Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.

a. To determine the operating time required for 80% conversion, we have to cal-
culate first the corresponding extent. Using Eq. 2.6.2,
Z ¼
y
A
(0)
s
A
f
A
¼ 0:32
From the operating curve (or the numerical solution of the design equation),
Z ¼ 0.32 is reached at t ¼ 4.25. To calculate the actual operating time, we
first have to determine the characteristic reaction time. We calculate C
0
,
which for an ideal gas is
C
0
¼
P
0
RT
0
¼
2 atm
ð82:05 10
3
L atm =mol KÞ(523 K)
¼ 4:66 10
2
mol=L
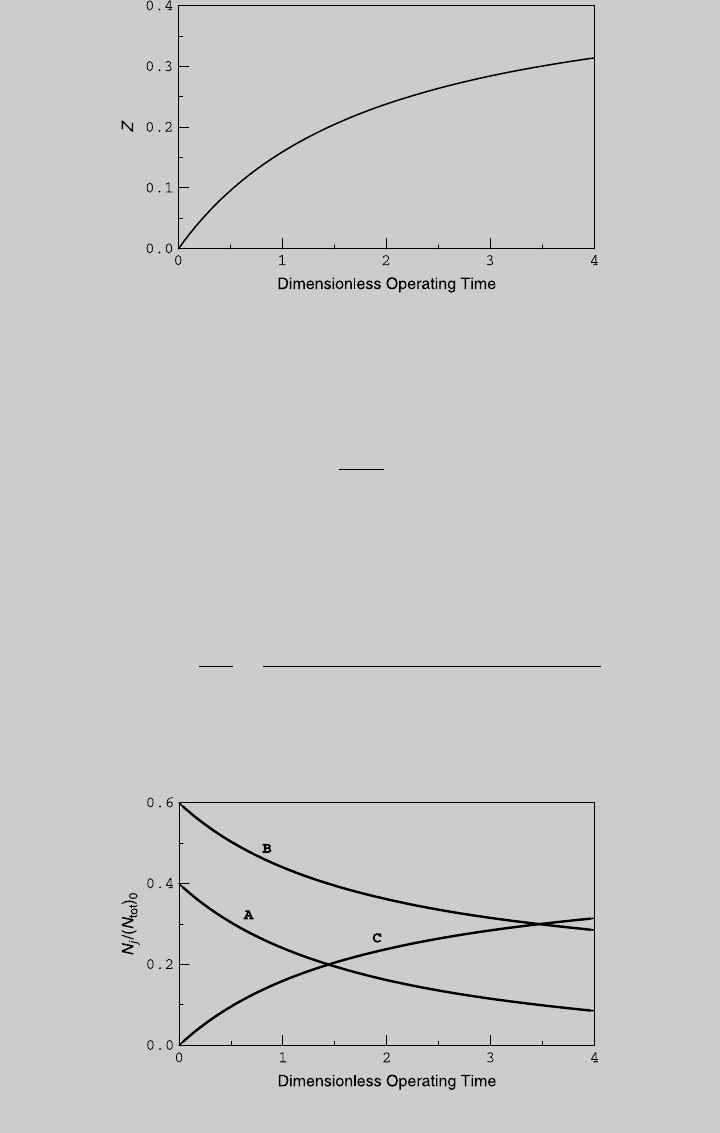
Figure E6.4.2 Species curves.
Figure E6.4.1 Reaction curve.
180 IDEAL BATCH REACTOR

Using the definition of the dimensionless time, (f),
t ¼
t
kC
0
¼ 114:0 min
b. Using Eq. 2.7.6, the reactor pressure at 80% conversion of reactant A is
P(t) ¼ P
0
[1 þ D Z(t)] ¼ 1:36 atm
c. Using (c), the concentration of product C is
C
C
¼ C
0
Z ¼ 1:49 10
2
mol=L
6.2.2 Gaseous, Variable-Volume Batch Reactors
In this section, we consider ideal batch reactors whose volume changes during the
operation, like the one shown schematically in Figure 6.2. In general, when a gas-
phase reaction results in a net change of the total number of moles, the volume of
the reactor changes during the operation if the reactor is maintained at a constant
pressure and temperature. For example, the combustion stroke of a car engine
cycle (while the valves are closed) is carried out in a variable-volume reactor; in
fact, the change in the volume is utilized to generate mechanical work. For such
operations, we should incorporate the changes in the reactor volume into the
design equation.
The variation of the reactor volume with the reaction extent is given by
Eq. 6.1.14. For isothermal-isobaric operations with single reactions, and when
the initial state is selected as the reference, Eq. 6.1.14 reduces to
V
R
(t)
V
R0
¼ 1 þ D Z(t)(6:2:21)
Substituting Eq. 6.2.21 into Eq. 6.1.1, the design equation becomes
dZ
dt
¼ r(1 þ D Z)
t
cr
C
0
(6:2:22)
Equation 6.2.22 is solved subject to the initial condition Z(0) ¼ 0. When r depends
only on Z, we can obtain analytical solutions by separating the variables and
integrating:
t ¼
C
0
t
cr
ð
Z(t)
0
dZ
r(1 þ D Z)
(6:2:23)
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 181
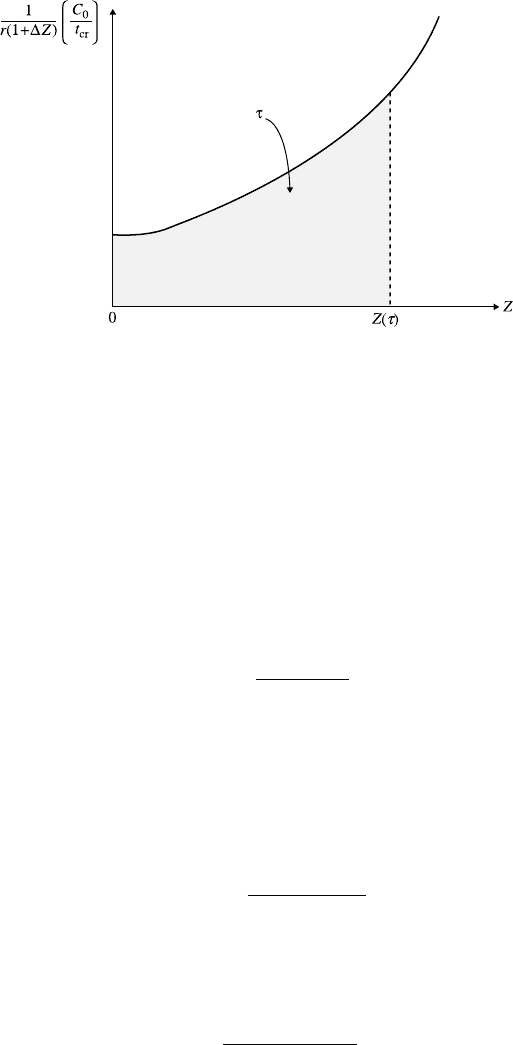
Equation 6.2.23 is the integral form of the design equation, and it is represented
graphically in Figure 6.6. Note that for the special case when D ¼ 0 (no change
in the total number of moles), Eq. 6.2.22 reduces to Eq. 6.2.5, the design equations
for a constant-volume batch reactor. Also, by multiplying both sides of Eq. 6.2.23
by t
cr
, we express the design equation in terms of the actual operating time (rather
than the dimensionless time):
t ¼ C
0
ð
Z(t)
0
dZ
r(1 þ D Z)
(6:2:24)
To solve the design equation, we have to express the reaction rate r in terms of Z
and, to do so we relate the species concentrations to the dimensionless extent. From
Eq. 6.1.11, for isothermal operations with single reactions, and when the reference
state is the initial state:
C
j
(t) ¼ C
0
y
j
(0) þ s
j
Z(t)
1 þ D Z(t)
(6:2:25)
Note that, when D=0, the species concentrations are not proportional to the reac-
tion extent. To determine Z(t) when C
j
(t) is given, we rearrange Eq. 6.2.25,
Z(t) ¼
C
j
(t) y
j
(0)C
0
s
j
C
0
DC
j
(t)
(6:2:26)
Below, we analyze the operation of variable-volume, gaseous batch reactors and
describe how to apply the design equation for different cases.
Figure 6.6 Graphical presentation of the design equation for variable-volume batch
reactor.
182 IDEAL BATCH REACTOR
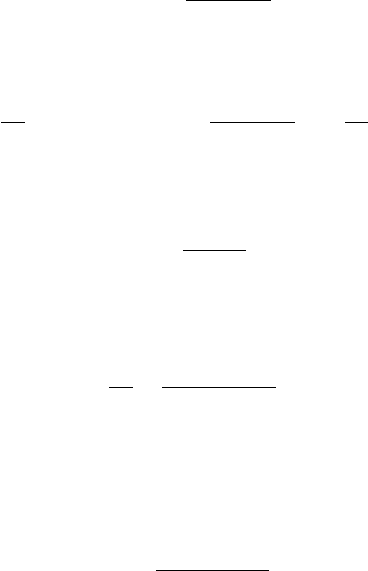
First, we discuss the application of the design equation for a variable-volume
batch reactor when the reaction rate is provided in the form of an algebraic
expression. We start by considering chemical reactions of the form
A ! Products
whose ra te expression is r ¼ kC
A
a
. For this case, s
A
¼ 21, and when the initial sta te is
the reference state, Eq. 6.2.25 is used to e xpress the species concentr ations. The ra te
ex pr ession becomes
r ¼ kC
0
a
y
A
(0) Z
1 þ D Z
a
(6:2:27)
Substituting Eq. 6.2.27 into Eq. 6.2.22, the design equation is
dZ
dt
¼ kC
0
a
(1 þ D Z)
y
A
(0) Z
1 þ D Z
a
t
cr
C
0
(6:2:28)
For ath-order r eactions, the char acteristic reaction time is
t
cr
¼
1
kC
0
a1
(6:2:29)
Substituting Eq. 6.2.29 into Eq. 6.2.28, the design equation reduces to
dZ
dt
¼
[ y
A
(0) Z]
a
(1 þ D Z)
a1
(6:2:30)
which can be solv ed subject to a specified initial condition, Z(0). Separa ting the vari-
ables and integrating,
t ¼
ð
Z(t)
Z(0)
(1 þ D Z)
a1
(1 Z)
a
dZ (6:2:31)
Equation 6.2.31 is the integral design equation for this case. Figure 6.7 shows the
reaction oper a ting curve of a second-order r ea ction (a ¼ 2) for different values of D.
Note that for larger values of D, longer oper a ting times are r equir ed for achieving a
given extent. Also, the curv e for D ¼ 0 repr esents the performance of a constant-
volume batch r ea ctor.
We continue the analysis of gaseous, variable-volume batch reactors and con-
sider now chemical reactions with two reactants of the general form
A þ bB ! Products
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 183

whose rate expression is of the form r ¼ kC
A
a
C
B
b
. For this case, s
A
¼ 21,
s
B
¼ 2b, and the initial composition, y
A
(0) and y
B
(0), is specified. When the initial
state is selected as the reference state, using Eq. 6.2.25, the rate expression becomes
r ¼ kC
0
aþb
[ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
(1 þ D Z)
aþb
(6:2:32)
Substituting Eq. 6.2.32 into Eq. 6.2.22, the design equation becomes
dZ
dt
¼ (1 þ D Z) kC
0
aþb
[ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
(1 þ D Z)
aþb
t
cr
C
0
(6:2:33)
Using Eq. 3.5.4, for rea ctions whose ov er all order is a þ b, the char acteristic reaction
time is
t
cr
¼
1
kC
0
aþb1
(6:2:34)
Substituting Eq. 6.2.34 into Eq. 6.2.33, the design equation r educes to
dZ
dt
¼
[ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
(1 þ D Z)
aþb1
(6:2:35)
Equation 6.2.35 is the differential design equa tion for this case and should be solved
subject to the initial condition that at t ¼ 0, Z ¼ 0. Separ ating the variables and
Figure 6.7 Operating curves of isothermal-isobaric batch reactor with second-order gas-
eous reaction A ! products for different values of D.
184 IDEAL BATCH REACTOR

integrating, w e obtain
t ¼
ð
Z(t)
0
(1 þ D Z)
aþb1
[ y
A
(0) Z]
a
[ y
B
(0) bZ]
b
dZ (6:2:36)
Equation 6.2.36 is the integr al design equation for this case.
The design equation for gaseous variable-volume batch reactors was derived
under two assumptions: (i) All the species are gaseous, and (ii) the mixture behaves
as an ideal gas. In some operations, one or more of the species (especially heavier
products generated by the reaction) may be saturated vapor. In this case, any
additional amount generated will be in a condensed phase (liquid). While the
ideal gas relation provides a reasonable approximation for the volume of species
in the vapor phase, it cannot be applied for their volume in the liquid phase.
Below, we modify the design equations for a variable-volume batch reactor with
saturated vapors.
Recognizing that, with the exception of operations at very high pressures (or near
critical conditions), the specific volume of a species in the liquid-phase is two to
three orders of magnitude smaller than its specific volume in the vapor phase,
the volume of the species that is in a liquid phase can be neglected. Hence,
when considering variations in the reactor volume, we should account only for
changes in the number of moles in the gas phase. Thus, the factor D used in the
design equation should be calculated on the basis of the change in the number
of moles in the gas phase only, D
gas
, not D of the reaction. The calculation of
D
gas
depends on the reaction extent when the two phases are in equilibrium, as
illustrated in the example below.
Example 6.5 The elementary gas-phase reaction
A þ B ! C
is carried out in an isothermal-isobaric (variable-volume) batch reactor. Its initial
volume is 234.5 L, the pressure is 1.2 atm, and the temperature is 708C. At this
temperature, k ¼ 3.2 L/mol min, and the vapor pressure of the product C is
0.3 atm. The reactor is filled with a stoichiometric mixture of A and B.
Determine:
a. The reaction extent when C starts to condense.
b. The operating time when C starts to condense.
c. The operating time needed for 80% conversion.
Solution At the beginning of the operation, all the species are gaseous, the
chemical reaction is
A(g) þ B(g) ! C(g)
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 185

and the stoichiometric coefficients are
s
A
¼1 s
B
¼1 s
C
¼ 1 D
gas
¼ D ¼1
We select the initial state as the reference state, and the initial composition is
y
A
(0) ¼ y
B
(0) ¼ 0.5 and y
C
(0) ¼ 0. Using Eqs. 2.7.4 and 2.7.6,
N
A
(t)
(N
tot
)
0
¼
N
B
(t)
(N
tot
)
0
¼ 0:5 Z(t) (a)
N
C
(t)
(N
tot
)
0
¼ Z(t) (b)
N
tot
(t)
(N
tot
)
0
¼ 1 Z(t) (c)
Using Eq. 6.1.16, the concentrations of the species are
C
A
¼ C
B
¼ C
0
0:5 Z
1 Z
(d)
C
C
¼ C
0
Z
1 Z
(e)
Applying ideal gas law, the initial (reference) total number of moles is
(N
tot
)
0
¼
P
0
V
R
0
RT
0
¼ 10 mol
and the reference concentration is
C
0
;
(N
tot
)
0
V
R
0
¼ 0:0426 mol=L
a. Condensation of product C commences when its partial pressure in the reac-
tor is 0.3 atm. Assuming ideal gas behavior,
P
C
(t) ¼ y
C
(t)P(t) ¼
N
C
(t)
N
tot
(t)
P(t)
Using the stoichiometric relations (b) and (c),
P
C
(t) ¼
Z(t)
1 Z(t)
(1:2) ¼ 0:3(f)
Solving (f), Z(t) ¼ 0.20.
186 IDEAL BATCH REACTOR

b. Using Eq. 6.2.22, the design equation for an isothermal-isobaric operation is
dZ
dt
¼ r(1 Z)
t
cr
C
0
0 Z 0:2 (g)
Using (d) and (e), the rate expression is
r ¼ kC
A
C
B
¼ kC
0
2
(0:5 Z)
2
(1 Z)
2
(h)
Using Eq. 3.5.4, for a second-order reaction, the characteristic reaction time is
t
cr
¼
1
kC
0
¼ 7:33 min (i)
and t ¼ kC
0
t. Substituting (h) and (i) into (g), the design equation reduces to
dZ
dt
¼
(0:5 Z)
2
1 Z
0 Z 0:2(j)
The dimensionless operating time for Z ¼ 0.20 is
t ¼
ð
0:2
0
1 Z
(0:5 Z)
2
dZ ¼ 1:177 (k)
and, the operating time is
t ¼
t
kC
0
¼ 8:63 min
c. For t . 1.178 (or Z . 0.2), product C is formed in a liquid phase; hence the
following reaction takes place in the reactor:
A(g) þ B(g) ! C(liquid) (l)
For this reaction, D
gas
¼ 22. Hence, a portion of C is in gas phase and a por-
tion in liquid phase (with negligible volume). For t . 1.178, the total
number of moles in the gas phase (for Z . 0.20) is
(N
tot
)
gas
¼ (N
tot
)
0
[1 þ (1)0:2 þ (2)(Z 0:2)] ¼ (N
tot
)
0
(1:2 2Z)
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 187

and, for isothermal-isobaric operation, the reactor volume varies according to
V
R
V
R
0
¼
N
tot
(N
tot
)
0
¼ 1:2 2Z (m)
Using (m), for t . 1.178 the design equation is
dZ
dt
¼ r(1:2 2Z)
t
cr
C
0
Z . 0:2 (n)
and the concentrations of the two reactants are
C
A
¼ C
B
¼ C
0
0:5 Z
1:2 2Z
Z . 0:2 (o)
Substituting (o) into (n), the design equation becomes
dZ
dt
¼
(0:5 Z)
2
(1:2 2Z)
Z . 0:2(p)
To determine the dimensionless operating time for 80% conversion
(Z ¼ 0.4), we separate the variables and integrate (numerically),
t ¼
ð
0:4
0:2
1:2 2Z
(0:5 Z)
2
dZ ¼ 3:5305 (q)
and the operating time for Z . 0.2 is
t ¼
t
kC
0
¼ 25:9 min
The total operating time needed for 80% conversion is t ¼ 8.63þ 25.9 ¼
34.53 min.
d. The reactor operation is described by two design equations, each applies for a
certain period during the operation: (j) for 0 Z 0.2 and (p) for Z . 0.2.
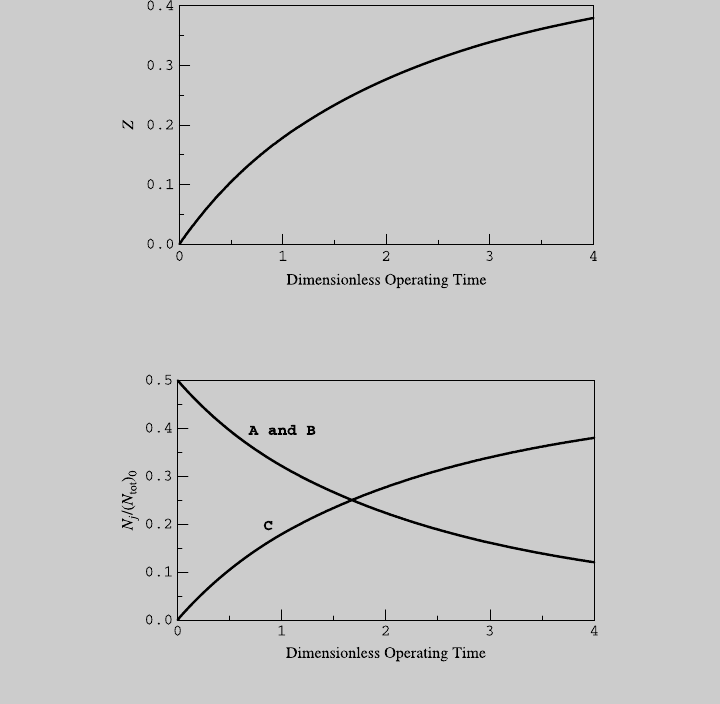
Figure E6.5.1 shows the reaction curve. We use (a) and (b) to determine the
species curves, shown in Figure E6.5.2. Note that in the second stage of the
operation, the D factor is smaller than in the first stage; consequently, the total
operating time is smaller than that calculated by ignoring the condensation of
species C. If we integrated (k) between 0 and 0.4, we would have obtained
t ¼ 5.61 and the operating time is 41.15 min.
188 IDEAL BATCH REACTOR
We conclude the discussion on the applications of the design equation of ideal
batch reactors with two important comments. First, note that in all the cases dis-
cussed above, the design equation provides only the reactor operating time
(either dimensionless or dimensional) needed to obtain a given extent level. The
design equation does not indicate what reactor size (volume) should be used.
The reactor size is determined by the required production rate and by the downtime
between batches (for loading, downloading, and cleaning). Second, one may ask
what extent level (or conversion) should be specified for a given operation. The
optimal extent level is determined by the cost of the reactants, the value of the pro-
ducts, the cost of the equipment, and the operating expenses. These issues are
discussed in Chapter 10.
6.2.3 Determination of the Reaction Rate Expression
In the preceding two sections, w e discussed how to apply the design equation when the
rate expr ession is known. In this section, we will describe methods to determine the reac-
tion ra te fro m opera ting da ta, and then determine the parameters of the ra te e xpr ession.
Figure E6.5.2 Species curves.
Figure E6.5.1 Reaction curves.
6.2 ISOTHERMAL OPERATIONS WITH SINGLE REACTIONS 189
