Coker A.K. Fortran Programs for Chemical Process Design, Analysis, and Simulation
Подождите немного. Документ загружается.


Instrument Sizing 357
the AP will cause it to flash. Therefore, the size of the valve and dis-
charge pipework must be increased to accommodate the larger volume.
Further flashing in the downstream piping will increase the gas-liquid
mixture through the two-phase regimes. If this is not arrested, slug flow
will develop along the pipeline resulting in the mechanical damage to
the pipework.
Discharge Reactive Forces
The total stress imposed on a safety valve or its piping is a result of
the sum of the following:
9 internal pressure
9 dead weight of piping
9 thermal expansion or contraction of either the discharge line
9 the equipment upon which the valve is mounted
9 the bending moment caused by the reaction thrust at the discharge
The discharge reactive force is based on the assumption that critical
flow of the gas or vapor is obtained at the outlet of the relief device and
the discharge is horizontal to the atmosphere. For any gas or vapor, the
reactive force can be expressed as:
I
10.5
W kT
F- (k + 1)M
_ w (5-56)
366
Relief Valve Location
The location of a relief device often influences its size and material
of construction. Relief devices must be sited so that the risk of blockage
by debris is minimized. It is poor practice to mount relief valves at the
end of long horizontal pipe through which there is no flow. The possi-
bility of liquid phase entering a vapor relief system must also be avoided.
Locating a relief device upstream of equipment that has AP due to flow
allows the relief valve to be smaller because increased AP is available.
Reduced costs of a relief system may be obtained by installing relief
valves in a more conducive position with regard to temperature and
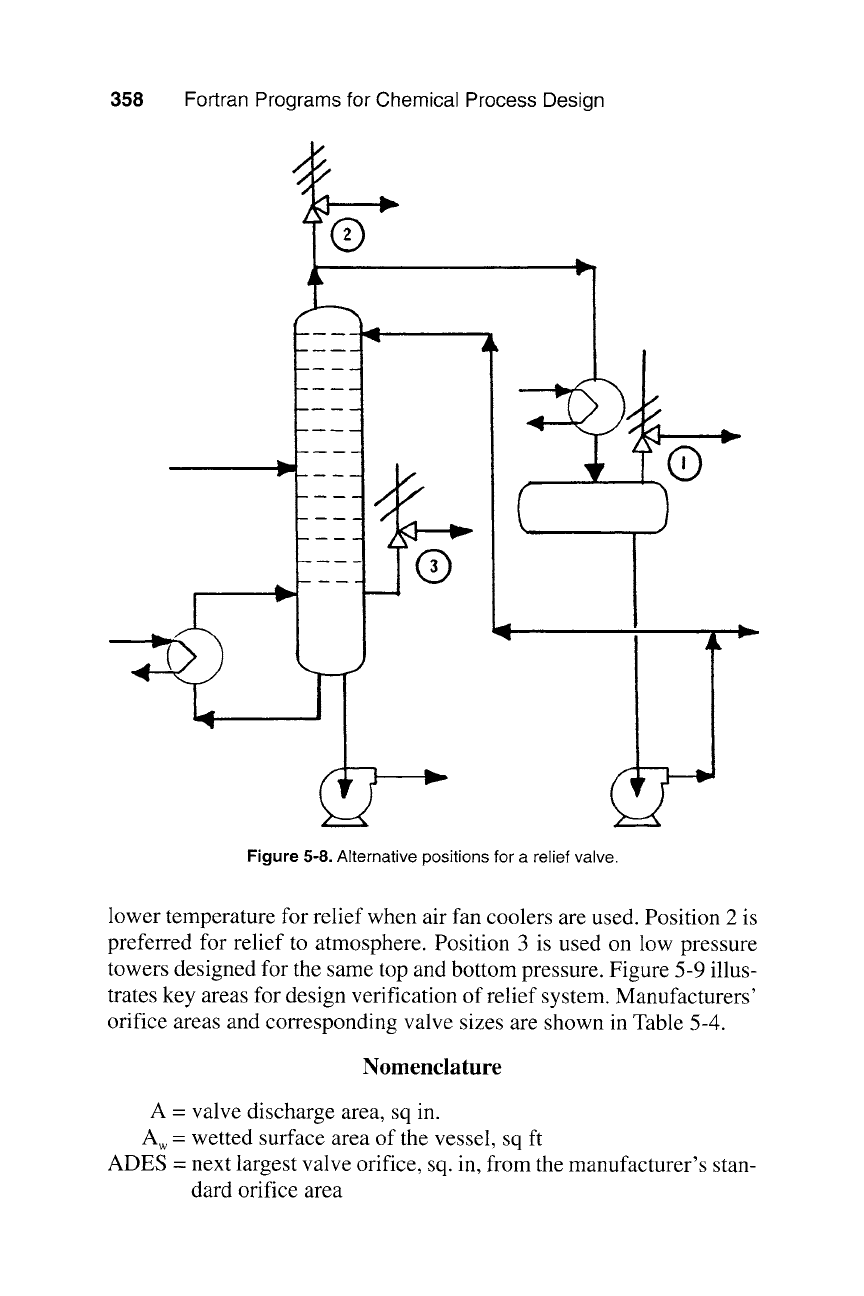
corrosion. Figure 5-8 shows alternative positions. Position 1 is preferred
when the relief valve is tied into a closed relief system because of ease
of maintenance. It assures a relatively short line to the relief header and
358
Fortran Programs for Chemical Process Design
/
/
Z
f,
|
i ii i| |
I i
(b
( )
(b
Figure 5-8. Alternative positions for a relief valve.
lower temperature for relief when air fan coolers are used. Position 2 is
preferred for relief to atmosphere. Position 3 is used on low pressure
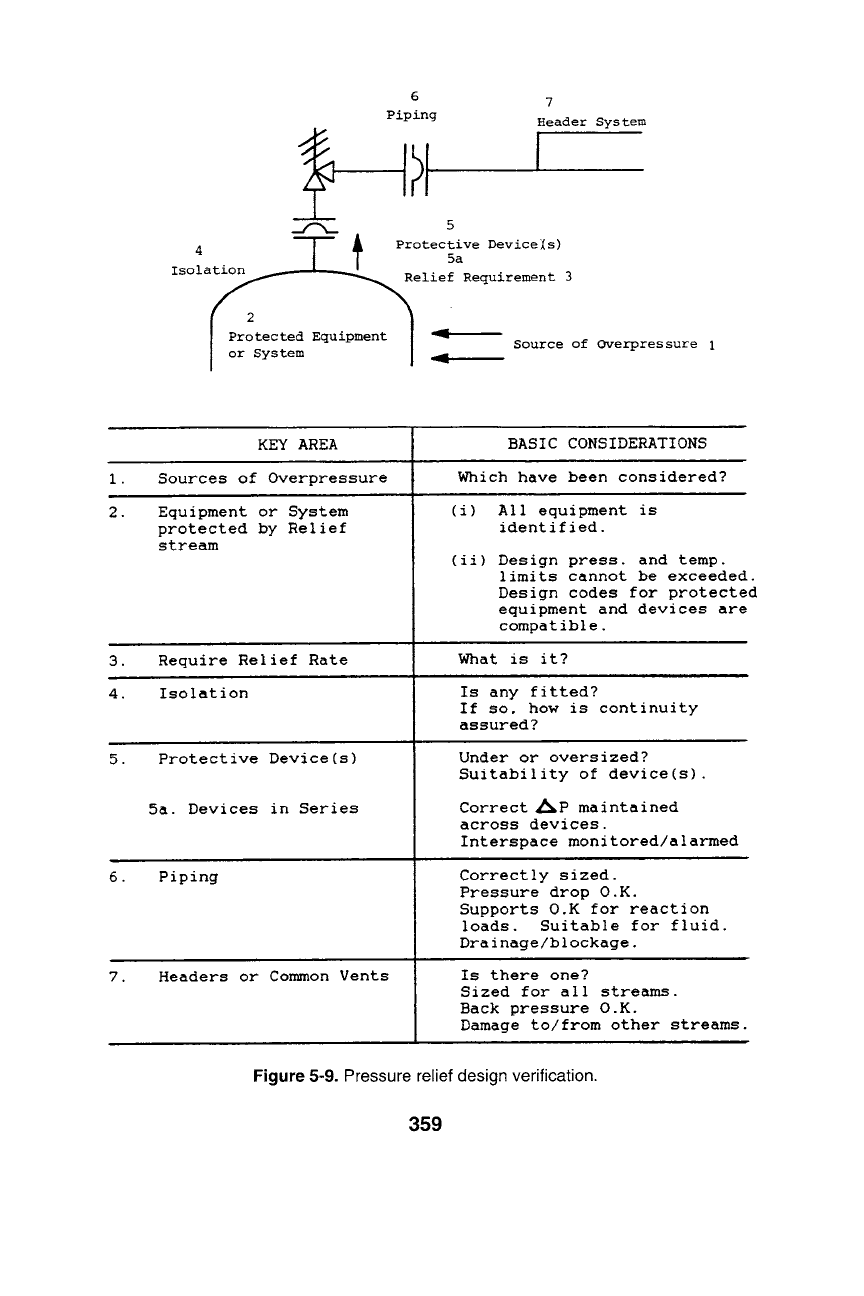
towers designed for the same top and bottom pressure. Figure 5-9 illus-
trates key areas for design verification of relief system. Manufacturers'
orifice areas and corresponding valve sizes are shown in Table 5-4.
Nomenclature
A - valve discharge area, sq in.
A w - wetted surface area of the vessel, sq ft
ADES - next largest valve orifice, sq. in, from the manufacturer's stan-
dard orifice area
6
Piping
7
Header System
,if i, ....
_f-h_ 5
4 ~A Protective Device X s)
5a
Isolation ~Relief Requirement 3
2
ected Equipment 1 ~" Source of Overpressure 1
I or System
I
i .
2.
3.
4.
5.
6.
7.
KEY AREA
Sources of Overpressure
Equipment or System
protected by Relief
stream
Require Relief Rate
Isolation
Protective Device(s)
5a. Devices in Series
Piping
Headers or Common Vents
BASIC CONSIDERATIONS
Which have been considered?
(i) All equipment is
identified.
(ii) Design press, and temp.
limits cannot be exceeded.
Design codes for protected
equipment and devices are
compatible.
What is it?
Is any fitted?
If so, how is continuity
assured?
Under or oversized?
Suitability of device(s).
Correct ~P maintained
across devices.
Interspace moni tored/alarmed
Correctly sized.
Pressure drop O.K.
Supports O.K for reaction
loads. Suitable for fluid.
Drainage/blockage.
Is there one?
Sized for all streams.
Back pressure O.K.
Damage to/from other streams.
Figure 5-9. Pressure relief design verification.
359

360
Fortran Programs for Chemical Process Design
Table 5-4
Orifice Sizes for Relief Valves
Orifice Designation
Orifice Area Valve Size
(sq. in.) (in.)
D 0.110 1-2
E 0.196 1-2
F 0.307 1.5-2
G 0.503 1.5-2.5
H 0.785 1.5-3 or 2-3
J 1.287 2-3 or 2.5-4
K 1.838 3-4
L 2.853 3-4 or 4-6
M 3.600 4-6
N 4.340 4-6
P 6.379 4-6
Q 11.050 6-8
R 16.000 6-8 or 6-10
T 26.000 8-10
C = coefficient determined by ratio of specific heats of the gas or
vapor at standard conditions
F = reaction force at the point of discharge to the atmosphere, lb
FI = factor for insulation (for non-insulated vessel FI is 1.0)
K = effective coefficient factor due to backpressure (0.975 for
vapor flow)
K b =
capacity correction factor due to backpressure is 1.0, when
backpressure is 55% of absolute relieving pressure
K d = coefficient of discharge (0.953)
K N = correction factor for Napier equation, K N, is 1.0, where
P < 1515 psia
Kp
-
overpressure correction factor for direct spring-operated valves
only, Kp is 1.0
KsH = correction factor due to amount of superheat in the steam. Val-
ues range from 0.88 (maximum superheat) to 1.0 for saturated
steam.
Kv = viscosity correction factor
K w = backpressure correction factor for balanced-bellows spring-
operated valves only (K w is 1.0)
k = ratio of specific heat capacities (Cp/C~)

Instrument Sizing 361
M = molecular weight
M w = molecular weight of vapor (or process fluid)
P = upstream relieving pressure, psia
Pb =
total backpressure, psig
Pi = set pressure at which relief valve begins to open, psig
Q = flow rate, in U.S. gallons per min
Qr = rate of heat input due to fire, Btu/hr
SGL = specific gravity of liquid at flowing temperature referred to
water as 1.0 at 70~
T = absolute temperature of inlet vapor ~176 + 460)
V, = required air capacity, standard ft3/min
W = flow of any gas or vapor, lb/hr
Z = compressibility factor
= latent heat of vaporization at the boiling point of the liquid,
Btu/ib
= absolute viscosity at the flowing temperature, cP
TWO-PHASE FLOW RELIEF SIZING
FOR RUNAWAY REACTION
Introduction
Many methods have been used to size relief systems: area/volume scal-
ing, mathematical modeling using reaction parameters and flow theory,
and empirical methods by the Factory Insurance Association (FIA). The
Design Institute for Emergency Relief Systems (DIERS) of the AIChE
has carried studies of sizing reactors undergoing runaway reactions. Intri-
cate laboratory equipment and analysis have resulted in better vent sizes.
A selection of relief venting as the basis of safe operation is based
upon the following considerations [14]:
9 compatibility of relief venting with the design and operation of the
plant/process
9 identifying the worst scenarios
9 type of reaction
9 means of measuring the reaction parameters during the runaway
reaction
9 relief sizing procedure
9 design of the relief system including discharge ducting and safe
discharge area

362 Fortran Programs for Chemical Process Design
Runaway Reactions
A runaway reaction occurs when an exothermic system becomes un-
controllable. The reaction leads to a rapid increase in the temperature
and pressure, which if not relieved can rupture the containing vessel. A
runaway reaction happens because the rate of reaction and therefore the
rate of heat generation increases exponentially with temperature. Alter-
natively, the rate of cooling increases only linearly with temperature.
Once the rate of heat generation exceeds available cooling, the rate of
increase in temperature becomes progressively faster. Runaway reac-
tions nearly always result in two-phase flow reliefs. Runaway reactions
are generally classified into three systems.
Vapor Systems
Boiling is attained before potential gaseous decomposition, that is,
the heat of reaction is removed by the latent heat of vaporization. The
reaction is tempered, and the total pressure in the reactor is equal to the
vapor pressure. The principal parameter determining the vent size is
the rate of the temperature rise at the relief set pressure.
Gassy Systems
Gaseous decomposition reaction occurs without tempering. The total
pressure in the reactor is equal to the gas pressure. The principal param-
eter determining the vent size is the maximum rate of pressure rise.
Hybrid Systems
Gaseous decomposition reaction occurs before boiling. The reaction
is still tempered by vapor stripping. The total pressure in the reactor is
the summation of the gas partial pressure and the vapor pressure. The
principal parameters determining the vent size are the rates of tempera-
ture and pressure rise corresponding to the tempering condition. A tem-
pered reactor contains a volatile fluid that vaporizes or flashes during
the relieving process. This vaporization removes energy via the heat of
vaporization and tempers the rate of temperature rise due to the exo-
thermic reaction.
Adiabatic Calorimetry
Two-phase flow calculations are complex when conditions change
rapidly as in runaway reactions. Because of this complexity, several
Instrument Sizing 363
experimental and analytical techniques have been developed over the
past decade to obtain the relevant data for the relief vent calculations.
The data required for the runaway calculations are determined with labo-
ratory tools such as the Differential Scanning Calorimetry (DSC), the
accelerating rate calorimeter (ARC), the vent sizing package (VSP),
and recently, the laboratory calorimetric Reactive System Screening Tool
(RSST). The RSST determines the type of reaction and principal
parameters required for sizing reactor vents for vapor, hybrid, and gassy
systems. Figures 5-10A and 5-10B show the principal features of the
RSST. The principal data for relief sizing calculations are shown in Fig-
ures 5-11A, 5-11B, 5-11 C, and 5-11D, the temperature rate at the set
(text continued on page 366)
Figure
5-10A. The RSST vessel.
Figure 5-10B. The test cell and associated equipment.
~0
i 'atmax,mumJ r
I
permitted pressure/ ~ |
IT at relief set ~
s
(~ t t+5 t+10
t+151t+20t+25
Time L
~ (min)
Venting time for vapour system j
E during which time the entire J
i0 vessel content is discharged Jr
0 1000 2000 3000 400(3
Time (rain)
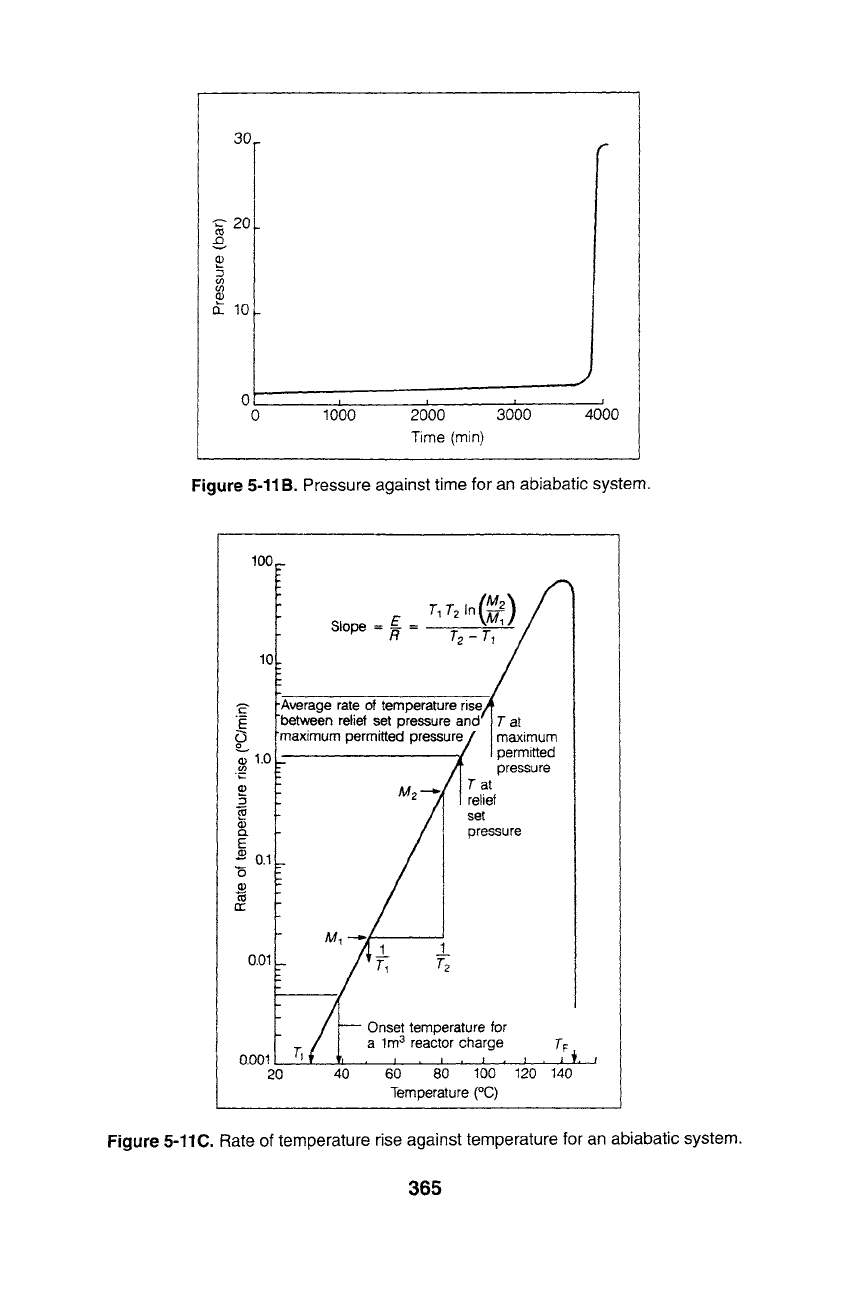
Figure 5-11A. Temperature against time for an abiabatic system.
364
CD
4-
J
0 - ! ! .
0 1000 2000 30()0 40C)0
Time (rain)
Figure
5-11B. Pressure against time for an abiabatic system.
100
10
Slope = E _ T1T21n(-~12) ~ ''~
R 7"2 T1/
-Average rate of temperature rise/
"between relief set pressure and T at
"maximum permitted pressure/' maximum
............. .~ permitted
1.0 /]- pressure
,, ....~/ I T at
= ,,',2 J/1 Irelief
"~ /I set
r~ ressure
E
0.11
5
Q)
K
M' --',,.t 1 •
o.o~ /' T, T2
/I
/ t"-- Onset temperature for
0.001 " / I a l m 3 reactor charge TF
' f. ~, . , 9 , . ,
.... ,. ,~. ,
20 40 60 80 100 120 140
Temperature (~
Figure
5-11C. Rate of temperature rise against temperature for an abiabatic system.
365
366
Fortran Programs for Chemical Process Design
#_
00
10
1[
20
Maximum //~dp l
/ dt
tmax
.. permitted pressure 1 /~-----'
- AP (vapour
A-T systems)
/I AP
/(gassy,
= ~ ;v / systew,,~)
Rel~ AT/ -
40
60 I I 80 I &O
120 140
Temperature (~
100
10
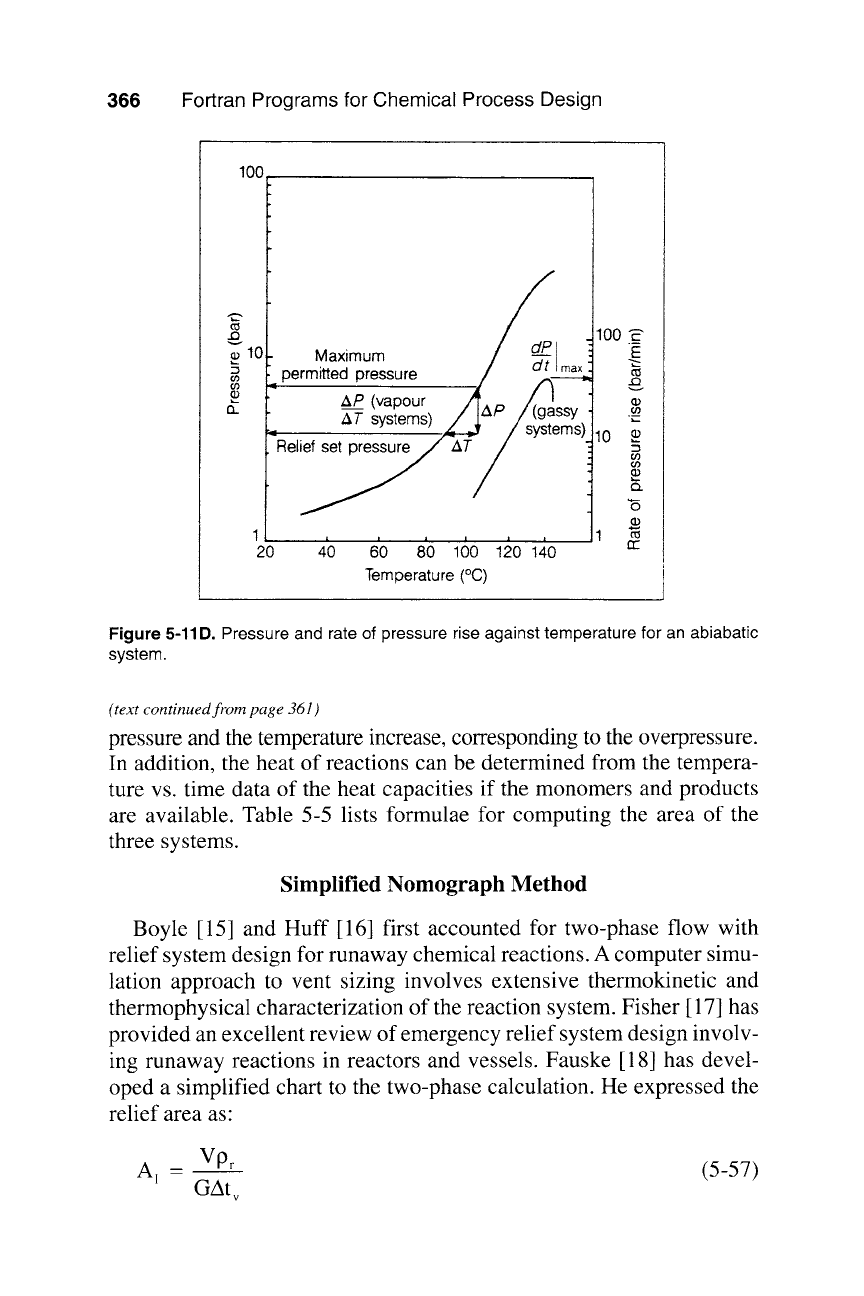
Figure 5-11D. Pressure and rate of pressure rise against temperature for an abiabatic
system.
(text continued from page 361)
pressure and the temperature increase, corresponding to the overpressure.
In addition, the heat of reactions can be determined from the tempera-
ture vs. time data of the heat capacities if the monomers and products
are available. Table 5-5 lists formulae for computing the area of the
three systems.
Simplified Nomograph Method
Boyle [15] and Huff [16] first accounted for two-phase flow with
relief system design for runaway chemical reactions. A computer simu-
lation approach to vent sizing involves extensive thermokinetic and
thermophysical characterization of the reaction system. Fisher [ 17] has
provided an excellent review of emergency relief system design involv-
ing runaway reactions in reactors and vessels. Fauske [18] has devel-
oped a simplified chart to the two-phase calculation. He expressed the
relief area as:
A~ = VPr (5-57)
GAt v
