Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.

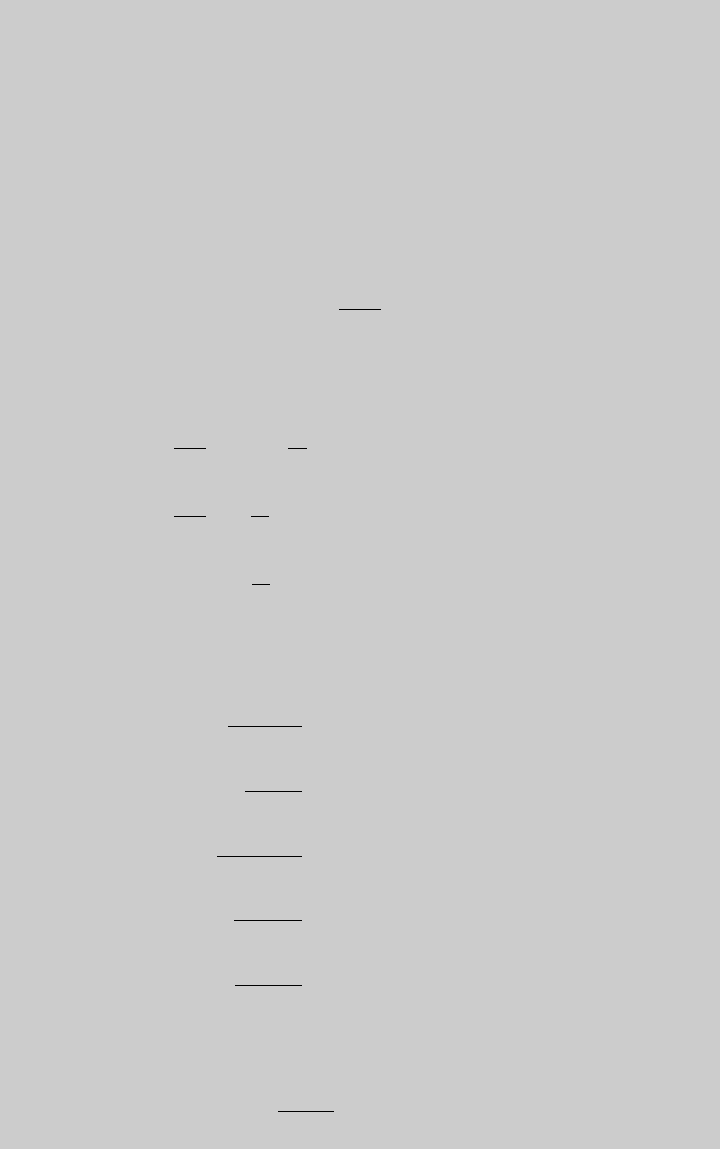
Using Eq. 6.1.12 to express the species concentrations, the rates of the three
chemical reactions are
r
1
¼ k
1
C
0
2
(0:6 Z
1
)(0:4 Z
1
3:5Z
2
) (d)
r
2
¼ k
2
C
0
2
(Z
1
Z
2
)(0:4 Z
1
3:5Z
2
) (e)
r
3
¼ k
3
C
0
2
(0:6 Z
1
)(0:4 Z
1
3:5Z
2
)(f)
We select Reaction 1 as the leading reaction, and, using Eq. 3.5.4, the charac-
teristic reaction time is
t
cr
¼
1
k
1
C
0
¼ 100 s (g)
Substituting (d), (e), (f), and (g) into (b) and (c), the design equations
become
dZ
1
dt
¼ 1 þ
k
3
k
1
(0:6 Z
1
)(0:4 Z
1
3:5Z
2
) (h)
dZ
2
dt
¼
k
2
k
1
(Z
1
Z
2
)(0:4 Z
1
3: 5Z
2
) þ
k
3
k
1
(0:6 Z
1
)(0:4 Z
1
3:5Z
2
) (i)
Once we solve the design equations, we use Eq. 2.7.4 to obtain the species
curves:
N
C
3
H
6
(t)
(N
tot
)
0
¼ 0:6 Z
1
(t)(j)
N
O
2
(t)
(N
tot
)
0
¼ 0:4 Z
1
(t) 3:5Z
2
(t) (k)
N
C
3
H
4
O
(t)
(N
tot
)
0
¼ Z
1
(t) Z
2
(t) (l)
N
H
2
O
(t)
(N
tot
)
0
¼ Z
1
(t) þ 2Z
2
(t) (m)
N
CO
2
(t)
(N
tot
)
0
¼ 3Z
2
(t) (n)
Using Eq. 2.7.6, the total molar content in the reactor is
N
tot
(t)
(N
tot
)
0
¼ 1 þ 0:5Z
2
(t) (o)
210 IDEAL BATCH REACTOR

We substitute the numerical values k
2
/k
1
¼ 0.25 and k
3
/k
1
¼ 0.1 into (h) and
(i), and solve them numerically subject to the initial conditions that at t ¼ 0,
Z
1
¼ Z
2
¼ 0. Figure E6.10.1 shows the reaction curves. We the apply ( j)
through (n) to determine the species curves, shown in Figure E6.10.2.
b. Using the definition of the conversion, at 75% conversion of oxygen,
N
O
2
¼ (1 0:75)N
O
2
(0) ¼ (1 0:75)0:4(N
tot
)
0
hence, N
O
2
=(N
tot
)
0
¼ 0.1. From the species operating curve (or tabulated
values of the numerical solution), this is reached at t ¼ 1.78. Using (g),
the operating time is
t ¼ tt
cr
¼ 178 s
c. From the reaction curves at t ¼ 1.78, Z
1
¼ 0.204 and Z
2
¼ 0.0275. Using ( j)
through (n) and (o), the mole fractions of the individual species are
y
C
3
H
6
¼ 0:391 y
O
2
¼ 0:099 y
C
3
H
4
O
¼ 0:174
y
H
2
O
¼ 0:255 y
CO
2
¼ 0:081
Figure E6.10.1 Reaction curves.
Figure E6.10.2 Species curves.
6.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 211

Example 6.11 Selective oxidation of ammonia is investigated in an isothermal
constant-volume batch reactor. The following gaseous chemical reactions take
place in the reactor:
Reaction 1: 4NH
3
þ 5O
2
! 4NO þ6H
2
O
Reaction 2: 4NH
3
þ 3O
2
! 2N
2
þ 6H
2
O
Reaction 3: 2NO þ O
2
! 2NO
2
Reaction 4: 4NH
3
þ 6NO ! 5N
2
þ 6H
2
O
The desired product is NO. A gas mixture consisting of 50% NH
3
and 50% O
2
(mol %) is charged into a 4-L reactor, and the initial pressure is 2 atm. The reac-
tor operates at 609 K.
a. Derive the design equations and plot the reaction and species operating
curves.
b. Determine the operating time at which the maximum amount of NO is
reached.
c. Determine the reactor composition at that time.
The rate expressions of the reactions are
r
1
¼ k
1
C
NH
3
C
O
2
2
r
2
¼ k
2
C
NH
3
C
O
2
r
3
¼ k
3
C
O
2
C
NO
2
r
4
¼ k
4
C
NH
3
2=3
C
NO
Data: At 609 K,
k
1
¼ 20 (L=mol)
2
min
1
k
2
¼ 0:04 (L=mol)min
1
k
3
¼ 40 (l=mol)
2
min
1
k
4
¼ 0:0274 (L=mol)
2=3
min
1
Solution The reactor design formulation of these chemical reactions was dis-
cussed in Example 4.3. Recall that there are three independent reactions and one
dependent reaction, and, following the heuristic rule, we select Reactions 1, 2,
and 3 as a set of independent reactions and Reaction 4 is a dependent reaction.
Hence, m ¼ 1, 2, 3, k ¼ 4. The stoichiometric coefficients of the independent
reactions are
(s
NH
3
)
1
¼4(s
O
2
)
1
¼5(s
NO
)
1
¼4(s
H
2
O
)
1
¼6(s
N
2
)
1
¼0(s
NO
2
)
1
¼0 D
1
¼1
(s
NH
3
)
2
¼4(s
O
2
)
2
¼3(s
NO
)
2
¼0(s
H
2
O
)
2
¼6(s
N
2
)
2
¼2(s
NO
2
)
2
¼0 D
2
¼1
(s
NH
3
)
3
¼0(s
O
2
)
3
¼1(s
NO
)
3
¼2(s
H
2
O
)
3
¼0(s
N
2
)
3
¼0(s
NO
2
)
3
¼2 D
3
¼1
212 IDEAL BATCH REACTOR

Also, from Example 4.3 the multipliers
a
k
m
’s of the dependent reaction and
the three independent reactions are a
43
¼ 0, a
42
¼ 2.5, and a
41
¼ 21.5. We
select the initial state as the reference state; hence, the reference concentration is
C
0
¼
P
0
RT
0
¼0:04mol =L (a)
The total molar content of the reference state is
(N
tot
)
0
¼V
R
0
C
0
¼0:16mol
The initial composition is
y
NH
3
(0)¼0:5 y
O
2
(0)¼0:5 y
NO
(0)¼y
H
2
O
(0)¼y
N
2
(0)¼y
NO
2
(0)¼0
a. To design the reactor, we write Eq. 6.1.1 for each independent reaction,
dZ
1
dt
¼ (r
1
1:5r
4
)
t
cr
C
0
(b)
dZ
2
dt
¼ (r
2
þ 2:5r
4
)
t
cr
C
0
(c)
dZ
3
dt
¼ r
3
t
cr
C
0
(d)
We use Eq. 6.1.12 to express the species concentrations, and the rates of the
four chemical reactions are
r
1
¼ k
1
C
0
3
[ y
NH
3
(0) 4Z
1
4Z
2
][ y
O
2
(0) 5Z
1
3Z
2
Z
3
]
2
(e)
r
2
¼ k
2
C
0
2
[ y
NH
3
(0) 4Z
1
4Z
2
][ y
O
2
(0) 5Z
1
3Z
2
Z
3
](f)
r
3
¼ k
3
C
0
3
[ y
O
2
(0) 5Z
1
3Z
2
Z
3
][ y
NO
(0) þ 4Z
1
2Z
3
]
2
(g)
r
4
¼ k
4
C
0
5=3
[ y
NH
3
(0) 4Z
1
4Z
2
]
2=3
[ y
NO
(0) þ 4Z
1
2Z
3
] (h)
We select Reaction 1, the leading reaction, and using Eq. 3.5.4, the charac-
teristic reaction time is
t
cr
¼
1
k
1
C
0
2
¼
1
(20)(0:04)
2
¼ 31:25 min (i)
6.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 213

Substituting (e) through (h) and (i) into (b), ( j), and (c), the design equations
are
dZ
1
dt
¼[y
NH
3
(0)4Z
1
4Z
2
][y
O
2
(0)5Z
1
3Z
2
Z
3
]
2
1:5
k
4
k
1
C
0
4=3
[y
NH
3
(0)4Z
1
4Z
2
]
2=3
[y
NO
(0)þ4Z
1
2Z
3
](j)
dZ
2
dt
¼
k
2
k
1
C
0
[y
NH
3
(0)4Z
1
4Z
2
][y
O
2
(0)5Z
1
3Z
2
Z
3
]
þ2:5
k
4
k
1
C
0
4=3
[y
NH
3
(0)4Z
1
4Z
2
]
2=3
[y
NO
(0)þ4Z
1
2Z
3
] (k)
dZ
3
dt
¼
k
3
k
1
[y
O
2
(0)5Z
1
3Z
2
Z
3
][y
NO
(0)þ4Z
1
2Z
3
]
2
(l)
Once we solve the design equations, we use Eq. 2.7.4, to obtain the species
curves:
N
NH
3
(N
tot
)
0
¼y
NH
3
(0)4Z
1
4Z
2
(m)
N
O
2
(N
tot
)
0
¼y
O
2
(0)5Z
1
3Z
2
Z
3
(n)
N
NO
(N
tot
)
0
¼y
NO
(0)4Z
1
2Z
3
(o)
N
H
2
O
(N
tot
)
0
¼y
H
2
O
(0)þ6Z
1
þ6Z
2
(p)
N
N
2
(N
tot
)
0
¼y
N
2
(0)þ2Z
2
(q)
N
NO
2
(N
tot
)
0
¼y
NO
2
(0)þ2Z
3
(r)
Using Eq. 2.7.6,
N
tot
(N
tot
)
0
¼1þZ
1
þZ
2
Z
3
(s)
The parameters of the design equations are
k
2
k
1
C
0
¼0:05
k
3
k
1
¼2
k
4
k
1
C
0
4=3
¼0:1
214 IDEAL BATCH REACTOR

We substitute these values into ( j), (k), and (l), and solve them numerically
subject to the initial conditions that at t ¼ 0, Z
1
¼ Z
2
¼ Z
3
¼ 0.
Figure E6.11.1 shows the solutions of the design equations—the reaction
operating curves. Next, we use (m) through (r) to determine the species
curves, shown in Figure E6.11.2.
b. From the species curves (or the table of calculated data), the maximum
amount of NO is achieved at t ¼ 1.275, and, using (i), the required operating
time is
t ¼ tt
cr
¼ 39:84 min
c. At t ¼ 1.275, Z
1
¼ 0.0395, Z
2
¼ 0.0232, and Z
3
¼ 0.0095. Substituting
these values into (m) through (r) and using (s), the species molar fractions are
y
NH
3
¼ 0:237 y
O
2
¼ 0:21 y
NO
¼ 0:132 y
H
2
O
¼ 0:357
y
N
2
¼ 0:044 y
NO
2
¼ 0:018
Figure E6.11.1 Reaction curves.
Figure E6.11.2 Species curves.
6.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 215

6.4 NONISOTHERMAL OPERATIONS
The design formulation of nonisothermal batch reactors with multiple reactions fol-
lows the procedure outlined in the previous section—we write Eq. 6.1.1 for each
independent reaction. However, since the reactor temperature may vary during
the operation, we should solve the design equations simultaneously with the
energy balance equation. Note that the energy balance equation (Eq. 6.1.17) con-
tains another variable—the temperature of the heating (or cooling) fluid, u
F
, that
may also vary during the operation (u
F
is constant only when the fluid either evap-
orates or condenses). In general, we have to write the energy balance equation for
the heating/cooling fluid to express changes in u
F
, but, in most batch reactor appli-
cations, u
F
is assumed constant. Because of the complex geometry of the heat-
transfer surface (shell or coil), the average of the inlet temperature and the outlet
temperature is usually used.
The procedure for setting up the energy balance equation goes as follows:
1. Define the reference state, and identify T
0
, C
0
,(N
tot
)
0
, and its species compo-
sitions, y
j
0
’s. Recall that, in most applications, the initial state is selected as
reference state.
2. Determine the specific molar heat capacity of the reference state,
^
c
p
0
.
3. Determine the dimensionless activation energies, g
i
’s, of all chemical
reactions.
4. Determine the dimensionless heat of reactions, DHR
m
’s, of each independent
reaction.
5. Determine the correction factor of the heat capacity, CF(Z
m
, u).
6. Determine (or specify) the dimensionless temperature of the heating/cooling
fluid, u
F
.
7. Specify the dimensionless heat-transfer number, HTN (using Eq. 6.1.26).
8. Determine (or specify) the initial dimensionless temperature, u(0).
9. Solve the energy balance equation simultaneously with the design equations
to obtain Z
m
’s and u as functions of the dimensionless operating time, t.
The design formulation of nonisothermal batch reactors consists of n
I
þ 1 non-
linear first-order differential equations whose initial values are specified. The sol-
utions of these equations provide Z
m
’s and u as functions of t. The examples
below illustrate the design of nonisothermal ideal batch reactors.
Example 6.12 The first-order, liquid-phase endothermic reactions
Reaction 1: A ! 2B
Reaction 2: B ! 2C þ D
216 IDEAL BATCH REACTOR

take place in aqueous solution. A 200-L solution with a concentration of
C
A
(0) ¼ 0.8 mol/L is charged into a batch reactor. Initially, the solution is at
478C, and the average temperature of the heating fluid is 678C.
a. Derive the reaction and species curves for isothermal operation. Determine
the operating time needed to maximize the production of product B, and
the amount produced.
b. Determine the heating (or cooling) load during the isothermal operation.
c. Estimate the average HTN for the isothermal operation.
d. Derive the reaction, temperature, and species curves for adiabatic operation.
Determine the operating time needed to maximize the production of product
B, and the amount produced.
e. Examine the effect of HTN on the reactor temperature, the two reactions, and
the production of product B.
Data:At478C,
k
1
¼ 0:04 min
1
k
2
¼ 0:03 min
1
DH
R1
(T
0
) ¼ 40 kcal=mol B DH
R2
(T
0
) ¼ 60 kcal=mol B
E
a1
¼ 7000 cal=mol E
a2
¼ 6000 cal=mol
r ¼ 1:0kg=L
c
p
¼ 1:0 kcal=kgK
Solution The stoichiometric coefficients of the chemical reactions are:
s
A
1
¼1 s
B
1
¼ 2 s
C
1
¼ 0 s
D
1
¼ 0 D
1
¼ 1
s
A
2
¼ 0 s
B2
¼1 s
C
2
¼ 2 s
D
2
¼ 1 D
2
¼ 2
The two reactions are independent, and there are no dependent reactions. We
select the initial state as the reference state; hence, T
0
¼ T(0) ¼ 320 K,
C
0
¼ C
A
(0) ¼ 0.8 mol/L, and the total molar content of the reference state is
(N
tot
)
0
¼ V
R
0
C
0
¼ 160 mol (a)
The initial composition is y
A
(0) ¼ 1.0, and y
B
(0) ¼ y
C
(0) ¼ y
D
(0) ¼ 0. Using
Eq. 6.1.23, for liquid-phase reactions, the specific molar heat capacity of the
reference state is
^
c
p
0
¼
M
(N
tot
)
0
c
p
¼ 1:25
kcal
mol K
6.4 NONISOTHERMAL OPERATIONS 217

The dimensionless activation energies of the two reactions are
g
1
¼
E
a
1
RT
0
¼ 14:15 g
2
¼
E
a
2
RT
0
¼ 9:44
The heat of reaction of Reaction 1 (for the chemical formula used) is
DH
R
1
(T
0
) ¼ 40
kcal
mol B
2
mol B
mol extent
¼ 80 kcal=mol extent
The dimensionless heat of reactions of the two chemical reactions are
DHR
1
¼
DH
R
1
(T
0
)
^
c
p
0
T
0
¼ 0:20 DHR
2
¼
DH
R
2
(T
0
)
^
c
p
0
T
0
¼ 0:15
For liquid-phase reactions (assuming constant heat capacity), CF(Z
m
, u) ¼ 1. We
write Eq. 6.1.1 for each independent reaction,
dZ
1
dt
¼ r
1
t
cr
C
0
(a)
dZ
2
dt
¼ r
2
t
cr
C
0
(b)
Using Eq. 6.1.12 to express the species concentrations, the reaction rates are
r
1
¼ k
1
(T
0
)C
0
(1 Z
1
)e
g
1
(u1)=u
(c)
r
2
¼ k
2
(T
0
)C
0
(2Z
1
Z
2
)e
g
2
(u1)=u
(d)
We select Reaction 1 as the leading reaction, for first-order reaction, the charac-
teristic reaction time is
t
cr
¼
1
k
1
(T
0
)
¼ 25 min (e)
Substituting (c), (d), and (e) into (a) and (b), the design equations become
dZ
1
dt
¼ (1 Z
1
)e
g
1
(u1)=u
(f)
dZ
2
dt
¼
k
2
(T
0
)
k
1
(T
0
)
(2Z
1
Z
2
)e
g
2
(u1)=u
(g)
218 IDEAL BATCH REACTOR

Using Eq. 6.1.17, the energy balance equation is
du
dt
¼
1
CF(Z, u)
HTN(u
F
u) DHR
1
dZ
1
dt
DHR
2
dZ
2
dt
(h)
We have to solve (f), (g), and (h) simultaneously, subject to the initial conditions
that at t ¼ 0, Z
1
(0) ¼ Z
2
(0) ¼ 0, and u(0) ¼ 1. Once we solve the design
equation, we use Eq. 2.7.4 to obtain the species curves:
N
A
(t)
(N
tot
)
0
¼ 1 Z
1
(t) (i)
N
B
(t)
(N
tot
)
0
¼ 2Z
1
(t) Z
2
(t)(j)
N
C
(t)
(N
tot
)
0
¼ 2Z
2
(t) (k)
N
D
(t)
(N
tot
)
0
¼ Z
2
(t) (l)
a. For isothermal operation, u ¼ 1, and (f) and (g) reduce to
dZ
1
dt
¼ (1 Z
1
) (m)
dZ
2
dt
¼
k
2
(T
0
)
k
1
(T
0
)
(2Z
1
Z
2
) (n)
We solve (m) and (n) numerically subject to the initial condition
Z
1
(0) ¼ Z
2
(0) ¼ 0. Figure E6.12.1 shows the reaction curves. We then use
(i) through (l) to determine the species curves, shown in Figure E6.12.2.
From the curve of product B, the highest N
B
/(N
tot
)
0
is 0.8437 and is reached
Figure E6.12.1 Reaction operating curves—isothermal operation.
6.4 NONISOTHERMAL OPERATIONS 219
