Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


c. Determine the operating times for 70 and 80% conversion, respectfully.
d. Determine the operating times for 70 and 80% conversion, when the initial
concentration is 3.5 mol/L.
Solution The stoichiometric coefficients of the chemical reaction are
s
A
¼2 s
B
¼ 1 D ¼1
We select the initial state as the reference state, and, since only reactant A is
charged into the reactor, C
0
¼ C
A
(0), y
A
(0) ¼ 1, y
B
(0) ¼ 0.
a. For a constant-volume reactor, using Eq. 6.1.12,
C
A
¼ C
0
(1 2Z)
and the reaction rate expression is
r ¼ kC
0
2
(1 2Z)
2
(a)
Substituting (a) into Eq. 6.2.1, the design equation becomes
dZ
dt
¼ kC
0
2
(1 2Z)
2
t
cr
C
0
(b)
We select the characteristic reaction time to be
t
cr
¼
1
kC
0
(c)
and t ¼ kC
0
t. Substituting (c) into (b), the design equation reduces to
dZ
dt
¼ (1 2Z)
2
(d)
We solve (d) subject to the initial condition Z(0) ¼ 0, by separating the vari-
ables and obtain
t ¼
Z(t)
1 2Z(t)
(e)
Hence, the solution is
Z(t) ¼
t
1 þ 2t
(f)
Figure E6.1.1 shows the reaction operating curve.
170 IDEAL BATCH REACTOR
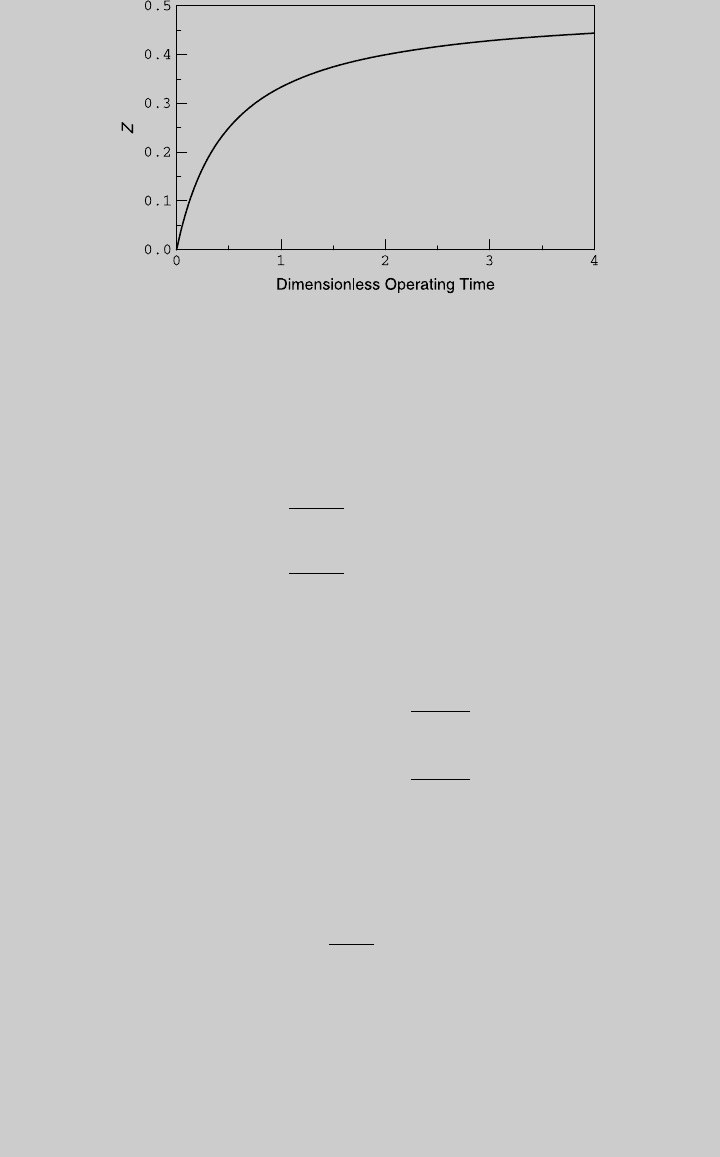
b. Once the reaction curve is known, we use Eq. 2.7.4 to obtain the species
curves:
N
A
(t)
(N
tot
)
0
¼ 1 2Z(t) (g)
N
B
(t)
(N
tot
)
0
¼ Z(t) (h)
Substituting (f) into (g) and (h), the species contents are
N
A
(t) ¼ (N
tot
)
0
1
1 þ 2t
(i)
N
B
(t) ¼ (N
tot
)
0
t
1 þ 2t
(j)
Figure E6.1.2 shows the species operating curves.
c. Using Eq. 2.6.2 to relate the conversion to extent,
Z ¼
y
A
(0)
s
A
f
A
¼ 0:50 f
A
The extents for 70 and 80% conversions are, respectively, Z ¼ 0.35 and
Z ¼ 0.4. Substituting these values in (e), the respective dimensionless oper-
ating times are t ¼ 1.167 and t ¼ 2.0. Using (c), for C
0
¼ 4 mol/L,
t
cr
¼ 0.5 h. The required operating times for 70% conversions is
t ¼ tt
cr
¼ (1:167)(0:5h) ¼ 0:583 h
Figure E6.1.1 Reaction curve.
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 171

The required operating times for 80% conversions is
t ¼ tt
cr
¼ (2)(0:5h) ¼ 1h
d. Using (c), for C
0
¼ 3.5 mol/L, t
cr
¼ 0.571 h, and the required operating
times for 70% conversions is
t ¼ tt
cr
¼ (1:167)(0:571 h) ¼ 0:666 h
The required operating times for 80% conversions is
t ¼ tt
cr
¼ (2)(0:571 h) ¼ 1:332 h
Example 6.2 At 800 K, dimethyl ether (CH
3
OCH
3
) decomposes according to
the first-order, irreversible, gas-phase reaction
CH
3
OCH
3
! CH
4
þ CO þ H
2
Dimethyl ether is introduced into an evacuated, constant-volume batch reactor,
and the initial pressure is 310 mm Hg. After 780 min, the reactor pressure is
490 mm Hg.
a. Derive the reaction and species operating curves.
b. Derive an expression for the reactor pressure as a function of operating time.
c. Determine the value of the reaction rate constant.
d. Determine the operating time for 80% conversion.
Figure E6.1.2 Species curves.
172 IDEAL BATCH REACTOR

Solution For convenience, the chemical reaction is represented symbolically by
A ! B þ C þ D
and its stoichiometric coefficients are
s
A
¼1 s
B
¼ 1 s
C
¼ 1 s
D
¼ 1 D ¼ 2
We select the initial state as the reference state, and, since only ether (reactant A) is
charged into the reactor, C
0
¼ C
A
(0), y
A
(0) ¼ 1, y
B
(0) ¼ 0, y
C
(0) ¼ 0, and
y
D
(0) ¼ 0.
a. The reaction is first-order, r ¼ kC
A
. For a constant-volume reactor, using
Eq. 6.1.12,
C
A
¼ C
0
(1 Z)
and
r ¼ kC
0
(1 Z) (a)
Substituting (a) into Eq. 6.2.4, the design equation reduces to
dZ
dt
¼ 1 Z (b)
where the characteristic reaction time is
t
cr
¼
1
k
(c)
and t ¼ kt. We solve (b) subject to the initial condition Z(0) ¼ 0, and the
solution is
Z(t) ¼ 1 e
t
(d)
or
t ¼ln [1 Z(t)] (e)
Equation (d) is the expression of the reaction operating curve. Using
Eq. 2.7.4, the species curves are
N
A
(t)
(N
tot
)
0
¼ 1 Z(t) ¼ e
t
N
B
(t)
(N
tot
)
0
¼
N
C
(t)
(N
tot
)
0
¼
N
D
(t)
(N
tot
)
0
¼ Z(t) ¼ 1 e
t
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 173

b. Next we derive the relation between the reactor pressure and the extent. At
high temperature and low pressure, ideal gas behavior can be assumed,
PV
R
¼ N
tot
RT
and, for a constant-volume isothermal reactor
P(t)
P(0)
¼
N
tot
(t)
N
tot
(0)
(f)
Using the relation between the total number of moles and the extent,
Eq. 2.7.6,
N
tot
(t) ¼ (N
tot
)
0
1 þ DZ(t)½ (g)
and substituting in (f),
P(t)
P(0)
¼ 1 þ 2Z(t) (h)
Substituting (d) into (h), we obtain
P(t)
P(0)
¼ 3 2e
t
¼ 3 2e
k t
(i)
c. At t ¼ 780 min,
P(t)
P(0)
¼
490
310
¼ 1:58
Hence, from (h), Z(t) ¼ 0.29, and from (e), t ¼ 0.343, and using (c),
k ¼
t
t
¼
0:343
780 min
¼ 4:4 10
4
min
1
d. Using the stoichiometric relation between the conversion and extent (Eq.
2.6.2), the extent for 90% conversion is
Z ¼
y
A
(0)
s
A
f
A
¼
1
1
0:90 ¼ 0:90
Substituting Z ¼ 0.90 in (e), t ¼ 2.303. To determine the actual operating
time t, use the definition of the dimensionless time, (e),
t ¼ tt
cr
¼ (2:303)(20 min) ¼ 46:06 min
174 IDEAL BATCH REACTOR

Example 6.3 A biological waste, A, is decomposed by an enzymatic reaction
A ! B þC
in aqueous solution. The rate expression of the reaction (Michaelis– Menten
equation) is
r ¼
kC
A
K
m
þ C
A
A solution with a concentration of 2 mol A/L is charged into a batch reactor. For
the enzyme type and concentration used, k ¼ 0.1 mol/L min and K
m
¼ 4 mol/L.
a. Derive and plot the reaction operating curve.
b. Derive and plot the operating curves for species A and B.
c. Determine how long should we operate the reactor to achieve 80%
conversion.
Solution This example illustrates how to apply the design equation for ideal
batch reactors with reactions whose rate expressions are not power functions
of the species concentrations.
a. For the chemical reaction, the stoichiometric coefficients are s
A
¼ 21,
s
B
¼ 1, and s
C
¼ 1. We select the initial state as the reference state, and,
since A is the only species charged into the reactor, C
0
¼ C
A
(0) and
y
A
(0) ¼ 1, y
B
(0) ¼ 0, y
C
(0) ¼ 0. The design equation is
dZ
dt
¼ r
t
cr
C
0
(a)
Using Eq. 6.1.12, the concentration of reactant A is
C
A
¼ C
0
(1 Z) (b)
and, substituting (b), the reaction rate expression is
r ¼ k
1 Z
K
m
=C
0
þ (1 Z)
(c)
Substituting (c) into (a), the design equation becomes
dZ
dt
¼ k
1 Z
K
m
=C
0
þ (1 Z)
t
cr
C
0
(d)
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 175
We select the characteristic reaction time to be
t
cr
¼
C
0
k
¼ 20 min (e)
(the same as the one obtained by the procedure described in Section 3.5).
Substituting (e) into (d), the design equation reduces to
dZ
dt
¼
1 Z
K
m
=C
0
þ (1 Z)
(f)
To obtain the reaction operating curve, Z(t) versus t, (f) is solved, subject to
the initial condition that Z(0) ¼ 0. In this case an analytical solution is
obtained by separating the variables and integrating:
t ¼ Z(t)
K
m
C
0
ln [1 Z(t)] (g)
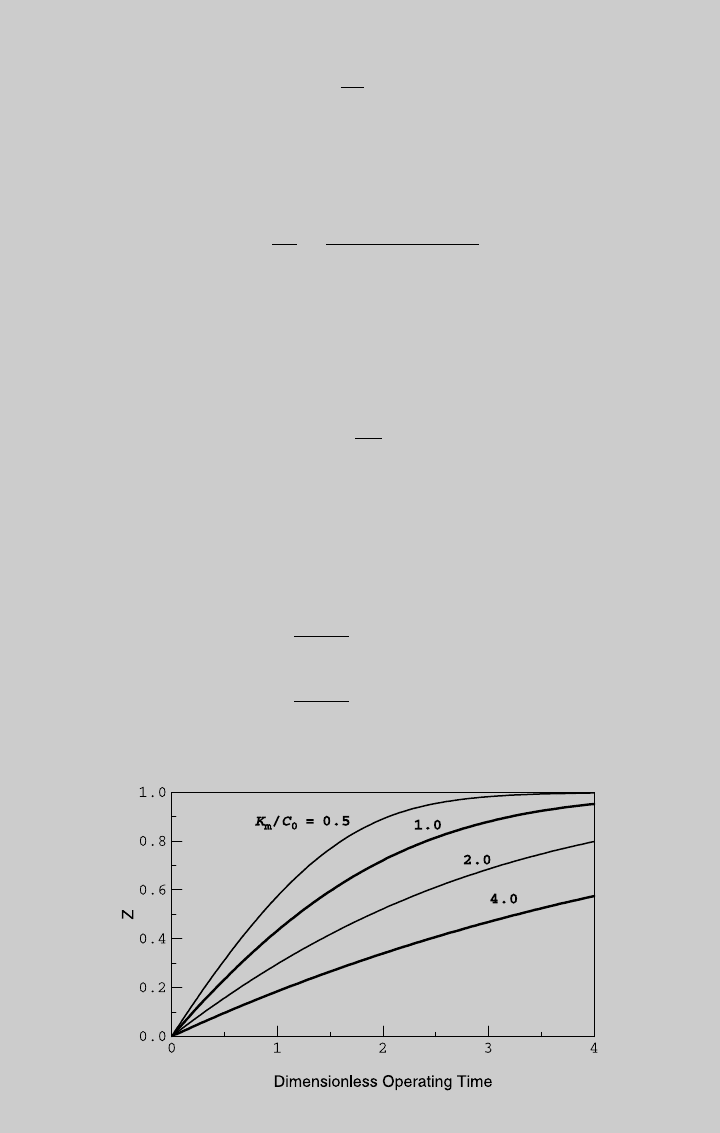
Figure E6.3.1 shows the reaction operating curve for different values of K
m
/C
0
.
b. Once the reaction operating curve is known, the species composition is
readily determine using Eq. 2.7.4,
N
A
(t)
(N
tot
)
0
¼ 1 Z(t)
N
B
(t)
(N
tot
)
0
¼ Z(t)
Figure E6.3.1 Reaction curve.
176 IDEAL BATCH REACTOR

c. Using the relation between the conversion and extent for a single reaction
(Eq. 2.6.2), for 80% conversion
Z ¼
y
A
(0)
s
A
f
A
¼ 0:80
Substituting Z ¼ 0.80 in (g) for K
m
/C
0
¼ 2, we obtain t ¼ 4.019. To deter-
mine the operating time t, use the definition of the dimensionless time, (e):
t ¼ tt
cr
¼ 80:4 min
We continue the analysis of ideal, isothermal, constant-volume batch reactors
with single reactions and consider now chemical reactions involving more than
one reactant. Consider the general reaction form
A þ bB ! Products (6:2:13)
that represents reactions between two reactants where b ¼ s
B
/s
A
. The stoichio-
metric coefficients of the reaction are s
A
¼ 21 and s
B
¼ 2 b. We consider here
chemical reactions whose rate expressions are of the form r ¼ kC
A
a
C
B
b
. We select
the initial state as the reference state and express the species concentrations using
Eq. 6.1.12:
C
A
(t) ¼ C
0
[ y
A
(0) Z(t)] (6:2:14)
C
B
(t) ¼ C
0
[ y
B
(0) bZ(t)] (6:2:15)
where y
A
(0) ¼ C
A
(0)/C
0
and y
B
(0) ¼ C
B
(0)/C
0
. Substituting these equations into
the rate expression,
r ¼ kC
0
aþb
[ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
(6:2:16)
Substituting Eq. 6.2.16 into Eq. 6.2.4, the design equation becomes
dZ
dt
¼ kC
0
aþb
[ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
t
cr
C
0
(6:2:17)
We select characteristic reaction time
t
cr
¼
1
kC
0
aþb1
(6:2:18)
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 177

(the same t
cr
as obtained by the procedure in Section 3.5), and the design equation
reduces to
dZ
dt
¼ [ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
(6:2:19)
We solve Eq. 6.2.19 numerically, subject to the initial condition that at t ¼ 0,
Z(0) ¼ 0. Figure 6.5 shows the reaction operating curves for reaction 6.2.13 with
a ¼ b ¼ 1, for different proportions of the reactants. Note that when the reactants
are in stoichiometric proportion, y
B
(0) ¼ by
A
(0), Eq. 6.2.19 reduces to
dZ
dt
¼ b
b
[ y
A
(0) Z]
aþb
(6:2:20)
Also, since reactant A is the limiting reactant, for all other cases by
A
(0)/y
B
(0) 1.
Example 6.4 Consider the gas-phase reaction
A þ B ! C
carried out in a 100-L isothermal constant-volume batch reactor operated at
2508C. The reaction is first order with respect to A and B. A gas mixture con-
sisting of 40% A and 60% B (by mole) is charged into the reactor whose initial
pressure is 2 atm. At 2508C, the reaction rate constant is k ¼ 800 L/mol min.
a. Derive and plot the dimensionless reaction and species operating curves.
b. Determine the operating time needed for 80% conversion.
c. Calculate the pressure of the reactor at 80% conversion.
d. Calculate the content of species C at 80% conversion.
Figure 6.5 Reaction operating curves a reaction of the form A þ B ! products.
178 IDEAL BATCH REACTOR

Solution The stoichiometric coefficients of the chemical reaction are
s
A
¼1 s
B
¼1 s
C
¼ 1 D ¼1
We select the initial state as the reference state; hence, C
0
¼ C
A
(0) þ
C
B
(0) þ C
C
(0), and y
A
(0) ¼ 0.4, y
B
(0) ¼ 0.6, and y
C
(0) ¼ 0. Using Eq. 6.1.12,
the species concentrations are
C
A
¼ C
0
(0:4 Z) (a)
C
B
¼ C
0
(0:6 Z) (b)
C
C
¼ C
0
Z (c)
The rate expression is r ¼ kC
A
C
B
, and using (a) and (b),
r ¼ kC
0
2
(0:4 Z)(0:6 Z) (d)
Substituting (d) into Eq. 6.2.1, the design equation becomes
dZ
dt
¼ kC
0
2
(0:4 Z)(0:6 Z)
t
cr
C
0
(e)
We define the characteristic reaction time by
t
cr
¼
1
kC
0
(f)
and the design equation reduces to
dZ
dt
¼ (0:4 Z)(0:6 Z) (g)
We solve (h) numerically, subject to the initial condition that at t ¼ 0, Z ¼ 0,
and plot the reaction operating curve, shown in Figure E6.4.1.
Once we know the reaction curve, we can determine readily the species curves
using Eq. 2.7.4:
N
A
(t)
(N
tot
)
0
¼ 0:4 Z(t) (h)
N
B
(t)
(N
tot
)
0
¼ 0:6 Z(t) (i)
N
C
(t)
(N
tot
)
0
¼ Z(t)(j)
Figure E6.4.2 shows the species curves.
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 179
