Kallen A. Understanding Biostatistics
Подождите немного. Документ загружается.


292 HAZARDS AND CENSORED DATA
intuitively ‘intensity’ is more neutral, whereas ‘hazard’ is associated with risks). We take a
deterministic view and assume we have a very large population for which a function F (t)
describes the fraction of individuals who have experienced the event up to time t (the CDF
for the corresponding stochastic variable). The (possibly time-dependent) intensity function
λ(t) for this event is defined by the differential equation (see also Section 2.7)
F
(t) = λ(t)F
c
(t).
In words, the rate at which new events occur in the population is proportional to the number
of individuals who have not yet experienced it, that is, the population still at risk. We assume
that an individual only can experience the event once. If we use the fact that F
c
(0) = 1, high-
school calculus shows that F
c
(t) = e
−(t)
, where (t) =
t
0
λ(s)ds is the cumulative intensity
(or hazard) function. The cumulative intensity (t) relates to the intensity λ(t) in the same
way as the CDF relates to the probability density.
We can write the differential equation above in the notation of Box 4.8 as
dF (t) = F
c
(t−)d(t), (11.1)
noting that the proportion at risk at time t is actually the left-hand limit F
c
(t−). At a jump
point of F (t) the relation, expressed as probabilities, reads
P(T = t) = P(T ≥ t)P (T = t|T ≥ t),
which means that the hazard d(t) = P(T = t|T ≥ t) is the conditional probability that we
have a jump at time t, given that it has not yet occurred. This observation helps us also
to understand the meaning of the hazard d(t) at points where F (t) is continuous, as the
instantaneous conditional probability that the event occurs now, given that it has not yet
occurred. Equation (11.1) therefore means that the jump size at time t is the product of the
number at risk and the hazard at that time.
From equation (11.1) we learn how to obtain F (t) from (t). For smooth functions this
is the differential equation mentioned above; in the general case we integrate the expression
to get an integral equation for F (t). We will discuss this integral equation and its connection
to the Kaplan–Meier estimate of the CDF further in Section 11.8.
More important at this point is that we have the relation d(t) = dF (t)/F
c
(t−). If we
multiply the numerator and denominator on the right-hand side by the sample size, this means
that d(t) = dN(t)/Y(t), where N(t) is the expected number of events that have occurred at
time t and Y(t) the expected number at risk at the same time t. This immediately leads us to
the Nelson–Aalen estimator
n
(t) of the cumulative hazard from the relation
d
n
(t) =
dN
n
(t)
Y
n
(t)
,
where N
n
(t) is the (cumulative) number of observed events at time t and Y
n
(t) the number ob-
served to be at risk for experiencing an event at the same time. Since d(t) is an instantaneous
probability, we can compute it from only those we observe, as long as we adjust N
n
(t) to mean
observed events only. This estimates the true hazard as long as any censoring mechanisms
operating are not related to the event we observe (more on this in the next section).

HAZARD MODELS FROM A POPULATION PERSPECTIVE 293
Box 11.1 The Gompertz distribution
A survival distribution much used to describe life expectancy in insurance contexts is
a distribution which was introduced in 1825 by Benjamin Gompertz in an article with
the title ‘On the Nature of the Function Expressive of the Law of Human Mortality’.
The distribution is motivated by assuming
the average exhaustion of a man’s power to avoid death to be such that at the end
of equal infinitely small intervals of time he lost equal portions of his remaining
power to oppose destruction which he had at the commencement of these intervals.
This assumption leads to the differential equation
d
dt
λ(t)
−1
=−bλ(t)
−1
(where t is age) which is equivalent to the simpler equation λ
(t) = bλ(t), and implies
that λ(t) = λe
bt
. The corresponding distribution is a generalization of the exponential
distribution (which is the case b = 0); when b/= 0 the survival function is given by
F
c
(t) = e
−λ(e
bt
−1)/b
.
There is a connection between this distribution and the logistic distribution in
Box 9.4, via the generalized logistic function defined by
F
(t) = F (t)(1 − F (t)
ν
)/ν,
with ν is a further parameter. If we let ν → 0, this equation becomes F
(t) =
−ln(F (t))F (t), the solution of which is the Gompertz function.
In 1874 W.M. Makeham expanded this to a distribution with one additional param-
eter, deriving what is known as the Gompertz–Makeham law of mortality, which states
that the death rate is the sum of an age-independent component (the Makeham term) and
an age-dependent component (the Gompertz function). This appeared to be particularly
suitable as an approximation to many empirical life tables and therefore soon became
an important tool in actuarial theory.
The parametric distributions F (t) that appear for time-to-event data are typically not the
same as those that appear in the analysis of other types of measurement data. The Gaussian
distribution is usually not relevant here, being too symmetric for most applications, but the
lognormal distribution is a candidate, at least in some situations. However, it has a long right-
hand tail, and for this type of data, in particular mortality data, tails are often to the left instead,
which calls for a different set of distributions.
It is natural to derive the relevant distribution from the behavior of the intensity
function. The simplest case is when there is a constant intensity λ, so that (t) = λt and
F (t) = 1 −e
−λt
. In such a case T has an exponential distribution with parameter λ, denoted
Exp(λ) (the same as (1, 1/λ)). Like the lognormal distribution, it has a long tail to the right. Its
mean value is 1/λ and the graph of the function (t) is a simple straight line through the origin
with slope λ. It has the important and interesting property that it has no memory. This means
294 HAZARDS AND CENSORED DATA
0
1
2
3
4
5
6
7
8
(a) (b)
Hazard
43210
Time
0
0.2
0.4
0.6
0.8
1
Survival Function
43210
Time
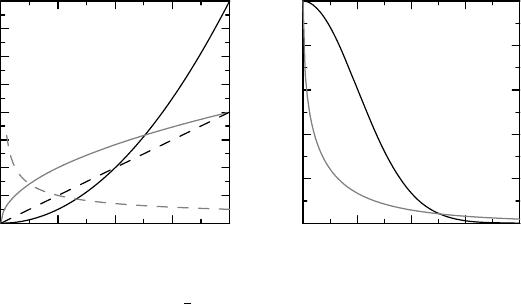
Figure 11.2 Illustration of the Weibull distribution. Dashed curves in (a) are graphs
of the hazard functions t and 1/
√
t, corresponding to Weibull distributions with ex-
ponents γ = 2 > 1 (black) and γ = 0.5 < 1 (gray), respectively. Solid curves in (a)
are the corresponding cumulative hazards, and the corresponding survival functions are
shown in (b).
that if the event has not occurred at time s, the time that remains until it occurs has the same
distribution as it had from the start; the distribution is insensitive to when we start our clock.
It is the only continuous distribution with this property. (In fact, since P(T>t+ s|T>t) =
F
c
(t + s)/F
c
(t), the ‘no-memory property’ is equivalent to the functional equation
F
c
(t + s) = F
c
(t)F
c
(s), for which the only (admissible) solution is F
c
(t) = e
−λt
.) This
property and the constancy of the intensity say the same thing: events occur completely at
random. So, when events occur completely at random, the time-to-event distribution is the
exponential distribution.
There are different directions in which we can generalize the exponential distribution
for time-to-event data. One such direction is the gamma distribution, which occurs when
there is a series of p events that build up the final event, and the times between individual
sub-events are independent with a common exponential distribution. Another general-
ization is the Weibull distribution (Figure 11.2), which we write Wei(λ, γ). Compared
to the exponential distribution it has an additional exponent γ on the time variable,
so that
F
c
(t) = e
−λt
γ
,(t) = λt
γ
.
The corresponding hazard λ(t) = λγt
γ−1
is increasing with time if γ>1 and decreasing with
time if γ<1. The Weibull family of distributions constitutes a flexible family for data when
one wants to understand how ‘non-exponential’ a particular distribution is. Furthermore,
P(ln T>t) = F
c
(e
t
) = e
−λe
γt
,
and this distribution of ln T is called the smallest extreme value distribution. We will see
later that some important models for time-to-event data (AFT models) are actually regression
models for ln T , which explains why this observation is important.
We close this section with a description of the likelihood for a parametric model for time-
to-event data in the presence of right-censoring. We want to express the likelihood in the

HAZARD MODELS FROM A POPULATION PERSPECTIVE 295
Box 11.2 The likelihood for parametric hazard models with right-censored data
In order to compute the likelihood for a parametric model for continuous survival data in
the presence of right-censoring, let F
c
(t, θ) = e
−(t,θ)
where (t, θ) is the cumulative
hazard for λ(t, θ). For complete data the likelihood is given by
L(θ) = dF (t
1
,θ) ...dF(t
n
,θ) = λ(t
1
,θ) ...λ(t
n
,θ)F
c
(t
1
,θ) ...F
c
(t
n
,θ),
where t
i
is the observed event time for subject i. If we also have another m observations
about which we only know that they are larger than τ, for a fixed number τ, then we
multiply this likelihood by F
c
(τ, θ) a total of m times. The resulting likelihood can be
written as
L(θ) =
i
λ(t
i
,θ)
δ
i
F
c
(t
i
,θ),
where δ
i
= 1 if this is an event (t
i
≤ τ) and δ
i
= 0 if the observation is censored and
we have that t
i
= τ for censored observations.
We now introduce a right-censoring process which is independent of the survival
time and with CDF C(t) and intensity function μ(t) not containing θ. What we observe
is the minimum of the time to event and the time to censoring, so what we observe
has a CDF Q(t, θ) which is such that Q
c
(t, θ) = F
c
(t, θ)C
c
(t). The likelihood L(θ)
now becomes
i
λ(t
i
,θ)
δ
i
μ(t
i
)
1−δ
i
Q
c
(t
i
,θ) =
i
λ(t
i
,θ)
δ
i
F
c
(t
i
,θ)
i
μ(t
i
)
1−δ
i
C
c
(t
i
).
The relevant (θ-free) part of the log-likelihood can therefore be written as
ln L(θ) =
i
(ln λ(t
i
,θ)N
i
(t) − (t
i
,θ)),
where we have written δ
i
as N
i
(t) instead. In integral notation this is equation (11.2).
intensity function instead of distribution function, in order to smoothly handle censoring. Let
λ(t, θ) be the intensity function. As is outlined in Box 11.2, we have that
ln L(θ) =
∞
0
ln λ(t, θ) dN
n
(t) −
∞
0
Y
n
(t)λ(t, θ)dt, (11.2)
where N
n
(t) and Y
n
(t) are as before. To find the maximum of this function, we differentiate.
The equation that determines the estimate of θ from data is then given by
∞
0
∂
θ
λ(t, θ)
λ(t, θ)
(dN
n
(t) − Y
n
(t)d(t, θ)) = 0. (11.3)
This equation will, in various forms, prove to be fundamental in the next chapter.
296 HAZARDS AND CENSORED DATA
11.4 The impact of competing risks
There is a special consideration for right-censored data which is not relevant to complete data,
namely an understanding of the censoring mechanism. We have already pointed out that the
Kaplan–Meier estimator estimates the CDF. Its use may be uncontroversial if we are studying
time to death and a data point is censored for an individual only when he has yet to die. If,
however, we want to study mortality in a particular disease, it is a complicating factor that
patients may die from other causes. It may be that understanding time to death in the particular
disease is what is important to the investigator, because that is what a particular treatment
targets, but from the patient’s perspective the situation may be different. He may want to learn
how much longer he is expected to live if he takes the treatment, especially if there is a price
for taking it, such as side-effects. It is also possible that there are some negative effects of a
treatment, which leads to an increased mortality in other diseases, such as heart attacks. The
cause of death may be less important to the patient.
Suppose we are studying a particular event, such as death in a specific disease, in the
presence of competing events which, when they occur in an individual, do not allow us to
study the event we are interested in. What we can always estimate is the function G(t), which
is the probability that an event has occurred by the time t (it has occurred in the interval (0,t]).
It is simply the proportion of individuals for whom this event has occurred, and is called the
cumulative incidence function (CIF) for the event. It is not a distribution function, since in
the presence of competing risks we expect to have that G(∞) < 1. But it has all the other
properties of a CDF, and is therefore called a sub-distribution function. If we denote by T the
time we can observe, which is when the first of the competing events occurs, the following
relation holds:
dG(t) = P(T
1
= t|T ≥ t)P(T ≥ t),
where T
1
denotes the time to the event we are interested in. If F (t) is the CDF for T , this
relation can be written as
dG(t) = F
c
(t−)d
∗
(t)
where d
∗
(t) is the event intensity among those that have not experienced any event yet. This
is not the same as the hazard d(t) defined earlier, which is associated only with the event.
The hazard d
∗
(t), on the other hand, is very much defined by what competing risks operate,
because it measures intensity on patients who so far have survived these. When we eliminate
one particular risk, it is not necessary that the hazards for the remaining ones are unchanged.
If we take away a risk factor for a particular cause of death, we may well affect other causes
as well: tobacco smoking not only induces lung cancer but also has effects on cardiovascular
death risks, as well as on other causes of death. If we give a drug that reduces blood clotting
we may reduce the risk of cerebral infarction, but increase the risk of cerebral hemorrhage,
leaving the overall risk of dying from a stroke unchanged.
We can estimate d
∗
(t) by the Nelson–Aalen ratio of number of observed events divided
by number at risk for this. The question then is when this estimates d(t), so that we can
deduce the CDF for the time variable we are interested in. This question assumes that there
is a well-defined stochastic variable T
1
, free of the environment, that we can discuss. This is
not necessarily the case.
THE IMPACT OF COMPETING RISKS 297
Example 11.2 Suppose we are studying a chronic but fluctuating disease for which there
naturally occur periods of symptom worsening, so-called exacerbations. Our treatment is
expected to reduce the intensity of the occurrence of such exacerbations, and we have a
placebo control. As part of the protocol the patient is allowed to discontinue participation in
the study whenever he wishes, and in doing so he should state his reasons for discontinuation.
One such reason may be a deterioration of the disease under study. Even though such a
withdrawal is not an exacerbation per se, not fulfilling the precise medical criteria for this, it
may well be closely related to it. It may, for example, precede it. If we study the time to the first
exacerbation and censor these withdrawals, we probably will not do a meaningful analysis. It
may be better to redefine the event under study as either exacerbation or discontinuation due
to deterioration of the disease.
If the variable T
1
exists, we have that d(t) = P(T
1
= t|T
1
≥ t). We can only measure
d
∗
(t), which works conditionally on T ≥ t, so if we want to substitute d
∗
(t) for d(t), we
need the cause-specific hazard to stay the same if we remove subjects experiencing competing
events. Essentially that means that our event and the competing events must act independently
of each other.
This, like the ITT analysis we discussed in Section 3.7, is something that is often dif-
ficult for medical researches to accept: ‘why can’t I analyze non-fatal and fatal myocardial
infarctions separately?’ The answer is that even though it may make medical sense to do this,
there is simply not enough data to do a valid statistical analysis. Instead we need to make
a fundamental assumption, that competing risks act independently of the event of interest,
so it is the shortcomings of statistics (or, rather, the amount of information) that hinder the
fulfillment of this wish.
If we can somehow assume independence, we can estimate (t) from data, considering
those individuals with a competing event to be censored, and thereby obtain the Kaplan–Meier
estimate of the CDF for T
1
. It may also be that we want to eliminate only some censoring
reasons. Suppose we are studying the events ‘death from myocardial infarction’ and ‘death
from other causes’, but also that patients in the study may be lost to follow-up. In order to be
able to estimate the CIF for death from myocardial infarction in the presence of other causes
of death, we want to eliminate the problem posed by those who were lost to follow-up and to
study termination. This is done by use of the formula
G(t) =
t
0
F
c
(s−)d(s),
where d(t) is cause-specific and is estimated from the Nelson–Aalen estimator, and F
c
(t)
is estimated by the Kaplan–Meier estimate for total survival. In this way we get an estimate
of the CIF, eliminating the study-specific censoring mechanisms.
One lesson from this discussion is that the Kaplan–Meier estimate relates to an abstract
time-to-event variable, free of any competing events. To describe the results of an analysis
in terms of the Kaplan–Meier estimate may therefore be somewhat artificial in situations
where there are competing risks that cannot be eliminated. In such a situation it may be more
relevant to compute something like the function G
1
(t)/G
c
2
(t). This represents the conditional
probability for the event prior to time t, provided that the none of the competing risks have
yet occurred. This is how the Kaplan–Meier estimate is sometimes (erroneously) interpreted.
The Kaplan–Meier estimate is an estimate of the CDF for the event in the absence of other,
competing, events. The suggested function provides the probability of obtaining an event of

298 HAZARDS AND CENSORED DATA
Box 11.3 Bernoulli’s model as differential equations
Bernoulli’s analysis did not discuss vaccination against smallpox, since Edward Jen-
ner’s work on inoculation with cowpox was still 30 years in the future. Instead it was
concerned with variolation, in which infectious material is inoculated into the skin of
the susceptible in order to induce a mild infection. This was not an altogether harmless
process; children could die from it, and it could trigger small epidemics. At the time
physicians argued about whether the benefits of inoculation outweighed the risks, and
the objective of Bernoulli’s paper was to provide some numbers to facilitate decision
making. As such it is one of the earliest attempts at evidence-based medicine.
In his analysis Bernoulli used the then recently developed methods of calculus.
Introducing the two unknowns x(a), the number of susceptibles at age a, and n(a), the
total number surviving to age a, he wrote down the two differential equations
x
(a) =−(λ + μ(a))x(a),n
(a) =−pλx(a) − μ(a)n(a),
subject to the initial conditions x(0) = n(0) = n. Here λ is the force of infection, μ(a) the
age-dependent death rate, and p is the probability of dying from smallpox. From this he
derived the following differential for the prevalence f (a) = x(a)/n(a) of susceptibles:
f
(a) = λf (a)(pf (a) − 1),f(0) = 1,
which we solve to get f (a) = 1/(p + (1 − p)e
λa
). Bernoulli could then estimate the
number of deaths due to smallpox, and deduce that in an environment free of smallpox
the fraction surviving to age a should be given by e
λa
/(p + (1 −p)e
λa
). He described
the result of his investigation by adding new columns to Halley’s tables, containing
‘would-be’ data.
the type under study, given that a competing event has not occurred. It will depend on the
environment in which it operates.
The competing risk discussion is important when we try to assess what the overall effect
would be if we took action to modify one of the competing risks. What does the elimination
of one particular cause of death do to overall mortality? This may well be the relevant health
economic question for a new drug treatment, whereas the relevant biological question may
address what happens in an environment free of competing risks. The former type of question
was actually where the mathematical theory of competing risks started, and its history preceded
mainstream statistics when, in the mid eighteenth century, Daniel Bernoulli of the (among
mathematicians) famous Bernoulli family, wanted to understand what influence smallpox
had on overall mortality. The specific question he addressed was the following. Published
life tables reflect the mortality of the population for which they were calculated, taking into
account all causes of death, including smallpox. How would these life tables change if, because
of mandatory vaccination, deaths from smallpox were entirely eliminated?
We will address this question in a way similar to Bernoulli’s approach, but investigating a
probability model, where Bernoulli used differential equations. An outline of his approach is
given in Box 11.3. His calculations were based on a life table compiled in 1693 by the English
astronomer Edmond Halley (of Halley’s comet) from the records of the then German city of
Breslau (now Wrocław in Poland) on the age at death of each individual in the town.

THE IMPACT OF COMPETING RISKS 299
For a person who is alive and still susceptible to smallpox (i.e., has not yet been infected),
at age a, there are two competing risks:
1. He may contract smallpox, from which he dies with probability p(a). Let S(a) denote
the distribution for the age at which the subject contracts smallpox in an ‘environment
free of other causes of death’.
2. He may die for reasons unrelated to smallpox. Let H(a) denote the lifetime CDF in a
‘smallpox-free environment’.
Bernoulli’s objective was to estimate the survival function H
c
(a). His argument was essentially
as follows. Someone who is alive at age a either is alive and susceptible to smallpox, which
occurs with probability H
c
(a)S
c
(a), or has had smallpox, survived it and not yet died from
other causes. The probability of having contracted smallpox and survived to age a is given by
a
0
(1 − p(α))dS(α), from which we deduce that the overall probability of survival to age a is
given by
l(a) = H
c
(a)
S
c
(a) +
a
0
(1 − p(α))dS(α)
.
Note the independence assumptions here. The left-hand side is (approximately) provided by
Halley’s table, so we obtain the expression
H
c
(a) =
l(a)
S
c
(a) +
a
0
(1 − p(α))dS(α)
.
If we assume, almost certainly incorrectly, that the probability of dying from smallpox is age-
independent, p(a) = p, we can write this as H
c
(a) = l(a)/((1 − p) + pS
c
(a)). Bernoulli also
assumed that there is an age-independent risk λ of contracting smallpox, so that S
c
(a) = e
−λa
,
and therefore arrived at the expression
H
c
(a) =
l(a)
1 − p + pe
−λa
.
To obtain parameter values he assumed that during one year smallpox attacks one in eight
individuals, so that λ = 1/8, and that it causes death in one in eight who are infected,
so that p = 1/8 as well. Under these assumptions the eradication of smallpox would be
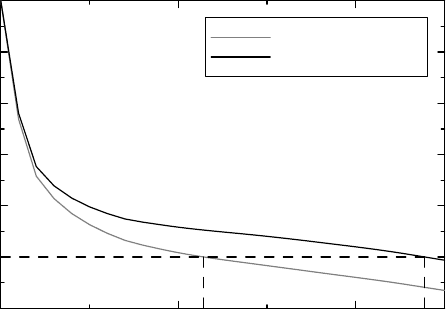
expected to increase the median age at death by 12.4 years from about 11.4 to 23.9 years,
which is illustrated in Figure 11.3. Under the model we can also compute the mean life
expectancy from Halley’s table, which predicts an increase of 3.1 years, from about 26.6
to 29.6 years.
As already noted, there are some strong assumptions in these calculations, an important
one being the independence between death from smallpox and death from other causes. But
this is a modeling exercise, and when we finally inspect the result and compare with our
predictions, we must bear in mind that this assumption may explain some of the discrepancy.
Likewise, when we discuss the modeling predictions we must assess how sensitive they are
to the assumptions, not only the independence assumption but also other assumptions made.
This makes modeling an endeavor with special problems, especially if you cannot test the
model before you need to make your decision. Climate modeling today is a case in point.
300 HAZARDS AND CENSORED DATA
0.4
0.5
0.6
0.7
0.8
0.9
1
20100
A
g
e (
y
ears)
Halley’s data
Bernouilli’s prediction
Figure 11.3 Life table data based on Halley’s data with and without smallpox. The gray
curve shows Halley’s tabulated data, the black curve Bernoulli’s prediction of how it would
change if smallpox were eradicated.
11.5 Heterogeneity in survival analysis
It is tempting to interpret the hazard, or the survival function, computed on population data
as individual risks. This is often not appropriate, basically because biological systems do
not consist of cloned, identical individuals living in identical environments; instead there is
biological variation. In fact, one of the purposes of a statistical analysis is to explain part of this
variation in terms of covariates. We have already seen that omitted covariates have different
effects in different statistical models. For some statistical models unobservable variation is
simply random noise with no effect on the size of the signal. This is true for methods such as
the t-test for means and ordinary linear regression. It is not true for time-to-event data because,
in a heterogeneous world, high-risk subjects will experience the event early and with time
we will gradually study more and more low-risk subjects. The hazard we see will therefore
decline with time because of a selection procedure, which may give us a false impression that
the individual hazards disappear with time. Although this may be true on a population level,
it may not apply on the individual level. Since the life expectancy is dependent on how frail,
or fragile, someone is, heterogeneity in this context is referred to as frailty.
To discuss this frailty problem we make a simple basic assumption about the nature of
the heterogeneity, which may or may not be realistic. At least it will help us understand
the problem and its consequences. The model in question, referred to as the frailty model,
assumes that individuals have proportional hazards, so that for each individual there is a
value θ, such that the hazard for that individual is given by θd(t), where d(t) is some
reference hazard, common to all individuals. The θ tries to capture the individual frailty,
which includes factors such as disease severity, genetic makeup or aspects of the (constant)
environment. We refer to the θs as frailties and assume that their distribution in the population
is described by a CDF P(θ). In order to be able to uniquely specify the reference hazard and
the frailty distribution, we assume that the latter has a known mean value. Let F (t) denote
the CDF corresponding to (t), and let F (t, θ) denote the CDF with cumulative hazard

HETEROGENEITY IN SURVIVAL ANALYSIS 301
Box 11.4 What can we deduce about frailty based on the outcome?
This box contains some mathematical details about the discussion on frailty the text.
If we randomly sample an individual from the population, the bivariate distribution
of the time-to-event variable T and the frailty θ has a density given by dF (t, θ)dP(θ).
The proportional hazards model means that
dF (t, θ)dP(θ) = [θF
c
(t−,θ)dP(θ)]d(t),
a relation that contains a lot of information. First, we can integrate out θ to obtain the
marginal density for T :
d(t) = S
0
(t)d(t), where S
0
(t) =
F
c
(t−,θ)θdP(θ).
Here F
c
(t−,θ)dP(θ) is the probability (density) that we get an individual with frailty θ
and for whom T ≥ t. It follows that F
c
(t−,θ)dP(θ)/
c
(t−) is the conditional density
for θ, given that T ≥ t, and therefore that S
0
(t)/
c
(t−) is the expected value of θ,given
that T ≥ t. The population hazard is therefore given by
d
(t) =
d(t)
c
(t−)
= E(θ|T ≥ t)d(t).
If we instead divide the bivariate density dF (t, θ)dP(θ) by the density d(t), we get
the conditional probability that the frailty is θ, given that we know that the outcome is
T = t. An alternative expression for this is obtained from above as
dF (t, θ)
d(t)
dP(θ) =
F
c
(t−,θ)θd(t)
S
0
(t)d(t)
dP(θ) =
F
c
(t−,θ)θdP(θ)
S
0
(t)
.
This is the density of the conditional frailty CDFs P(θ|T = t) shown in Figure 11.5.
function θ(t). Before proceeding, we note that the famous Cox model, which is so often
used in the analysis of survival data, has precisely this set-up, except that instead of a frailty
distribution it tries to explain the different frailties in terms of measured covariates. More
precisely, it assumes that there is a relation of the form θ = e
zβ
, where z are the covariates
and β are regression coefficients that are to be estimated. This model is discussed in some
detail in Section 12.6 and our derivation of it will build on the observations we make in the
current section.
Now back to the basic setup. As seen in Box 11.4, we can derive some useful information
about the frailty of an individual based on knowledge about when his event occurred. The
marginal CDF for the time-to-event variable T is obtained by integrating θ out:
(t) =
F (t, θ)dP(θ).
This is the distribution we see in the population, and with some mathematical trickery, outlined
in Box 11.4, we can show that the corresponding population hazard is given by the expression
d
(t) = E(θ|T ≥ t)d(t).
