Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


follows second-order kinetics. For a feed rate of 4 m
3
/h of Reactant A at
5 atm and 3508C, an experimental reactor (2.5 cm ID pipe 2 m long)
gives 60% conversion. You are assigned to design a commercial plant to
treat 320 m
3
/h of feed consisting of 50% A, 50% inert at 35 atm, and
3508C to obtain 80% conversion. Assume plug-flow, negligible pressure
drop, and ideal gas behavior.
a. How many 5-m length of 20 cm ID pipes are required?
b. Should they be placed in parallel or in series?
7.4
2
Formic acid is fed at a rate of 8 mol/h into a 10-L tubular reactor operated
at 1508C and 1 atm. The acid decomposes according to the first-order,
chemical reaction
HCOOH ! H
2
O þ CO
At 1508C, k ¼ 2.46 min
21
. Assume ideal-gas behavior.
a. Derive the design equation.
b. Plot the reaction curve and the species curves.
c. Determine the conversion of the formic acid.
7.5
4
Methanol is produced by the gas-phase reaction
CO þ 2H
2
! CH
3
OH
However, at the reactor operating conditions, the undesirable reaction
CO þ 3H
2
! CH
4
þ H
2
O
is also taking place. Both reactions are second-order (each is first-order with
respect to each reactant) and k
2
/k
1
¼ 1.2. A synthesis gas stream is fed into
a plug-flow reactor operated at 4508C and 5 atm. Plot the reaction and
species curves for isothermal operation with a feed consisting of 50%
CO and 50% H
2
(mole basis).
7.6
4
Below is a simplified kinetic model of the cracking of propane to produce
ethylene
C
3
H
8
! C
2
H
4
þ CH
4
r
1
¼k
1
C
C
3
H
8
C
3
H
8
C
3
H
6
þH
2
r
2
¼k
2
C
C
3
H
8
r
2
¼
k
2
K
2
C
C
3
H
6
C
H
2
C
3
H
8
þC
2
H
4
! C
2
H
6
þC
3
H
6
r
3
¼k
3
C
C
3
H
8
C
C
2
H
4
2C
3
H
6
! 3C
2
H
4
r
4
¼k
4
C
C
3
H
6
C
3
H
6
C
2
H
2
þCH
4
r
5
¼k
5
C
C
3
H
8
r
5
¼
k
5
K
5
C
C
2
H
2
C
C
H
4
C
2
H
4
þC
2
H
2
! C
4
H
6
r
6
¼k
6
C
C
2
H
4
C
C
2
H
2
310 PLUG-FLOW REACTOR
At 8008C, k
1
¼ 2.341 s
21
, k
2
¼ 2.12 s
21
, K
2
¼ 1000, k
3
¼ 23.63 m
3
/
kmol s, k
4
¼ 0.816 s
21
, k
5
¼ 0.305 s
21
, K
5
¼ 2000, and k
6
¼ 4.06 10
3
m
3
/kmol s. Design an isothermal plug-flow reactor for cracking of propane
to be operated at 2 atm and 8008C. Derive and plot the reaction and species
curves. Assume propane is fed to the reactor.
7.7
4
The liquid-phase chemical reactions
Reactions 1 & 2: A þ B
C
Reaction 3: C þ B ! D
take place in a plug-flow reactor, operating isothermally at 908C. A 200
L/min stream with C
A
¼ 2 mol/L, C
B
¼ 2 mol/L is to be processed in
the reactor. Based on the data below, derive and plot the reaction and
species operating curves.
a. Derive and solve the design equations and plot the reaction and species
curves.
b. Determine the reactor volume needed for 70% conversion of reactant B.
c. Determine the production rates of species C and D for f
B
¼ 0.7.
d. If we want to maximize the production of C, what should be the reactor
volume?
e. What is the maximum yield of C?
Data: The rate expressions of the chemical reactions are:
r
1
¼ k
1
C
A
C
B
r
2
¼ k
2
C
C
r
3
¼ k
3
C
C
C
B
At 908C, k
1
¼ 3 L mol
1
min
1
, k
2
¼ 0:5 min
1
k
3
¼ 1 L mol
1
min
1
7.8
4
The first-order gas-phase reaction
A ! B þC
takes place in a plug-flow reactor. A stream consisting of 90% A and 10% I
(% mole) is fed into a 200 L reactor at a rate of 50 L/s. The feed is at 731 K
and 3 atm. Based on the data below, derive the reaction and species curves
and the temperature curve for each of the operations below. Determine:
a. Determine the conversion of A when the reactor is operated
isothermally.
b. Determine the heating load in (a).
c. Determine the local HTN and average isothermal HTN.
d. Determine the conversion of A and the outlet temperature when the
reactor is operated adiabatically.
PROBLEMS 311
e. Determine the conversion of A and the outlet temperature when HTN is
25% of the value estimated in (c).
Data: At 731 K, k ¼ 0:2s
1
, E
a
¼ 12,000 cal=mol
DH
R
¼10,000 cal=mol extent
^
c
p
A
¼ 25 cal=mol K
^
c
p
B
¼ 15 cal=mol K
^
c
p
C
¼ 18 cal=mol K
^
c
p
I
¼ 9 cal=mol K
7.9
4
The elementary liquid-phase reactions
A þ B ! C
C þ B ! D
take place in a plug-flow reactor with a diameter of 10 cm. A solution (with
C
A
¼ 2 mol/L, C
B
¼ 2 mol/L) at 808C is fed into the reactor at a rate of
200 L/min.
a. Determine the length of the reactor for maximum production of product
C if the reactor is operated isothermally.
b. Determine the heating/cooling load in (a).
c. Determine the local and average isothermal HTN.
d. Determine the length of the reactor for maximum production of product
C if the reactor is operated adiabatically.
e. Plot the temperature profile along the reactor in (d).
f. Determine the length of the reactor for maximum production of product
C if the shell of the reactor is maintained at 808C, and the HTN is half of
the value estimated in (c).
g. Plot the temperature profile along the reactor in (f).
Data:At808C,
k
1
¼ 0:1 L mol
1
min
1
E
a
1
¼ 12,000 cal =mol
k
2
¼ 0:2 L mol
1
min
1
E
a
2
¼ 16,000 cal=mol
DH
R
1
¼15,000cal=mol extent DH
R
2
¼10,000cal=mol extent
Density ¼ 900 g/L Heat capacity ¼ 0.8cal/g8C
7.10
4
The irreversible gas-phase reactions (both are first-order)
A ! 2V
V ! 2W
Take place in a tubular reactor (plug-flow). A stream of species A is fed into
a 10-cm ID reactor at a rate of 20 L/min. The feed is at 3 atm and 731 K
(C
A
¼ 0.05 mol/L). Based on the data below,
312 PLUG-FLOW REACTOR
a. Determine the length of an isothermal reactor to maximize the pro-
duction of Product V. What is the maximum production rate of
Product V?
b. Determine the conversion of Reactant A and the yield of Product V
in (a).
c. Determine the heating load in (a), and estimate the average isothermal
HTN.
d. Determine the length of an adiabatic reactor to maximize the production
of Product V.
e. What is the maximum production rate of V in (d)?
f. Determine the length of a reactor whose wall temperature is 750 K if the
HTN is 25% the value of the isothermal, that provides maximum pro-
duction rate of Product V. What is the production rate of Product V?
Data: At 731 K, k
1
¼ 2 min
1
, k
2
¼ 0:5 min
1
E
a
1
¼ 8000 cal=mol E
a
2
¼ 12,000 cal=mol
DH
R
1
¼ 3000 cal=mol of V DH
R
2
¼ 4,500 cal=mol of W
^
c
p
A
¼ 65 cal=mol K
^
c
p
V
¼ 40 cal=mol K
^
c
p
W
¼ 25 cal=mol K
7.11
4
Cracking of naphtha cut to produce olefins is a common process in the
petrochemical industry. The cracking reactions are represented by the
simplified elementary gas-phase reactions:
C
10
H
22
! C
4
H
10
þ C
6
H
12
C
4
H
10
! C
3
H
6
þ CH
4
C
6
H
12
! C
2
H
4
þ C
4
H
8
C
4
H
8
! 2C
2
H
4
The reactions take place in a 90 m long 10 cm ID tubular reactor (plug flow)
placed in a furnace chamber. The reactor wall is maintained at 8008C. A gas
mixture at T ¼ 7008C and P ¼ 5 atm, consisting of 90% naphtha (C
10
H
22
)
and 10% I, is fed into the reactor at a rate of 100 L/s. Based on the data
below:
a. Estimate the HTN for isothermal operation at T ¼ 7008C.
b. Plot the reaction and species curves and the temperature profile for
“actual” isothermal operation.
c. What should the reactor length be to optimize the production rate of
butane?
d. Determine the production rate of ethylene and propylene in (c).
e. Determine the heating load of the reactor in (c).
f. Estimate the needed value of the heat-transfer coefficient.
PROBLEMS 313
Data: r
1
¼ k
1
C
C
10
H
22
at 7008C, k
1
¼ 2:0s
1
E
a
1
¼ 25,000 cal=mol
r
2
¼k
2
C
C
4
H
10
at 7008C, k
2
¼0:5s
1
E
a
2
¼35,000 cal=mol
r
3
¼k
3
C
C
6
H
12
at 7008C, k
3
¼0:01s
1
E
a
3
¼40,000 cal=mol
r
4
¼k
4
C
C
4
H
8
at 7008C, k
4
¼0:001s
1
E
a
4
¼45,000 cal=mol
DH
R
1
(T
0
) ¼20,000 cal=mol extent DH
R
2
(T
0
) ¼35,000 cal=mol extent
DH
R
3
(T
0
) ¼45,000 cal=mol extent DH
R
4
(T
0
) ¼55,000 cal=mol extent
^
c
p
C
10
H
22
¼280 cal=mol8C
^
c
p
C
6
H
12
¼180 cal=mol 8 C
^
c
p
CH
4
¼20 cal=mol8C
^
c
p
C
4
H
10
¼150 cal=mol8 C
^
c
p
C
4
H
8
¼140 cal=mol8C
^
c
p
I
¼10 cal=mol8C
^
c
p
C
3
H
6
¼120 cal=mol8C
^
c
p
C
2
H
4
¼30 cal=mol8 C
7.12
4
The first-order gas-phase reaction
A ! B þC
takes place in a plug-flow reactor. A stream consisting of species 90% A
and 10% I (% mole) is fed into a 200-L reactor at a rate of 20 L/s. The
feed is at 731 K and 3 atm. Based on the data below, calculate:
a. Determine the conversion of Reactant A when the reactor is operated
isothermally.
b. Determine the heating/cooling load in (a).
c. Determine the local HTN and estimate the average HTN for isothermal
operation (T
F
¼ 710 K).
d. Determine the conversion of reactant A and the outlet temperature when
the reactor is operated adiabatically.
e. Determine the conversion of reactant A if HTN is 25% of the isothermal
HTN, and the temperature of the cooling fluid is 710 K.
Data: At 731 K, k ¼ 0:2s
1
, E
a
¼ 12,000 cal=mol
DH
R
¼10,000 cal=mol extent
^
c
p
A
¼ 25 cal=mol8K
^
c
p
B
¼15 cal=mol8K
^
c
p
C
¼18 cal=mol8K
^
c
p
I
¼9 cal=mol8K
7.13
2
The second-order, gas-phase reaction
A ! B þC
is carried out in a cascade of two tubular reactors connected in series.
Reactant A is fed at a rate of 100 mol/h into the first reactor, whose
volume is 1000 L. The molar flow rate of reactant A at the exit of the
first reactor is 60 mol/h, and its flow rate at the exit of the second reactor
314 PLUG-FLOW REACTOR
is 20 mol/h. The temperature throughout the system is 1508C, and the
pressure is 2 atm. Determine the volume of the second reactor by:
a. Taking the inlet stream to the first reactor as the reference stream.
b. Taking the inlet stream to the second reactor as the reference stream.
7.14
4
The elementary liquid-phase reactions
A þ B ! C
C þ B ! D
D þ B ! E
take place in a 0.1-m ID tubular reactor. A solution (C
A
¼ C
B
¼ 2 mol/L)
at 808C is fed into the reactor at a rate of 100 L/min. Based on the data
below, derive the reaction operating curves and the temperature curve for
each of the operations below.
a. Determine the reactor length needed to maximize the production of
Product C for isothermal operation at 80 8 C.
b. Determine the heating load on the reactor in (a).
c. Determine the local HTN, and estimate the average HTN for isothermal
operation (T
F
¼ 658C).
d. Determine the reactor length needed to maximize production of product
C for adiabatic operation. What is the outlet temperature?
e. Determine the reactor length needed to maximize the production of
product C with HTN 25% of average isothermal HTN, and T
F
¼ 658C.
Data:At808C,
k
1
¼ 0:1 L mol
1
min
1
E
a
1
¼ 6000 cal=mol
k
2
¼ 0:2 L mol
1
min
1
E
a
2
¼ 8000 cal=mol
k
3
¼ 0:3 L mol
1
min
1
E
a
3
¼ 10,000 cal =mol
DH
R
1
¼15,000 cal=mol extent DH
R
2
¼10,000 cal=mol extent
DH
R
3
¼8000 cal=mol extent
Density of the solution ¼ 1000 g/L
Heat Capacity of the solution ¼ 1 cal/g8C.
7.15
4
The elementary gas-phase reversible chemical reactions
Reactions 1 & 2: A þB
C
Reactions 3 & 4: C þ B
D
Reactions 5 & 6: D þ B
E
take place in a tubular reactor. A stream consisting of 50% A and 50%
B (mole %) at 600 K and 5 atm, is fed into the reactor at a rate of
PROBLEMS 315
1000 L/min. Based on the data below, derive the reaction operating curves
and the temperature curve for each of the operations below.
a. Determine the reactor volume needed to maximize the production of
product C for isothermal operation at 600 K.
b. Determine the heating load on the reactor in (a).
c. Determine the local HTN, and estimate the average HTN for isothermal
operation (T
F
¼ 580 K).
d. Determine the reactor volume needed to maximize production of
Product C for adiabatic operation. What is the outlet temperature?
e. Determine the reactor length needed to maximize the production of
Product C with HTN 25% of average isothermal HTN, and T
F
¼ 580 K.
Data: At 600 K, k
1
¼ 100; k
3
¼ 200; k
5
¼ 300 L mol
1
min
1
k
2
¼ 0:3; k
4
¼ 0:5; k
6
¼ 0:6 min
1
E
a
1
¼ 6,000 cal=mol E
a
2
¼ 12,000 cal=mol
E
a
3
¼ 8,000 cal=mol E
a
4
¼ 13,000 cal=mol
E
a
5
¼ 10,000 cal=mol E
a
6
¼ 14,000 cal=mol
DH
R
1
¼6,000 cal=mol extent DH
R
3
¼5,000 cal=mol extent
DH
R
5
¼4,000 cal=mol extent
^
c
p
A
¼ 25 cal=mol8K
^
c
p
B
¼ 10 cal=mol8K
^
c
p
C
¼ 30 cal=mol8K
^
c
p
D
¼ 35 cal=mol8K
^
c
p
E
¼ 40 cal=mol8K
316 PLUG-FLOW REACTOR
8
CONTINUOUS STIRRED- TANK
REACTOR
The continuous stirred-tank reactor (CSTR) is a mathematical model that describes
an important class of continuous reactors—continuous, steady, well-agitated tank
reactors. The CSTR model is based on two assumptions:
†
Steady-state operation
†
Uniform conditions (concentrations and temperature) exist throughout the
reactor volume (due to good mixing)
These conditions are readily achieved in small-scale agitated reactors. However, in
large industrial reactors, it is not easy to achieve good mixing, and special care
should be given to the design of the tank and agitator. Furthermore, even when
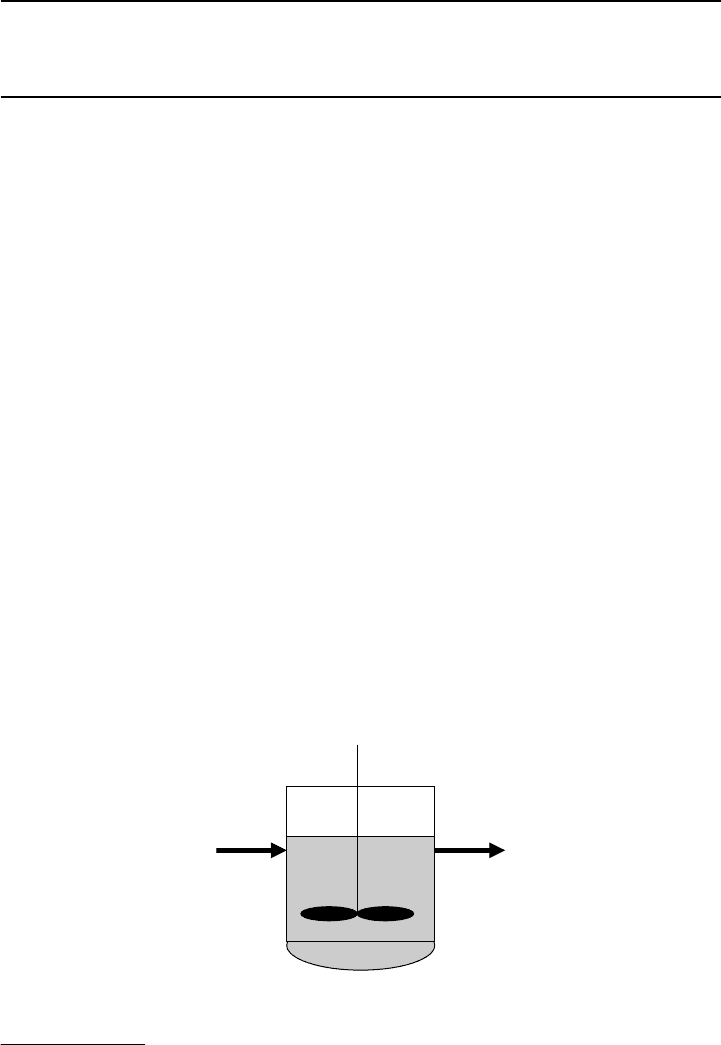
Figure 8.1 Schematic description liquid-phase CS TR.
Principles of Chemical Reactor Analysis and Design, Second Edition. By Uzi Mann
Copyright # 2009 John Wiley & Sons, Inc.
317
the same conditions exist in most sections of the reactor, the conditions near the
reactor inlet and near the reactor wall are different from those in the remainder
of the reactor. Since these zones usually represent a small portion of the reactor,
the CSTR model provides a reasonable description of the well-agitated large reac-
tors. Figure 8.1 shows schematically a liquid-phase CSTR.
8.1 DESIGN EQUATIONS AND AUXILIARY RELATIONS
The design equation of a CSTR was derived in Chapter 4. The design equation,
written for the mth-independent reaction is
Z
m
out
Z
m
in
¼ r
m
out
þ
X
n
D
k
a
km
r
k
out
!
t
t
cr
C
0
(8:1:1)
where Z
m
is the dimensionless extent of the mth-independent reaction, defined by
Eq. 2.7.2:
Z
m
¼
_
X
m
(F
tot
)
0
(8:1:2)
and t is the dimensionless space time of the reactor defined by Eq. 4.4.8:
t ¼
V
R
v
0
t
cr
(8:1:3)
where t
cr
is a conveniently selected characteristic reaction time (defined
by Eq. 3.5.1), and C
0
is a conveniently selected reference concentration
defined by
C
0
¼
(F
tot
)
0
v
0
(8:1:4)
where (F
tot
)
0
and v
0
are, respectively, the total molar flow rate and the volumetric
flow rate of the reference stream.
As discussed in Chapter 4, to describe the operation of a CSTR with multiple
reactions, we have to write Eq. 8.1.1 for each independent chemical reaction.
The solution of the design equations (the relationships between Z
m
out
’s and t )pro-
vide the reaction operating curves and describe the reactor operation. To solve the
design equations, we have to express the rates of the chemical reactions that take
place in the reactor in terms of Z
m
’s and t. Below, we derive the auxiliary relations
used in the design equations.
The volume-based rate expression of the ith chemical reaction (Eq. 3.3.1 and
Eq. 3.3.6) is
r
i
¼ k
i
(T
0
)e
g
i
(u1)=u
h
i
(C
j
,
s) (8:1:5)
318 CONTINUOUS STIRRED-TANK REACTOR

where k
i
(T
0
) is the reaction rate constant at reference temperature T
0
, g
i
is the
dimensionless activation energy (g
i
¼ E
a
i
=RT
0
), and h
i
(C
j
’s) is a function of the
species concentrations, given by the rate expression. For a CSTR, the concentration
of species j is the same everywhere inside the reactor and is equal to the outlet
concentration:
C
j
out
¼
F
j
out
v
out
(8:1:6)
where F
j
out
and v
out
are, respectively, the molar flow rate of species j and the volu-
metric flow rate at the reactor outlet. Using Eq. 2.7.7,
F
j
out
¼ (F
tot
)
0
(F
tot
)
in
(F
tot
)
0
y
j
in
þ
X
n
I
m
(s
j
)
m
Z
m
out
"#
and the outlet concentration of species j is
C
j
out
¼
(F
tot
)
0
v
out
(F
tot
)
in
(F
tot
)
0
y
j
in
þ
X
n
I
m
(s
j
)
m
Z
m
out
"#
(8:1:7)
When the inlet stream is selected as the reference stream, Eq. 8.1.7 reduces to
C
j
out
¼
(F
tot
)
0
v
out
y
j
0
þ
X
n
I
m
(s
j
)
m
Z
m
out
"#
(8:1:8)
For liquid-phase reactions, the density of the reacting fluid is assumed to be con-
stant; hence, v
out
¼ v
0
, and Eq. 8.1.8 reduces to
C
j
out
¼ C
0
y
j
0
þ
X
n
I
m
(s
j
)
m
Z
m
out
"#
(8:1:9)
Equation (8.1.9) provides the species concentrations in terms of the extents of the
independent reactions for liquid-phase reactions.
For gas-phase reactions, the volumetric flow rate depends on the total molar flow
rate and the temperature and pressure in the reactor. Assuming ideal-gas behavior,
the volumetric flow rate is
v
out
¼ v
0
(F
tot
)
out
(F
tot
)
0
T
out
T
0
P
0
P
out
(8:1:10)
Using Eq. 2.7.9 to express the total molar flow rate in terms of the extents of the
independent reactions,
F
tot
¼ (F
tot
)
0
(F
tot
)
in
(F
tot
)
0
þ
X
n
I
m
D
m
Z
m
"#
8.1 DESIGN EQUATIONS AND AUXILIARY RELATIONS 319
