Klipp E., Herwig R., Kowald A., Wierling C., Lehrach H. Systems Biology in Practice: Concepts, Implementation and Application
Подождите немного. Документ загружается.


1. There is a system s of spins s
k
with k =1,…,N, where N is large (N >> 1). Each
spin has the value of either 1 or –1.
2. Spins can exchange their values by random interactions, which lead to spin rever-
sals.
3. The energy E (s) of the spin system is
E s
P
k<l
J
kl
s
k
s
l
: (10-24)
The values J
kl
are elements of the exchange interaction matrix with normally dis-
tributed random values and a probability density P
D
of
P
D
J
kl
2 p
1
2
N 1
1
2
exp J
2
kl
N 1
2
: (10-25)
According to Eqs. (10-24) and (10-25), the mean spin-glass energy is zero, [E] =0,
and for one-spin reversal the mean square root of energy variation is equal to 2.
The interesting feature of the spin-glass concept is the large number of local en-
ergy minima M, where a local energy minimum is defined as a state s
L
at which any
one-spin reversal would increase the energy E. Furthermore, there is a global energy
minimum E
0
with E
0
&0.8 N.
A spin-glass model of evolution represents a model sequence as a vector S
i
of
spins and a population as a set S ={S
1
,…,S
n
)ofn sequences. Each sequence has a
selective value that depends on its energy:
fS
i
exp bES
i
(10-26)
for a choice of J
kl
. b is a parameter for the selection intensity.
Spins s
ik
and s
il
interact according to the interaction matrix J
kl
. The selective value of
a sequence becomes maximal and its energy minimal when the combinations of spins
in the sequences provide maximally cooperative interactions for a given matrix J
kl
.
To this end we again consider an evolutionary process with subsequent genera-
tions (compare Section 10.2.1.4). The initial population is generated by chance. New
generations are obtained by selection and mutation. Selection occurs with respect
to the selective value defined in Eq. (10-26). Mutation is a sign reversal of a spin
s
ik
? –s
ik
with certain probability P.
The evolutionary process can be followed by considering for successive genera-
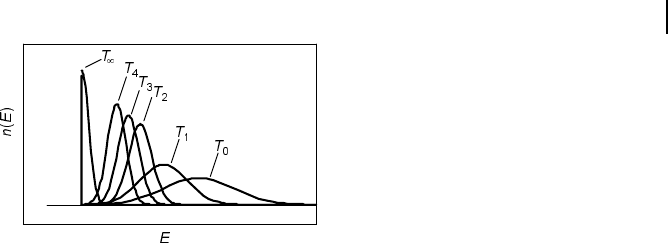
tions the number of sequences possessing certain energy n(E), as illustrated in
Fig. 10.5.
In the spin-glass-type of evolution, the system converges to one of the local energy
minima E
L
, which can be different for different runs of simulation. One may compare
this evolutionary method with the sequential method of energy minimization, i.e.,
consequent changes of symbols (s
ik
? –s
ik
) of one sequence and fixation of only suc-
cessful reversals. The sequential search is computationally simpler than the evolution
search. Nevertheless, the evolution search results on average in a deeper local energy
minimum E
L
, because different valleys in the energy landscape that are never reached
350
10 Evolution and Self-organization
in the sequential method are simultaneously examined in the evolution process.
Thus, in the spin-glass case, the evolutionary search has a certain advantage with re-
spect to the sequential search: it results on average in the greater selective value.
10.2.2
Neutral Theory of Molecular Evolution
The neutral theory of molecular evolution introduced by Kimura (1983) states that
mutations are mostly neutral or only slightly disadvantageous. The historical back-
ground for this statement was the deciphering of the genetic code and the structure
of DNA by Watson and Crick (1953a–c) and the understanding of the principle of
protein synthesis. In addition, the evolutionary rate of amino acid substitutions and
protein polymorphism was estimated. The assumption of neutrality of mutations
agrees both with the mutational molecular substitution rate observed experimentally
and with the fact that the rate of the substitutions for the less biologically important
part of macromolecules is greater than for the active centers of macromolecules.
The mathematical models of the neutral theory are essentially stochastic, i.e., a re-
latively small population size plays an important role in the fixation of the neutral
mutations. The features of neutral selection can easily be explained using the game
of neutral evolution.
Consider populations (of sequences or organisms or, in the following example, of
balls) with a finite population size n. The rules describing the evolutionary process
are as follows. (1) The population contains black and white balls with a total popula-
tion size n. (2) The next generation is created in two steps. First, all balls are dupli-
cated preserving their color. A black ball has a black offspring, a white ball a white
one. Secondly, half of the population is removed irrespective of the “age” of a ball
(i.e., whether it is an offspring or a parent ball) and with equal probability for black
and white balls.
The state of the population is given by the number l of black balls. Consequently,
there are n–lwhite balls. The evolution is characterized by the probability P
lm
for
the transition from a state with l black balls to a state with m black balls in the next
generation. P
lm
can be calculated by applying combinatorial considerations:
351
10.2 Other Mathematical Models of Evolution
Fig. 10.5 Schematic representation of
sequence distributions n(E) for subsequent
generations T
0
< T
1
<…<T
4
.ForT
?
the system
is trapped into a local energy minimum E
L
. The
global energy minimum is E
0
.

P
lm
2l
m
2n2l
nm
2n
n
; if 2l n m 2l
0; if 2l < m or m < 2ln
8
>
>
>
>
>
<
>
>
>
>
>
:
(10-27)
with
a
b
a!
a b!b!
.
Possible evolutionary processes for a population of size 10 with initially five black
and five white balls are illustrated in Fig. 10.6.
The matrix P
lm
determines the random Markovian process, which can be consid-
ered as an example of a simple stochastic genetic process. Concerning the behavior
of this process, the following can be stated. (1) The process always converges to one
of two states, l = 0 (only white balls) or l = n (only black balls). (2) For a large popula-
tion size n, the characteristic number of generations needed to converge to either of
these states is equal to 2 n. Thus, although this evolution is purely neutral (black and
white balls have equal chances of survival), only one species is selected.
It can be questioned how progressive evolution is possible if molecular substitu-
tions are neutral. To answer this question, M. Kimura uses the concept of gene dupli-
cation developed by Ohno (1970). According to Kimura, gene duplications create un-
necessary, surplus DNA sequences, which in turn drift further because of random
mutations, providing the raw material for creation of new, biologically useful genes.
The evolutionary concepts of the neutral theory came from interpretations of bio-
logical experiments; this theory was strongly empirically inspired. The other type of
theory, a more abstract one, was proposed by Stuart A. Kauffman: NK automata or
Boolean networks.
10.2.3
Boolean Network Models
Boolean models are based on proposition logic founded by George Boole (1815–
1864). This type of logic entails the principle of bivalence: any statement is either
true or false. A third possibility or contradictions are excluded. Statements can be
352
10 Evolution and Self-organization
Fig. 10.6 Representative runs for the game of neutral evolution.
Starting with five black balls and five white balls at generation T
0
(bot-
tom line), the system converges within several generations to a state
with either only black balls (left panel) or only white balls (second
and forth panel from left). For the third and fifth panels, the final state
is not yet decided at generation T
10
.

combined using the operators “and,” “or,” or “not” and combinations thereof. The
truth-value of combined statements depends only on the truth-value of the indivi-
dual statements and their connection.
Boolean logic is applied to biological processes such as regulation of gene expres-
sion in the framework of Kauffman’s NK Boolean networks (Kauffman and Weinber-
ger 1989; Kauffman and Johnsen 1991; Kauffman 1993; Kauffman and Macready
1995). Genes are the elements of the network. Levels of gene expression are approxi-
mated by only two states: each gene is either expressed (is assigned the value “1”) or
not expressed (“0”). The network has N elements or nodes. Each element has K in-
puts (regulatory interactions) and one output. Input and output have binary values
(1 or 0). The values are updated in discrete time steps.
Boolean networks always have a finite (although possibly large) number of possi-
ble states and hence only a finite number of possible state changes. The state
changes of an individual element are specified by rules that relate the output to the
inputs. There are 2
2
K
possible rules for a node with K inputs. The labeling of the
rules corresponds to numbers representing the respective binary numbers of output
or to their meaning in normal life (and, or) (see Tabs. 10.1 and 10.2).
Tab. 10.1 Boolean rules for K = 1. The rules represent a functional dependence of the output of an
element from the input. The second row indicates verbal notions for the rules, and the last row pre-
sents the numbering of the rules according to the decimal value of the binary number correspond-
ing to the output values.
Input Output
A0 AnotA1
00011
10101
Rule 0123
Tab. 10.2 Boolean rules for K = 2. The rules represent a functional dependence of the output of an
element from the input (e.g., “xor” = “exclusive or,” “nor” = “not or,” “nand” = “not and”). The
second row indicates verbal notions for the rules, and the last row presents the numbering of the
rules according to the decimal value of the binary number corresponding to the output values.
Input Output
A B 0 and A B xor or nor not B not A nand 1
000000000011111 111
010000111100001 111
100011001100110 011
110101010101010 101
Rule 0123456789 101112 131415
353
10.2 Other Mathematical Models of Evolution

Example 10-5
The network
(10-28)
has the connectivity K = 1. Let A = const., B = f (A) = notA, C = f (B) = not B, and
D = f (C)=C,with the initial state (A,B,C,D)(t
0
) = (1,0,0,0). The following states are
(A,B,C,D)(t
1
) = (1,0,0,1)
(A,B,C,D)(t
2
) = (1,0,1,1)
…
(A,B,C,D)(t
i
) = (1,0,1,1) for i =2,…,?.
After two steps the system has attained a fix point.
Example 10-6
(10-29)
Another typical structure of a Boolean network with K = 1 is the closed loop,
where the input of the first element is the output of the last element.
Periodic behavior is possible if all elements obey rules 1 or 2. Assume, for exam-
ple, that all elements follow rule 1 and the initial state is (ABCDE) = (10000). The
states for the following time steps will be (01000), (00100), (00010), (00001), and
again (10000), which closes the cycle. Rules 0 or 3 break the periodic behavior,
since the output of the respective element is no longer dependent on the input.
Example 10-7
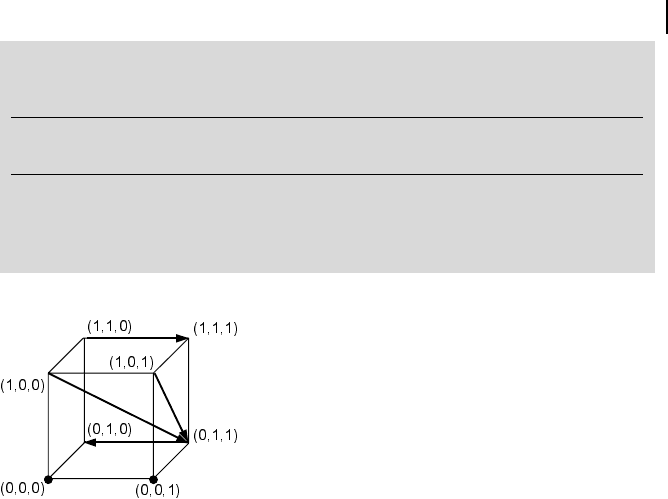
The network
(10-30)
has N = 3 elements and may assume 2
N
= 8 different states. Let the rules be as fol-
lows:
At 1Atand Bt
Bt 1Ator Bt
Ct 1Ator not Btand Ct
354
10 Evolution and Self-organization
The following table lists the possible actual states and the respective following
states for (ABC).
It can be seen that the states (000), (001), (010), and (111) are fix points, since
they will not be left. The states (000), (001), (100), (101), and (110) cannot be
reached from other states (see also Fig. 10.7).
An NK automaton is an autonomous random network of N elements with K in-
puts and one output per element. The Boolean elements of the network and the con-
nections between elements are chosen randomly. There are no external inputs to the
network. The number of elements N is assumed to be large. Since N and K are finite,
the number of possible states is also finite – although possibly too large to be in-
spected in reasonable time.
In reverse engineering of gene expression networks, it is of interest how many
gene expression states have to be measured independently to find out the network
connections. It has been estimated that this number is about 2
N
for fully connected
Boolean networks, about K72
K
log(N) for networks with connectivity K, and about
log(N) in the case of pairwise correlation of gene expression values.
10.3
Prediction of Biological Systems from Optimality Principles
Biological systems developed through evolution by mutation and selection. Evolution
can be considered as an optimization process that took place over millions of years.
It seems that present-day systems show properties that are optimal with respect to
certain selective conditions. Hence, system properties may be predicted from mathe-
matical models based on optimality criteria.
Evolution is considered to be without aim or direction, but it forces development
of species towards maximizing fitness. The formulation of a function that measures
fitness is not straightforward. Several optimality criteria have been proposed. For cel-
355
10.3 Prediction of Biological Systems from Optimality Principles
Fig. 10.7 The eight possible states for a network of N =3
elements are represented as corners of a cube. The possible
transitions are symbolized by arrows.
Actual state 000 001 010 011 100 101 110 111
Next state 000 001 010 010 011 011 111 111

lular reaction systems, they involve, e.g., (1) maximization of steady-state fluxes,
(2) minimization of the concentrations of metabolic intermediates, (3) minimization
of transition times, (4) maximization of sensitivity to an external signal, and (5) opti-
mization of thermodynamic efficiencies (Heinrich and Schuster 1998).
Evolution and optimization of cellular systems are subject to physical constraints.
Such constraints include, for example, differences in free energy for participants of a
reaction expressed by the equilibrium constant, diffusion limitations in the move-
ment of compounds through the cell, structural requirements in the composition of
macromolecules, or the stoichiometry of metabolic systems.
In order to take into account biological constraints, the concept of a cost function
has been introduced (Reich 1983). The following have been suggested as cost func-
tions: the total amount of enzyme in a cell or the pathway under consideration
(Reich 1983), the total energy utilization (Stucki 1980), or the evolutionary effort
(Heinrich and Holzhutter 1985) counting the number of mutations or events neces-
sary to attain a certain state.
In the following three sections we will study how metabolic networks should be
designed if they were designed according to optimality principles. We investigate the
consequences of the demand for rapid conversion of substrate into product on the
catalytic properties of single enzymes and on the appropriate amount of enzymes in
a metabolic pathway. In the first two sections, we determine conditions that yield
maximal steady-state fluxes. In the third section, an example for temporal regulation
of pathway properties is studied.
10.3.1
Optimization of Catalytic Properties of Single Enzymes
An important function of enzymes is to increase the rate of a reaction. Therefore,
evolutionary pressure should lead towards a maximization of the reaction rate
v ? max (Pettersson 1989, 1992; Heinrich and Hoffmann 1991; Wilhelm et al.
1994). High reaction rates may be achieved only if the kinetic properties of the en-
zymes are suitably adapted. We identify the optimal kinetic parameters that maxi-
mize rates for the reversible conversion of substrate S into product P (Klipp and
Heinrich 1994).
There are two constraints to be considered. First, the action of an enzyme cannot
alter the thermodynamic equilibrium constant for the conversion of S to P (Eq. (5-13)).
Changes of kinetic properties must obey the thermodynamic constraint. Second, the
values of the kinetic parameters are limited by physical constraints even for the best
enzymes. Their maximal possible values are denoted by k
max
, and we consider all
rate constants to be normalized by their respective k
max
, such that the maximal
values of the normalized kinetic constants are one.
For a reaction that can be described with linear kinetics
(10-31)
with the thermodynamic equilibrium constant
356
10 Evolution and Self-organization

q k
1
=k
1
; (10-32)
the rate equation reads
v E
total
S k
1
P k
1
E
total
k
1
S q P
E
total
k
1
S
P
q
: (10-33)
It is easy to see that v becomes maximal for fixed values of E, S, P, and q,ifk
1
and k
–1
become maximal. Note that usually only one of the two rate constants may
attain its maximal value. The value of the other, submaximal constant is given by
Eq. (10-32).
For the reversible reaction obeying the Michaelis-Menten kinetics (Eq. (5-34)) with
the reaction rate given in Eq. (5-36), the optimal result depends on the value of P.
For P ^ 1/q the rate becomes maximal if k
1
, k
2
, and k
–2
assume maximal values and
k
–1
is submaximal (R
1
in Fig. 10.8). For P 6 q we obtain submaximal values of only
k
–2
(R
2
). For 1/q < P<qthe optimal solution is characterized by submaximal values
of k
–1
and k
–2
, with k
–1
=
P=q
p
and k
–2
=
1=Pq
p
(R
3
).
Comparison of the optimal state with a reference state can assess the effect of the
optimization. One choice for a reference state is k
1
= k
2
= 1 and k
–1
= k
–2
=1/
q
p
,
i.e., equal distribution of the free energy difference represented by the equilibrium
constant on the first and the second step. The respective reference rate reads
v
ref
Sq P
S 1q P 1
q
p
and the optimal rate in regions R
1
,R
2
, and R
3
read
v
opt;R
1
Sq P
S1q1Pq
, v
opt;R
2
Sq P
S1q q P
, and v
opt;R
3
Sq P
S1q 2
Pq
p
.
For example, in the case P = q and q = 100, the maximal rate for optimal kinetic con-
stants is v
max
S 1
S 3
and the reference rate calculates as v
ref
S 1
S 11:1
, which is
lower than the maximal rate.
357
10.3 Prediction of Biological Systems from Optimality Principles
Fig. 10.8 Subdivision of the plane of substrate and product concen-
trations (S, P) into regions of different solutions for the optimal mi-
croscopic rate constants (schematic representation). The dashed
lines indicate the function Sq = P. (a) Solution regions for the two-
step mechanism. (b) Solution regions for the three-step mechanism.

For the reversible three-step mechanism involving the binding of the substrate to
the enzyme, the isomerization of the ES complex to an EP complex, and the release
of product from the enzyme
(10-34)
the reaction rate is given as
v E
total
S k
1
k
2
k
3
P k
1
k
2
k
3
k
2
k
3
k
1
k
3
k
1
k
2
S k
1
k
2
k
3
k
2
P k
3
k
2
k
1
k
2
(10-35)
It turns out that the optimal solution for this mechanism depends on the values
of both S and P; there are 10 different solutions (see Tab. 10.3 and Fig. 10.8).
There are three solutions with a submaximal value of one backward rate constant,
three solutions with submaximal values of two backward rate constants, three solu-
tions with submaximal values of one backward and one forward rate constant, and
one solution with submaximal values of all three backward rate constants. The con-
358
10 Evolution and Self-organization
Tab. 10.3 Optimal solutions for the rate constants of the three-step enzymatic reaction as func-
tions of the concentrations of substrate and product for q 6 1.
Solution k
1
k
–1
k
2
k
–2
k
3
k
–3
R
1
11/q 1111
R
2
1111/q 11
R
3
111111/q
R
4
1
P=q
p
111
1=Pq
p
R
5
1
S P
q 1 P
s
1
1 P
qS P
s
11
R
6
111
2P
q 1 S
s
1
1 S
2 Pq
s
R
7
2 q 1 P
S
r
11
2 1 P
qS
s
11
R
8
11
2 q 1 S
P
r
11
2 1 S
qP
s
R
9
1
2 S P
q
s
11
2 q S P
p
1
R
10
1*1
P
qk
2
1
1
1
qk
1
k
2
* k
–1
is the solution to the equation k
4
1
k
3
1
k
1
P
q
SP
q
0

straint imposed by the thermodynamic equilibrium constant leads to the following
effects. At very low substrate and product concentrations, maximal rate is achieved
by improving the binding of S and P to the enzyme (the so-called high (S,P)-affinity
solution). If S or P is present in very high concentrations, they are weakly bound
(low S-orP-affinity solutions). For intermediate values of S and P, only backward
constants assume submaximal values. For concentrations of S and P equal to unity,
the optimal solution reads
k
1
k
2
k
3
q
1=3
and k
1
k
2
k
3
1 : (10-36)
This case represents an equal distribution of the drop in free energy on all three
elementary steps.
10.3.2
Optimal Distribution of Enzyme Concentrations in a Metabolic Pathway
By means of regulated gene expression and protein degradation, cells can adjust the
amount of enzyme allocated to the reactions of a metabolic pathway according to the
current metabolic supply and demand. In many cases the individual amounts of en-
zymes should be regulated such that the metabolic fluxes necessary for the mainte-
nance of cell functions can be achieved while the total enzyme amount is low. One
reason for this is that enzymes are osmotically active substances. One strategy to
achieve osmotic balance is, therefore, to keep the total amount of enzyme con-
strained. Furthermore, enzyme synthesis is expensive for the cell, with respect to
both energy and material. It is therefore reasonable to assume that various pathways
or even individual reactions compete for the available resources.
In theoretical terms we can study how a maximal steady-state flux through a path-
way is achieved with a given fixed total amount of enzyme (Klipp and Heinrich
1999). The optimization problem is to distribute the total protein concentration
E
total
P
r
i1
E
i
optimally among the r reactions. We will exemplify this for the simple
unbranched pathway presented in Eq. (5-180). To assess the effect of optimization
we will compare the optimal state to a reference state where the given total concen-
tration of enzymes is distributed uniformly such that E
i
E
total
=r.
The optimal enzyme concentrations E
opt
i
in states of maximal steady-state flux J
can be determined by the variational equation
i
iE
i
J l
X
r
i1
E
i
E
total
! !
0 i 1; ::; r
; (10-37)
where l denotes the Lagrange multiplier. From this equation it follows that
iJ
iE
i
l ; (10-38)
359
10.3 Prediction of Biological Systems from Optimality Principles
