Holik H. (ed.) Handbook of Paper and Board
Подождите немного. Документ загружается.


Substances of different chemical composition and different biological origin are
used for fluorescent whitening of paper. Derivatives of diaminostilbene disulfonic
acid have proved the most popular in industry because of their fastness properties.
The active ingredient content is generally between 20 and 27%. Their central part
is disulfonated diaminostilbene. The types differ in the number of sulfonic acid
groups in the side groups. The high-substantivity types with only 2 sulfo groups
make up about 11% of the market while tetrasulfonated derivatives with medium
substantivity are about 80%. The rest is low-substantive, hexasulfonated types.
The paper industry today needs fluorescent whitening agents to obtain high
degrees of whiteness at reasonable cost, especially if the FWA (OBA) are combined
with shading dyestuffs. This in turn leads to sharper contrasts in the printed image
and thus helps to reduce the toner consumption of copiers, for example. Similarly,
the color brilliance of color prints is improved. For application in the stock, high or
medium-substantivity types are generally used, being 1% of the commercial-grade
product. For surface application in the size or film press, medium or low-sub-
stantivity types are used, the normal addition being 1.5% and in exceptional cases
up to 3.5%. Low or medium-substantivity FWA are also the preferred products for
coating application, the amount required being up to 1.5% up to 3.5 % in excep-
tional cases. A maximum amount of FWA can be used in each application as the
shade becomes greener. This will lead to a graying effect on the brightness.
3.6.3
Chelating Agents – Complexing Agents [3–5]
The presence of heavy metals negatively influences many production processes
and paper properties. In particular, they interfere with the bleaching agents in
chemical and mechanical pulp production, reducing their effect, so the task of
chelating agents is to counteract this detrimental impact. Chelating agents can also
reduce or stop uncontrolled decomposition of a hydrogen peroxide solution, which
would result in loss of bleaching effectiveness.
Chelating agents that contain amino and carboxyl groups mask metal ions effec-
tively. Chemical compounds of this type are nitrilotriacetic acid (NTA), ethylene-
diaminetetraacetic acid (EDTA), diethylenetriaminepentaacetic acid (DTPA), and
hydroxyethylethylenediaminetriacetic acid (HEEDTA). Other complexing agents
include the soluble salts of oxalic acid, citric acid, tartaric acid, gluconic acid,
amines, and ammonia.
Both the demand for increased brightness and the technology trend to elemental
chlorine-free and total chlorine-free bleached pulps has led to an increased use of
chelating agents today, dominated by DTPA and EDTA.
In view of environmental protection, there exists a controversial discussion
about the biodegradability of chelating agents. The results of newer investigations
show that, under certain conditions, EDTA and, with some restrictions, DTPA, are
fairly biodegradable. Their degree of biodegradability is still under discussion. It is
strongly influenced by pH (optimum at pH 8.5–9), by ultraviolet light and by the
effectiveness of adapted microorganisms. Up to 90% biodegradability of EDTA is
reached in some pulp and paper mills.
3 Chemical Additives82

Of less importance are chelating agents based on polyphosphates, phospho-
nates, and hydroxycarboxylates as well as nitrogene free products like citrate, ta-
trate and gluconate.
3.6.4
Sizing Agents [2, 3, 9–11]
3.6.4.1 Fundamental Aspects of Paper Sizing
Sizing makes the native fiber network hydrophobic and thus prevents or reduces
the penetration of water or other aqueous liquids into the paper. Sizing prevents
the spreading and strike through of ink or printing colors. Papermaking fibers
have a strong tendency to interact with water. This property is important for the
development of strong interfiber hydrogen bonds, especially during drying, and is
also the reason why paper loses its strength when rewetted. A high absorbency is
important for a few paper grades such as toweling and tissue. Also corrugated
medium paper must be “absorbent” to a certain degree to convert properly in the
corrugating process. On the other hand such properties are disadvantageous for
many paper grades e.g. liquid packaging, top layer of corrugated board, writing
and printing papers and most of the specialty papers. The water and liquid absorb-
ency can be reduced by the addition of sizing agents to the paper stock and/or by
their application to the paper surface.
Since the invention of paper about 2000 years ago, it has been treated or sat-
urated with mucilage and rice starch for surface glazing properties, so that people
could write on it. After 1280 A.D. animal glue was the principal sizing agent.
Alum (potassium aluminum sulfate) was used to harden the applied glue. Alum
rosin sizing entered the scene in 1806 (Moritz Friedrich Illig). In 1876, paper-
maker’s alum, Al
2
(SO
4
)
3
, was introduced. Since the 1950s the various forms of
rosin size (paste, dispersed, fortified), alkyl ketene dimmer size (AKD), alkenyl
succinic anhydride size (ASA) and polymers mainly based on styrene acrylate and
styrene maleinate (PSA) have come to the market. Today, beside starch for strength
improvement (Fig. 3.2) and polymer binders for paper coating, sizing agents are
the most important quality improving additives in paper manufacturing. Amongst
the specialty chemicals, sizing agents represent a share of 10% in dry mass
(Fig. 3.3.) and a total worldwide turnover of approximately 950 V 10
6
€.
3.6.4.2 Product Classes
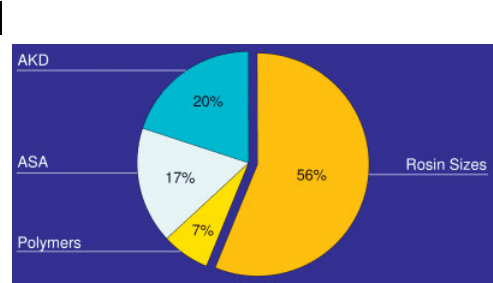
The main classes of chemicals and their consumption as a proportion of the total
worldwide usage of 350000 tons dry p. a. is shown in Fig. 3.10. Because of the
trend to neutral or slight alkaline papermaking and the increasing use of calcium
carbonate as a filler and coating pigment, the proportion and also the absolute
volume of rosin will decline further and the synthetic products will steadily
grow.
3.6 Functional Chemicals 83
3.6.4.2.1 Rosin-based Sizing Agents
These are still the most widely used of all wet-end sizes, because they are cheap to
produce and are produced from regenerative raw materials. They are mainly used
in acid and much less in pseudo-neutral pH conditions. They have the advantage
that the degree of sizing can be controlled very easily, and the sizing effect is fully
cured when the paper leaves the machine. In practical use there are different
preparations of rosin, for which the following terms are used:
• Free rosin: Resin acids whose carboxyl groups are in the completely protonated
form.
• Dispersed size: High (95–100%) free rosin dispersion of resin acids. The dis-
persed size particles are stabilized by surfactants, starch, polymers, or pro-
teins.
• Rosin soap size: Rosin size composed of the sodium salts of resin acids. Formed
by neutralizing (saponifying) resin acids with NaOH or some other sodium
base, such as Na
2
CO
3
. Different forms of soap sizes are available, including
paste, extended, and dry sizes.
• Extended size: A 50% solids product consisting of a 50:50 mixture of completely
neutralized resin acids and urea.
• Fortified size: Rosin size that has been reacted with fumaric acid or maleic anhy-
dride to form the Diels-Alder adduct. The reaction product contains extra car-
boxyl groups and produces more efficient sizing response than the unreacted
resin acids. It is used as a starting material for other size products.
• Dry size: A 100% neutralized, dry rosin size product that dissolves easily in
water.
• Paste size: An 80 % neutralized rosin size product marketed as a 70–80% solids
paste.
3.6.4.2.2 Synthetic Sizing Materials
These were only developed in the second half of the last century. Driving forces
have been the increasing use of the cost and performance effective calcium carbon-
Fig. 3.10 Product groups of sizing agents worldwide in 2004
(% shares of solid or active substances).
3 Chemical Additives
84

ate as filler and coating pigment and also the need to produce paper with high and
permanent paper strength. To enable its use the paper has to be produced under
alkaline conditions. Other paper grades must have very hard sizing in order to
resist edge wicking in liquid packaging applications or photographic base paper, or
to exhibit hot water resistance for gypsum board. Many mills also seek an alter-
native sizing procedure to the wet-end application, e.g. in the form of a surface
treatment in the drying section, to obtain higher efficiency.
• Alkyl Ketene Dimer (AKD) was the first synthetic size and appeared in the patent
literature in 1953. Structurally, alkyl ketene dimers are unsaturated lactones. In
the manufacturing process a frequently used synthesis route is: the acid chloride
of a carboxylic acid is prepared (fatty acids, C
16–22
homologs treated with, e.g.
phosphorus trichloride, phosphorus pentachloride, thionyl chloride or phos-
gene), followed by intermolecular lactone ring condensation via a labile inter-
mediate carboxylic acid obtained by dehydrohalogenation of the acid chloride
with triethanolamine in an organic solvent. Newly developed processes work
without any solvent. To handle and to use AKD in a paper mill, the wax must be
converted into tiny particles (0.5–2 mm) dispersed in water. The emulsification is
usually effected in a hot (75–90 °C) solution containing the cationic starch stabi-
lizer (alternatively a cationic polymer e.g. polydadmac, polyvinylamine) and a
small amount of surfactant, e. g. sodium lignin sulfonate, to which are fed AKD
flakes. After melting the AKD, the mixture is forced through a microfluidizer
and cooled. Small amounts of a promoter (cationic polymer with low molecular
weight and high charge density) and biocide can also be included. The solid
content of the commercial products varies between 6 and 30%. Some scientists
believe that AKD reacts with the hydroxy groups of cellulose to form b-keto
esters.
• Alkenyl Succinic Anhydride (ASA), as a synthetic sizing agent for paper, was first
synthesized in 1974. It is composed of an unsaturated alkenic hydrocarbon back-
bone coupled to succinic anhydride. It is usually manufactured in two stages:
First an unsaturated linear or branched olefin, e.g. a 1-alkene, is isomerized by
moving its double bond randomly from its a-position. This will yield an ASA
product that is liquid at room temperature, a condition important for easy emul-
sification of the size on application in the paper mill. In the second step, the
mixture of isomerized alkenes is reacted with maleic anhydride to produce the
ASA raw material. Normally ASA is a yellowish oily product. This 100% active
substance can be stored as such for a long time but must be well protected from
water or humidity.
• Polymeric Sizing Agents (PSA) are mainly based on styrene-acrylates, polyure-
thanes or styrene-maleic acid anhydride. They are either water-based dispersions
or aqueous solutions. These products were developed around 1960. Since that
time an increasing number of size-presses for strength improvement with
starch have been installed.
The styrene-maleinate polymers are anionic, aqueous solutions with a solid con-
tent of 20–30% and an alkaline pH (mainly containing the ammonium salt).
They are specially designed for surface application. Their advantages are very
3.6 Functional Chemicals 85

good compatibility with all other anionic chemicals (e.g. native and anionic
starches, CMC, optical brighteners, anionic polyacrylamides) and high stability
in the size-press or in water doctors. The disadvantages of styrene-maleinate
polymers are that the sizing effect depends on a certain amount of alum in the
base paper, the improvement of printability on wood-free white papers is lim-
ited, and these products are not suitable for wet-end addition (internal sizing).
In respect of this, the cationic styrene-acrylate dispersions are much more com-
monly used. They possess high paper stock affinity and the sizing effect is
largely independent of the alum content and pH in the stock suspension. There-
fore they can be used for nearly all paper grades, depending on the cationic
density of the polymer. With increasing cationic density a reduction in the opti-
cal brightening and the paper brightness will occur. Most products of this class
are suitable for wet-end and surface application. The sizing effect is developed
within the paper machine. The sizing is improved by intense contact drying on
hot metal surfaces (e. g. drying cylinders). The sizing obtained is resistant to
acids and alkalis. The surface properties of the paper are improved, especially
pick strength and printability and linting and dusting are also reduced.
3.6.4.3 Application Advice
• Rosin: The average addition rate is 1% solid calculated on paper, the individual
rate depending on the required degree of sizing. With these products this degree
can be adjusted very precisely and is fully obtained directly after the paper has
been dried. The product is usually added to the stock suspension upstream of
the headbox and then fixed chemically, in the simplest case with aluminum
sulfate (alum). The retention of rosin size is in the region of 70 % if the usual
fixing agents are employed. Rosin sizes are not usually suitable for surface ap-
plications.
• AKD: As little as 0.005–0.008% reacted AKD is believed to yield sufficient
monomolecular coverage of the paper stock, while dosage rates are considerably
higher and typically vary from 0.05 to 0.2%, calculated as solid AKD wax based
on paper stock. The stability to hydrolysis of most commercial products has been
improved significantly in recent years. The ketone resulting from the hydrolytic
decomposition of AKD is a solid (melting point 80–85 °C) which can be retained
in the sheet but does not contribute to sizing. The risk of the formation of
deposits is relatively low. For cost efficient sizing, the curing (orientation of AKD
particles) must be initiated appropriately because chemical reaction alone is not
sufficient. The curing can be improved by drying the paper web at high contact
temperatures and to a very low moisture content e.g. before the size press,
typically to 1–3%, especially on fast-running machines. Another way to speed
up the curing before converting is to carry out AKD sizing in the presence of a
so-called promoter resin, e.g. polyamide epichlorhydrin, modified polyethyleni-
mine, polyvinylamine.
• ASA: This will not dissolve in water and, prior to application in papermaking, it
must be emulsified on site at the paper mill. There, a small amount (3–6%) of
3 Chemical Additives86

an activator is added, plus cationic starch and a synthetic cationic polymer which
serve as stabilizers. Activators are surface-active agents that promote effective
emulsification at low mechanical energy. Emulsion stability is best with syn-
thetic polymer, and sizing efficiency is best with cationic starch, improving with
the amount of starch used. The ratio of starch to ASA is usually in the range 2:1
to 4:1. Emulsification is primarily carried out in a continuously operated special
automated equipment, and can be affected by both low and high shear proce-
dures. Particle sizes in the range 0.5 to 2 mm can be obtained. Emulsification
imparts a cationic charge on the oil droplets which helps to increase the stability
as well as improving ASA retention on anionic fibers and sizing efficiency. Lin-
ear hydrocarbons are more hydrophobic than branched ones. However, the
greater the hydrophobicity of the ASA molecule, the more difficult is the prepa-
ration of a stable quality ASA emulsion.
ASA undergoes the normal reactions of acid anhydrides. Of particular interest
in conjunction with sizing is the reaction with alcoholic hydroxy groups to yield
an ester, and the hydrolysis with water. Both reactions occur in the papermaking
system. ASA is highly reactive, and the reactions proceed rapidly and irreversi-
bly. Although this would provide satisfactory development of sizing on a paper
machine, the hydrolysis of ASA is undesirable. The hydrolysis accelerates with
pH, time, and temperature and leads to deposits and runnability problems on
the paper machine. In order to limit the hydrolysis of ASA emulsions, the pH
can be lowered immediately after the emulsification by addition of aluminum
sulfate.
High solids retention is extremely important in ASA sizing. If this is guaranteed
and ASA is applied correctly, a relatively small dosage of 0.1–0.2 % based on
solid furnish is required to produce the desired degree of sizing. Careful selec-
tion of retention aid is therefore an important part of the ASA sizing process.
The great advantage in favor of ASA is the high rate of cure in comparison with
AKD size. The reaction with cellulose hydroxy groups takes place rapidly in the
dryer section of the paper machine at less than 5% web humidity. More than
90% of the attainable sizing potential is, in most cases, achieved before the size
press unit. The reaction rate is influenced by the same parameters as the hydrol-
ysis, that is primarily by pH, previous residence time, and stock temperature.
The best conditions obviously depend upon the balance between the reaction
rates with cellulose hydroxy groups and hydrolysis of ASA, and optimum sizing
can be achieved at neutral rather than at higher pH, depending on the full set of
system conditions.
• PSA: For internal sizing (wet-end addition) quantities of 0.5 to 1% (solid product
on dry paper stock) are required to obtain a hard sized sheet, depending on the
nature of the stock. The product is best metered continuously to the stock sus-
pension before the pressure screens. The polymer must be prediluted with an
in-line mixer to a concentration of 1–5%. A cationic retention aid, e. g. modified
polyethylenimine, cationic polyacrylamide or polyvinylamine improves the fixa-
tion/retention and therefore gives a better sizing effect.
3.6 Functional Chemicals 87

For surface application, first the compatibility with the other chemicals used in
the preparation and also the shear stability have to be checked (e.g. with starch,
optical brightener, dyes). It is advisable to meter the undiluted polymer by
means of a piston pump to the circuit of the size-press, e.g. before the feed
pump. To achieve a good effect, the PSA must penetrate sufficiently into the
paper, e. g. an adequate quantity of sizing solution must be taken up. Quantities
of 2–4 g l
–1
solid polymer in combination with 40–80 g l
–1
starch are normally
sufficient to achieve a hard sized paper sheet; 1–2 g l
–1
solid polymer is suffi-
cient for final sizing of a paper that has been presized internally.
3.6.4.4 Requirements and Measurements of Sized Papers
The purpose of sizing is to modify the surface of the fibers to control penetration
of aqueous liquids into the paper. The penetration usually correlates with the ab-
sorbency, repellency (hydrophobicity), and spreading of the aqueous liquid con-
cerned. Control of these important properties may be required for three pur-
poses:
1. Control of the penetration rate of the aqueous phase in a converting operation
such as size press treatment, coating, glueing (influences also the machine
runnability).
2. Control of liquid absorption or wetting in a printing process.
3. Control of the serviceability of many grades of paper and board, e.g. milk/juice
carton, packaging papers, wallpaper, printing writing papers, etc.
Besides a certain degree of water and ink resistance the papermakers and their
customers often look for improvements in dimensional stability, surface strength
(pick and rub), internal bond, linting, dusting, stiffness, smoothness, porosity, or
friction coefficient, depending on the converting process and use of the paper.
Several test methods are used to measure the degree of water and ink resis-
tance:
• Water drop absorption: the time required for defined droplets of water to be ab-
sorbed by paper.
• Ink flotation test: the time required for writing ink to penetrate through a floating
piece of paper and to change the color of the upper surface.
• Hercules sizing test: the decrease in reflectance of the opposite side of a paper
sheet which has been covered with a given amount of ink.
• Cobb test: the amount of water absorbed by a given area of paper in a specified
period of time.
• Pen and ink feathering test: the extent of spreading of lines drawn on a paper
sample with a steel pen and ink.
• Contact angle: the tangential angle from the horizontal which the base of a drop
of liquid develops when carefully placed on a paper sheet.
• Edge penetration/edge wicking test: the lineal penetration of liquid in the in-plane
dimension of the paper.
3 Chemical Additives88

3.6.5
Dry-Strength Resins (DSR) [3, 9–11, 22–24]
Some of the specific mechanical properties important for paper are tensile
strength, tearing resistance, folding endurance, bending resistance, burst and sur-
face strength, internal bond and compression resistance. It is generally accepted
that the major factors contributing to dry strength development in paper manu-
facture include Van der Waals forces, hydrogen bonding and ionic bonding. Be-
cause of the special effect of water on paper strength, it is common to distinguish
between “dry” and “wet” strength properties of paper. The two subjects are ob-
viously related, but wet strength will be treated as a separate topic. Paper strength
is affected by many furnish and process variables. On the furnish side, longer
softwood fibers produce stronger papers than shorter hardwood fibers. Fillers re-
duce strength. Alkaline pH conditions in the wet-end produce stronger papers,
especially after aging, than do acid pH conditions. On the process side, both in-
creased refining and wet pressing increase paper strength. The basic factors that
influence paper strength are: individual fiber strength, interfiber bond strength,
the number of interfiber bonds (bonded area) and the distribution of fibers (sheet
formation). While the first factor cannot be influenced by strength additives, the
remaining three factors can be strongly influenced by such products.
Many water soluble, hydrogen bonding polymers will act as dry strength ad-
ditives. In fact, wood fibers contain their own natural dry strength additive in the
form of hemicelluloses. It is well-known that the removal of hemicelluloses from
wood fibers makes it more difficult to develop their bonding characteristics.
• Starch: In general starch derivatives represent the most common and by far the
largest amount of dry strength additives (Fig. 3.1).
• Vegetable Gums: Much less important but also used are water soluble vegetable
gums, such as locust bean gum and guar gum. These highly hydrophilic poly-
mers have chemical structures which are similar to cellulose, enabling them to
participate in extensive hydrogen bonding with fiber surfaces. The natural mate-
rials are nonionic and are not retained by fibers to any extent. Consequently,
successful commercial products all have cationic groups attached to the main
chain, which increases the attraction between gum molecules and fibers and
results in improved polymer retention. Due to the combination of high retention
and effective dry strength enhancement only 0.1–0.35% of the material has to be
added in most instances.
• Polyacrylamide Resins: The fully synthetic DSR are of growing importance. In
Japan they already form the largest proportion of the total consumption of dry
strength resins. Their worldwide consumption is an average of 300 g dry poly-
mer per ton of paper or 3% of the total amount of specialty chemicals (see
Fig. 3.3). In the group of synthetic dry strength resins the polyacrylamide-based
products are still dominant today. Since anionic polyacrylamides are negatively
charged, they are not directly attracted to paper fibers. A cationic substance, such
as alum or polyamide epichlorohydrin resin, must be used to promote their
retention. To avoid the need for a cationic promoter, it is possible to incorporate
3.6 Functional Chemicals 89

cationic groups (e.g. methacryuloyloxethyl trimethyl ammonium methosulfate,
dimethyldiallyl ammonium chloride, vinyl benzyl trimethyl ammonium chlo-
ride, 3-acrylamido-3-methyl butyl trimethyl ammonium chloride) directly into
the polyacrylamide backbone by copolymerization. When used as dry strength
additives, typically 10% of the monomers will be charged and their molecular
weight will be between 100000 and 500 000. This range is low enough that the
polymers will not bridge between particles and cause flocculation, and high
enough to retard migration of the polymer into the fiber pores with concomitant
loss of activity.
• Polyvinylformamide/Polyvinylamine Resins (PVF/PVAm): Recently developed new
types of polymers in the form of polyvinylformamide and polyvinylamine are
coming into use. All these water soluble polymers contain primary amino
groups that can form hydrogen bonds with surface cellulose molecules in fibers
and improve interfiber bonding. These polymers enable the papermaker to
achieve combinations of paper properties that cannot be obtained through refin-
ing alone. For example, strength properties can be improved without affecting
sheet bulk and/or appearance properties.
The new DSRs are produced by polymerizing vinylformamide and then hydro-
lyzing it. This results in a chain type macromolecule with primary amino groups
without using an additional monomer. These polymers can be varied within a
very wide range of molar mass and charge density to optimize the performance.
Medium or low charged polyvinylamines of medium molecular length give high
performing dry strength resins. Further improvements in cost-performance can
be achieved by product combinations, e.g. low/middle charged cationic poly-
vinylamine plus low molecular, middle/high charged anionic polyacrylamide or
cationic polyvinylamide plus anionic polyvinylformamide.
These products have no remaining monomers, and do not contain formalde-
hyde or organically bound chlorine, therefore they do not release chlorine to the
effluent. The environmental advantages of this new group of DSRs is underlined
by the fact that they have been approved by the German health authorities (BfR-
Bundesinstitut für Risikobewertung) for food packaging paper and board. PVF
and PVAm products also conform to the requirements of the United States Food
and Drug Administration (FDA) regulation 21 CFR 176.170 (Components of
paper and board in contact with aqueous and fatty foods), according to its cur-
rent status. It may be added at levels of up to 1.5% solid polymer, expressed as a
proportion of the dry, finished paper.
Polyacrylamide- and polyvinylamine-based DSRs are mainly supplied as ready to
use aqueous solutions or emulsions (10–40%) or as water soluble powders that
must be dissolved prior to use. No other preparatory steps are necessary. The
most beneficial way of application is to meter them continuously to the thick
paper stock at a point of thorough mixing, e.g. at stock dilution. Addition rates
of 0.1 to 0.5% of solid material are adequate for most uses. Excessive use can
overcationize the stock suspension and reduce the effectiveness of the DSR and
of other cationic additives.
3 Chemical Additives90

3.6.6
Wet Strength Resins (WSR) [2, 9–11, 21–24]
Certain types of paper can only fulfil their purpose if they have adequate wet
strength. Such papers include, for example, filter papers, hygienic papers, papers
for bags, label papers, wallpapers, laminate base papers, packaging papers for
moist goods and all papers which, in the course of further processing and use, risk
breaking when rewetted. The required wet strength (up to 50% of the dry paper
strength can be retained) is obtained with the aid of wet-strength resins. For ex-
tremely high wet-strength properties the most common WSR are urea formal-
dehyde resins (UF-resins) and melamine formaldehyde resins (MF-resins), These
chemicals need acid pH conditions and the presence of alum in the papermaking
process. For neutral pH conditions polyamide-epichlorohydrin resins (PAE-resins)
are mainly used (e.g. for hygiene and laminate papers); polyethylenimine products
are used for specialty papers such as industrial filter papers and shoe board.
The total consumption of wet strength resins, together with insolubilizers for
coating (see 3.6.9.3.5.2), accounts for about 0.07% of the worldwide paper produc-
tion or 7% of all specialty chemicals, calculated on the active ingredient (Fig. 3.3).
Over the past years, consumption of PAE-resins has increased overproportionately.
This is partly due to the trend from acid manufacturing conditions to the neutral
pH range, where polyamide-epichlorohydrin resins are more effective than urea
formaldehyde and melamine formaldehyde resins. However, the increasing im-
portance of PAE-resins is no doubt also largely a result of the formaldehyde con-
troversy of the early eighties. PAE-resins account today for about 45%, urea for-
maldehyde resins 15%, melamine formaldehyde resins 10 %, glyoxal resins 15 %
and the remaining 15% are others, e. g. ammonium zirconium carbonate (as in-
solubilizer) and newly developed products e.g. polyvinylamines.
There are two theories regarding the mechanism of wet strength. The first states
that the wet strength effect is due, at least in part, to a reaction between the resin
and the cellulose, which leads to the formation of ether bonds. The second theory
assumes that the wet-strength resins crosslink on exposure to heat in the dryer
section to form a three-dimensional network, wrap themselves around the points
where the fibers intersect and thus protect the points of intersection from water
penetration and swelling. Given the short contact times with the steam-heated
cylinder surfaces (less than a second in the case of the yankee cylinder used in
hygienic paper production), wet-strength agents require a high level of reactivity to
allow crosslinking to take place and bonds to form. At the same time, the wet-
strength resins have to have a selective effect if they are not to react with the
surplus hydroxy groups in the paper stock suspension. Therefore a healthy balance
between reactivity and selectivity has to be found, so that the chemical reactions
(crosslinking, formation of covalent bonds) are not completed at the end of the
paper manufacturing process but continue during storage until maximum wet
strength is reached one to three weeks later. This gradual curing should not be
looked upon as a disadvantage as it is essential for good recycling of the paper
machine broke.
3.6 Functional Chemicals 91
