Holik H. (ed.) Handbook of Paper and Board
Подождите немного. Документ загружается.


3.4.3
Starch Application
As can be seen from Fig. 3.5, 80% of total starch consumption in the paper in-
dustry is used for surface application, either for surface sizing (62 %), or for spray-
ing (3%) on the wet paper web or between two layers in board manufacturing, or
as a coating binder (15%). The main role of surface size is to promote surface
properties, e.g. strengthen the paper surface and to bind particles such as fibers
and pigments more strongly to the surface. Additionally, the starch is expected to
add internal strength to the sheet through a liquid penetration in the z-direction.
Opposite to wet-end application, where starch retention is crucial, starch applied to
the surface is generally 100 % retained.
For production of woodfree uncoated and coated fine papers up to 40 kg starch
per ton of paper are applied. 3 to 10 kg starch is added at the wet end, with the aim
of internal strength improvement and retention increase. The major share of the
starch is added to the sheet in surface treatment. A mass balance on a typical fine
paper machine has shown, that more than 90% of the added starch is retained in
the final paper product. Losses occur mainly during the sheet forming process in
the wire section due to insufficient retention. Starch which is not held back in the
paper is discharged with the process effluents to the waste water treatment plant,
where a complete biodegradation process follows.
Packaging paper, e.g. testliner and corrugating medium, made from 100 % re-
covered paper, can only be produced economically and in the required quality by
adding cost effective dry-strength agents, i.e. biosynthetic starch products. There-
fore these papers are produced with an average starch consumption of 40 kg t
–1
,
mainly by surface application. A further 25 kg (t corrugated box board)
–1
is applied
as an adhesive in the converting plant. This means that a high amount of starch is
returned to the production process via recovered papers, where it is nearly not
retained in the paper sheet. Therefore this uncontrolled starch quantity leads to a
considerable load in the white water circuit (usual COD levels from 5.000 to
30.000 (mg O
2
) l
–1
) and finally also in the waste water. By applying starch with a
size-press to the paper surface, the paper strength will be increased by 30 to 60 %.
Additional energy is necessary for extra drying of the paper web and the paper
machine runnability may decrease.
To improve productivity and reduce production costs, effective and well con-
trolled starch application at the wet end is required (see Fig. 3.6). New practical
experience shows that a starch-polyvinylamine complex can be an alternative to a
surface treatment by size-press for strength improvement. A holistic picture of the
application of starch based on analytical investigations (in produced paper, exhaust
air, clarified waste water and soil) shows that starch is an extraordinary, envir-
onmentally friendly additive with a diversified functionality.
3 Chemical Additives72

3.5
Aluminum Compounds [3, 9]
Aluminum compounds are widely used in the paper industry: in rosin sizing, as
drainage and retention aids, and as fixing agents for anionic trash (dissolved or-
ganic compounds e.g. extracts from wood fibers, degraded starch). Important ap-
plications are the treatment of circuit water and effluents or the thickening of
sludge, where the flocculation and precipitation effects of aluminum ions are uti-
lised. The most frequently applied aluminum compounds are aluminum sulfate,
polyaluminum chloride (PAC), polyaluminum hydroxidechloride sulfate, and re-
cently also (poly)-aluminum nitrate and polyaluminum nitrate sulfate. Aluminum
compounds have a favorable price-performance ratio. An analysis of the alumi-
num concentration in the production of a wood-containing printing paper (at neu-
tral pH, with 0.45% aluminum sulfate added to the white water) showed an alumi-
num level in the water circuit (at a pH of approx. 7.5) of about 0.1 mg l
–1
, max-
imum 0.2 mg l
–1
(determined by UV/VIS spectroscopy). This indicates that the
paper retains nearly all of the aluminum added in the form of aluminum hydrox-
ide. The anionic counterions sulfate and chloride, however, are nearly fully dis-
charged into the effluent. The paper retains the share of ions dissolved in its
residual moisture at the end of the drying section. Sulfate and chloride ions are
nonvolatile, i.e. they are not released into the atmosphere. The loss via paper-
making residues is negligible. The two anions may be considered environmentally
harmless. As sulfate promotes the corrosion of concrete, there may be statutory
discharge limits for this substance (500 mg sulfate l
–1
). There are usually no re-
strictions, however, on the discharge of chloride.
The use of polyaluminum nitrate and polyaluminum nitrate sulfate is fairly new.
The two compounds help to prevent or combat malodours caused by the conver-
sion of sulfate into hydrogen sulfide under anaerobic conditions. The results of
mill trials suggest that nitrate is converted into elemental nitrogen because no
nitrate could be detected in the effluent – nitrate was found only in the white water.
According to information from mills using large quantities of polyaluminum ni-
trate, a minor discharge of nitrate into the effluent can occur [3].
3.6
Functional Chemicals
All chemical additives which are used to obtain certain properties of the finished
paper, e. g. color, improved resistance to ink or water, gloss, printability, strength,
are called “functional chemicals” (see Fig. 3.3.).
3.6 Functional Chemicals 73

3.6.1
Coloring Materials (Dyes) [3, 9–11]
3.6.1.1 General
Historically, dyes are the oldest class of synthetic specialty chemicals employed in
the production of paper. Dyes are added to paper at a statistical average rate of only
150 g chromophore per 1 ton of paper and board worldwide. The turnover in dyes
(total 500 V 10
6
€) accounts for 5% of the specialty chemicals applied to paper but
only for 1,5% in view of solid mass addition (Fig. 3.2 and 3.3).
Colorants are used in paper manufacture for a variety of reasons, including eye
appeal, color coding, and brand identification. The selection of suitable colorants
depends for instance upon end-use requirements, physical and chemical proper-
ties, and handling characteristics. Coloring is done to achieve the following two
general goals:
• to produce paper having a given color or shade
• to produce white paper having a desired tint.
Coloring is rather tricky:
• Process variables may interfere with dyes.
• Specific dyes react differently in different furnishes.
• Dyes may adversely interact with other additives – primarily through charge
interaction – and interfere with them.
Additional parameters have to be considered:
• In the case of food contact, bleeding has to be assessed according to DIN EN
646
• For advertisement use the light fastness is important
• Dyes have to meet the toxicological and ecological requirements, e.g. German
“Technical Regulations of Risky Substances” and EU guideline 2002/61/EG
(19. 07. 2002). Therefore, no listed aromatic amines are released after reductive
cleavage of any of the azo-bonds.
3.6.1.2 Classes of Coloring Material
3.6.1.2.1 Anionic Direct Dyes
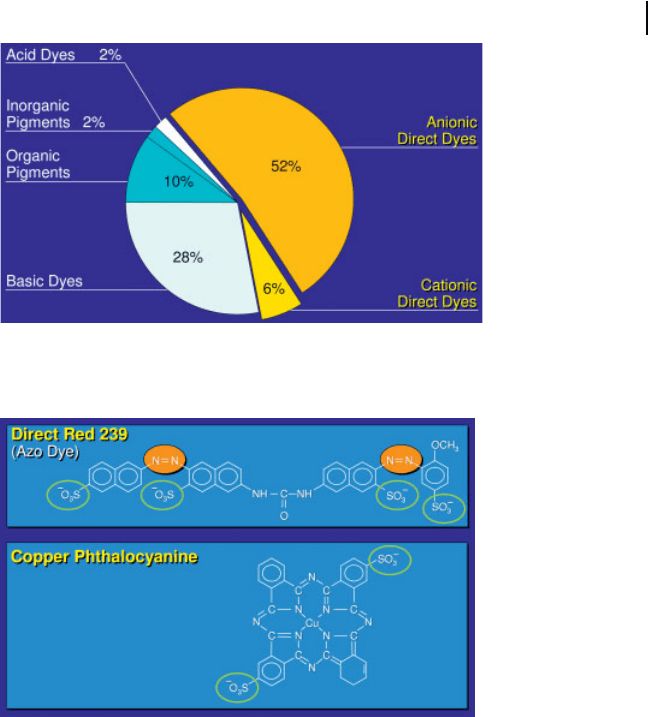
These are the dominant class of dyes. They account for 52% of the total worldwide
turnover, followed by basic dyes with 28 % share (Fig. 3.7). Anionic direct dyes are
the sodium salts of azo dyes containing sulfo groups to give water solubility. An-
other group are copper phthalocyanines which also contain sulfo groups (Fig. 3.8).
They have a high affinity to bleached chemical pulps, and often do not need addi-
tional fixing agents, unless very deep shades with good bleed fastness are required
(e.g. deeply colored napkins). Suitable fixatives are condensation products of or-
ganic amides with formaldehyde, however, they are not very well suited for the
dyeing of wood-containing stocks because the incrusting components (binding
3 Chemical Additives74
substances) of wood prevent uniform dyeing. Therefore a mottling tendency is
occasionally observed with stock mixtures (bleached chemical and mechanical
pulp; recovered paper). The lightfastness is usually good, but the colors attainable
may not be as brilliant as with acid and basic dyes.
3.6.1.2.2 Cationic Direct Dyes
These retain the planar molecular structure of acid direct dyes, but cationic groups
have been incorporated in the structure. This is to increase their affinity to paper
fibers. They are moderately adsorbed on bleached lignin-containing stocks, and
even in the case of deep shades, alum or a fixative is usually not required. These
dyes produce fairly good bleed fastness. Combination dyeing with anionic direct
dyes can be carried out to improve the fixation of the dyes on the paper machine
Fig. 3.8 Chemical structure of two direct dyes.
Fig. 3.7 Classes of coloring materials in 2004 (market shares
at world wide turnover of 500 V 10
6
€).
3.6 Functional Chemicals
75

and to reduce the contamination of the circuit process water and the waste water.
In most cases combination dyeing leads also to an improved bleed fastness.
3.6.1.2.3 Basic Dyes
These are the salts (chlorides, hydrochlorides, sulfates, and oxalates) of color bases.
They are soluble in acid aqueous solutions, and this is often the reason for the use
of acetic acid to produce concentrated dye solutions. Such liquid dye formulations
often contain 5–25% acetic acid and 5–20% of a glycol derivative for stability
reasons, along with ca. 25% chromophor. The chromophors have a very high affin-
ity for paper fibers, especially to unbleached and mechanical pulps (lignin-contain-
ing fibrous material) and also for fillers. This is due to their high cationic charge
density, corresponding to a 99 % fixation in the finished paper. Salt formation gives
rise to very stable color lakes that are insoluble in water.
Anionic fixatives, e.g., the sodium salt of condensation products of formalde-
hyde and naphthalene sulfonic acid, also form color lakes of this type with cationic
dyes. They are used to improve the fixation of basic dyes on bleached pulps that
contain only small amounts of lignin. Better dye fixation always results in richer
colors, improved bleed fastness, and less effluent staining.
Basic dyes are mainly used in the production of packaging papers (liner, tes-
tliner, corrugating medium; 55 % of total volume), for tinting of wood-containing
printing papers (20%) and colored, wood-containing printing/writing papers
(15%).Within the class of basic dyes, brown grades have the greatest volume. The
main reason for this is that recovered paper is nearly the sole source of packaging
paper in many countries. Brown dyes in testliner should imitate unbleached kraft-
liner, enhance the visual appearance and characterize certain qualities. Two typical
representatives of these dyes are “Basic Brown 1”, an azo-dye, and “Basic Brown
19”, a methine dye, corresponding to the international color index (Fig. 3.9). The
strong cationic charge centers of both dyes give a very high affinity to the paper
stock. Because of the molecule doubling the methine dye shows even better fixa-
Fig. 3.9 Chemical structure of two basic brown dyes.
3 Chemical Additives
76

tion. Basic dyes are brilliant and tinctorially strong, resulting in good fastness to
water and steam on groundwood-containing furnishes. Disadvantages are poor
affinity to bleached furnishes, mottling tendency (i.e., parts of the paper stock or
stock mixture are dyed more deeply than others), and poor lightfastness.
3.6.1.2.4 Acid Dyes
These are all water-soluble salts (usually in the form of the sodium or potassium
salt) of colored organic acids which dissociate in water to form colored anions.
Most acid dyes are azo dyes and are similar to direct dyes. These two groups
overlap with no distinct boundary. Acid dyes generally contain more acid groups
which gives rise to their greater solubility in water, compared to direct dyes.
Acid dyes have little affinity to paper fibers. Although the dye molecules pene-
trate well into the capillaries of the fibers, they must be fixed with aluminum
sulfate (pH ca. 4.5) and cationic fixing agents, e.g., condensation products of di-
cyandiamide with formaldehyde, polyamines, polyvinylamines, in order to achieve
a satisfactory dye yield. Further disadvantages are relatively strongly colored waste
water/effluent and poor bleed fastness. Advantages of acid dyes are good solubility,
no tendency to mottle on stock mixtures, and they are very well suited to dip and
surface dyeing. Acid dyes are mainly used for (certain) writing and printing pa-
pers, decorative crepe paper, and carbon paper. The importance of acid dyes for
paper dyeing has decreased tremendously because of their poor cost/performance
ratio and the above mentioned waste water problems.
3.6.1.2.5 Colored Pigments
These fall into three classes: natural inorganic pigments, synthetic inorganic pig-
ments, and synthetic organic pigments. The third class is by far the most im-
portant for the paper industry. The natural inorganic pigments such as ocher, terra
sienna, or umber are of little significance at present. The synthetic inorganic pig-
ments (iron oxide, cadmium, chromium oxide) the organic pigments (azo and
polycyclic), and the metal complex pigments (phthalocyanine) are the important
ones.
In the pigments the colorant is present in a water insoluble, finely dispersed
form. The binding of the pigment to the fibers is improved by rosin sizing and the
use of aluminum sulfate. The pH of the system must be kept below 5.0 in order to
keep the alum in an active form for retention. Fixing agents and effective retention
aids must be employed during papermaking in neutral or slightly alkaline systems.
The lightfastness of pigments is excellent and mottling does not usually occur. The
low tinctorial value of the colored pigments means that high loading levels are
required to achieve strong colors, which leads to a weakening of the sheet and to
high costs. Therefore these pigments are only used for specialty papers where a
very high lightfastness is essential, e. g. bookkeeping and label papers, document
and laminate base papers, lightfast printing and writing papers.
3.6 Functional Chemicals 77

Colored pigments are applied more often in surface coloring and coating for-
mulations where they are superior to soluble dyes, because they do not follow the
water into the paper sheet when the starch or size or pigment coating is applied.
3.6.1.3 Dyeing Processes
3.6.1.3.1 Stock Dyeing
This is also called internal dyeing and is the most widely used paper dyeing proc-
ess. Because of clean working conditions and the most efficient usage, the dyes are
now mostly added continuously and fully automatically into the stock flow. More
seldomly the dyes are metered batchwise in the pulper or mixing chest.
The choice of dye and the fixing and dyeing conditions largely depend on the raw
materials used in papermaking (recycled fibers, stone groundwood, TMP, CTMP,
unbleached or bleached chemical pulp, type and portion of filler) and on its prepa-
ration process e.g. a higher degree of beating of the pulp results in a deeper
coloring. Fillers increase the required amount of dyes because they absorb dyes
and, at the same time, reduce the coloration owing to their covering power and
brightness. Fillers also lead to an increase in the two-sidedness of the paper
sheet.
In continuous dyeing, the point of addition is also determined by a few factors
e.g. high consistency dyeing at a stock consistency of 3–4 % (before mixing with
white water ahead of the headbox) and alternatively low consistency dyeing at a
stock consistency of 0.5–1.5% (in front of the mixing pump or pressure screens).
The pH conditions are very important. The addition of aluminum sulfate usually
promotes the absorption of dyes and yields less colored waste water and effluent.
In general, there is a trend towards paper production in the neutral or alkaline pH
range. These conditions need dyes with a very good affinity to the paper stock in a
neutral medium and/or very effective fixatives and retention aids.
The two stock dyeing processes have the following advantages and disadvan-
tages:
Batch addition has the advantage of thorough mixing of the additives with the
paper stock and optimal fixation due to longer contact time between the fiber and
dye. The disadvantages are that the time required for color correction and color
change is relatively long (loss of productivity). The handling of the dyes is more
problematic with regard to clean working conditions and to an exact and regular
metering control system. An integration of a continuous on-machine dye shade
measurement with the metering of the dyes is not possible (less productivity).
Continuous addition has the advantages of a short length in the stock line that
must be cleaned when the color is changed, and of less broke because the desired
shade is attained more quickly (higher productivity). However, a lower color yield
(low contact time) is obtained for intensely colored papers. The more complex
equipment required for this dyeing process must be taken into consideration as
well. On the other hand, control of the shade of the paper produced by continuous
color measurements in the paper machine, and fast adjustment of the feeding
pumps, lead to less broke and thus higher productivity.
3 Chemical Additives78

A specialty of stock dyeing is the so-called tinting, mainly used for printing and
writing papers. This procedure basically consists of counteracting the slight yellow
tinge of all paper stocks by adding a violet dye or a combination of pure blue and a
brillant red dye, which leads to a slightly blue shade. The human eyes perceive this
shade as more bright.
3.6.1.3.2 Surface Coloring
Here liquid dyes are added e.g. during the size preparation for size press or film
press application. Other additives e.g. starch, synthetic sizing agents, optical
brighteners are also applied in this way. For such product combinations negative
interaction of any kind must be avoided. An essential prerequisite for uniform
dyeing is adequate and, above all, uniform absorbency of the paper. The advan-
tages of this process are: quick changeover of shades, the possibility of only one-
paper-side dyeing (typical paper grades for this are liner and testliner), absence of
dyes in the water circuit, and, in the case of papers with higher basis weights
(> 80 g m
–2
), the saving of dyes. Nevertheless, compared with stock dyeing – the
classical dyeing process – surface dyeing has gained acceptance only in individual
cases because really uniform dyeing of the paper is difficult to achieve. It is occa-
sionally advisable to combine stock and surface dyeing, e. g. to correct two-sided-
ness. The bleed fastness of surface dyed papers is generally lower than for internal
dyed papers.
3.6.1.3.3 Dip Dyeing
A small group of specialty papers, called effect papers (flower crepe paper, tissue
paper), is noted for its intense brilliant shades. The paper is passed through a
dipping bath containing an aqueous solution of the dye or dye combinations. The
excess dye liquor is pressed off between two rolls and the wet paper is creped, if
required, before drying. Acid dyes are usually used because they have high sol-
ubility and bright shades. The low affinity of these dyes for fibers results in uni-
form dyeing, even in the case of papers with greatly varying fiber composition. The
bleed fastness of dip dyed paper is poor, corresponding to that of surface-colored
paper and even poorer than stock dyed paper.
3.6.1.3.4 Surface Coloring by Coating
In the usual coating process, the surface of the paper or board is covered with
white pigments. In the case of colored coatings, the starting material is also a white
pigment coating mixture, and the desired shade is attained by adding a dispersion
of a colored organic or inorganic pigment. This coloring method and these colored
pigments are mainly used for specialty paper and board e.g. for labels, documents,
impressive image brochures and packaging materials. E.g. for a bronze-glazed
paper surface aluminum or brass powder is added to the coating color, which
produces a silver or gold effect or, in combination with soluble dyes, a metallic
3.6 Functional Chemicals 79

effect. Water-soluble dyes can be used only to a limited extent because, even with
the use of fixatives, bleeding cannot be prevented (migration into the base paper
and into moist surfaces). Also, the inadequate lightfastness compared to pigments
limits the use of these dyes.
3.6.1.4 Requirements of Colored Paper and Board
Depending on the intended purpose of the paper, different fastness properties are
required:
3.6.1.4.1 Light Fastness
This is defined as the fastness of a dyed paper to the action of light. It is deter-
mined by both the dye used and the raw materials of the paper. The degree of
lightfastness is specified by a test method according to DIN 54 003, which is also
used in the textile industry. Originally lightfastness was tested by exposing the
dyed material to sunlight under defined conditions. Today artificial light with a
radiation spectrum similar to sunlight is used (Xeno test apparatus or Fade-Ome-
ter). The lightfastness cannot be given as an absolute value but can only be ex-
pressed in relation to a standard which is exposed simultaneously. In the textile
and paper industry, the Blue Wool Scale is used as a standard for comparison. It
consists of blue dyeings with lightfastness ratings from 1–8. Rating 1 signifies the
lowest, and rating 8 the highest lightfastness. A dyed paper with a lightfastness of
1 will change its shade after one hour “sunlight” exposure. However, the shade will
not fade completely until it has been exposed for several hours, depending on the
depth of shade and the fibrous material. Since paper is normally not subjected to
such severe exposure, a lightfastness of 1 is sufficient for all short-lived paper
grades e.g. newsprint, magazines and grades based on mechanical pulps (e. g. for
coating base paper, notepad) and/or mixed recovered paper (e. g. for liner, test-
liner). A lightfastness of 3 corresponds to a resistance of several days exposure.
Dyes with this lightfastness rating can be used for most paper articles, provided
that the paper stock, too, has approximately the same lightfastness e. g. for all
kinds of high grade printing and writing papers. Dyed paper with a lightfastness
rating of 5 does not undergo any change in shade, even on exposure to direct
sunlight for several weeks, this rating is needed e.g. for document paper, photo-
graphic paper, laminating base paper.
3.6.1.4.2 Bleed Fastness
This is required for paper and board that are used for food-packaging and tissue
papers (napkins and hygienic papers). According to the regulations, tests must be
carried out in each case to determine whether and to what extent dye can migrate
from a colored paper onto the packed foodstuff or to human skin. The paper may
come into contact with water, dilute fruit acid, grease, oil, alcohol or alkali. A test
specimen is placed between two uncolored glassfiber papers moistened by dipping
3 Chemical Additives80

into the test solution. This sandwich is then placed between two glass plates of the
same size. The whole is then wrapped airtight in a polyethylene film and loaded
with a certain weight. The specimen is kept in this condition for 24 h at 20 °C.
After drying, the coloration of the glassfiber paper is compared with the Grey Scale
to assessing the fastness to bleeding according to DIN 54 002. It should be empha-
sized that the bleed fastness is not only dependent on the dye but also on the
fibrous material and the type of dye fixation. A reliable prediction can be made
only on the basis of tests with the paper in question. For napkins and hygiene
papers a number of direct dyes are suitable and for wood-containing papers and
testliner a good bleeding fastness can be obtained with basic dyes.
3.6.1.4.3 Other Properties
Rub resistance is required for album and wrapping paper and for cover board.
Direct dyes are suitable for this purpose.
Acid resistance is required for parchment and vulcanized fiber base paper, writ-
ing, and printing paper. Organic and inorganic pigments, selected representatives
of all colorant groups, are suitable for this purpose.
Solvent resistance is required for labeling paper (packaging of perfumes, medi-
cines, and spirits). Special pigments are suitable for this purpose.
Heat resistance is required for cable and core paper and laminate paper. Organic
and inorganic pigments are suitable for this purpose.
3.6.2
Optical Brightening Agents (OBA) – Fluorescent Whitening Agents (FWA) [2, 9, 11]
OBAs, also called FWAs, represent 3% by value and only 1 % of the total dry
amount of specialty chemicals (see Fig. 3.2 and 3.3). They increase the whiteness/
brightness of paper and are preferably added to the stock. They are very effective
when used with highly bleached pulps, and much less effective, or even ineffective,
when applied to unbleached chemical pulps and mechanical pulps. OBA are also
used in surface applications such as surface sizing and paper coatings.
OBA absorb light in the ultraviolet spectrum range (below 370 nm) and re-emit
the light in the visible blue range (peaking at 457 nm). This results in a fluorescent
effect with bright white in daylight masking the inherent yellowness of the raw
materials. Any material that absorbs ultraviolet light will lower the efficiency of
fluorescent whiteners. For example, lignin absorbs ultraviolet light and the higher
the lignin content of the pulp, the less effective is the OBA. Hence, mechanical
pulps and unbleached pulps are less susceptible to whitening with OBA. Some
filler clays tend to counteract the fluorescence and reduce the effect of OBA. Fillers
such as calcium carbonate and aluminum trihydrate reflect ultraviolet light,
thereby enhancing the effect of OBA. A high pH (above 6) also helps to achieve
maximum whiteness. On the other hand TiO
2
absorbs UV, thus OBA cannot be
used in conjunction with high TiO
2
loading.
3.6 Functional Chemicals 81
