxy-
propyl methyl cellulose (HPMC) films with various anti-
microbial agents have been reported (16).
Antimicrobial Agent
The widely used method to inhibit the growth of undesir-
able microorganisms is the use of chemical agents exhibit-
ing antimicrobial activity. These chemicals may be either
synthetic compounds intentionally added to foods or natu-
rally occurring, biologically derived substances. The selec-
tion of an antimicrobial agent depends primarily on its
activity against the target microorganisms, compatibility
with the packaging material, and the heat stability during
the thermal process (4–6).
Table 1. A List of Synthetic Antimicrobial Polymers with Their Active Compounds (12)
Active Compound Biologically Active Polymer
Quaternary ammonium
compounds (QACs)
Poly[{dimethylalkyl-3-(and -4-)vinylbenzyl}alkylammonium chlorides]
Polyionenes (in-chain QAC of p-xylene spacer, in-chain QAC of hexamethylene spacer)
Hydroxyterechelic polybutadienes carrying covalently-bonded QAC
Polysiloxanes with pendant primary alcohol groups
Biguanide group Poly(hexamethylene biguanide hydrochloride)s
Polymer of methacrylate containing biguanide units
Quaternary pyridinium salt 4-[2,29-Bis(ethoxycarbonyl)ethyl]-1-alkylpyridinium bromide
Poly[iminoethyleneimino-2-[(1-butyl)pyridine-4-ylmethyl]malonyliminoethyleneiminoadipoyl bromide]
Phosponium salt Poly{trialkyl-3-[(and-4-)vinylbenzyl]phosphonium chloride}
Poly([ethylenebis(diphenylphosphonio)]ethylene dibromide)
Poly{[4-(2-tributylphosphonioethyl)styrene chloride]-co-4-(2-chloroethyl)styreme}
Poly{[4-(3-tributylphosphoniopropyl)styrene chloride]-co-4-(3-chloropropyl)styreme}
Sulfonium salt Poly[p-vinylbenzyltetramethlenesulfonium tetrafluoroborate]
2-(4u-Thiazolyl) benzimidazol
(TBZ)
Poly[N-Acryloyl-2-(4u-thiazolyl)benzimidazol]
Poly[2,4,4u-trichloro-2u-acryloyloxydiphenyl ether]
CH
3
CH
3
N
+
Cl
−
R
CH
2
CH=CH
2
CH
3
Br
−
CH
3
N
+
CH
2
CH
2
CH
3
CH
3
N
+
m
m
(CH
2
)
6
n
(CH
2
)
6
NH C NH
NH
NHC
NH
HCl
(a)
(b)
(c)
(d)
Figure 1. Monomers with antimicrobial activities. (a) Dimethy-
lalkyl-3-(and -4-)vinylbenzyl ammonium chlorides, (b and c)
polyionenes, and (d) poly(hexamethylene biguinide hydrochlor-
ide) (12).
54 ANTIMICROBIAL PACKAGING
Currently, synthesized chemical preservatives have
been widely employed to control the number of micro-
organisms in the packaged food, but increasing consumer
awareness of potential health risk associated with the
synthetic agent requires a substituent that is available in
the nature. The natural antimicrobial agents such as
bacteriocins and spice extract have long been used as
constituents of foods and showed negligible toxicity.
Therefore, natural antimicrobial agents are on the way
of replacing the synthetic antimicrobial agents as food
preservatives for direct deposition and also as ingredients
of packaging materials for migration into food.
Organic Acid. Sorbic acid, benzoic acid, propionic acid,
and their salts have been widely used for preventing
microbial deterioration of food. They are on the food
additive list in many countries and have been used under
the control of regulation for their potential health risk.
High polarity of organic acids causes them to be incompa-
tible with the apolar LDPE. Acid anhydrides were thought
to be more compatible than free acids and their salts
because of their lower polarity. Organic acid antimicro-
bials could be incorporated into the biopolymeric film for
their compatibility.
Inorganic Metallic Ion.
Silver Ion. Silver ions in the microbial cell inhibit
metabolic enzymes and have strong antimicrobial activity.
Silver ions are generally incorporated into polymer films
in the form of silver substituted zeolite, an ionic bonded
metallic compound in which the sodium ions present has
been substituted with the silver ions. Silver-substituted
zeolite is thermo-stable and can be applied to the thermal
process of synthetic polymers like polyethylene, polypro-
pylene, and nylon as well as biopolymeric films (11, 17,
18). Silver ions are released from the silver-substituted
zeolite incorporated in the film matrix to the food materi-
als for antimicrobial action (18).
Titanium Ion. Titanium dioxide (TiO
2
) is nontoxic and
has been approved by the U.S. Food and Drug Adminis-
tration for the use in foods and food contact materials (19).
Ultraviolet energy shifts the energy level of TiO
2
to the
excited state. On its way back to ground state, TiO
2
releases absorbed energy to the food material (20). The
released energy may form very reactive radicals and
singlet oxygen which have antimicrobial function. In this
system titanium ion is not a migrating agent but trapped
in the film structure (20). Currently there is considerable
interest in the self-disinfecting property of TiO
2
to satisfy
hygienic design requirements in food processing equip-
ment surfaces (21, 22).
Natural Compounds. The naturally occurring antimi-
crobials are enzymes, peptides, oils, and other materials
that have been present self-defense functions against
microbial contamination. Typical examples are bacterio-
cins in lactic acid bacteria, lysozymes in egg white,
flavonoids in plant extracts, and essential oils.
Bacteriocins. Bacteriocins are small bacterial peptides
that show strong antimicrobial activity against closely
related bacteria. Antimicrobial effectiveness of bacterio-
cins in food is related to specific antimicrobial activity of
the bacteriocin, amount of the bacteriocin used, number of
microorganisms, process conditions, interaction with food
components, and pH and temperature of the product (23).
Nisin is a polypeptide produced by Lactococcus lactis
spp. It has been approved as a food additive with GRAS
status in over 50 countries worldwide. It has a relatively
broad spectrum of antimicrobial activity against various
lactic acid bacteria and other Gram-positive bacteria (24).
It is particularly effective against heat-resistant bacterial
spores of Clostridium botulinum (25). It is not effective
against gram-negative bacteria because it cannot pene-
trate outer lipid membrane of cell to the cytoplasm (24).
Use of nisin in conjunction with ethylenediamine tetra-
acetic acid (EDTA) may increase the antimicrobial effec-
tiveness against Gram-negative bacteria (26). Nisin has
been incorporated into biopolymer films such as soy
protein, corn zein, chitosan, and methylcellulose films
for antimicrobial edible coating applications (27, 28). The
thermal processing in film production may reduce anti-
microbial activity of nisin (28).
Lysozyme. Lysozyme, also known as muramidase or N-
acetylmuramichydrolase, is a relatively small enzyme
from hen egg white. Lysozyme is effective against spoilage
and pathogenic microorganisms by disintegrating the cell
wall structure. Antimicrobial activity of lysozyme is at-
tributed to the function that catalyzes the hydrolysis of
the b-1, 4 glycosidic bond between N-acetylmuramic acid
and 2-cetyl-amino-2-deoxy-
D-glucose residues in the bac-
terial cell wall (29, 30). Lysozyme demonstrates a strong
antibacterial potential against Gram-positive bacteria
such as Listeria monocytogenes (30). Lysozyme has been
applied to biopolymer films such as chitosan, whey pro-
tein, fish gelatin, corn zein, and sodium alginate films in
either purified or unpurified forms alone and in combina-
tion with EDTA and other bacteriocins (31, 32). Antimi-
crobial activity of immobilized lysozyme in polyvinyl
alcohol film also has been reported against various spoi-
lage and pathogenic bacteria (33).
Plant Extracts. Antimicrobial activity of phytochemicals
in the extracts of spices and herbs has been demonstrated
by many researchers (5, 6, 31). In many cases, the
concentration of biologically active component in spices
and herbs and their essential oils are too low to be
effectively used for packaging applications due to their
limited sensory acceptance at high concentration level
(23). They may be used in junction with other biologically
active agents and contribute to total hurdle technology
system for microbial control. Many phytochemical com-
pounds in essential oils and extracts responsible for anti-
microbial activity are phenolic compounds.
1. Essential Oils. Plant extracts of coffee, green tea,
spices, and herbs (i.e., cinnamon, cloves, mustard
seed, oregano, rosemary, thyme and vanillin) have
been evidenced for their antimicrobial activity
against a wide spectrum of microorganisms (5, 34).
The antimicrobial activities of the spices are
ANTIMICROBIAL PACKAGING 55

associated with phenolic compounds in their essen-
tial oil fraction. The antimicrobial essential oil com-
ponents of cinnamon and cloves are eugenol and
cinnamaldehyde, respectively. Terpenes such as car-
vacrol, p-cymene, and thymol are the major volatile
components of oregano and thyme (23). Many essen-
tial oils are volatile and can be applied to volatile
migration when they are incorporated into the
packaging film (31).
2. Allyl Isothiocyanate (AIT). Allyl isothiocyanate is a
naturally occurring nonphenolic volatile compound
responsible for the flavor of horseradish, wasabi,
and mustard. Volatilized AIT has an effective anti-
microbial activity against variety of pathogens when
used at low concentration. AIT causes metabolite
leakage by affecting cell membranes (35). Antimi-
crobial activity of AIT-incorporated gelatin film,
nylon 6,6 film, and PVDC/PVC copolymer film has
been reported against spoilage and pathogenic mi-
croorganisms (36).
COMMERCIAL APPLICATIONS
Commercialized Antimicrobial Packaging
Even though many research works have been conducted
all over the world, there are only a few commercialized
products of antimicrobial food packaging materials. This
is because of strict hygienic regulation on food packaging,
high price, and limited consumer perception and accep-
tance on their effectiveness. The area of food preparation
and utensil products has less strict regulation in applying
the antimicrobial materials into real practices and has
seen a higher number of commercial products—for exam-
ple, kitchen board and gloves. Table 2 lists some examples
of commercial products available in market. The list is not
exclusive and covers only those observed in the writers’
eye for the last 20 years. Some products available in a
country or area may not be allowed for sale in other
countries because of the difference in food safety
regulation.
In a practical sense, ethanol emitter and plant extracts
have acquired wider acceptance, depending on people’s
attitude and corresponding country’s legislation. Some
ethanol emitters combine the oxygen-scavenging capacity
to inhibit aerobic spoilage organisms. Ag-containing cera-
mic materials have been applied in plastic films and
containers where people generally recognize silver as
hygiene-improving and antitoxic material. SO
2
generator
may be used in postharvest handling and storage of
agricultural products, where agricultural safety practices
are applied. A ClO
2
-generating plastic tube, Knick’n’-
clean
s
, which is activated by bending to mix two fluids
in it, is marketed for the use in refrigerators for a limited
time period of about 30 days. A ClO
2
-emitting film (Micro-
Gardet) claimed to produce antimicrobial microatmo-
sphere for 10 days keeping freshness of foods and
breaking down ethylene gas. Until 2008, ready-to-eat
food applications of this system await FDA clearance in
the United States. Triclosan is not approved as a food
additive and is incorporated into a plastic master batch,
which is converted into food-contact household equip-
ments such as cutting board and hand gloves.
Edible coating being a component of food may be
applied for antimicrobial function when it contains safe
Table 2. Some Commercialized Antimicrobial Packaging or Food-Contact Materials in Food Storage, Packaging,
and Preparation
Active Compound or Releasing
Compound Matrix or Form Application Trade Name and/or Producer
Ag-substituted zeolite or
zirconium
PVC, LLDPE, PE, rubber Wrap, film, kitchen board,
kitchen glove
Agion
s
(Agion Technologies,
USA), Zeomict (Sinanen
Zeomic Co., Japan),
Cleanaidt (Gyunghyang
Industrial Co., Korea)
Chlorine dioxide Polyolefin Film, compartmented stick for
refrigerator
MicroGardet (BarrierSafe
Solutions International Inc.,
USA), Knick’n’clean
s
(Helrik Bobke, Germany)
Ethanol Silicon dioxide Sachet Ethicapt (Freund Corp.,
Japan), Agelesst type SE
(Mitsubishi Gas Chemical.,
Japan)
Sulfur dioxide Laminated plastic sheet with
Na
2
S
2
O
5
Sheet or pad for postharvest
storage of grape fruits
Uvasyt (Grapetek, South
Africa)
Triclosan Polymer, rubber, etc. Food preparation equipment
and supplies, kitchen sponge
and gloves
Microban
s
(Microban
International Ltd., USA),
Ultra-Fresh
s
(Thomson
Research Associates,
Canada)
Wasabi (Japanese horseradish)
extract
Encapsulation in cyclodextrin Coated PET film, tablet Wasapowert (Sekisui Plastics
Co., Japan)
56 ANTIMICROBIAL PACKAGING
bioactive compounds, organic acids, or edible essential oils
of plant or spice origins. Because the coating may be
understood as a food component, there would be no barrier
of application in the regulations if all active/inactive
ingredients are food grade. The potential edible coating
matrices include chitosan, hydroxy propyl methyl cellu-
lose, and alginate gels (29).
Effectiveness of Antimicrobial Food Packaging
Many studies showed potential benefits of antimicrobial
packaging for fresh fruits and vegetables, milk, beverages,
cheese, ham, meat and fish (37, 38). Packaging materials
with nisin, chitosan, or acids typically resulted in mar-
ginal microbial count reduction of 1.5–2.5 log10 compared
to control (37). The reduction or suppression of microbial
growth by the antimicrobial packaging is varied too much
to conclude that any specific system or condition is uni-
versally optimal or better than others. It is generally
accepted that the results in microbial media or buffer
solutions cannot be directly applied to the real food
applications. Conducting test with real food sample is
needed for practical or commercial application.
The effectiveness of microbial inhibition by antimicro-
bial packaging is generally improved when combined with
other preservation hurdles such as modified atmosphere,
low pH, high-pressure treatment, and low temperature
(38–40). Analysis of microbial spoilage kinetics with anti-
microbial packaging showed that the microbial growth was
suppressed more with lower storage temperature (41). The
microbial inhibition of antimicrobial packaging at the low
temperature could be represented by low cell concentra-
tion, extended lag time, and reduced growth rate.
When the temperature dependence of microbial lag
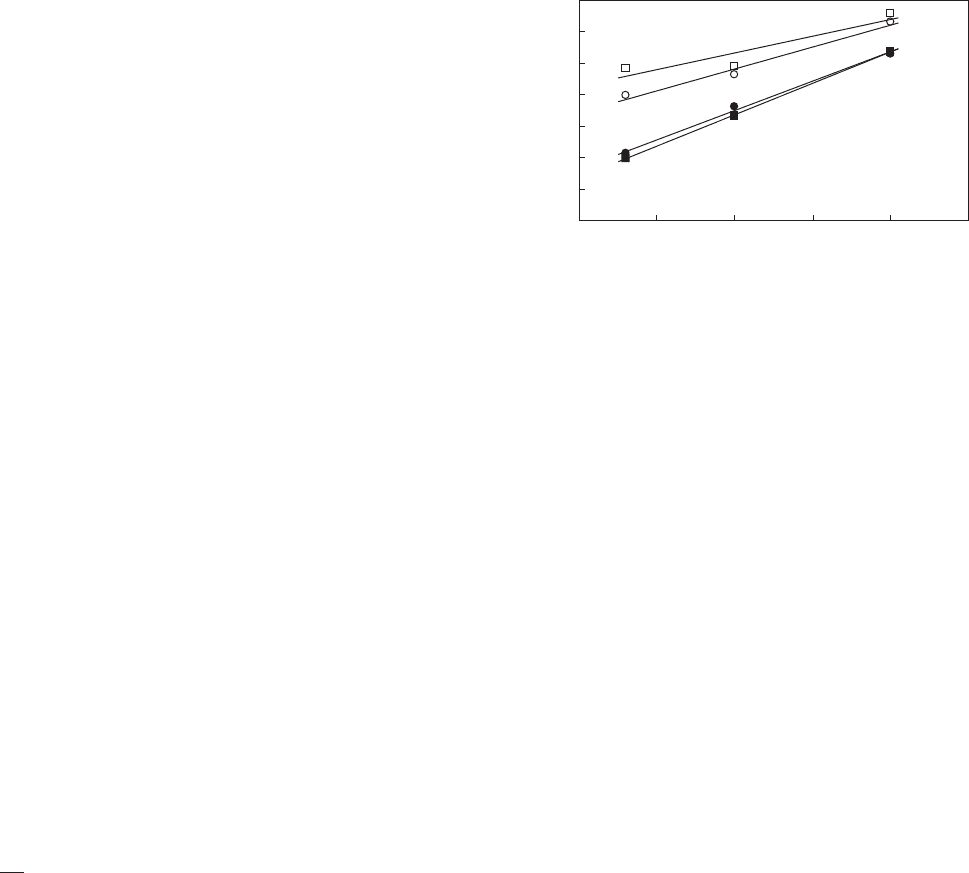
time was analyzed by square root kinetic model [equation
(1)], antimicrobial packaging showed higher temperature
dependence (increased b value) with increase of hypothe-
tical minimum growth temperature (increased T
min
) (41):
ffiffiffiffiffiffiffi
1
t
lag
s
¼ b ðT T
min
Þð1Þ
where T is the temperature (in 1C), b is slope parameter
representing temperature effect, and T
min
is respective
theoretical minimum temperatures for growth estimated
by extrapolation of the regression line to the temperature
axis (Figure 2).
The effectiveness of antimicrobial packaging may vary
with food type, release kinetics of antimicrobial agents,
and environmental factors. Many antimicrobial packaging
systems that show effective activity with microbial culture
media may not work with real foods. Some food compo-
nents such as fatty acids, amino acids, sulfates, and/or
enzymes may diminish the activity of the antimicrobial
packaging films (11, 42). Food–package interaction may
change the mechanism and effectiveness of microbial
inhibition of the antimicrobial packaging system (29).
There are possibilities that the incorporation of antimi-
crobials into the plastic material may change its mechan-
ical, barrier, and optical properties. Sensory properties of
food may also be affected by food–package interaction. For
example, film with plant extract such wasabi essential oil
may alter flavor of the contained food. The antimicrobial
packaging may have to be designed, selected, and custo-
mized for each food type (43). The packaging system has to
be selected after consideration on the primarily concerned
target microorganism, spoilage organisms, its activity
spectrum, microbial inhibition mode, food properties, re-
lease of active agent, and storage conditions.
Regulations
Antimicrobial packaging to be marketed in the United
States is subject to the food additive approval process if its
components are reasonably expected to migrate to foods
for effective microbial inhibition (4). There are two pro-
cesses of food contact substance approval in FDA: food
contact notification process and food additive petition
process. Packaging material that has no intended techni-
cal effect on the food may be approved by the former
process, in which a manufacturer should notify the FDA to
market a new product at least 120 days before its intro-
duction and can sell it unless the FDA objects to the
notification. However, the material that exerts antimicro-
bial effect on the food through migration or controlled
release needs to be processed through the food additive
petition process. Food contact substance notification is
specific only to the manufacturer named in the notifica-
tion, and it does not apply generically to the product
category. Due to complicated and strict regulation on
approval of antimicrobial food packaging materials, their
applications in food packaging is very limited in the
United States. Chlorine-dioxide-releasing material seems
to reach the commercial stage (42). Some silver incorpo-
rated materials are approved for food-contact purpose (6).
Until 2004, plastic packaging regulations in the EU did
not allow any specific provisions for antimicrobial packa-
ging and, thus, required that any potential antimicrobial
component in the antimicrobial food packaging should be
covered by ‘‘positive lists,’’ which would have specific
0
0.2
0.4
0.6
0.8
1
1.2
1.4
0 5 10 15 20 25
Temperature (°C)
(1/tlag)
1/2
Figure 2. Effect of antimicrobial packaging (incorporated with
chitosan and nisin) on temperature dependence of microbial lag
time. (&) Yeasts of orange juice in plain package; (’) yeasts of
orange juice in the antimicrobial package; (
) aerobic bacteria of
milk in plain package; (
) aerobic bacteria of milk in the anti-
microbial package. (From data of Ref. (41)).
ANTIMICROBIAL PACKAGING 57
