Francoise J.-P., Naber G.L., Tsun T.S. (editors) Encyclopedia of Mathematical Physics
Подождите немного. Документ загружается.


of particles from the same cluster be defined by the
equation
ðT
a
ðp
1
; ...; p
n
Þf Þðx
1
; ...; x
N
Þ¼f ðx
0
1
; ...; x
0
N
Þ
where x
0
j
= x
j
þ p
l
if j 2C
l
. The operator H
a
com-
mutes with the operators T
a
(p
1
, ..., p
n
) for all
vectors p
1
, ..., p
n
2 R
d
. Let the subspace X
a
be
determined by the condition
X
j2C
l
m
j
x
j
¼ 0; l ¼ 1; ...; n
and let X
a
be the orthogonal complement to X
a
in
X
cm
with respect to scalar product [6]. Clearly,
dim X
a
= (N #(a))d, dim X
a
= (#(a) 1)d. Then
the space H splits into the tensor product
L
2
ðX
cm
Þ¼L
2
ðX
a
ÞL
2
ðX
a
Þ½10
In what follows, x
a
and x
a
are the orthogonal
projections of x 2 X
cm
on the subspaces X
a
and
X
a
, respectively. The ‘‘external’’ variable x
a
=
(x
1
, x
2
, ..., x
n
), where
x
l
¼ M
1
l
X
j2C
l
m
j
x
j
; M
l
¼
X
j2C
l
m
j
describes positions of centers of masses of the clusters.
The ‘‘internal’’ variable x
a
is the set of numbers x
j
x
l
for all j 2C
l
and all l = 1, ..., n. Of course, for each l
only jC
l
j1(jC
l
j is the number of particles in a cluster
C
l
)ofvariablesx
j
x
l
are independent. Set
K
a
¼
x
a
¼
X
n
l¼1
ð2M
l
Þ
1
x
l
and
H
a
¼
x
a
þ V
a
Then
H
a
¼ K
a
I þ I H
a
Note that eigenvalues
a, n
of the operator H
a
are
sums over l = 1, ..., n of eigenvalues of the operators
HðC
l
Þ¼H
0
ðC
l
Þþ
X
2C
l
V
describing each cluster. Similarly, eigenfunctions
a, n
of
H
a
are products of eigenfunctions of these operators.
We usually write a instead of a couple {a, n}. In the
following, the index a labels all cluster decompositions
with #(a) 2. The eigenvalues
a
of the operators H
a
(
a
= 0if#(a) = N) are called thresholds of the
Schro¨dinger operator [8]. If all functions V
(x
) !0
as jx
j!1, then the essential spectrum of the operator
H consists of the interval [
0
, 1), where
0
¼ min
a
a
(the Hunziker–Van Winter–Zhislin theorem). More-
over, the eigenvalues of the operator H may
accumulate at its thresholds only.
The fundamental result of scattering theory for
the N-particle Schro¨ dinger operator can be formu-
lated as follows. Let P
a
be the orthogonal proje ction
in L
2
(X
a
) on the subspace H
(p)
a
spanned by all
eigenvectors
a, n
of H
a
, and let P
a
= I P
a
, where
the tensor product is defined by eqn [10]. Then P
a
commutes with the operator H
a
. Set also K
0
= H
0
,
P
0
= I. Suppose that for all
jV
ðx
Þj Cð1 þjx
jÞ
;>1 ½11
(the short-range assumption). Then, for all a, the
wave operators
W
a
¼ W
ðH; H
a
; P
a
Þ¼
s-lim
t!1
e
iHt
e
iH
a
t
P
a
exist and are isometric on the ranges Ran P
a
of
projections P
a
. The subspaces Ran W
a
are mutually
orthogonal, and scattering is asymptotically complete:
M
a
Ran W
a
¼H
ðacÞ
The singular continuous spectrum of H is empty, so
the absolutely continuous subspace H
(ac)
of the
operator H can be replaced by HH
(p)
, where
H
(p)
is spanned by all eigenvectors of H.
These results can be reformulated in terms
of scattering theory in a couple of spaces.
Suppose that, for every a, eigenvectors
a, n
are
normalized and orthogonal if the corresponding
eigenvalues
a, n
coincide. Let us introduce an
auxiliary space
^
H¼
M
a
H
a
; H
a
¼H
a
¼ L
2
ðX
a
Þ½12
and an auxiliary operator
^
H ¼
M
a
K
a
; K
a
¼ K
a
þ
a
½13
in this space. Here and below, the sums are taken
over all a. We define an identification
^
J :
ˆ
H!H by
the relations
^
J ¼
X
a
J
a
; J
a
f
a
¼ f
a
a
½14
where the tensor product is the same as in [10].In
particular, J
0
= I. Since H
a
J
a
= J
a
K
a
, the wave
operators W
(H,
^
H;
^
J) exist and are isometric and
complete, that is,
Ran W
ðH;
^
H;
^
JÞ¼H
ðacÞ
Thus, for states orthogonal to eigenvectors of
H, evolution of an N-particle system decomposes
N-Particle Quantum Scattering 587

asymptotically into a sum of evolutions which
are ‘‘free’’ in external variables x
a
and are
determined by eigenvalues and eigenfunctions of
the H amiltonians H
a
in internal variables x
a
.Tobe
more precise, we have that, for all f 2H
(ac)
and
t !1,
expðiHtÞf ¼
X
a
expðiK
a
tÞf
a
a
þ oð1Þ½15
where
f
a
¼ W
ðH; K
a
; J
a
Þ
f
and the term o(1) t ends to zero in H.Thewave
operator W
(H, K
a
; J
a
) describes the scattering
channel where a system of N interacting particles
splits up asymptotically (for t !1) into non-
interacting clusters C
1
, ..., C
n
, n 2, and particles
from the same cluster C
l
are in the bound state (if
there are more than one particle in C
l
)givenbythe
function
a
(x
a
). Somewhat loosely speaking, this
implies that the continuous spectrum of the
operator H consists of branches starting from all
its thresholds.
Note that the scattering problem can equivalently
be formulated without the separation of center-
of-mass motion. In this case, a trivial decomposition
with #(a) = 1 should be added, and the set of
thresholds of the operator H includes eigenvalues of
the operator H.
The existence of the wave operators and their
isometricity can be obtained by the Cook method.
Only the asymptotic completeness is a difficult
mathematical problem. It can be solved within the
framework of the smooth method, which requires a
study of boundary values of resolvents as the
spectral parameter z approaches the continuous
spectrum or, equivalently, a study of a large-time
behavior of evolution operators.
The scattering operator
S ¼ W
þ
ðH;
^
H;
^
JÞ
W
ðH;
^
H;
^
JÞ
is unitary on the space
^
H and commutes with the
operator
^
H. Its component S
ab
: H
b
!H
a
describes
a process wh ere a system in a state b as t !1
goes over in a state a as t !þ1. Diagonalizing
the operator
^
H by a unitary operator
^
F,(
^
F
^
Hf)() = (
^
Ff)(), >
0
, we obtain the
scattering matrix S() defined by the equation
(
^
FSf )() = S()(
^
Ff)(). In its turn, the scatte ring
matrix is also a matrix operator with components
S
ab
(). For N 3, t he structure of the scattering
matrix is essentially m ore complicated than for
N = 2. This is discussed i n some detail i n the next
section.
Resolvent Equations for Three-Particle
Systems
Let the Hamiltonian H be defined by eqns [2]–[4],
where N = 3, and let the configuration space of each
particle be R
d
, d 3. The operator H acts in the
space H= L
2
(X
cm
), where the subspace X
cm
R
3d
is distinguished by condition [5]. Let R
0
(z) =
(H
0
z)
1
, R(z) = (H z)
1
. Since V(x) does not
tend to 0 as jxj!1, x 2 X
cm
, in the three-particle
case, the resolvent equation
RðzÞ¼R
0
ðzÞR
0
ðzÞVRðzÞ½16
is not Fredholm even for Im z 6¼ 0.
To overcome this difficulty, Faddeev (1965)
derived a system of equations for compone nts of
the resolvent. The entries of this system are
constructed in terms of three Hamiltonians
H
¼ H
0
þ V
= (12), (13), (23), containing only one pair inter-
action each, and their resolvents R
(z) = (H
z)
1
.
Let us write down the resolvent equation for each
pair H
, H
RðzÞ¼R
ðzÞR
ðzÞ
X
6¼
V
RðzÞ
We multiply it by jV
j
1=2
and set
r
0
ðzÞ¼jV
j
1=2
R
ðzÞ; r
ðzÞ¼jV
j
1=2
RðzÞ
t
;
ðzÞ¼0; t
;
ðzÞ¼jV
j
1=2
R
ðzÞðV
Þ
1=2
where (V
)
1=2
= V
jV
j
1=2
. This yields a system of
equations
r
ðzÞ¼r
0
ðzÞ
X
6¼
t
;
ðzÞr
ðzÞ½17
for the operators r
(z). Note that the resolvent R(z)
can be recovered from its components r
(z) by the
formula
RðzÞ¼R
0
ðzÞR
0
ðzÞ
X
ðV
Þ
1=2
r
ðzÞ
It is convenient to rewrite eqn [17] in the matrix
notation
rðzÞ¼r
0
ðzÞtðzÞrðzÞ½18
where r
0
(z) = {r
0
(z)}, r(z) = {r
(z)} are the ‘‘vector’’
operators in the three-component space L
(3)
2
(X
cm
)and
t(z) = {t
,
(z)} is the ‘‘matrix’’ operator in this space.
The advantage of eqn [17] compared to [16] is
that the operators t
,
(z) are compact for Im z 6¼ 0.
This can be deduced from the fact that the product
V
(x
)V
(x
), where 6¼ tends to 0 as
588 N-Particle Quantum Scattering

jxj!1, x 2 X
cm
, provided that V
(x
) !0as
jx
j!1 for all . Moreover, the homogeneous
equation [17] has only a trivial solution. Indeed, if
for some z with Im z 6¼ 0
f
¼
X
6¼
t
;
ðzÞf
½19
then the function
u ¼
X
ðV
Þ
1=2
f
satisfies the equation u = R
0
(z)Vu. Since the
operator H is self-adjoint, this implies that u = 0
and hence f
= 0 for all . According to the
Fredholm alternative, eqns [17] for r
(z)or[18]
for r(z) can be solved if Im z 6¼ 0, that is,
rðzÞ¼ðI þ tðzÞÞ
1
r
0
ðzÞ½20
This equation allows one to deduce the existence of
necessary boundary values of the ‘‘sandwiched’’
resolvent R (z) from similar results for the resolvents
R
(z) of the ‘‘two-particle’ ’ operators H
.Inits
turn, R
(z) can be expressed in terms of the resolvent
R
(z) of the operator H
acting in the space L
2
(R
d
).
Indeed, in the ‘‘mixed’’ representation (
, x
), where
the Fourier transform in the variable x
is performed
and the variable
is dual to x
,wehave
ðR
ðzÞf Þð
; x
Þ¼ðR
ðz ð2m
Þ
1
j
j
2
Þf Þ
ð
; x
Þ½21
The passage to the limit Im z !0 requires that
assumption [11] be satisfied for >2. Moreover,
we have to suppose that the operators H
do not
have the so-called zero-energy resonances as well as
eigenvalues embedded in the con tinuous spectrum.
Then the operator functions hx
i
l
R
(z)hx
i
l
, l > 1,
hx
i= (1 þjx
j
2
)
1=2
, are analytic in the complex
plane cut along [0, 1), they have poles only at the
points
, n
, and are continuous up to the cut, the
point z = 0 included. In particular, it follows from
eqn [21] that, if the operators H
do not have
negative eigenvalues, then the operator functions
hx
i
l
R
(z)hx
i
l
, l > 1, are also analytic in the
complex plane cut along [0, 1) and are continuous
up to the cut.
The next result is of genuinely three-particle nature
and is crucial for the study of the operator t(z). The
operator functions hx
i
l
R
0
(z)hx
i
l
, 6¼ , l > 1,
are continuous in norm up to the cut along [0, 1).
Now it follows from eqn [20] that the operator-
valued functions r
(z)jV
j
1=2
are continuous up to
the cut (0, 1)exceptpoints 2 (0, 1), where the
homogeneous equation [19] for z = i0 has a
nontrivial solution. The set N = N
þ
[N
of such
points 2 (0, 1) is closed and has Lebesgue measure
zero. In particular, the operators hx
i
l
, l > 1,
are H-smooth on any compact subinterval of
= (0, 1)nN. Therefore, the smooth method of
scattering theory can be d irectly applied. It yields
the existence and completen ess of the wave
operators W
(H, H
0
). In this case, three-particles
are necessarily asymptotically free.
‘‘Two-particle’’ channels of scatter ing arise if the
operators H
have negative eigenvalues. To simplify
notation, we assume that every H
has exactly one
eigenvalue
< 0. Moreover, it is supposed that the
corresponding eigenfunction
(x
) tends to zero
sufficiently rapidly as jx
j!1. Analytically, the
appearance of new channels is due to new singula-
rities of the resolvents. Indeed, in this case
R
ðzÞ¼ð
zÞ
1
P
þ
^
R
ðzÞ
where the function
^
R
(z) is analytic and continuous
up to the cut in the complex plane cut along [0, 1).
It follows from eqn [21] that in this case the
resolvent R
(z) contains the additional term
ðð2m
Þ
1
j
j
2
þ
zÞ
1
P
which is analytic only in the complex plane cut
along [
, 1). To take these terms into account,
system [17] should be further rearranged. This yields
the following result. Let us set
G
0
¼hx
i
l
ðI P
Þ; G
1
¼hx
i
l
ðJ
Þ
X
6¼
V
½22
Then, for all ,, i, j = 0, 1, a suitable l > 1 and
0
= min{
}, the operator functions G
i
R(z)G
j
are
norm continuous as z approaches the cut (
0
,1 )at
the points of =(
0
,1)nN, where N is again a
closed set of measure zero. In particular,
the operators G
0
and G
1
are H-smooth on any
compact subinterval of .
In the multichannel case, to fit scattering for the
Hamiltonian H into the framework of smooth
theory, it is convenient to reformulate the result in
terms of scattering theory in a couple of spaces. Let
the space
^
H, the operator
^
H, and the identification
^
J
be defined by eqns [12], [13], and [14], respectively,
where the index a takes four values a = 0, and
= (12), (13), (23). One, further, needs to introduce
auxiliary identifications
J
0
¼ I
X
P
and
^
J ¼ J
0
M
J
N-Particle Quantum Scattering 589

The H- (and
^
H-) smoothness of operators [22] imply
that the wave operators
W
ðH;
^
H;
^
JÞ and W
ð
^
H; H;
^
J
Þ
exist.
The operators W
(H,
^
H;
^
J) are isometric because
s-lim
jtj!1
P
expðiH
0
tÞ¼0 ½23
and the operators P
P
are compact for 6¼ .
Using that the operator
^
J
^
J
I ¼
X
6¼
P
P
is compact (whereas
^
J
^
J
I is not), we see that the
operators W
(
^
H, H;
^
J
) are also isometric. Finally,
we remark that, by eqn [23],
W
ðH;
^
H;
^
JÞ¼W
ðH;
^
H;
^
JÞ
This implies the asymptotic completeness.
Let us discuss properties of the scattering matrix
in the one-channel case where the pair operators H
do not have negative eigenvalues. The scattering
matrix S():L
2
(S
2d1
) !L
2
(S
2d1
), >0, is of
course a unitary operator, but in contrast to the
two-particle case the operator S() I is not
compact because its kernel contains the Dirac
functions (
0
). Nevertheless, the structure of
its singularities can be explicitly described. Actually,
let S
() be the ‘‘two-particle’’ scattering matrix for
the pair H
0
, H
. Then
SðÞ¼S
12
ðÞS
23
ðÞS
13
ðÞ
~
SðÞ
where the operator
~
S() I is compact.
The approach described briefly in this section
relies on a kind of an advanced perturbation theory
where the free problem is determined by the set of
all sub-Hamiltonians. Its generalization to the case
of an arbitrary number of particles meets with
numerous difficulties. A different, nonperturbative,
approach which works well for any number of
particles will be discussed in the next section.
A purely time-dependent method in three-particle
scattering is exposed in Enss (1983).
Nonperturbative Approach
Now N and d are arbitrary. In the nonperturbative
approach (see Graf (1990), Sigal and Soffer (1989),
and Yafaev (1993)) the operators H and H
0
as well
as the Hamiltonians of all subsystems are treated on
an equal basis. It is supposed that all pair potentials
satisfy condition [11]. No assumptions on subsys-
tems are required.
The starting point of this approach is the limiting-
absorption principle, which claims that the operator
hxi
l
, x 2 X
cm
,forl > 1=2isH-smooth on any
compact interval not containing the thresholds and
eigenvalues of H. Its proof relies on the Mourre
commutator method (see Cycon et al. (1987)). To be
more precise, it is deduced from the following estimate:
ið½H; Af ; f Þckf k
2
; c ¼ cðÞ > 0
f 2 Eð
ÞH ½24
for the commutator of H with the generator of
translations
A ¼i
X
j
ðx
j
@
j
þ @
j
x
j
Þ
Here x
j
are coordinates of x 2 X
cm
in some orthonor-
mal (with respect to scalar product [6])basisinX
cm
,
is neither a threshold nor an eigenvalue of the operator
H and
is a sufficiently small interval. Very roughly
speaking, the Mourre estimate [24] means that,
similarly to the two-particle case, the observable
ðAe
iHt
f ; e
iHt
f Þ
is a strictly increasing function of t for all f 2H
(ac)
.
The limiting-absorption principle implies that the
singular continuous spectrum of the operator H is
empty, but it is not sufficient for scattering theory. If
the limiting-absorption principle were true for the
critical value l = 1=2, then it would imply asymptotic
completeness. Unfortunately, the operator hxi
1=2
is
definitely not smooth even with respect to the free
operator H
0
. However, by introducing an auxiliary
differential operator we can fix this problem. This
leads to the radiation estimates. These estimates look
differently in different regions of the configuration
space. Choose any cluster decomposition a =
(C
1
, ..., C
n
). The radiation estimate morally implies
that the motion of a system is asymptotically free in
the variable x
a
(describing the relative motion of
clusters) in the region where particles from each
cluster C
l
, l = 1, ..., n, are close to each other
compared to distances between different clusters.
On the contrary, this motion is very complicated in
the variable x
a
pertaining to bound states of different
clusters. In particular, the radiation estimate is the
same as for the two-particle case in the ‘‘free’’ region
where all particles are far from each other.
To be more precise, let r
a
= r
x
a
be the gradient
in the variable x
a
and let r
?
a
,
r
?
a
u
ðxÞ¼ðr
a
uÞðxÞjx
a
j
2
hðr
a
uÞðxÞ; x
a
ix
a
be its orthogonal projection in X
a
on the subspace
orthogonal to the vector x
a
. Let
a
be the
590 N-Particle Quantum Scattering

characteristic function of a closed cone Y
a
X
cm
satisfying the condition Y
a
\ X
b
= ; for all b such
that X
a
6 X
b
. Then the operator
G
a
¼
a
hxi
1=2
r
?
a
is H-smooth on .
A proof of the radiation estimates is based on
the consideration of the commutator of H with
some differential operator M = i
P
(m
(j)
@
j
þ @
j
m
(j)
),
where m
(j)
= @m=@x
j
.Herem (it depends on a)isa
specially constructed function satisfying the follow-
ing properties:
1. m(x) is homogeneous (for jxj1) of order 1;
2. for any b it does not depend on x
b
in some
conical neighborhood of the subspace X
b
;
3. m(x) is convex; and
4. m(x) =
a
jx
a
j,
a
1, on support of the function
a
.
Note that we can set m(x) = jxj in the case of the
operator H
0
.
Due to properties (1) and (2) the commutator
[V, M] is a short-range function (estimated by
hxi
1"
for ">0). Due to properties (3) and (4)
the commutator [H
0
, M] cG
a
G
a
, c > 0, up to
short-range terms. The estimate
½H; McG
a
G
a
c
1
hxi
1"
implies that the operator G
a
is H-smooth on .
The main difficulty in the N-particle problem is
that pair potentials V
(x
) do not tend to zero as
jxj!1. The idea of the proof of asymptotic
completeness is to introduce auxiliary wave opera-
tors such that ‘‘effective’’ perturbations are decaying
functions. This requires a suitable smooth partition
of unity. Moreover, it is convenient to choose
auxiliary identifications as first-order differential
operators rather than operators of multiplication.
Unfortunately, altho ugh such identifications allow
one to ‘‘kill’’ directions where the potentials V
(x
)
do not tend to zero, their commutators with the
operator H
0
have coefficients decaying at infinity
only as jxj
1
.
Thus, we introduce differen tial operators
M
a
¼i
X
m
ðjÞ
a
@
j
þ @
j
m
ðjÞ
a
with coefficients m
(j)
a
= @m
a
=@x
j
. The functions m
a
satisfy properties (1), (2) formulated above and
5. m
a
(x) = 0 in some conical neighborhoods of the
subspaces X
b
such that X
a
6 X
b
. To put it
differently, m
a
(x) = 0 in some conical neighbor-
hood of the subspace where x
i
= x
j
for some i, j
belonging to different clusters C
1
, ..., C
n
.
Let the operator H
a
be defined by eqn [9]. Given
the limiting-absorption principle and the radiation
estimates, we first check the existenc e of auxiliary
wave operators
W
ðH; H
a
; M
a
E
a
ðÞÞ
and
W
ðH
a
; H; M
a
EðÞÞ ½25
Here we use that according to (5) coefficients of the
differential operator (V V
a
)M
a
are, under assump-
tion [11], short-range (in the configuration space
X
cm
). By property (2), the function [V
a
, M
a
] is also
short-range. Thus, the operator VM
a
M
a
V
a
can be
taken into account by the limiting-absor ption
principle. The commutator [H
0
, M
a
] factori zes into
a product of H
a
- and H-smooth operators accordi ng
to the radiation estimates.
Similar arguments show that, for
P
a
m
a
= m and
M =
P
a
M
a
(the sums here are taken over all
possible breakups of the N-particle system), the
wave operator (observable)
W
ðH; H; MEðÞÞ ½26
also exists. Moreover, it can be easily achieved
that m(x) 1. Then it follows from the Mourre
estimate that operator [26] is positive definite
on the subspace E()H and hence its range
coincides with this subspace. It means that for
all f 2 E()H
lim
t!1
kexpðiHtÞf M expðiHtÞg
k¼0 ½27
if f = W
(H, H; ME())g
.
The existence of wave operators [25] implies that
for any g
= E()g
and g
a
= W
(H
a
, H; M
a
E())g
lim
t!1
kM expðiHtÞg
X
a
expðiH
a
tÞg
a
k¼0 ½28
Combining eqns [27] and [28],weseethat
exp ( iHt)f decomposes asymptotically into sim-
pler evolutions exp (iH
a
t)g
a
. T his is one of the
equivalent formulations of asymptotic complete-
ness and leads to eqn [15].
Finally, we note that eqn [15] can be rewritten as
expðiHtÞf ¼
X
a
expði
a
ðx
a
; tÞÞð2itÞ
d
a
=2
^
f
a
ðx
a
=ð2tÞÞ
a
ðx
a
Þþoð1Þ½29
N-Particle Quantum Scattering 591

where t !1, d
a
= dim X
a
,
^
f
a
is the Fourier trans-
form of f
a
and
a
ðx
a
; tÞ¼x
2
a
ð4tÞ
1
a
t ½30
Long-Range Interactions: New Channels
The multiparticle problem acquires a long-range
character if pair potentials decay as Coulomb
potentials or slower. Similarly to the two-particle
problem, for long-range potentials the definition of
wave operators should be naturally modified. As in
the short-range case, only the asymptotic complete-
ness is a really difficult mathematical problem.
Assume that pair potentials satisf y condition
jð@
V
Þðx
Þj Cð1 þjx
jÞ
jj
;>
ffiffiffi
3
p
1
for all jj
0
and sufficiently large
0
. Then only
phase factors in eqn [29] should be modified.
Actually, instead of eqn [30] we should set
a
ðx
a
; tÞ¼x
2
a
ð4tÞ
1
a
t t
Z
1
0
V
a
ðsx
a
; 0Þds
where V
a
(x) = V(x) V
a
(x) and as usual x = (x
a
, x
a
).
As shown in Derezin
´
ski (1993), with this definition of
wave operators, the asymptotic completeness holds.
On the contrary, if pair potentials decay slower
than j xj
1=2
, then the traditional picture of scatter-
ing bre aks down (see Yafaev (1996)). Actu ally, a
three-particle system might h ave additional scatter-
ing channels intermediary between the c hannel
where three particles are asymptotically free and
the channels where a couple of particles form a
bound state. In these additional channels, the
bound state of a couple of particles depends on a
position of the third particle, and it is destroyed
asymptotically.
See also: Quantum Mechanical Scattering Theory;
Schro
¨
dinger Operators.
Further Reading
Cycon H, Froese R, Kirsh W, and Simon B (1987) Schro¨dinger
Operators, Texts and Monographs in Physics. Berlin: Springer.
Derezin
´
ski J (1993) Asymptotic completeness of long-range
quantum systems. Annals of Mathematics 138: 427–473.
Derezin
´
ski J and Ge´rard C (1997) Scattering Theory of Classical
and Quantum N Particle Systems. Berlin: Springer.
Enss V (1983) Completeness of three-body quantum scattering.
In: Blanchard P and Streit L (eds.) Dynamics and Processes,
Springer Lecture Notes in Mathematics, vol. 1031, pp. 62–88.
Faddeev LD (1965) Mathematical Aspects of the Three Body
Problem in Quantum Scattering Theory, Israel Program of Sci.
Transl.
Graf GM (1990) Asymptotic completeness for N-body short-
range quantum systems: a new proof. Communications in
Mathematical Physics 132: 73–101.
Reed M and Simon B (1979) Methods of Modern Mathematical
Physics III. New York: Academic Press.
Sigal IM and Soffer A (1987) The N-particle scattering problem:
asymptotic completeness for short-range systems. Annals of
Mathematics 126: 35–108.
Yafaev DR (1993) Radiation conditions and scattering theory for
N-particle Hamiltonians, Communications in Mathematical
Physics 154: 523–554.
Yafaev DR (1996) New channels of scattering for three-body
quantum systems with long-range potentials, Duke Mathema-
tical Journal 82: 553–584.
Yafaev DR (2000) Scattering Theory: Some Old and New
Problems, Springer Lecture Notes in Mathematics, vol. 1735.
Nuclear Magnetic Resonance
P T Callaghan, Victoria University of Wellington,
Wellington, New Zealand
ª 2006 Elsevier Ltd. All rights reserved.
Introduction
The existence of nuclear spin and its associated
magnetism was first suggested by Wolfgang Pauli in
1924, a conjecture based on the fine details of
atomic spectra, the so-ca lled hyperfine structure.
The interaction of this nuclear magnetism with an
external magnetic field was predicted to result in a
finite number of discrete energy levels known as the
Zeeman structure. However, the first direct
excitation of transitions between nuclear Zeeman
levels was by Isador Rabi in 1933, using radio-
frequency (RF) waves in an atomic beam apparatus.
In 1945, Felix Bloch and co-workers at Stanford,
and Edward Purcell and co-workers at MIT,
performed the first nuclear magnetic resonance
(NMR) experiments in condensed matter, with the
RF response of the hydrogen nucleus (proton) being
directly detected.
The early prospects for this new technique were
limited to precise measurements of magnetic fields
and nuclear magnetic moments. However, three
transformational discoveries intervened to set
NMR on a course that would result in initially
unimaginable contributions to physics, chemistry,
592 Nuclear Magnetic Resonance

engineering, medicine, geology, food science, and
biochemistry. In 1950, it was found that atomic
nuclei at different sites of a molecular orbital had
slightly different resonant frequencies, a phenom-
enon known as ‘‘chemical shift.’’ In the a same year,
Erwin Hahn discovered the spin echo, thus opening
the possibility that multiple RF pulse trains could be
used to remove unwanted nuclear spin interactions
while being used to manipulate spin coherences with
exquisite resolution. In addition, in 1951, using this
spin echo, Herbert Gutowsky and Charles Slichter
revealed a hitherto unobserved scalar spin–spin
interaction between nuclei, mediated by the mole-
cular orbital electrons.
The discovery of the chemical shift and the scalar
coupling would immediately revolutionize chemis-
try. Further discoveries of nuclear quadrupole
interactions and through- space dipol ar interactions
would add to the capacity of NMR to provide
insight regarding structure and order in the solid and
liquid crystalline state. But the spin echo would
provide a platform for new advances in science in
every one of the six decades following the discovery
of NMR in 1945. These were successively diffusion
and flow NMR, multidimensional NMR, magnetic
resonance imaging, protein structure NMR, ex situ
NMR, and quantum computing NMR.
Resonant Excitation and Detection
In quantum-mechanical language, the Zeeman
Hamiltonian H for a nuclear spin experiencing a
magnetic field B
0
along the laboratory z-axis may be
written as
H ¼B
0
I
z
½1
being the (nuclear) gyromagnetic ratio while I
z
is the
operator for the z-component of angular momentum,
with eigenvalues mh, m lying in the range I, I þ
1, ..., I. I is the angular momentum quantum
number, being either integer or half-integer. From the
Schro¨ dinger equation, it can be seen that the eigenkets
of H precess about the z-axis at a rate B
0
,the
frequency corresponding to the energy difference
between the 2I þ 1 Zeeman levels. For convenience,
we shall take the eigenvalues of I
z
to be simply m,
dropping the factor h, and leading to a Hamiltonian
expressed in frequency rather than in energy units.
Resonant excitation between the Zeeman levels is
achieved by the application of an RF (!) magne tic
field of amplitude 2B
1
linearly polarized normal to
B
0
such that the total Hamiltonian becomes
H ¼B
0
I
z
2B
1
cos !tI
x
½2
This excitation is easily applied by means of a
transversely oriented antenna coil, the same coil
generally being used to detect the nuclear spin
response. In the frame of reference rotating about
B
0
at !, the Hamiltonian transforms to
H ¼ B
0
!
I
z
B
1
I
x
B
1
expði2!tI
z
ÞI
x
expði2!tI
z
Þ½3
At resonance, ! = !
0
= B
0
. The last term in eqn [3]
averages to zero and may be neglected (the
Heisenberg condition) provided ! B
1
,thatis,
B
0
B
1
.GivenB
0
of the order of tesla and B
1
of
the o rder of millitesla, this condition is easily
satisfied. Hence, from the perspective of the
rotating frame, the spins at resonance see only the
static magnetic interaction B
1
I
x
, s o that applica-
tion of this resonant RF field causes spins to nutate
about the rotating frame x-axis at a rate B
1
. Thus,
by application of RF pulses of different duration,
and phases, one may produce arbitrary reorienta-
tion of the spins about various axes in the rotating
frame.
With the spin system disturbed from equilibrium,
the NMR ‘‘signal’’ is detected via the subsequent
free precession, and usually via the same antenna
coil used for resonant excitation, Semiclassically, the
phenomenon may be pictured as follows. RF
excitation nutates an initial z-magnetization into
the transverse plane of the rotating frame. Such
transverse magnetization corresponds the laboratory
frame to a magnetization precessi ng at the Larmor
frequency, thus inducing an oscillating emf in the
receiver coil. In the next section, we see how to
describe this phenomenon in the language of
quantum mechanics.
Typically, NMR is performed using the nuclei of
common atoms in organic molecules, (
1
H,
2
H,
13
C,
15
N,
19
F,
31
P) although for inorganic matter a wider
class of nuclei are available. Of all these, the
proton is most abundant and most sensitive,
having the highest gyromagnetic ratio, ,ofall
stable nuclei.
The Quantum Statistics of the
Spin Ensemble
The nuclear Zee man energy in typically available
laboratory magnetic fields, B
0
h, is many orders of
magnitude smaller than the Boltzmann energy, k
B
T,
except at millikelvin temperatures. At room tem-
perature in thermal equilibrium, the fractional
difference in populations between the Zeeman levels
Nuclear Magnetic Resonance 593

is normally very small, for example, for protons,
about 10
5
. Of course, the total number of spins
available may be very large, for example, on the
order of 10
20
.
The signal in magnetic resonance is detected as a
collective effect of the large ensemble of nucl ear
spins. The natural language of quantum statistics is
that of the density matrix, ; the time-dependent
expectation value for any observable represented
by an operator O is then, tr(O(t)), the diagonal
sum of the product of O and . The time evolution
of the density matrix is given by the Liouville
equation
i
@
@t
¼ H;½ ½4
where [ , ] is a commutator. For a constant Hamilto-
nian, this equation gives
ðtÞ¼expðiHtÞð0ÞexpðiHt Þ½5
Physical solutions to the density matrix (Liouville
space) are (2I þ 1)
2
(square) matrices formed in
the (2I þ 1)-dimensional angular momentum
eigenbasis. Generally, we may write the density
matrix in a representation o f irreducible tensor
operators. One very convenient representation is
the set formed by taking products of spin
operators. For example, in the case of spin-1/2
where Liouville space is 2
2
-dimensional, we may
write
ðtÞ¼
1
2
I þ a
x
I
x
þ a
y
I
y
þ a
z
I
z
½6
where I is the identity operator. The operators I
x
and I
y
provide the off-diagonal elements of and
define the degree of phase coherence in the
ensemble, while the operator I
z
defines the degree
to which the diagonal elements differ, thus defining
the polarization. a
x
and a
y
give the amount of ‘‘one-
quantum coherence’’ in the ensemble while a
z
gives
the polarization. In thermal equilibrium a
x
= a
y
= 0,
and the spin ensemble exists in a state of
pure longitudinal polarization given, in the high-
temperature approximation, B
0
h << k
B
T,by
eqbm
ð0Þ
1
ð2I þ 1Þ
I þ
hB
0
ð2I þ 1Þk
B
T
I
z
½7
This is the starting point for all NMR experiments
(Figure 1).
Consider then the detection of precession via the
Faraday induction. The size of the signal observed
will be proportional to the size of the transverse
magnetization M = tr[(I
x
þ iI
y
)(t)] present in the
rotating frame, this magnetization producing an
induced emf with real and imaginary components
because of the capacity of heterodyne receivers to
detect quadrature phase. In the laboratory frame,
the detected signal has a prefactor of B
0
reflecting the
Faraday induction, which, taken together with the
dependence of the initial equilibrium magnetization on
B
0
, gives an overall NMR sensitivity (B
0
)
2
, helping
to explain in part why high magnetic fields are
advantageous. Take the simple example for I = 1=2,
where a single 90
resonant RF pulse is applied to the
spin system, subsequent free precession occurring
under the Zeeman Hamiltonian. The density matrix
at detection is
ðtÞ¼expði!
0
tI
z
Þexp i
2
I
x
eqbm
ð0Þ
exp i
2
I
x
expði!
0
tI
z
Þ
¼expði!
0
tI
z
Þexp i
2
I
x
a
eqbm
I
z
exp i
2
I
x
expði!
0
tI
z
Þ
¼expði!
0
tI
z
Þa
eqbm
I
y
expði!
0
tI
z
Þ
¼a
eqbm
I
y
cosð!
0
tÞþa
eqbm
I
x
sinð!
0
tÞ½8
Noting tr(I
2
x
) = tr(I
2
y
) = tr(I
2
z
) = (1=3)(2I þ 1)I(I þ 1)
and tr(I
I
) = 0, the signal may easily be calcu lated
as S(t):a
eqbm
exp(i!
0
t), correspo nding, upon Fourier
transformation, to a unique frequency at !
0
. Note
that a basis consisting of products of angular
momentum operators are easy to handle since all
evolution properties follow from the usual angular
momentum commutation algebra.
The spin echo pulse scheme of Figure 2 is one of
the most important in NMR. It allows one to
refocus dephasing effects caused by inhomoge-
neous broadening, for example, due to the hetero-
geneity of the magnetic field across the sample.
Rewriting the density m atrix equation in the
rotating frame, replacing the Zeeman precession
I = 2 I = 1/2m
m
–2
–1
0
1
2
–1/2
1/2
γ
B
0
γ B
0
Figure 1 Schematic Zeeman levels for the case I = 2 and
I = 1=2. The bold lines indicate the relative population in each
state in thermal equilibrium.
594 Nuclear Magnetic Resonance
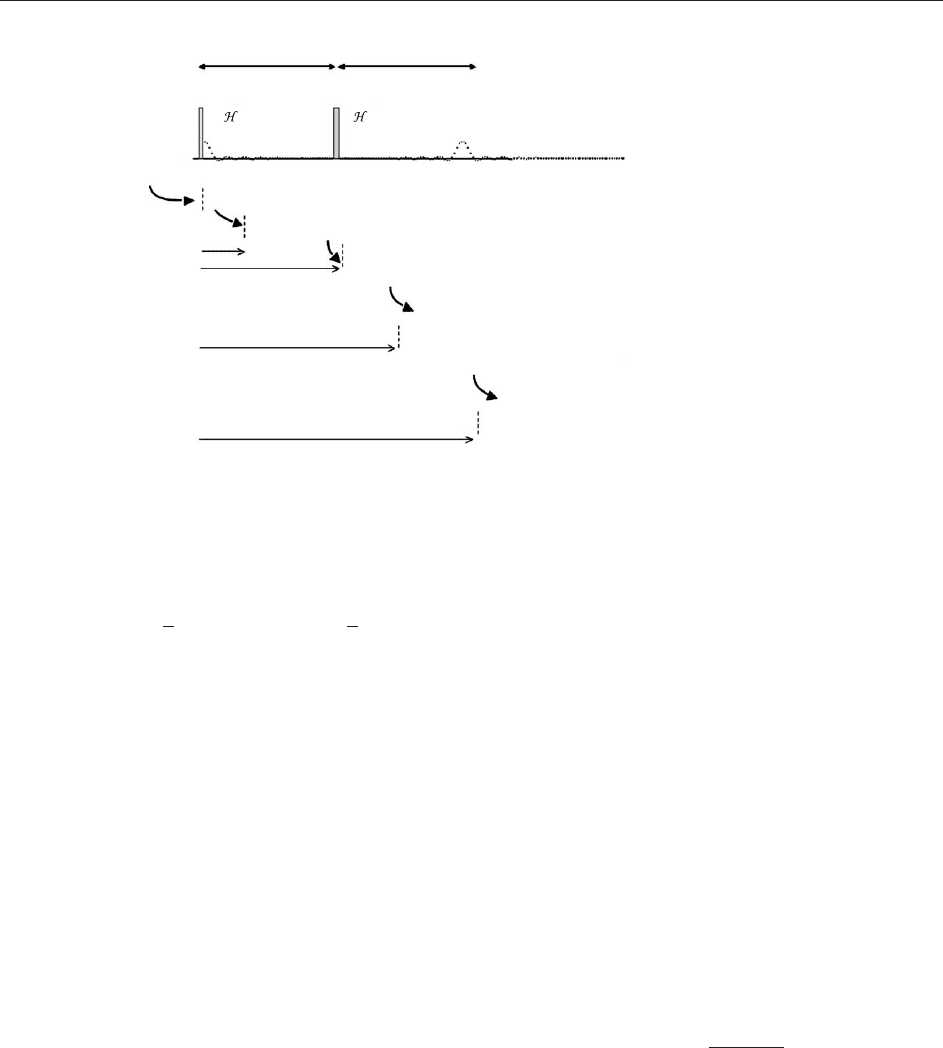
by its residual o ffset, and accounting for both RF
pulses,
rot
ð2Þ¼expði!
0
I
z
Þexpði I
y
Þexpði!
0
I
z
Þ
exp i
2
I
x
eqbm
ð0Þexp i
2
I
x
expði!
0
I
z
ÞexpðiI
y
Þ
expði!
0
I
z
Þ
¼a
eqbm
I
y
½9
Details of the density matrix evolution are given in
Figure 2. The inversion pulse has the effect of
completely reversing all the phase shifts that occur
during the first interval, resulting in an echo signal
when the two time periods are equal. Note the use
of nested operators representing the successive
influences of RF pulses (assumed to be ideal
rotations) and Hamiltonian evolutions. The overall
influence of the RF pulses is to render the effective
Hamiltonian zero in this case.
This echo sequence (and its equivalent multiple RF
train, the Carr–Purcell–Meiboom–Gill sequence) allows
one to remove the effect of magnetic field inhomo-
geneities so as to investigate the underlying homoge-
neous broadening and associated signal damping.
Spin Relaxation
The free precession of nuclear spins does not
continue indefinitely. Ultimately the off-diagonal
elements of the density matrix lose phase coherence
while the diagon al elements gradually return to their
thermal equilibr ium state, two processes known,
respectively, as T
2
(spin–spin) and T
1
(spin–lattice)
relaxation. The rate of relaxation depends on
interactions between the spins themselves and
between the spins and their thermal environment.
The process of T
1
relaxation requires f luctuations
that induce transitions between the Zeeman levels.
Clearly the relevant quantum-mechanical opera-
tors must possess a nonzero m atrix element
coupling the Zeeman levels, and the frequency of
those fluctuations must match the energy gap
spacing. Predominant in causing such relaxation
in diamagnetic environments are the internuclear
dipolar interactions, while in paramagnetic envir-
onments, dipolar interactions between nuclear and
electronic spins are effective. One simple way of
representing these processes is by the spectral
density function, the Fourier power transform of
their fluctuations, dipolar interactions causing
spin–lattice relaxation due to fluctuations at !
0
and 2!
0
. For a fluctuating interaction with correla-
tion time,
c
, that spectral density may approx-
imateaLorentzianoftheform
Jð!Þ=
c
1 þ !
2
2
c
½10
Thus, as the rate of molecular motions varies, due to
the influence of temperature on
c
, the T
1
relaxation
rate will be a maximum when !
0
c
= 1. Both solids
(!
0
c
1) and liquids (!
0
c
1) have long T
1
relaxation times while soft solids or complex liquids
may have faster relaxation. T
1
relaxation manifests
as an exponential return to equilibrium values of
longitudinal magnetization. Typical vales range
from hundreds of millis econds to hours, and the
need to re-establish equilibrium between repetitions
of the experiment can severely limit signal averaging
180°
y
90°
x
ττ
τ
τ
+ t
2
τ
=
–Δω
0
I
z
rot
=
–Δω
0
I
z
rot
ρ
rot
(0)
–
= a
eqbm
I
z
ρ
rot
(0)
+
= a
eqbm
I
y
ρ
rot
(2τ) = a
eqbm
I
y
ρ
rot
(t ) = a
eqbm
I
y
cos(Δω
0
t ) + a
eqbm
I
x
sin(Δω
0
t )
ρ
rot
(τ)
+
= a
eqbm
I
y
cos(Δω
0
τ ) – a
eqbm
I
x
sin(Δω
0
τ)
ρ
rot
(τ + t ) = a
eqbm
I
y
cos(Δω
0
τ)cos(Δω
0
t ) + a
eqbm
I
x
cos(Δω
0
τ)sin(Δω
0
t )
–a
eqbm
I
x
sin(Δω
0
τ)cos(Δω
0
t ) + a
eqbm
I
y
sin
2
(Δω
0
τ)sin(Δω
0
t )
t
Figure 2 Spin echo pulse scheme showing the evolution of the density matrix.
Nuclear Magnetic Resonance 595

and hen ce available signal-to-noise ratios. Note that
T
1
relaxation occurs by stimulated emission.
Spontaneous emission is effectively absent from
nuclear spin systems owing to the long-radiation
wavelength.
The case of T
2
(spin–spin) relax ation is inherently
more complex. First, the definition of ‘‘loss of phase
coherence’’ depends on the particular RF pulse
sequence employed. Second, the simple perturbation
theory description applied to T
1
relaxation only
works in the fast motion limit, where the T
2
relaxation rate may be shown to depend on spectral
density terms not only at !
0
and 2!
0
but also ! = 0.
In consequence, T
2
T
1
. T
2
relaxation is sensitive
to static components. These static components may
dominate in soft solids and solids. Indeed, any term
in the Hamiltonian which spreads sp in phases, and
which cannot be recovered by means of a judicious
RF pulse train, will contribute to T
2
relaxation.
Suppose the effective frequency distribution causing
dephasing is described by an ensemble second
moment <!
2
>, and exhibits fluctuat ions about a
mean of zero with correlation time,
c
. Then we may
identify two limiting cases: in the slow motion limit
<!
2
>
1=2
c
1, the decay of the detected magne-
tization is Gaussian, and given by a factor
exp(1=2 < !
2
> t
2
). In solids, the proton T
2
relaxation may take place in a few tens of micro-
seconds. In the fast motion limit <!
2
>
1=2
c
1,
the decay of the detected magnetization is exponential,
and given by a factor exp(<!
2
>
c
t). Liquid state
T
2
values approach T
1
under extreme narrowing
conditions.
The Details of the Nuclear
Spin Hamiltonian
Atomic nuclei interact with their environment, with
surrounding electrons, and with other nuclear spins.
It is precisely this feature that provides such a
sensitive probe of material structure and dynamics.
For a material immersed in a steady magnetic field
B
0
along the laboratory z -axis, the Hamiltonian for
the ith nuclear spin can be written
H ¼B
0
I
iz
I
i
:S
¼
:B
0
þ
X
j
J I
i
:I
j
þ
X
j
I
i
:D
¼
:I
j
þ I
i
:Q
¼
:I
i
½11
It is the variety of the terms in the nuclear spin
Hamiltonian that imparts power to NMR. The
first is the nuclear Zeeman interaction w ith the
applied magnetic field. In modern laboratory
superconducting magnets, this interaction can be
as large as 1000 MHz, although in earth field
applications it can be as small as 2.5 kHz. Given that
the sensitivity and resolution of NMR generally
improve with increasing magnetic field, the range of
100–1000 MHz is typically the operating regime of
choice. All other terms in the nuclear spin Hamiltonian
are smaller and thus act as first-order perturbations
only, projecting their quantum operators into the
zeroth-order Zeeman eigenbasis, the quantum frame
of the operator I
z
. Because several of the terms in
H depend on the orientation of the local nuclear
environment (e.g., the molecular orbital) with respect
to the magnetic field, these terms will fluctuate in the
presence of reorientational motions. By the Heisenberg
uncertainty principle, fluctuations faster in frequency
than the size of the Hamiltonian contribution,
expressed in frequency units, will result in an averaging
to the mean, a phenomenon known as ‘‘mo tional
averaging.’ ’
The term I
i
.S
¼
.B
0
is the chemical shift that occurs
for nuclei in molecular atoms, or the knight shift for
nuclei in metals. It is typically a few ppm to several
100 ppm (i.e., 100’s Hz to 10 kHz), depending
on the nucleus. S
¼
=
¼
is a tensor whose principal
axes (1, 2, 3) are associated with the local symmetry
axis of the molecular orbital (bond) in the vicinity
of the nucleus. For a liquid state molecule tumbling
rapidly and isotropically, only the averaged trace
of
¼
,
i
= (1=3)(
11
þ
22
þ
33
) survives under
motional averaging, giving a fixed freque ncy shift
i
B
0
I
iz
. However, in a solid-state environment,
the remaining terms also contribute to the aniso-
tropic chemical shift
H
CS
¼
i
B
0
I
iz
1
2
ð3 cos
2
1Þ
ð
33
i
ÞB
0
I
iz
½12
where is the polar angle between the magnetic
field and the principal axis (the axis ‘‘3’’).
The scalar coupling term,
P
j
JI
i
.I
j
causes each
(ith spin) energy leve l to be sensitive to the quantum
states of the neighboring j-spins, the coupling
constant J being typically tens to hundreds of hertz
for nearby spins, but reducing rapidly with greater
distance in the molecular orbital. Note that the
operator
P
j
JI
i
.I
j
is nondi agonal in the zeroth-order
representation, but provided that the chemical shift
between the I and j spins is larger than the coupling
frequency (known in chemistry as an AX spin
system), the operator reduces to
P
j
JI
iz
I
jz
the effect
being to split the i -spin resonance in to a multiplet,
depending on the state of the nearby j-spin. For m
identical nearby j-spins, the multiplet bears a simple
596 Nuclear Magnetic Resonance
