Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


(Z
2
Z
1
)(1 þ Z
2
)
1 Z
2
¼
(Z
3
Z
2
)(1 þ Z
3
)
1 Z
3
(n)
Substituting Z
3
¼ 0.8, we solve (m) and (n) numerically and obtain
Z
1
¼ 0.4445 and Z
2
¼ 0.6716. Now that we know the values of Z
1
and Z
2
,
we use ( j), (k), or (l) to calculate the dimensionless space time of each reac-
tor. We obtain t
1
¼ t
2
¼ t
3
¼ 1.156. To calculate the volume of each reac-
tor, we use Eq. 8.1.3 and (b):
V
R
¼
tv
0
k
¼ 48:15 L
The total volume of the cascade is 144.45 L.
d. To determine Z
1
and Z
2
for an optimized cascade of three CSTRs, we take the
derivative of (t
1
þ t
2
þ t
3
) with respect to Z
1
and Z
2
and equate them to
zero:
@(t
1
þ t
2
þ t
3
)
@Z
1
¼ 0
@(t
1
þ t
2
þ t
3
)
@Z
2
¼ 0 (o)
Substituting ( j), (k), and (l) and taking the derivatives, the two equations in
(o) become
(1 þ 2Z
1
)(1 Z
1
) þ Z
1
(1 þ Z
1
)
1 þ Z
2
1 Z
2
(1 Z
1
)
2
¼ 0(p)
(1 þ 2Z
2
Z
1
)(1 Z
2
) þ (Z
2
Z
1
)(1 þ Z
2
)
1 þ Z
3
1 Z
3
(1 Z
2
)
2
¼ 0 (q)
We substitute Z
3
¼ 0.8, solve (p) and (q) numerically, and obtain
Z
1
¼ 0.4152 and Z
2
¼ 0.6580. Now that we know the values of Z
1
and Z
2
,
we use ( j), (k), and (l) to calculate the dimensionless space time of each reac-
tor. We find that t
1
¼ 1.005, t
2
¼ 1.177, and t
3
¼ 1.278. Using (g), the
volumes of the three reactors are V
R
1
¼ 41:88 L, V
R
2
¼ 49:04 L, and
V
R
3
¼ 53:25 L. The total volume of the cascade is 144.17 L.
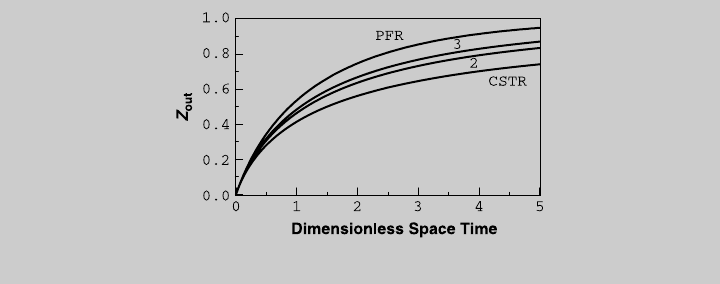
Figure E8.5.1 shows the reaction curve for a cascade of equal-size tanks, and
compare them to those of a plug-flow reactor and a single CSTR.
e. The total volume of the cascade for the different cases is summarized below:
A single CSTR (from Example 8.1) 300.00 L
A cascade of two equal-size CSTRs 172.92 L
An optimized cascade of two CSTRs 172.67 L
A cascade of three equal-size CSTRs 144.45 L
An optimized cascade of three CSTRs 144.17 L
A plug-flow reactor (from Example 7.1) 100.08 L
340 CONTINUOUS STIRRED-TANK REACTOR
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS
When more than one chemical reaction takes place in the reactor, we have to deter-
mine how many independent reactions there are (and how many design equations
are needed) and select a set of independent reactions. Next, we have to identify all
the reactions that actually take place (including dependent reactions) and express
their rates. We write Eq. 8.1.1 for each independent chemical reaction. To solve
the design equations (obtain relationships between Z
m
out
’s and t), we express the
rates of the individual chemical reactions in terms of the Z
m
out
’s and t. Since the
temperature is constant, the energy balance equation is used to determine the heat-
ing load. The procedure for designing isothermal CSTRs with multiple reactions
goes as follows:
1. Identify all the chemical reactions that take place in the reactor and define
the stoichiometric coefficients of each species in each reaction.
2. Determine the number of independent chemical reactions.
3. Select a set of independent reactions among the reactions whose rate
expressions are given.
4. For each dependent reaction, determine its a
km
multipliers with each of the
independent reactions, using Eq. 2.4.9.
5. Select a reference stream [determine (F
tot
)
0
, C
0
, v
0
] and the reference species
compositions, y
j
0
’s.
6. Write Eq. 8.1.1 for each independent chemical reaction.
7. Select a leading (or desirable) reaction and determine the expression form
and value of its characteristic reaction time, t
cr
.
8. Express the reaction rates in terms of the dimensionless extents of the inde-
pendent reactions, Z
m
out
’s.
9. Specify the inlet conditions (Z
m
in
’s).
10. Solve the design equations (determine Z
m
out
’s as functions of t) and obtain
the reaction operating curves.
Figure E8.5.1 Comparison of reaction curves for cascade of equal-size tanks.
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 341

11. Determine the species operating curves, using Eq. 2.7.8.
12. Determine the reactor volume based on the most desirable value of t, using
Eq. 8.1.3.
Below, we describe the design formulation of isothermal CSTRs with multiple
reactions for various types of chemical reactions (reversible, series, parallel, etc.).
In most cases, we solve the equations numerically using mathematical software.
In some simple cases, we obtain analytical solutions.
Example 8.6 The reversible, gas-phase chemical reaction
A
2B
takes place in a CSTR operated at 2 atm and 1208C. The forward reaction is first
order, and the backward reaction is second order. We want to process a
100-mol/min stream of pure A and achieve a level of 80% of the equilibrium
conversion. At the operating conditions (1208C), k
1
¼ 0.1 min
21
and
k
2
¼ 0.322 L/mol min
21
.
a. Derive the design equation and plot the dimensionless reaction operating
curve.
b. What is the equilibrium composition at 1208C in a CSTR for k
2
C
0
/k
1
¼ 0.5?
c. What is the required reactor volume needed to reach 80% of the equilibrium
conversion?
Solution We treat a reversible reaction as two separate reactions; a forward
reaction and a reverse reaction. But there is only one independent reaction.
We select the forward reaction (Reaction 1) as the independent reaction
and the reverse reaction as the dependent reaction. Hence, the index of the
independent reaction is m ¼ 1, the index of the dependent reaction is k ¼ 2,
and, a
21
¼ 21. The stoichiometric coefficients of the independent reaction are
s
A
1
¼1 s
B
1
¼ 2 D
1
¼ 1
We select the inlet stream as the reference stream; hence Z
1
in
¼ 0, and since only
reactant A is fed into the reactor, y
A
0
¼ 1 and y
B
0
¼ 0. The reference concen-
tration is
C
0
¼
P
0
RT
0
¼ 0:0602 mol=L (a)
Using Eq. 8.1.4, the volumetric flow rate of the reference stream is
v
0
¼
(F
tot
)
0
C
0
¼ 1612 L=min (b)
342 CONTINUOUS STIRRED-TANK REACTOR
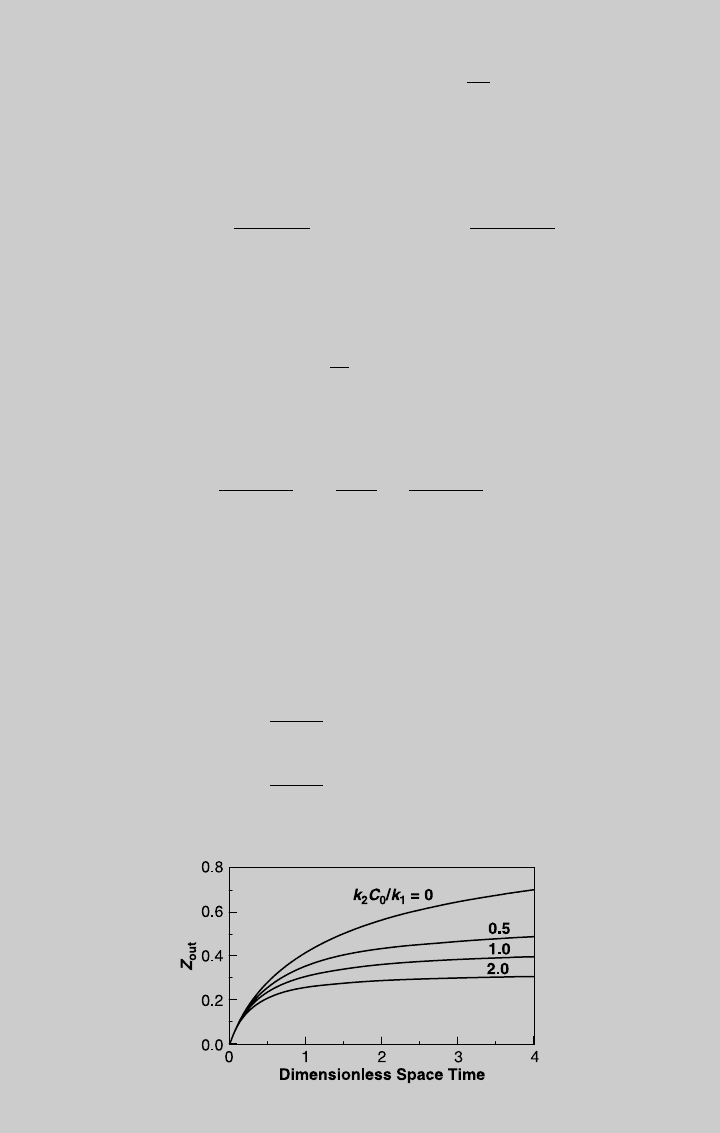
We write Eq. 8.1.1 for the independent reaction:
Z
1
out
Z
1
in
¼ (r
1
out
r
2
out
)t
t
cr
C
0
(c)
We use Eq. 8.1.13 to express the concentrations of the two species, and the rates
of the two reactions are
r
1
¼ k
1
C
0
y
A0
Z
1
1 þ Z
1
r
2
¼ k
2
C
0
2
y
b0
þ 2Z
1
1 þ Z
1
2
(d)
We select Reaction 1 as the leading reaction and using Eq. 3.5.4, define the
characteristic reaction time as
t
cr
¼
1
k
1
¼ 10 min (e)
Substituting (d) and (e) into (c), the design equation reduces to
Z
1
out
1 Z
1
out
1 þ Z
1
out
k
2
C
0
k
1
2Z
1
out
1 þ Z
1
out
2
"#
t ¼ 0(f)
We solve (f) numerically for different values of t using a mathematical
software. Figure E8.6.1 shows the reaction curve for various values of
k
2
C
0
/k
1
. Note that the curve for k
2
C
0
/k
1
¼ 0 represents the solution of the irre-
versible reaction. Once we have the reaction operating curve, we use Eq. 2.7.8 to
obtain the species curves
F
A
out
(F
tot
)
0
¼ y
A
0
Z
out
F
B
out
(F
tot
)
0
¼ y
B
0
þ 2Z
out
Figure E8.6.1 Comparison of reaction operating curves.
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 343

b. At equilibrium, r
1
¼ r
2
, and we obtain
Z
1
eq
¼ 1 þ 4
k
2
C
0
k
1
0:5
(g)
For k
2
C
0
/k
1
¼ 0.5, Z
1
eq
¼ 0:577, and the mole fractions of the species are
y
A
eq
¼
y
A
0
Z
1
eq
1 þ D
1
Z
1
eq
¼ 0:268
y
B
eq
¼
y
B
0
þ 2Z
1
eq
1 þ D
1
Z
1
eq
¼ 0:732
(h)
c. The extent for 80% of the equilibrium extent is 0.4616. From the operating
curve for k
2
C
0
/k
1
¼ 0.5, an extent of 0.4616 is reached at t ¼ 2.74. Using
Eq. 8.1.3 and (e), the required reactor volume is
V
R
¼ v
0
t
cr
t ¼ 2740 L
Example 8.7 An organic solution containing reactant A (C
A
¼ 2:0 mol=L) is
fed into a 1200-L CSTR, where the following chemical reactions take place:
Reaction 1: 2A ! B
Reaction 2: B ! C þ 2D
Reaction 1 is a second-order reaction, and Reaction 2 is first order. Product B is
the desired product. The feed rate is 100 L/min, and, at the operating tempera-
ture, k
1
¼ 10 L mol
21
h
21
and k
2
¼ 4h
21
.
a. Derive the design equations and plot the reaction and species operating
curves.
b. Determine the conversion of reactant A, the yield of product B, and the
production rates of products B and C for the given feed flow rate.
c. Determine the feed flow rate that provides the highest production rate of
product B.
d. Repeat (b) for the optimal feed rate.
Solution The stoichiometric coefficients of the chemical reactions are
s
A
1
¼2 s
B
1
¼ 1 s
C
1
¼ 0 s
D
1
¼ 0 D
1
¼1
s
A
2
¼ 0 s
B
2
¼1 s
C
2
¼ 1 s
D
2
¼ 2 D
2
¼ 2
The two reactions are independent, and there are no dependent reactions. We
select the inlet steam as the reference stream; hence, Z
1
in
¼ Z
2
in
¼ 0. Since
only reactant A is fed into the reactor, C
0
¼ C
A
in
¼ 2 mol=L, y
A
0
¼ 1, and
344 CONTINUOUS STIRRED-TANK REACTOR

y
B
0
¼ y
C
0
¼ y
D
0
¼ 0. The molar flow rate of the reference stream is
(F
tot
)
0
¼ v
0
C
0
¼ 200 mol=min
a. We write Eq. 8.1.1 for each independent reaction:
Z
1
out
¼ r
1
out
t
t
cr
C
0
(a)
Z
2
out
¼ r
2
out
t
t
cr
C
0
(b)
We use Eq. 8.1.9 to express the species concentrations, and the reaction
rates are
r
1
out
¼ k
1
C
0
2
(1 2Z
1
out
)
2
(c)
r
2
out
¼ k
2
C
0
(Z
1
out
Z
2
out
) (d)
We select Reaction 1 as the leading reaction and using Eq. 3.5.4, define the
characteristic reaction time by
t
cr
¼
1
k
1
C
0
¼ 3 min (e)
Substituting (c), (d), and (e) into (a) and (b), the design equations become
Z
1
out
(1 2Z
1
out
)
2
t ¼ 0(f)
Z
2
out
k
2
k
1
C
0
(Z
1
out
Z
2
out
)t ¼ 0 (g)
Once we solve (f) and (g), we can determine the species operating curves
using Eq. 2.7.8:
F
A
out
(F
tot
)
0
¼ 1 2Z
1
out
(h)
F
B
out
(F
tot
)
0
¼ Z
1
out
Z
2
out
(i)
F
C
out
(F
tot
)
0
¼ Z
2
out
(j)
F
D
out
(F
tot
)
0
¼ 2Z
2
out
(k)
In this case we can solve (f) and (g) analytically,
Z
1
out
¼
(1 þ 4t)
ffiffiffiffiffiffiffiffiffiffiffiffiffi
1 þ 8t
p
8t
(l)
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 345
Z
2
out
¼
0:2t
1 þ 0:2t
(1 þ 4t)
ffiffiffiffiffiffiffiffiffiffiffiffiffi
1 þ 8t
p
8t
(m)
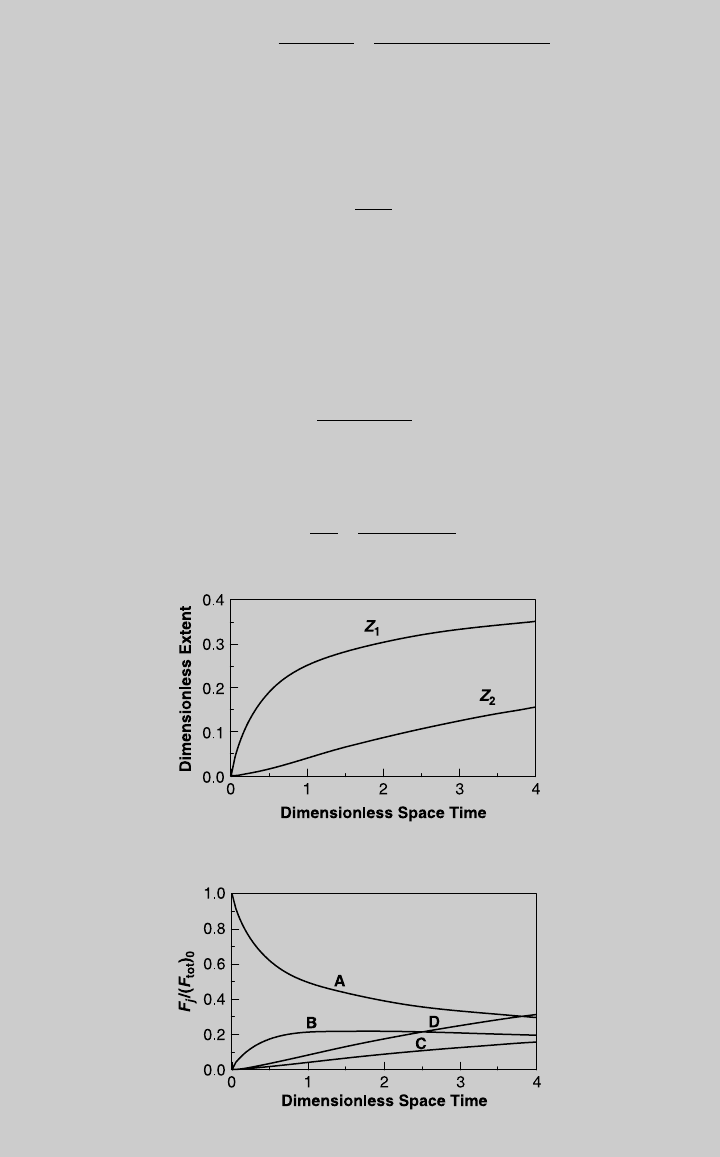
Figure E8.7.1 shows the two reaction curves. We substitute (l) and (m) into
(h) through (k) and obtain the species curves shown in Figure E8.7.2.
b. Using Eq. 8.1.3 and (e), for the given feed volumetric rate,
t ¼
V
R
v
0
t
cr
¼ 4
Substituting t ¼ 4 in (f) and (g), Z
1
out
¼ 0:3517 and Z
2
out
¼ 0:1563. Using
(h) through (k), the species flow rates at the reactor outlet are
F
B
out
¼ 39:1 mol=min, F
B
out
¼ 39:1 mol=min, F
C
out
¼ 31:3 mol=min, and
F
D
out
¼ 62:5 mol=min.
The conversion of reactant A is
f
A
out
¼
F
A
0
F
A
out
F
A
0
¼ 0:704
Using Eq. 2.6.14, the yield of product B is
h
B
out
¼
2
1
F
B
out
F
B
in
F
A
0
¼ 0:391
Figure E8.7.1 Reaction operating curves.
Figure E8.7.2 Species operating curves.
346 CONTINUOUS STIRRED-TANK REACTOR

c. From the curve of product B, the highest F
B
out
is reached at t ¼ 1.75, and
using (f) and (g), Z
1
out
¼ 0:295 and Z
2
out
¼ 0:0764. Using Eq. 8.1.3 and
(e), for t ¼ 1.75, the volumetric feed flow rate is
v
0
¼
V
R
tt
cr
¼ 228:6L=min
The optimal feed molar flow rate is
(F
tot
)
0
¼ v
0
C
0
¼ 457:1 mol=min
d. Using (h) through (k), the species flow rates at the reactor outlet are
F
A
out
¼ 187:6 mol=min, F
B
out
¼ 99:3 mol=min, F
C
out
¼ 34:9 mol=min, F
D
out
¼
69:8 mol=min. The conversion of reactant A is
f
A
out
¼
F
A
in
F
A
out
F
A
in
¼ 0:590
The yield of product B is
h
B
out
¼
2
1
F
B
out
F
B
in
F
A
in
¼ 0:435
The following table provides a comparison between the given and the
optimal operations:
Example 8.8 Product B is produced in a CSTR where the following gas-
phase, first-order chemical reactions take place:
Reaction 1: A ! 2B
Reaction 2: B ! C þ D
A stream of reactant A (C
A
in
¼ 0:04 mol=L) is fed into a 200-L CSTR at a rate
of 100 L/min. At the reactor operating temperature, k
1
¼ 2 min
21
and
k
2
¼ 1 min
21
.
Given Operation Optimal Operation
Reactor volume (L) 1200 1200
Feed flow rate (L/min) 100 229.9
Conversion of reactant A 0.704 0.590
Yield of product B 0.391 0.435
Flow rate of product B (mol/min) 39.1 99.3
Flow rate of product C (mol/min) 31.3 34.9
Flow rate of product D (mol/min) 62.5 69.8
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 347
a. Derive the design equations and plot the reaction and species curves.
b. Determine the conversion of reactant A, the yield of product B, and the pro-
duction rate of products B and C for the given feed rate.
c. Determine the reactor volume that provides the highest production rate of
product B.
d. Determine the conversion of reactant A and the yield of product B for the
optimal reactor.
Solution This is an example of series (consecutive) chemical reactions. The
stoichiometric coefficients of the two reactions are
s
A
1
¼1 s
B
1
¼ 2 s
C
1
¼ 0 s
D
1
¼ 0 D
1
¼ 1
s
A
2
¼ 0 s
B
2
¼1 s
C
2
¼ 1 s
D
2
¼ 1 D
2
¼ 1
Since each reaction has a species that does not participate in the other, the two
reactions are independent, and there is no dependent reaction. We select the
inlet stream as the reference stream; hence, Z
1
in
¼ Z
2
in
¼ 0. Since only reactant
A is fed into the reactor, C
0
¼C
A
in
¼ 0:04 mol=L, y
A
0
¼ 1, and y
B
0
¼ y
C
0
¼
y
D
0
¼ 0. The molar flow rate of the reference stream is
(F
tot
)
0
¼ v
0
C
0
¼ 4 mol= min
We write Eq. 8.1.1 for each independent reaction,
Z
1
out
¼ r
1
out
t
t
cr
C
0
(a)
Z
2
out
¼ r
2
out
t
t
cr
C
0
(b)
a. We use Eq. 8.1.13 to express the species concentrations, and the reaction
rates are
r
1
out
¼ k
1
C
0
1 Z
1
out
1 þ Z
1
out
þ Z
2
out
(c)
r
2
out
¼ k
2
C
0
2Z
1
out
Z
2
out
1 þ Z
1
out
þ Z
2
out
(d)
We define the characteristic reaction time on the basis of Reaction 1; hence,
t
cr
¼
1
k
1
¼ 0:5 min (e)
Substituting (c), (d), and (e) into (a) and (b), the design equations reduce to
Z
1
out
1 Z
1
out
1 þ Z
1
out
þ Z
2
out
t ¼ 0(f)
348 CONTINUOUS STIRRED-TANK REACTOR
Z
2
out
k
2
k
1
2Z
1
out
Z
2
out
1 þ Z
1
out
þ Z
2
out
t ¼ 0 (g)
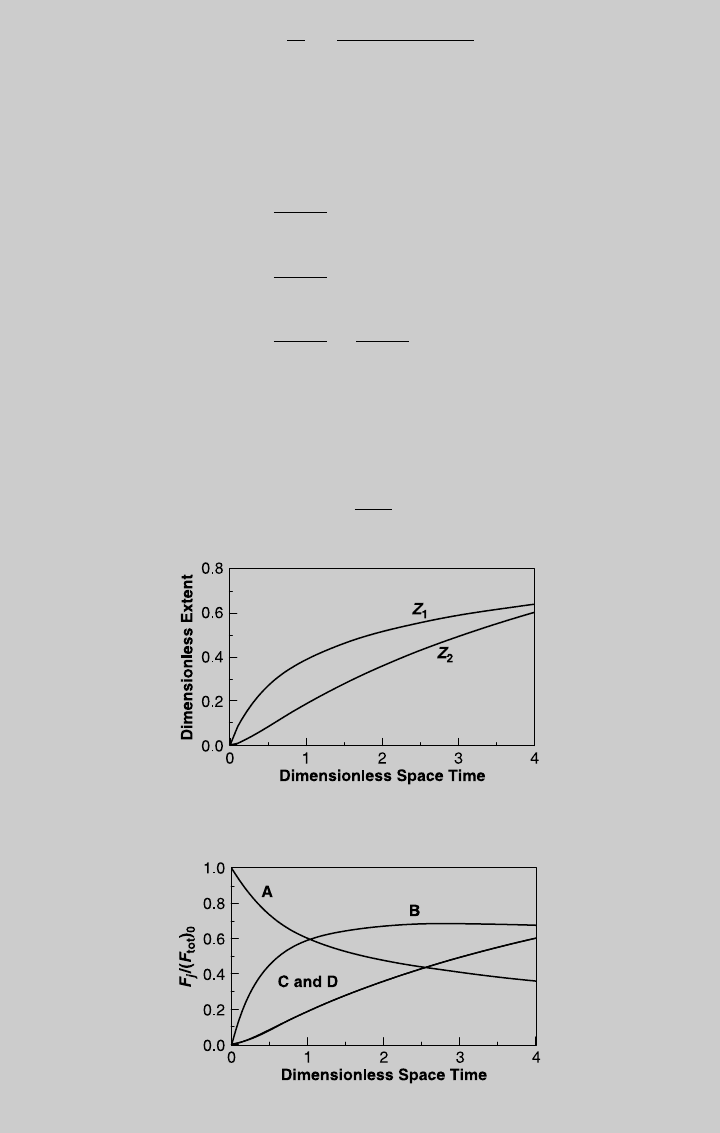
We solve (f) and (g) numerically for different values of t. The reaction
curves are shown in Figure E8.8.1. Once we have Z
1
and Z
2
as a function
of t, we use Eq. 2.7.8 to determine the species curves:
F
A
out
(F
tot
)
0
¼ 1 Z
1
out
(h)
F
B
out
(F
tot
)
0
¼ 2Z
1
out
Z
2
out
(i)
F
C
out
(F
tot
)
0
¼
F
D
out
(F
tot
)
0
¼ Z
2
out
(j)
Figure E8.8.2 shows the species curves.
b. For the given feed flow rate, using Eq. 8.1.3 and (e), the dimensionless space
time of the current operation is
t ¼
V
R
v
0
t
cr
¼ 4
Figure E8.8.1 Reaction operating curves.
Figure E8.8.2 Species operating curves.
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 349
