Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


Example 7.7 Diels-Alder reactions are organic reactions between a hydro-
carbon with two double bonds and another unsaturated hydrocarbon to form a
cyclic molecule. However, in many cases, the species with the two double
bonds reacts with itself to form an undesired product. A gaseous mixture con-
taining 50% butadiene (B) and 50% acrolein (A) is fed at a rate of 1 mol/min
into a plug-flow reactor, where the following reactions take place:
Reaction 1: A þ B ! P
Reaction 2: 2B ! W
The rate expressions of the chemical reactions are: r
1
¼ k
1
C
A
C
B
and r
2
¼ k
2
C
B
2
.
The reactor operates at 3308C and 2 atm. At these conditions, k
1
¼ 5.86
L/mol min
21
and k
2
¼ 0.72 L/mol min
21
. Assume ideal-gas behavior.
a. Derive the design equations and plot the reaction curves.
b. Derive and plot the species curves.
c. Determine the reactor volume needed to achieve 80% conversion of
reactant B.
Solution The stoichiometric coefficients of the chemical reactions are
s
A
1
¼1 s
B
1
¼1 s
P
1
¼ 1 s
W
1
¼ 0 D
1
¼1
s
A
2
¼ 0 s
B
2
¼2 s
P
2
¼ 0 s
W
2
¼ 1 D
2
¼1
Since each chemical reaction has a species that does not appear in the other, the
two reactions are independent, and there is no dependent reaction in this case.
We select the inlet stream as the reference stream; hence, Z
1
in
¼ Z
2
in
¼ 0:
C
0
¼
P
0
RT
0
¼
2 atm
(82:05 10
3
L atm =mol K)(603 K)
¼ 4:04 10
2
mol=L (a)
v
0
¼
(F
tot
)
0
C
0
¼ 24:75 L=min
The feed composition is y
A
0
¼ 0:5 and y
B
0
¼ 0:5.
a. We write Eq. 7.1.1 for each independent reaction:
dZ
1
dt
¼ r
1
t
cr
C
0
(b)
dZ
2
dt
¼ r
2
t
cr
C
0
(c)
270 PLUG-FLOW REACTOR

We use Eq. 7.1.14 to express the species concentrations, and the reaction
rates are
r
1
¼ k
1
C
2
0
( y
A
0
Z
1
)( y
B
0
Z
1
2Z
2
)
(1 Z
1
Z
2
)
2
(d)
r
2
¼ k
2
C
2
0
y
B
0
Z
1
2Z
2
1 Z
1
Z
2
2
(e)
We select Reaction 1 as the leading reaction; the characteristic reaction
time is
t
cr
¼
1
k
1
C
0
¼ 4:22 min ( f)
Substituting (d), (e), and (f) into (b) and (c), the design equations reduce to
dZ
1
dt
¼
( y
A
0
Z
1
)( y
B
0
Z
1
2Z
2
)
(1 Z
1
Z
2
)
2
(g)
dZ
2
dt
¼
k
2
k
1
y
B
0
Z
1
2Z
2
1 Z
1
Z
2
2
(h)
From the given data, k
2
/k
1
¼ 0.123, and we solve (g) and (h) numerically
subject to the initial conditions that at t ¼ 0, Z
1
¼ Z
2
¼ 0. Figure E7.7.1
shows the reaction curves.
b. Now that the reaction curves are known, we obtain the species curves by
using Eq. 2.7.8:
F
A
(F
tot
)
0
¼ y
A
0
Z
1
(i)
Figure E7.7.1 Reaction operating curves.
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 271

F
B
(F
tot
)
0
¼ y
B
0
Z
1
2Z
2
(j)
F
P
(F
tot
)
0
¼ y
P
0
þ Z
1
(k)
F
W
(F
tot
)
0
¼ y
W
0
þ Z
2
(l)
Figure E7.7.2 shows the species curves.
c. Using Eq. 2.6.2, for 80% conversion of reactant B, F
B
=F
B
0
¼ 0:20 and
F
B
=(F
tot
)
0
¼ 0:10. From the species operating curve of reactant B,
F
B
=(F
tot
)
0
¼ 0:10 is achieved at t ¼ 2.80. Using Eq. 7.1.3 and (f), the
required reactor volume is
V
R
¼ tv
0
t
cr
¼ 292:7 L (m)
Example 7.8 The following simplified reversible reactions were proposed for
gas-phase cracking of hydrocarbons:
Reactions 1 & 2: A
2B
Reactions 3 & 4: A þ B
C
Reactions 5 & 6: A þ C
D
Species C is the desired product. We want to design an isothermal, isobaric
tubular reactor (plug-flow reactor) to be operated at 4898C and 5 atm. A stream
of reactant A at a rate of 1 mol/s is available in the plant.
a. Derive the design equations and plot the reaction operating curves.
b. Plot the species operating curves.
Figure E7.7.2 Species operating curves.
272 PLUG-FLOW REACTOR

c. Determine the maximum production rate of product C and the required reac-
tor volume to achieve it.
d. Determine the mole fractions of all the species at equilibrium (t ¼ 1).
e. Repeat parts a, b, and d for a feed stream that consists only of species D.
The reaction rates are:
r
1
¼ k
1
C
A
r
2
¼ k
2
C
2
B
r
3
¼ k
3
C
A
C
B
r
4
¼ k
4
C
C
r
5
¼ k
5
C
A
C
C
r
6
¼ k
6
C
D
Data: At 4898C
k
1
¼ 0:2 min
1
k
2
¼ 0:1L=mol min
1
k
3
¼ 50 L=mol min
1
k
4
¼ 0:8 min
1
k
5
¼ 125 L=mol min
1
k
6
¼ 4 min
1
Solution This is an example of series–parallel reversible reactions. The reactor
design formulation of these chemical reactions was discussed in Example 4.1.
Here, we complete the design for an isothermal plug-flow reactor and obtain
the reaction and species curves. Recall that we select Reactions 1, 3, and 5 as
a set of independent reactions. Hence, the indices of the independent reactions
are m ¼ 1, 3, and 5, the indices of the dependent reactions are k ¼ 2, 4, and
6, and we express the design equations in terms of Z
1
, Z
3
, and Z
5
. The stoichio-
metric coefficients of the three independent reactions are
s
A
1
¼1 s
B
1
¼ 2 s
C
1
¼ 0 s
D
1
¼ 0 D
1
¼1
s
A
3
¼1 s
B
3
¼1 s
C
3
¼ 1 s
D
3
¼ 0 D
3
¼1
s
A
5
¼1 s
B
5
¼ 0 s
C
5
¼1 s
D
5
¼ 1 D
5
¼1
The multipliers of the independent reactions to obtain the dependent reactions
are
a
21
¼1 a
23
¼ 0 a
25
¼ 0
a
41
¼ 0 a
43
¼1 a
45
¼ 0
a
61
¼ 0 a
63
¼ 0 a
65
¼1
We select the inlet stream as the reference stream; hence, Z
1
in
¼ Z
3
in
¼ Z
5
in
¼ 0:
C
0
¼
P
0
RT
0
¼
5
(82:05 10
3
L atm=mol K)(762 K)
¼ 0:08 mol=L (a)
and
v
0
¼
(F
tot
)
0
C
0
¼ 12:5L=s ¼ 750 L=min
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 273

We write Eq. 7.1.1 for each independent reaction:
dZ
1
dt
¼ (r
1
r
2
)
t
cr
C
0
(b)
dZ
3
dt
¼ (r
3
r
4
)
t
cr
C
0
(c)
dZ
5
dt
¼ (r
5
r
6
)
t
cr
C
0
(d)
We use Eq. 7.1.15 to express the species concentrations, and the rates of the six
chemical reactions are
r
1
¼ k
1
C
0
y
A
0
Z
1
Z
3
Z
5
1 þ Z
1
Z
3
Z
5
(e)
r
2
¼ k
2
C
0
2
y
B
0
þ 2Z
1
Z
3
1 þ Z
1
Z
3
Z
5
2
(f)
r
3
¼ k
3
C
0
2
( y
A
0
Z
1
Z
3
Z
5
)( y
B
0
þ 2Z
1
Z
3
)
(1 þ Z
1
Z
3
Z
5
)
2
(g)
r
4
¼ k
4
C
0
y
C
0
þ Z
3
Z
5
1 þ Z
1
Z
3
Z
5
(h)
r
5
¼ k
5
C
0
2
( y
A
0
Z
1
Z
3
Z
5
)( y
C
0
þ Z
3
Z
5
)
(1 þ Z
1
Z
3
Z
5
)
2
(i)
r
6
¼ k
6
C
0
y
D
0
þ Z
5
1 þ Z
1
Z
3
Z
5
(j)
We select Reaction 1 and define the characteristic reaction time by
t
cr
¼
1
k
1
¼ 5 min (k)
Substituting (e) through ( j) and (k) into (b), (c), and (d), the design equations reduce to
dZ
1
dt
¼
y
A
0
Z
1
Z
3
Z
5
1þZ
1
Z
3
Z
5
k
2
C
0
k
1
y
B
0
þ2Z
1
Z
3
1þZ
1
Z
3
Z
5
2
(l)
dZ
3
dt
¼
k
3
C
0
k
1
(y
A
0
Z
1
Z
3
Z
5
)(y
B
0
þ2Z
1
Z
3
)
(1þZ
1
Z
3
Z
5
)
2
k
4
k
1
y
C
0
þZ
3
Z
5
1þZ
1
Z
3
Z
5
(m)
dZ
5
dt
¼
k
5
C
0
k
1
(y
A
0
Z
1
Z
3
Z
5
)(y
C
0
þZ
3
Z
5
)
(1þZ
1
Z
3
Z
5
)
2
k
6
k
1
y
D
0
þZ
5
1þZ
1
Z
3
Z
5
(n)
F or the given kinetic da ta,
k
2
C
0
k
1
¼0:04
k
3
C
0
k
1
¼20
k
4
k
1
¼4
k
5
C
0
k
1
¼50
k
6
k
1
¼20 (o)
274 PLUG-FLOW REACTOR
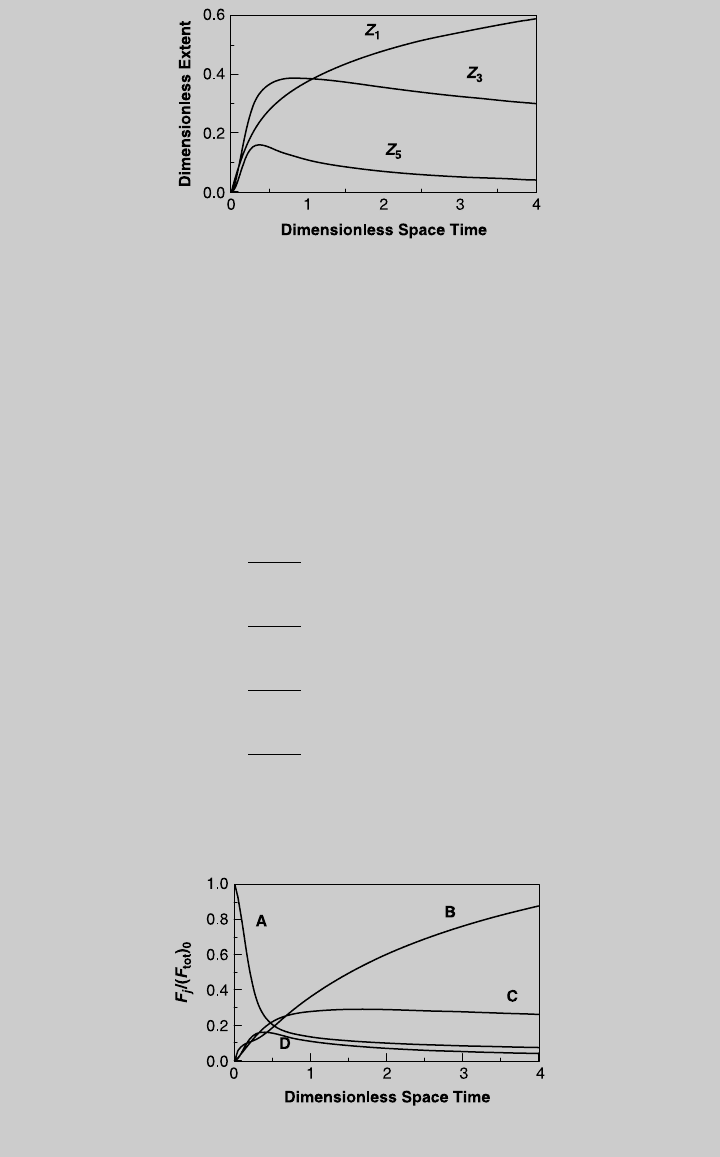
a. When a stream of reactant A is fed into the reactor, y
A
0
¼ 1,
y
B
0
¼ y
C
0
¼ y
D
0
¼ 0. We substitute these values into (l), (m), and (n), and
solve them numerically, subject to the initial conditions that at t ¼ 0,
Z
1
¼ Z
3
¼ Z
5
¼ 0. Figure E7.8.1 shows the solution—the reaction curves
for the three independent chemical reactions.
b. Now that the reaction curves are known, we use Eq. 2.7.8 to obtain the
species curves:
F
A
(F
tot
)
0
¼ y
A
0
Z
1
Z
3
Z
5
(p)
F
B
(F
tot
)
0
¼ y
B
0
þ 2Z
1
Z
3
(q)
F
C
(F
tot
)
0
¼ y
C
0
þ Z
3
Z
5
(r)
F
D
(F
tot
)
0
¼ y
D
0
þ Z
5
(s)
Figure E7.8.2 shows the species curves.
Figure E7.8.1 Reaction operating curves.
Figure E7.8.2 Species operating curves.
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 275

c. From the curve of product C (or tabulated calculated values), the maximum
value of F
C
out
=(F
tot
)
0
is 0.290, and it is reached at t ¼ 1.64. The highest
production rate of product C is
F
C
out
¼ (0:29)(F
tot
)
0
¼ 0:29 mol=s
Using Eq. 7.1.3 and (k), the reactor volume required to maximize the pro-
duction rate of product B is
V
R
¼ tv
0
t
cr
¼ 6150 L
d. We calculate the extents of the independent reactions at equilibrium by equat-
ing (l), (m), and (n) to zero. We obtain a set of nonlinear algebraic equations
whose solutions are
Z
1
eq
¼ 0:2451 Z
3
eq
¼ 0:3693 Z
5
eq
¼ 0:2100
Substituting these values into (p) through (s), we obtain
F
A
eq
(F
tot
)
0
¼ 0:1755
F
B
eq
(F
tot
)
0
¼ 0:1209
F
C
eq
(F
tot
)
0
¼ 0:1593
F
D
eq
(F
tot
)
0
¼ 0:210
Using Eq. 2.7.10, the total molar flow rate at equilibrium,
F
tot
(F
tot
)
0
¼ 1 Z
1
eq
Z
3
eq
Z
5
eq
¼ 0:6657 (t)
and the corresponding mole fractions are
y
A
eq
¼ 0:2636 y
B
eq
¼ 0:1816 y
C
eq
¼ 0:2393 y
D
eq
¼ 0:3155
e. The reactor design calculations when the feed stream consists of species D
proceeds in the same way as in parts a and b. The only difference is that
now y
A
0
¼ y
B
0
¼ y
C
0
¼ 0 and y
D
0
¼ 1. Substituting these values into (l),
(m), and (n), we solve them numerically. Figure E7.8.3 shows the reaction
curves for this case. Note that in this case, the extents of Reactions 3 and
5 are negative since they proceed in the reverse direction. Once we have
the reaction curves, we use (p) through (s) to determine the species curves,
shown in Figure E7.8.4. To determine the equilibrium composition, we
equate (l), (m), and (n) to zero and obtain a set of nonlinear algebraic
equations whose solutions are
Z
1
eq
¼ 0:1128 Z
3
eq
¼0:0766 Z
5
eq
¼0:4749
276 PLUG-FLOW REACTOR
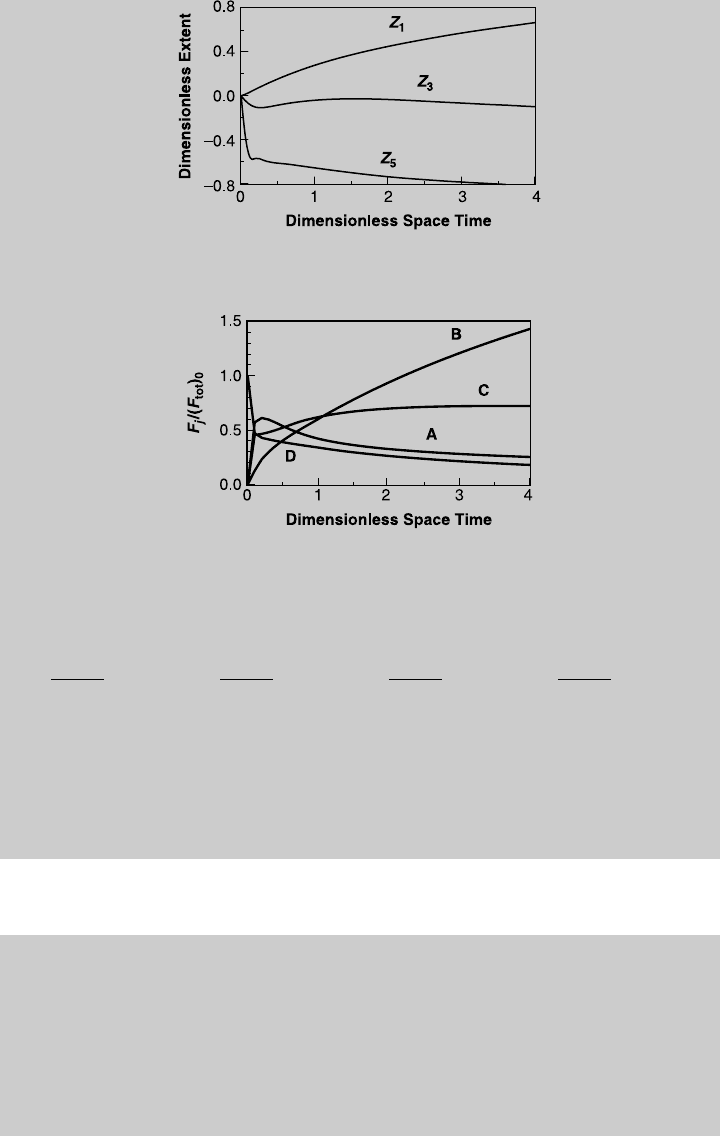
Substituting these values into (p) through (s), we obtain
F
A
eq
(F
tot
)
0
¼ 0:4388
F
B
eq
(F
tot
)
0
¼ 0:3021
F
C
eq
(F
tot
)
0
¼ 0:3986
F
D
eq
(F
tot
)
0
¼ 0:5251
The corresponding mole fractions at equilibrium are
y
A
eq
¼ 0:2637 y
B
eq
¼ 0:1815 y
C
eq
¼ 0:2393 y
D
eq
¼ 0:3155
Note that, as expected, the equilibrium composition is independent of the
feed composition.
Example 7.9 Ammonia oxidation is carried out in an isothermal plug-flow
reactor. The following gas-phase chemical reactions take place in the reactor:
Reaction 1: 4NH
3
þ 5O
2
! 4NO þ 6H
2
O
Reaction 2: 4NH
3
þ 3O
2
! 2N
2
þ 6H
2
O
Reaction 3: 2NO þ O
2
! 2NO
2
Reaction 4: 4NH
3
þ 6NO ! 5N
2
þ 6H
2
O
Figure E7.8.3 Reaction operating curves for feed of species D.
Figure E7.8.4 Species operating curves for feed of species D.
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 277

The desired product is NO. A stream consisting of 50% NH
3
and 50% O
2
(mol%) at 609 K and 2 atm is fed into the reactor at a rate of 240 L/min.
Based on the rate data below:
a. Derive the design equations and plot the reaction and species operating
curves.
b. Determine the volume of the plug-flow reactor for optimal production of NO;
what is the flow rate of each species at the reactor exit?
The rate expressions of the reactions are:
r
1
¼ k
1
C
NH
3
C
O
2
2
r
2
¼ k
2
C
NH
3
C
O
2
r
3
¼ k
3
C
O
2
C
NO
2
r
4
¼ k
4
C
NH
3
2=3
C
NO
Data: At 609 K:
k
1
¼ 20 (L=mol)
2
min
1
k
2
¼ 0:04 (L=mol)min
1
k
3
¼ 40 (L=mol)
2
min
1
k
4
¼ 0:0274 (L=mol)
2=3
min
1
Solution The reactor design formulation of these chemical reactions was dis-
cussed in Example 4.3. Here, we complete the design for an isothermal plug-
flow reactor and obtain the dimensionless reaction and species curves. Recall
that there are three independent reactions and one dependent reaction, and, fol-
lowing the heuristic rule, we select a set of three reactions from the given reac-
tions. We select Reactions 1, 2, and 3 as a set of independent reactions; hence,
m ¼ 1, 2, 3, and k ¼ 4, and we express the design equations in terms of Z
1
, Z
2
,
and Z
3
. The stoichiometric coefficients of the independent reactions are
(s
NH
3
)
1
¼4(s
O
2
)
1
¼5(s
NO
)
1
¼ 4(s
H
2
O
)
1
¼ 6(s
N
2
)
1
¼ 0
(s
NO
2
)
1
¼ 0 D
1
¼ 1
(s
NH
3
)
2
¼4(s
O
2
)
2
¼3(s
NO
)
2
¼ 0(s
H
2
O
)
2
¼ 6(s
N
2
)
2
¼ 2
(s
NO
2
)
2
¼ 0 D
2
¼ 1
(s
NH
3
)
3
¼ 0(s
O
2
)
3
¼1(s
NO
)
3
¼2(s
H
2
O
)
3
¼ 0(s
N
2
)
3
¼ 0
(s
NO
2
)
3
¼ 2 D
3
¼1
Recall from Example 4.3 that the multipliers a
km
’s of the dependent reaction
(Reaction 4) and the three independent reactions are a
43
¼ 0, a
42
¼ 2.5, and
a
41
¼ 21.5. We select the feed stream as the reference stream; hence,
Z
1
in
¼ Z
2
in
¼ Z
3
in
¼ 0, and the reference concentration is
C
0
¼
P
0
RT
0
¼
2
(0:08206 L atm=mol K)(609 K)
¼ 0:04 mol=L (a)
278 PLUG-FLOW REACTOR

The molar flow rate of the reference stream is
(F
tot
)
0
¼ v
0
C
0
¼ 9:6 mol=min
For the selected reference stream, y
NH
3
(0) ¼ 0:5, y
O
2
(0) ¼ 0:5, y
NO
(0)
y
N
2
(0) ¼ y
NO
2
(0) ¼ 0 y
N
2
(0) ¼ y
NO
2
(0) ¼ 0.
a. To design a plug-flow reactor, we write Eq. 7.1.1 for each independent
reaction:
dZ
1
dt
¼ (r
1
1: 5r
4
)
t
cr
C
0
(b)
dZ
2
dt
¼ (r
2
þ 2: 5 r
4
)
t
cr
C
0
(c)
dZ
3
dt
¼ r
3
t
cr
C
0
(d)
We select Reaction 1 as the reference reaction, and define characteristic
reaction time by
t
cr
¼
1
k
1
C
0
2
¼ 31:21 min (e)
We use Eq. 7.1.15 to express the species concentrations, and the rates of the
four chemical reactions are
r
1
¼ k
1
C
0
3
0:5 4Z
1
4Z
2
1 þ Z
1
þ Z
2
Z
3
0:5 5Z
1
3Z
2
Z
3
1 þ Z
1
þ Z
2
Z
3
2
(f)
r
2
¼ k
2
C
0
2
0:5 4Z
1
4Z
2
1 þ Z
1
þ Z
2
Z
3
0:5 5Z
1
3Z
2
Z
3
1 þ Z
1
þ Z
2
Z
3
(g)
r
3
¼ k
3
C
0
3
0:5 5Z
1
3Z
2
Z
3
1 þ Z
1
þ Z
2
Z
3
4Z
1
2Z
3
1 þ Z
1
þ Z
2
Z
3
2
(h)
r
4
¼ k
4
C
5=3
0
0:5 4Z
1
4Z
2
1 þ Z
1
þ Z
2
Z
3
2=3
4Z
1
2Z
3
1 þ Z
1
þ Z
2
Z
3
(i)
7.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 279
