Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


where
(TERM) ¼ HTN 1
_
mv
0
2
A
2
F
0
^
c
p
0
T
0
1 þ
X
n
I
m
D
m
Z
m
!
2
u
P
0
P
2
2
4
3
5
(u
F
u)
2f
_
mv
0
3
t
cr
DA
3
F
0
^
c
p
0
T
0
1 þ
X
n
I
m
D
m
Z
m
!
2
u
2
P
0
P
2
X
n
I
m
DHR
m
1
_
mv
0
2
A
2
F
0
RT
0
1 þ
X
n
I
m
D
m
Z
m
!
u
P
0
P
2
"#
dZ
m
dt
_
mv
0
2
A
2
F
0
^
c
p
0
T
0
1 þ
X
n
I
m
D
m
Z
m
!
u
2
P
0
P
2
X
n
I
m
D
m
dZ
m
dt
and
(Denominator) ¼ CF(Z
m
, u)1
_
mv
0
2
A
2
F
0
RT
0
1þ
X
n
I
m
D
m
Z
m
!
u
P
0
P
2
"#
þ
_
mv
0
2
A
2
F
0
^
c
p
0
T
0
1þ
X
n
I
m
D
m
Z
m
!
2
u
P
0
P
2
Equation 7.5.16 is the dimensionless, differential energy balance equation for
cylindrical tubular flow reactors, relating the temperature, u, to the extents of the
independent reactions, Z
m
’s, and P/P
0
as functions of space time t. To design a
plug-flow reactor, we have to solve design equations (Eq. 7.1.1), the energy balance
equation (Eq. 7.5.16), and the momentum balance (Eq. 7.5.12), simultaneously
subject to specified initial conditions.
Note that Eq. 7.5.16 was derived from the energy balance equation (first law of
thermodynamics), and it does not impose a limit on the value of u. However, the
second law of thermodynamics imposes a restriction on the conversion of thermal
energy to kinetic energy. For compressible fluids in tubes with uniform cross-sec-
tional area, the velocity cannot exceed the sound velocity; hence,
u
1
MW
^
c
p
RT
^
c
p
R
0:5
(7:5:17)
Using Eq. 7.5.14, the second law imposes the following restriction
P
P
0
v
0
A
2
MW
0
RT
0
CF(Z
m
, u)
^
c
p
0
CF(Z
m
, u)
^
c
p
0
R
1 þ
X
n
I
m
D
m
Z
m
!
u
"#
0:5
(7:5:18)
300 PLUG-FLOW REACTOR

For flow through a packed-bed reactor, the pressure drop is expressed in terms of
the Ergun equation:
dP
dL
¼ 150
(1
1
)
2
1
3
mu
(fd
p
)
2
þ 1:75
(1
1
)
1
3
ru
2
fd
p
(7:5:19)
where d
p
is the particle diameter, f is the shape factor, and
1
is the bed void frac-
tion. Noting that ru ¼ G, the mass velocity, is constant, and, from Eq. 7.1.3, dL ¼
(v
0
t
cr
/A) dt, Eq. 7.5.19 reduces to
d(P=P
0
)
dt
¼
v
2
0
t
cr
AP
0
150
(1
1
)
2
1
3
m
(fd
p
)
2
þ1:75
(1
1
)G
1
3
fd
p
1þ
X
n
I
m
D
m
Z
m
!
u
P
0
P
(7:5:20)
Equation 7.5.20 provides a relation for the change in the pressure along a packed-
bed reactor. In most applications, the gas velocity in packed-bed reactors is much
lower than the sound velocity. Hence, to formulate the reactor design, we should
solve design equations (Eq. 7.1.1), energy balance equation (Eq. 7.1.16), and
momentum balance equation (Eq. 7.5.20) simultaneously, subject to specified
initial conditions. The solutions provide Z
m
, u, and P/P
0
as functions of dimension-
less space time, t.
Example 7.12 A heavy hydrocarbon feedstock is being cracked in a tubular
reactor placed in a furnace that maintains the wall of the reactor at 980 K. The
cracking is represented by the following first-order chemical reactions:
Reaction 1: A ! B þ C
Reaction 2: B ! 2D
Reaction 3: C ! F þ G
Reaction 4: G ! 2F
The feed stream consists of 90% species A and 10% species I (mole percent), its
temperature is 900 K, its pressure is 5 atm, and it is fed to the reactor at a rate of
276 L/s. The inside diameter of the reactor is 10 cm, and its surface can be
assumed “smooth.” Accounting for pressure drop along the reactor:
a. Derive the reaction and species curves for isothermal operation and deter-
mine the local and average heat-transfer number (HTN).
b. Determine the reaction curves, the temperature curve, the pressure curve, and
the species curves when the average isothermal HTN is maintained in the
reactor.
c. What should be the reactor length to maximize the production of product C?
7.5 EFFECTS OF PRESSURE DROP 301

d. What reactor length should be specified if the pressure drop effect in neg-
lected?
Data: The species molecular weights (in g/mol) are
MW
A
¼ 104 MW
B
¼ 56 MW
C
¼ 48 MW
D
¼ 28
MW
F
¼ 16 MW
G
¼ 32 MW
I
¼ 28
At 900 K the reaction rate constants (in s
21
)are
k
1
¼ 2:0 k
2
¼ 1:0 k
3
¼ 0:3 k
4
¼ 0:2
The activation energies of the chemical reactions rate (in kcal/mol) are
E
a
1
¼ 24:0 E
a
2
¼ 32:0 E
a
3
¼ 45:0 E
a
4
¼ 55:0
At 900 K, the heats of reaction of the chemical reactions (in kcal/mol) are
DH
R
1
¼ 40:0 DH
R
2
¼ 50:0 DH
R
3
¼ 60:0 DH
R
4
¼ 90:0
The specific molar heat capacities of the species (in cal/mol K) are
^
c
pA
¼ 70
^
c
pB
¼ 40
^
c
pC
¼ 50
^
c
pD
¼ 30
^
c
pF
¼ 25
^
c
pG
¼ 40
^
c
pI
¼ 12
The estimated average Reynolds number in the reactor is 10
6
, and the friction
factor is f ¼ 0.005.
Solution The four given reactions are independent, and there are no dependent
reactions. The stoichiometric coefficients of the chemical reactions are
s
A
1
¼1 s
B
1
¼ 1 s
C
1
¼ 1 s
D
1
¼ 0 s
F
1
¼ 0 s
G
1
¼ 0
s
I
1
¼ 0 D
1
¼ 1
s
A
2
¼ 0 s
B
2
¼1 s
C
2
¼ 0 s
D
2
¼ 2 s
F
2
¼ 0 s
G
2
¼ 0
s
I
2
¼ 0 D
2
¼ 1
s
A
3
¼ 0 s
B
3
¼ 0 s
C
3
¼1 s
D
3
¼ 0 s
F
3
¼ 1 s
G
3
¼ 1
s
I
3
¼ 0 D
3
¼ 1
s
A
4
¼ 0 s
B
4
¼ 0 s
C
4
¼ 0 s
D
4
¼ 0 s
F
4
¼ 2 s
G
4
¼1
s
I
4
¼ 0 D
4
¼ 1
302 PLUG-FLOW REACTOR

We select the feed stream as the reference stream, and the reference temperature
is T
0
¼ 900 K. Since the operating temperature is high, we assume ideal-gas
behavior; hence, the reference concentration is
C
0
¼
P
0
RT
0
¼ 0:0677 mol=L
The composition of the reference stream is y
A
0
¼ 0.9 and y
I
0
¼ 0.1. The average
molecular of the reference stream is
MW
0
¼
X
n
I
j
y
j
0
MW
j
¼ 96:4g=mol
The density of the reference stream is
r
0
¼
(F
tot
)
0
MW
0
v
0
¼ 6:52 g=L
Using 5.2.30, the specific molar heat capacity of the reference stream is
^
c
p
0
¼
X
n
I
j
y
j
0
^
c
p
j
¼ 64:2 cal=mol K
Using 5.2.23, the dimensionless heats of reaction are
DHR
1
¼ 0:692 DHR
2
¼ 0:865 DHR
3
¼ 1:038 DHR
4
¼ 1:558
Using 3.3.5, the dimensionless activation energies of the chemical reactions are
g
1
¼
E
a
1
RT
0
¼ 13:42 g
2
¼ 17:89 g
3
¼ 25:16 g
4
¼ 30:76
Using Eq. 7.1.1, the reactor design equations are
dZ
1
dt
¼ r
1
t
cr
C
0
(a)
dZ
2
dt
¼ r
2
t
cr
C
0
(b)
dZ
3
dt
¼ r
3
t
cr
C
0
(c)
dZ
4
dt
¼ r
4
t
cr
C
0
(d)
7.5 EFFECTS OF PRESSURE DROP 303

Using Eq. 7.1.15, for gas-phase reactions, the local concentration of species j is
C
j
¼ C
0
y
j
0
þ
P
n
I
m
(s
j
)
m
Z
m
1 þ
P
n
I
m
D
m
Z
m
u
P
P
0
(e)
The rates of the chemical reactions are
r
1
¼ k
1
(T
0
)C
0
y
A
0
Z
1
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
1
(u1)=u
(f)
r
2
¼ k
2
(T
0
)C
0
Z
1
Z
2
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
2
(u1)=u
(g)
r
3
¼ k
3
(T
0
)C
0
Z
1
Z
3
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
3
(u1)=u
(h)
r
4
¼ k
4
(T
0
)C
0
Z
3
Z
4
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
4
(u1)=u
(i)
We select the characteristic reaction time on the basis of Reaction 1:
t
cr
¼
1
k
1
(T
0
)
¼ 0:5s (j)
Substituting (e) through (j) into (a), (b), (c), and (d), the design equations
reduce to
dZ
1
dt
¼
y
A
0
Z
1
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
1
(u1)=u
(k)
dZ
2
dt
¼
k
2
(T
0
)
k
1
(T
0
)
Z
1
Z
2
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
2
(u1)=u
(l)
dZ
3
dt
¼
k
3
(T
0
)
k
1
(T
0
)
Z
1
Z
3
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
3
(u1)=u
(m)
dZ
4
dt
¼
k
4
(T
0
)
k
1
(T
0
)
Z
3
Z
4
(1 þ Z
1
þ Z
2
þ Z
3
þ Z
4
)u
P
P
0
e
g
4
(u1)=u
(n)
Since u appears in the design equations, we have to use the energy balance
equation to express the changes in temperature. Assuming negligible kinetic
energy and friction work, the energy balance equation is given by Eq. 7.1.16,
du
dt
¼
1
CF(z
m
, u)
HTN(u
F
u)DHR
1
dZ
1
dt
DHR
2
dZ
2
dt
DHR
3
dZ
3
dt
DHR
4
dZ
4
dt
(o)
304 PLUG-FLOW REACTOR

For gas-phase reactions, the local correction factor of the heat capacity of the
reacting fluid is
CF(Z
m
, u) ¼
1
(F
tot
)
0
^
c
p
0
(F
A
^
c
pA
þF
B
^
c
pB
þF
C
^
c
pC
þF
D
^
c
pD
þF
F
^
c
pF
þF
G
^
c
pG
þF
I
^
c
pI
)
(p)
where F
j
is expressed by Eq. 2.7.8. Similarly, since P/P
0
appears in the design
equations, we have to use the momentum balance equation to express the change
in the pressure. Assuming negligible effect due to inertia changes, we use
Eq. 7.5.4,
d(P=P
0
)
dt
¼ 2f
r
0
u
3
0
t
cr
DP
0
(1þZ
1
þZ
2
þZ
3
þZ
4
)u
P
0
P
(q)
where u
0
is the velocity at the reactor inlet:
u
0
¼
4v
0
PD
2
¼37:7m=s
To comply with the second law of thermodynamics, the local velocity in the
reactor cannot exceed the sound velocity. For ideal gas, the sound velocity is
u
s
¼
^
c
p
^
c
p
R
RT
0
u
MW
0:5
(r)
Combining (r) with Eq. 7.5.14, the local pressure should satisfy the following
condition:
P
P
0
u
0
(1þZ
1
þZ
2
þZ
3
þZ
4
)u
^
c
p
^
c
p
R
RT
0
u
MW
0:5
(s)
Note that both c
ˆ
p
and MW vary along the reactor as the reactions proceed, and
they are given by
MW ¼
X
J
j
y
j
MW
j
^
c
p
¼
X
J
j
y
j
^
c
p
j
(u)
7.5 EFFECTS OF PRESSURE DROP 305
where y
j
is the local species molar faction given by
y
j
¼
F
j
F
tot
¼
y
j
0
þ
P
n
I
m
(s
j
)
m
Z
m
1þ
P
n
I
m
D
m
Z
m
To design the reactor, we have to solve (k), (l), (m), (n), simultaneously with (o)
and (q), and verify that (s) is satisfied. We solve these equations numerically,
subject to the initial conditions that at t ¼ 0, Z
1
¼ Z
2
¼ Z
3
¼ Z
4
¼ 0, u ¼ 1,
and P/P
0
¼ 1.
a. For isothermal operation, u ¼ 1, and we use (o) to determine the local
HTN
iso
:
HTN
iso
¼
1
u
F
1
DHR
1
dZ
1
dt
þDHR
2
dZ
2
dt
þDHR
3
dZ
3
dt
þDHR
4
dZ
4
dt
(t)
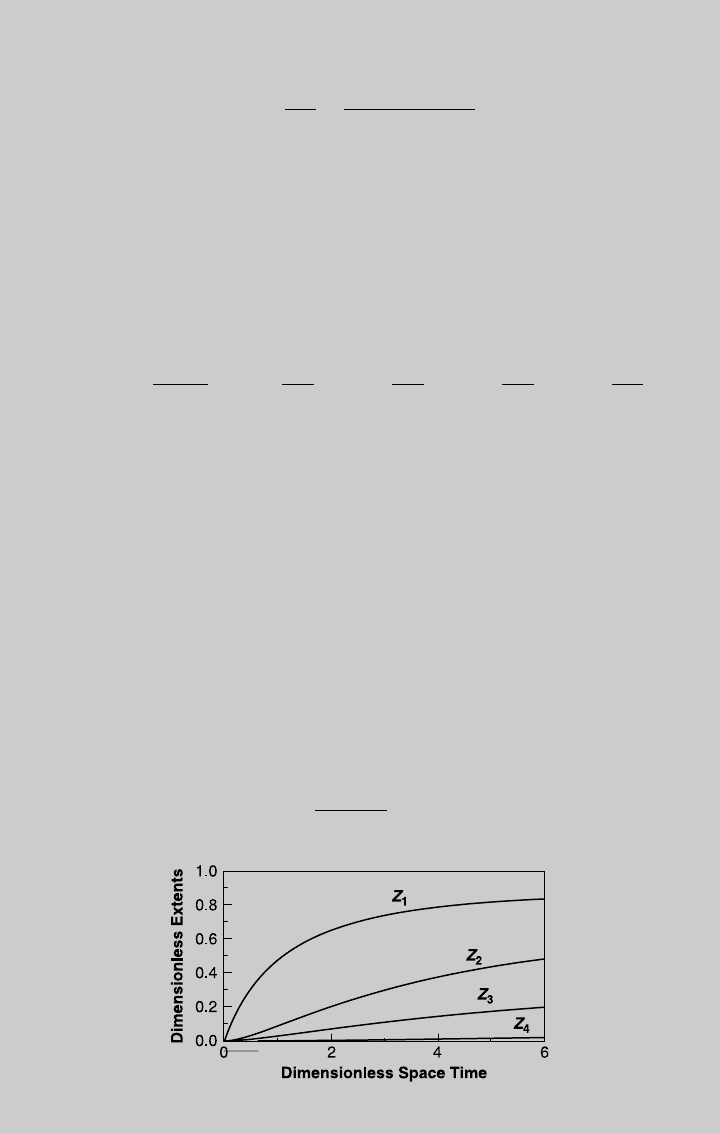
Figure E7.12.1 shows the reaction curves, Figure E7.12.2 shows the pressure
curve, and Figure E7.12.3 shows the HTN curve for this case. The average
isothermal HTN is 5.6.
b. Now that the HTN is known (HTN
ave
¼ 5.6), we solve (k), (l), (m), and (n)
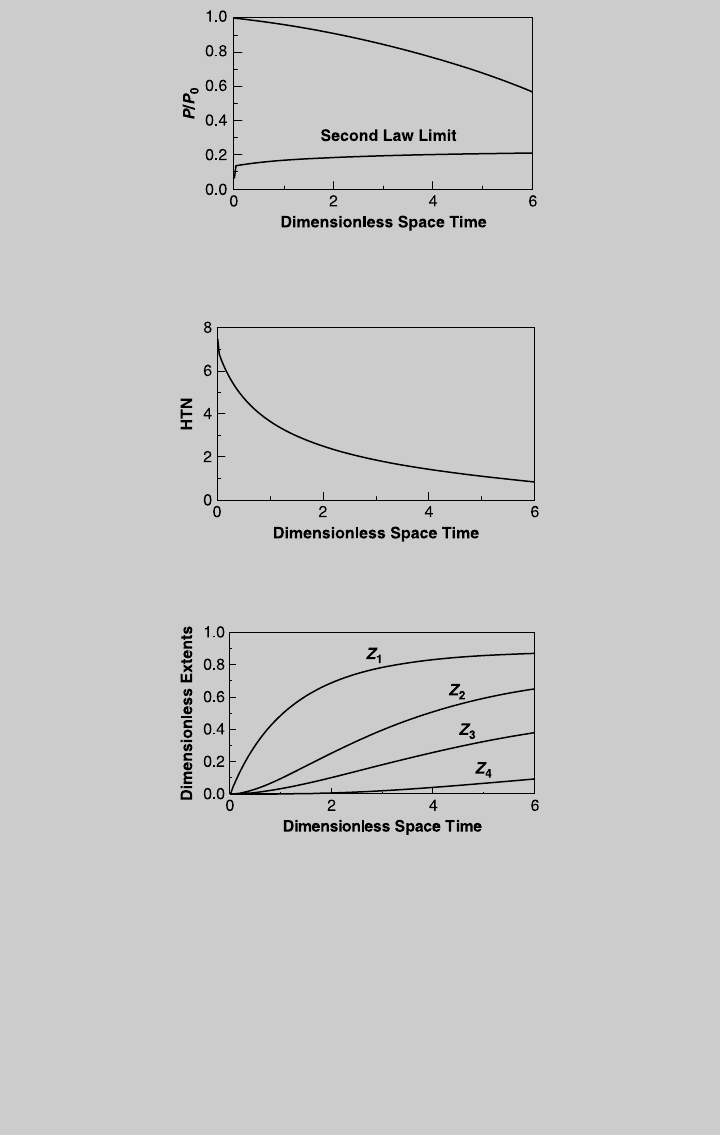
simultaneously with (o) and (q). Figure E7.12.4 shows the actual reaction
curves, Figure E7.12.5 shows the temperature curve, Figure E7.12.6 shows
the species curves, and Figure E7.12.7 shows the pressure curve. Note that
for the specified HTN, the temperature increases along the reactor except
for a short section near the inlet. Also note that, at any point, the pressure
is well above the limit imposed by the second law; hence, the assumption
of negligible kinetic energy is justified. c. From Figure E7.12.6, the highest
production of product C is achieved at t ¼ 2.73, with F
C
=(F
tot
)
0
¼0:6038, or
F
C
¼ 12.1 mol/s. The length of the reactor
L ¼
4(tv
0
t
cr
)
pD
2
¼51:4m
Figure E7.12.1 Reaction operating curves—isothermal operation.
306 PLUG-FLOW REACTOR
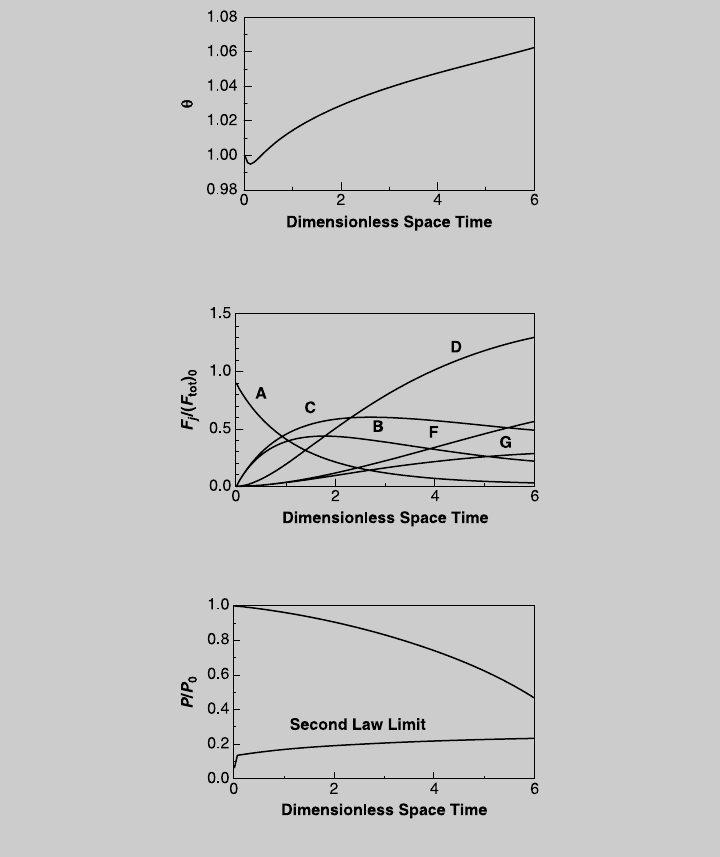
d. To examine the effect of the pressure drop on the reactor performance, we
carry out the design while neglecting the pressure drop. We solve ( j), (k),
(l), and (m) simultaneously with (o), and d(P/P
0
)/dt ¼ 0. From the curve
of product C (not shown here), we determine that the highest production is
achieved at t ¼ 2.29, with F
C
/(F
tot
)
0
¼ 0.5917, or F
C
¼ 11.86 mol/s.
From Figure E7.12.6 (the design formulation with pressure drop effect),
at t ¼ 2.29, F
C
/(F
tot
)
0
¼ 0.598, or F
C
¼ 11.98 mol/s. Hence, in this
case, the error due to neglecting the effect of pressure drop is only 1%.
Figure E7.12.3 HTN curve—isothermal operation.
Figure E7.12.4 Reaction operating curves—actual operation.
Figure E7.12.2 Pressure curve—isothermal operation.
7.5 EFFECTS OF PRESSURE DROP 307
7.6 SUMMARY
In this chapter, we analyzed the operation of plug-flow reactors. We covered the
following topics:
1. The underlying assumptions of the plug-flow reactor model and when they
are satisfied in practice
2. The design equations, the energy balance equation, and the auxiliary relations
for species concentrations
Figure E7.12.5 Temperature curve—actual operation.
Figure E7.12.6 Species operating curves—actual operation.
Figure E7.12.7 Pressure curve—actual operation.
308 PLUG-FLOW REACTOR
3. The reaction operating curves and species operating curves
4. Design and operation of isothermal reactors with single reactions
5. Method to determine the rate expression and its parameters
6. Design and operation of isothermal reactors with multiple reactions
7. Described a procedure to estimate the range of the HTN
8. Design and operation of nonisothermal reactors with multiple reactions
9. Design and operation of gas-phase plug-flow reactors with multiple reactions
where the pressure drop along the reactor is not negligible
PROBLEMS*
7.1
2
At 6508C, phosphine vapor decomposes according to the first-order,
chemical reaction
4PH
3
! P
4
(g) þ 6H
2
Phosphine is fed into a plug-flow reactor at a rate of 10 mol/h in a stream
which consists of 2/3 phosphine and 1/3 inert. The reactor is operated at
6508C and 11.4 atm. At 6508C, k ¼ 10 hr
21
.
a. Derive and solve the design equation.
b. Plot the reaction and species operating curves.
c. Determine the needed reactor volumes for 75% and 90% conversion.
7.2
2
An aqueous feed containing Reactants A and B (C
A
0
¼ 0:1 mol=L,
C
B
0
¼ 0:2 mol=L) is fed at a rate of 400 L/min into a plug-flow reactor,
where the reaction
A þ B ! R
takes place. The rate expression of the reaction is r ¼ 200C
A
C
B
mol/L min.
a. Derive the design equation and plot the reaction curve.
b. Derive and plot the species curves.
c. Determine the needed reactor volumes for 90%, 99%, and 99.9%
conversion.
7.3
2
The homogeneous, gas-phase chemical reaction
A ! 3R
*
Subscript 1 indicates simple problems that require application of equations provided in the text.
Subscript 2 indicates problems whose solutions require some more in-depth analysis and modifi-
cations of given equations. Subscript 3 indicates problems whose solutions require more comprehen-
sive analysis and involve application of several concepts. Subscript 4 indicates problems that require
the use of a mathematical software or the writing of a computer code to obtain numerical solutions.
PROBLEMS 309
