Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


At t ¼ 4, the solutions of (f) and (g) are Z
1
out
¼ 0:6406 and Z
2
out
¼ 0:6037.
Using (h) through (j), the species molar flow rates at the reactor outlet
are F
A
out
¼ 1:438 mol=min, F
B
out
¼ 2:71 mol=min , and F
C
out
¼ F
D
out
¼
2:42 mol=min. The conversion of reactant A is
f
A
out
;
F
A
in
F
A
out
F
A
in
¼ 0:641
Using Eq. 2.6.14, the yield of product B is
h
B
out
¼
1
2
F
B
out
F
B
in
F
A
in
¼ 0:339
c. From the curve of product B, the highest F
B
out
is reached at t ¼ 3.0. Using
Eq. 8.1.3 and (e), the optimal reactor volume is
V
R
¼ v
0
t
cr
t ¼ (100 L=min )(0:5 min )(3:0) ¼ 150 L
d. At t ¼ 3, the solutions of (f) and (g) are Z
1
out
¼ 0:5901 and Z
2
out
¼ 0:4939.
Using (h) through ( j), the species molar flow rates at the reactor
outlet are F
A
out
¼ 1:64 mol=min, F
B
out
¼ 2:75 mol=min, and F
C
out
¼ F
D
out
¼
1:98 mol=min. The conversion of reactant A is
f
A
out
¼
F
A
in
F
A
out
F
A
in
¼ 0:59
The yield of product B is
h
B
out
¼
1
2
F
B
out
F
B
in
F
A
in
¼ 0:343
The following table provides a comparison between the given and the optimal
reactors:
We see that the volume of the optimal reactor is 25% smaller than the given reac-
tor. While processing the same feed rate, the conversion of reactant A is slightly
lower, the production rate of product B is slightly higher, and the production rate
of products C and D is reduced by about 20%.
Given Reactor Optimal Reactor
Reactor volume (L) 200 150
Volumetric feed rate (L/min) 100 100
Conversion of reactant A 0.641 0.590
Flow rate of product B (mol/min) 2.71 2.75
Flow rate of product C (mol/min) 2.41 1.98
350 CONTINUOUS STIRRED-TANK REACTOR

Example 8.9 A chemical plant generates a stream of reactant A and a stream of
reactant B that presently are being discarded. The engineering department
suggested to use an available 2000-L CSTR to produce valuable product C,
by combining these streams. The reactor operates at 1848C and 1.5 atm. At
these conditions, the following gas-phase reactions take place:
Reactions 1 & 2: A þ B
C
Reaction 3: C þ B
!
D
The reaction rates are r
1
¼ k
1
C
A
C
B
; r
2
¼ k
2
C
C
; and r
3
¼ k
3
C
A
C
B
. The available
pumping equipment in the plant can provide a maximum feed rate of 800 L/min.
a. Determine the proportion of reactants A and B in the feed to maximize the
production of product C.
b. Determine the rates reactant A and reactant B are fed to the reactor.
c. Determine the flow rates of all species at the reactor outlet (at optimal feed
composition).
d. Determine the conversions of reactants A and B and the yield of product C.
Data: At 184 8C, k
1
¼20L=mol min
1
, k
2
¼0:16min
1
, k
3
¼L=molmin
1
Solution We have here two independent reactions and one dependent reaction.
We select Reactions 1 and 3 as the independent reactions and Reaction 2 as the
dependent reaction; hence, m ¼ 1, 3, and k ¼ 2. We use Eq. 2.4.9 and obtain
a
21
¼ 21, a
23
¼ 0. The stoichiometric coefficients of the two independent reac-
tions are
s
A
1
¼1 s
B
1
¼1 s
C
1
¼ 1 s
D
1
¼ 0 D
1
¼1
s
A
3
¼ 0 s
B
3
¼1 s
C
3
¼1 s
D
3
¼ 1 D
3
¼1
We select the inlet stream as the reference stream and denote y
A
0
and y
B
0
to be
the mole fractions of the two reactants in the feed stream ( y
C
0
¼ y
D
0
¼ 0).
The concentration and molar flow rate of the reference stream are
C
0
¼
P
RT
¼ 0:04 mol=L
(F
tot
)
0
¼ v
0
C
0
¼ 32 mol=min
We select Reaction 1 as the leading reaction, and using Eq. 3.5.4, the character-
istic reaction time is
t
cr
¼
1
k
1
C
0
(a)
For the given reactor,
t ¼
V
R
v
0
k
1
C
0
¼ 2 (b)
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 351
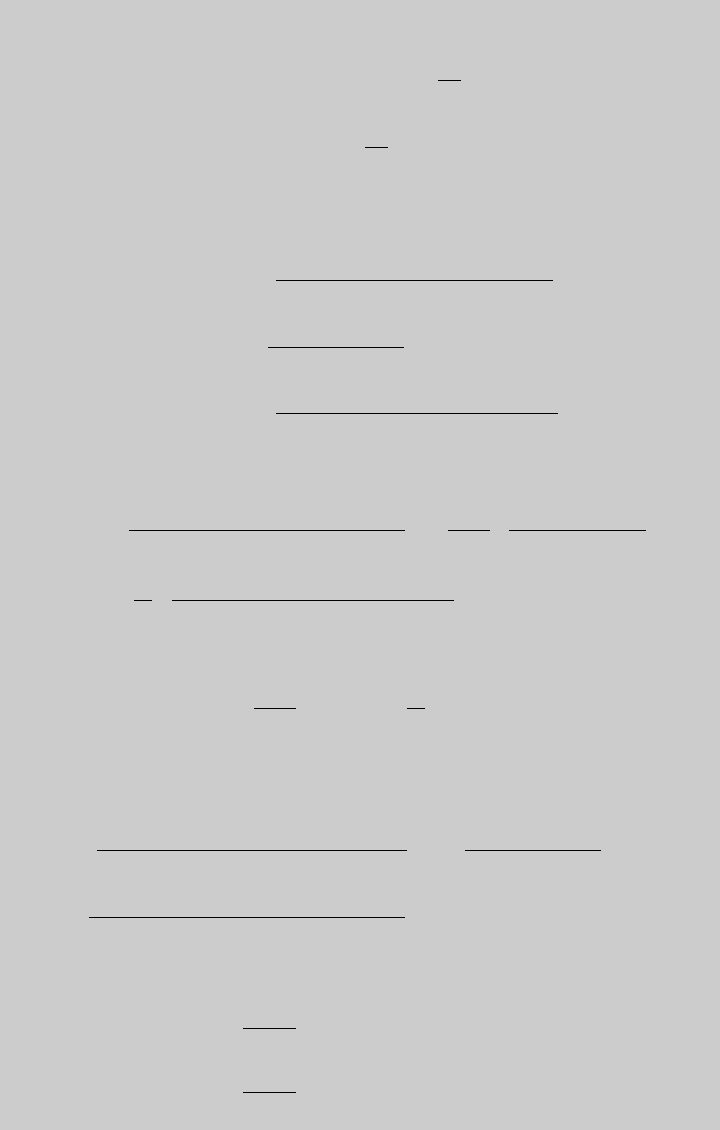
We write Eq. 8.1.1, for each independent reaction,
Z
1
out
¼ (r
1
out
r
2
out
)t
t
cr
C
0
(c)
Z
3
out
¼ r
3
out
t
t
cr
C
0
(d)
Using Eq. 8.1.13 to express the species concentrations, the rates of the
reactions are
r
1
out
¼ k
1
C
0
2
( y
A
0
Z
1
out
)( y
B
0
Z
1
out
Z
3
out
)
(1 Z
1
out
Z
3
out
)
2
(e)
r
2
out
¼ k
2
C
0
Z
1
out
Z
3
out
1 Z
1
out
Z
3
out
(f)
r
3
out
¼ k
3
C
0
2
(Z
1
out
Z
3
out
)( y
B
0
Z
1
out
Z
3
out
)
(1 Z
1
out
Z
3
out
)
2
(g)
Substituting (e), (f), (g), and (a) into (c) and (d), the design equations become
Z
1
out
¼
( y
A
0
Z
1
out
)( y
B
0
Z
1
out
Z
3
out
)
(1 Z
1
out
Z
3
out
)
2
k
2
k
1
C
0
Z
1
out
Z
3
out
1 Z
1
out
Z
3
out
t (h)
Z
3
out
¼
k
3
k
1
(Z
1
out
Z
3
out
)( y
B
0
Z
1
out
Z
3
out
)
(1 Z
1
out
Z
3
out
)
2
t (i)
Using the given data,
k
2
k
1
C
0
¼ 0:2
k
3
k
1
¼ 2
Substituting these values and noting that y
B
0
¼ 1 y
A
0
, the design equations
reduce to
Z
1
out
2
( y
A
0
Z
1
out
)(1 y
A
0
Z
1
out
Z
3
out
)
(1 Z
1
out
Z
3
out
)
2
0: 2
Z
1
out
Z
3
out
1 Z
1
out
Z
3
out
¼ 0(j)
Z
3
out
4
(Z
1
out
Z
3
out
)(1 y
A
0
Z
1
out
Z
3
out
)
(1 Z
1
out
Z
3
out
)
2
¼ 0 (k)
Once we solve ( j) and (k), we use Eq. 2.7.8 to obtain the species operating curve:
F
A
out
(F
tot
)
0
¼ y
A
0
Z
1
out
(l)
F
B
out
(F
tot
)
0
¼ y
B
0
Z
1
out
Z
3
out
(m)
352 CONTINUOUS STIRRED-TANK REACTOR
F
C
out
(F
tot
)
0
¼ Z
1
out
Z
3
out
(n)
F
D
out
(F
tot
)
0
¼ Z
3
out
(o)
a. To determine the optimal feed composition, we solve ( j) and (k) for different
values of y
A
in
and calculate F
C
out
=(F
tot
)
0
by (o). The following table provides
the results of these calculations:
The Figure E8.9.1 shows the plot of F
C
out
=(F
tot
)
0
versus y
A
in
. From the graph,
maximum production rate of product C is achieved for y
A
in
¼ 0:65.
b. The optimal feed rates of reactants A and B are
F
A
in
¼ y
A
in
(F
tot
)
0
¼ 20:8 mol=min
F
B
in
¼ y
B
in
(F
tot
)
0
¼ 11:2 mol=min
c. For the optimal reactant proportion, the solution of ( j) and (k) is Z
1
¼ 0.157
and Z
3
¼ 0.0708. From (h) through (k), the flow rates of the individual
species at the reactor outlet are F
A
out
¼ 15:8 mol=min, F
B
out
¼
3:91 mol=min, F
C
out
¼ 2:76 mol=min, and F
D
out
¼ 2:27 mol=min.
y
A
in
Z
1
out
Z
3
out
F
C
out
=ðF
tot
Þ
0
0.50 0.1956 0.1197 0.0759
0.55 0.1856 0.1039 0.0817
0.60 0.1726 0.0873 0.0853
0.65 0.1571 0.0708 0.0864
0.70 0.1398 0.0550 0.0847
0.75 0.1207 0.0405 0.0802
0.80 0.1000 0.0276 0.0724
Figure E8.9.1 Optimal reactant proportion.
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 353

d. The conversions of reactants A and B are
f
A
out
¼
F
A
in
F
A
out
F
A
in
¼ 0:24
f
B
out
¼
F
B
in
F
B
out
F
B
in
¼ 0:651
Using Eq. 2.6.14, the yield of product C is
h
B
out
¼
1
1
F
C
out
F
C
in
F
A
in
¼ 0:132
Example 8.10 The following reversible reactions (mechanism) were proposed
for gas-phase cracking of hydrocarbons:
Reactions 1 & 2: A
2B
Reactions 3 & 4: A þ B
C
Reactions 5 & 6: A þ C
D
The desired product is B. We want to design an isothermal CSTR to be operated
at 4898C and 5 atm. A stream of reactant A at a rate of 1 mol/s is available in the
plant.
a. Derive the design equations and plot the reaction and species operating
curves.
b. Determine the volume of the reactor that provides the highest production rate
of product B.
c. Determine the equilibrium composition.
d. Repeat parts (a) for a feed stream that consists of species D.
The rate expressions of the chemical reactions are:
r
1
¼ k
1
C
A
r
2
¼ k
2
C
B
2
r
3
¼ k
3
C
A
C
B
r
4
¼ k
4
C
C
r
5
¼ k
5
C
A
C
C
r
6
¼ k
6
C
D
Data: At 4898C,
k
1
¼ 2 min
1
k
2
¼ 20 L=mol min
1
k
3
¼ 50 L=mol min
1
k
4
¼ 0:8 min
1
k
5
¼ 125 L=mol min
1
k
6
¼ 2 min
1
Solution We have considered this reaction scheme in Examples 4.1 and 7.8
but the values of the rate constants are different here. We select the three forward
reactions as the set of independent reactions. Hence, the indices of the indepen-
dent reactions are m ¼ 1, 3, 5, and those of the dependent rea ctions are k ¼ 2, 4, 6.
354 CONTINUOUS STIRRED-TANK REACTOR

The factors a
km
’s of the dependent reactions are a
21
¼ 21, a
43
¼ 21, and
a
65
¼ 21, and all the others are zero. The stoichiometric coefficients of the
independent reactions are
s
A
1
¼1 s
B
1
¼ 2 s
C
1
¼ 0 s
D
1
¼ 0 D
1
¼ 1
s
A
3
¼1 s
B
3
¼1 s
C
3
¼ 1 s
D
3
¼ 0 D
3
¼1
s
A
5
¼1 s
B
5
¼ 0 s
C
5
¼1 s
D
5
¼ 1 D
5
¼1
We write Eq. 8.1.1 for each independent reaction:
Z
1
out
Z
1
in
¼ (r
1
out
r
2
out
)t
t
cr
C
0
(a)
Z
3
out
Z
3
in
¼ (r
3
out
r
4
out
)t
t
cr
C
0
(b)
Z
5
out
Z
5
in
¼ (r
5
out
r
6
out
)t
t
cr
C
0
(c)
We select the inlet stream as the reference stream; hence, T
0
¼ 762 K, y
A
0
¼ 1,
y
B
0
¼ y
C
0
¼ y
D
0
¼ 0, and Z
1
in
¼ Z
3
in
¼ Z
5
in
¼ 0. The reference concentration
and volumetric flow rate are
C
0
¼
P
0
RT
0
¼ 0:08 mol=L
v
0
¼
(F
tot
)
0
C
0
¼ 750 L= min
We use Eq. 8.1.13 to express the species concentration, and the rates of the
chemical reactions are
r
1
¼ k
1
C
0
y
A
0
Z
1
out
Z
3
out
Z
5
out
1 þ Z
1
out
Z
3
out
Z
5
out
(d)
r
2
¼ k
2
C
0
2
y
B
0
þ 2Z
1
out
Z
3
out
1 þ Z
1
out
Z
3
out
Z
5
out
2
(e)
r
3
¼ k
3
C
0
2
( y
A
0
Z
1
out
Z
3
out
Z
5
out
)( y
B
0
þ 2Z
1
out
Z
3
out
)
(1 þ Z
1
out
Z
3
out
Z
5
out
)
2
(f)
r
4
¼ k
4
C
0
y
C
0
þ Z
3
out
Z
5
out
1 þ Z
1
out
Z
3
out
Z
5
out
(g)
r
5
¼ k
5
C
0
2
( y
A
0
Z
1
out
Z
3
out
Z
5
out
)( y
C
0
þ Z
3
out
Z
5
out
)
(1 þ Z
1
out
Z
3
out
Z
5
out
)
2
(h)
r
6
¼ k
6
C
0
y
D
0
þ Z
5
out
1 þ Z
1
out
Z
3
out
Z
5
out
(i)
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 355

We select Reaction 1 as the leading reaction; using Eq. 3.5.4 the characteristic
reaction time is
t
cr
¼
1
k
1
¼ 0:5 min ( j)
Substituting (d) through (i) and into (a), (b), and (c), and using (i), the design
equations become
Z
1
out
t
y
A
0
Z
1
out
Z
3
out
Z
5
out
1þZ
1
out
Z
3
out
Z
5
out
þt
k
2
C
0
k
1
y
B
0
þ2Z
1
out
Z
3
out
1þZ
1
out
Z
3
out
Z
5
out
2
¼0 (k)
Z
3
out
t
k
3
C
0
k
1
(y
A
0
Z
1
out
Z
3
out
Z
5
out
)(y
B
0
þ2Z
1
out
Z
3
out
)
(1þZ
1
out
Z
3
out
Z
5
out
)
2
þ
t
k
4
k
1
y
C
0
þZ
3
out
Z
5
out
1þZ
1
out
Z
3
out
Z
5
out
¼0 (l)
Z
5
out
t
k
5
C
0
k
1
(y
A
0
Z
1
out
Z
3
out
Z
5
out
)(y
C
0
þZ
3
out
Z
5
out
)
(1þZ
1
out
Z
3
out
Z
5
out
)
2
þ
t
k
6
k
1
y
D
0
þZ
5
out
1þZ
1
out
Z
3
out
Z
5
out
¼0 (m)
The parameters are
k
2
C
0
k
1
¼ 0:8
k
3
C
0
k
1
¼ 0:2
k
4
k
1
¼ 0:4
k
5
C
0
k
1
¼ 5
k
6
k
1
¼ 1
Once we solve the design equations, we use Eq. 2.7.8 to determine the species
operating curves:
F
A
out
(F
tot
)
0
¼ y
A
0
Z
1
out
Z
3
out
Z
5
out
(n)
F
B
out
(F
tot
)
0
¼ y
B
0
þ2Z
1
out
Z
3
out
(o)
F
C
out
(F
tot
)
0
¼ y
C
0
þZ
3
out
Z
5
out
(p)
F
D
out
(F
tot
)
0
¼ y
D
0
þZ
5
out
(q)
a. For the given feed, y
A
0
¼ 1 and y
B
0
¼ y
C
0
¼ y
D
0
¼ 0, We substitute these
values into (k), (l), and (m) and solve them numerically for different
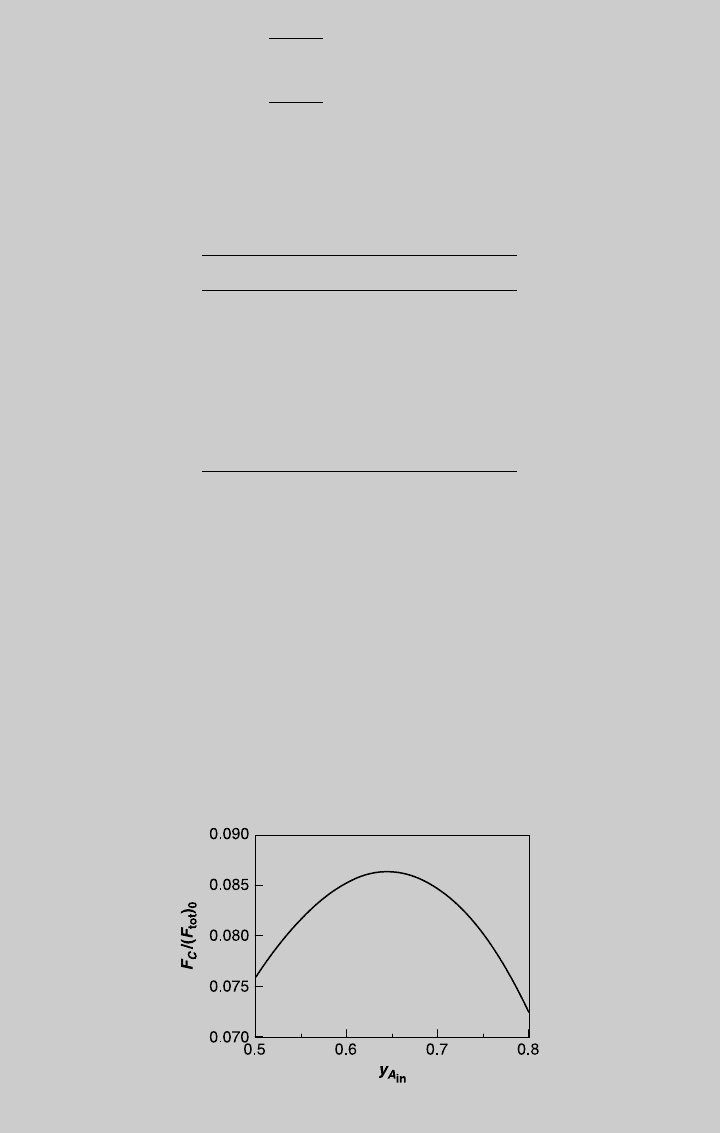
values of t. Figure E8.10.1 shows the reaction operating curves. We then
use (n) through (q) to obtain the species curves, shown in Figure E8.10.2.
356 CONTINUOUS STIRRED-TANK REACTOR
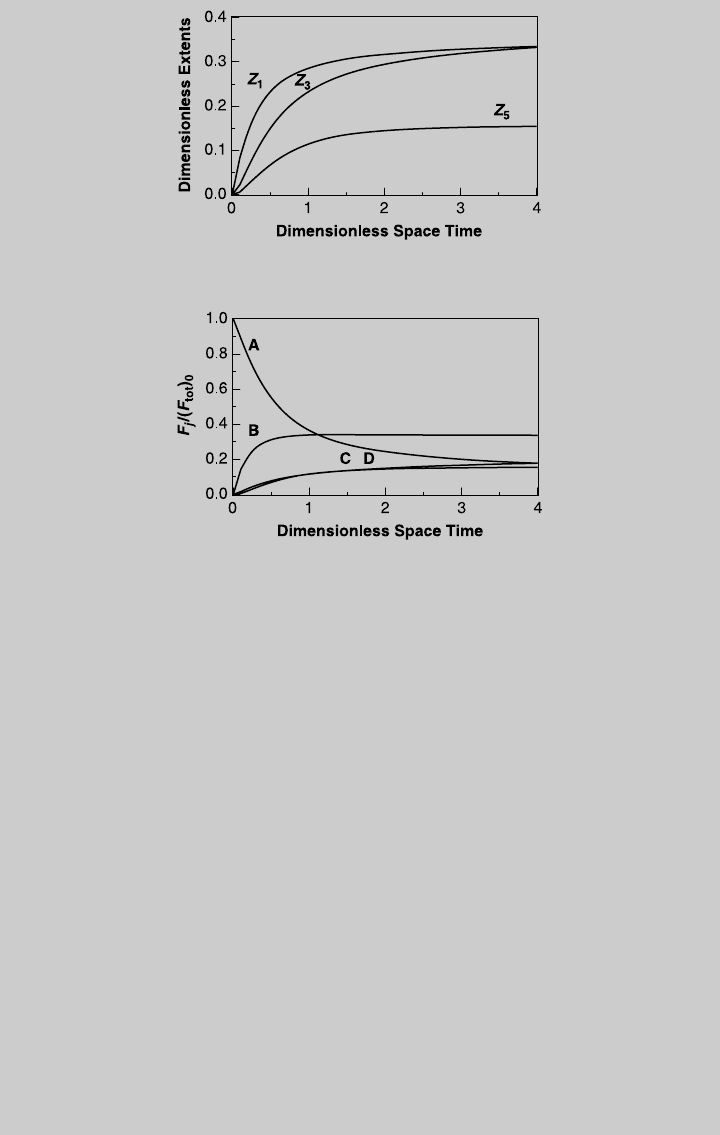
b. From the curve of product B, the highest F
B
out
is reached at t ¼ 1.48, where
F
B
=F
(tot)0
¼ 0:3407. At that space time, Z
1
¼ 0.3061, Z
3
¼ 0.2704, and
Z
5
¼ 0.1351. The production rate of product B is 0.34 mol/s. Using
Eq. 8.1.3 and (j), the reactor volume is
V
R
¼ t v
0
t
cr
¼ 8555 L
c. To estimate the equilibrium composition, we solve the design equations
for t ! 1.Fort ¼ 100, the mole fractions of the various species are:
y
Aeq
¼ 0.1371; y
Beq
¼ 0.4086; y
Ceq
¼ 0.2705; y
Deq
¼ 0.1838.
d. When the feed stream consists of species D, the reactor design proceeds in the
same way as in parts (a). The only difference is that, now, y
A
0
¼ y
B
0
¼
y
C
0
¼ 0 and y
D
0
¼ 1. Substituting these values into (k), (l), and (m), we
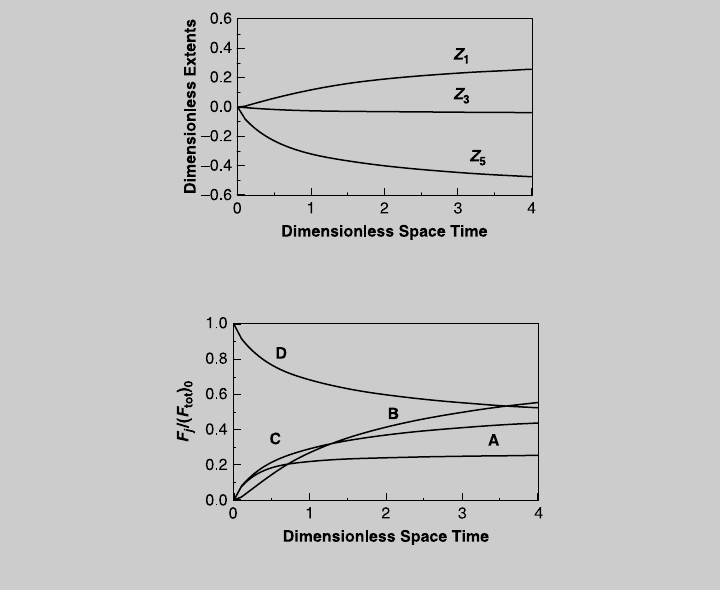
solve them numerically. Figure E8.10.3 shows the reaction operating
curves. We then use (n) through (r) to obtain the species curves, shown in
Figure E8.10.4. Note that in this case, the extents of Reactions 3 and 5 are
negative, since they proceed reversely. To estimate the equilibrium compo-
sition, we solve the design equations for t ¼ 100. The mole fractions of
the various species are: y
Aeq
¼ 0.1344; y
Beq
¼ 0.4039; y
Ceq
¼ 0.2725;
y
Deq
¼ 0.1891. The compositions are very close to those estimated in part (c).
Figure E8.10.1 Reaction operating curves—feed of species A.
Figure E8.10.2 Species operating curves—feed of species A.
8.3 ISOTHERMAL OPERATIONS WITH MULTIPLE REACTIONS 357
8.4 NONISOTHERMAL OPERATIONS
The design formulation of nonisothermal CSTRs with multiple reactions follows
the same procedure outlined in the previous section—we write the design equation,
Eq. 8.1.1, for each independent reaction. However, since the reactor temperature,
T
out
, is not known, we should solve the design equations simultaneously with
the energy balance equation (Eq. 8.1.14).
Note that by definition, the temperature of a CSTR is the same everywhere;
therefore, all CSTRs are, in principle, “isothermal.” Usually, the inlet temperature
is specified, and, as a part of the design, we have to determine the reactor tempera-
ture. When the reactor temperature is the same as the inlet temperature, T
out
¼ T
in
,
we refer to a CSTR as an isothermal reactor. But when T
out
= T
in
,wehaveto
determine the reactor temperature. Also, note that the energy balance equation con-
tains another variable—the temperature of the heating (or cooling) fluid, u
F
.
Because of the complex geometry of the heat-transfer surface in a CSTR, an aver-
age of the heating/cooling fluid’s inlet and outlet temperatures is usually used.
The procedure for setting up the energy balance equation goes as follows:
1. Select and define the temperature of the reference stream, T
0
.
2. Determine the specific molar heat capacity of the reference stream,
^
c
p
0
.
Figure E8.10.3 Reaction operating curves—feed of species D.
Figure E8.10.4 Species operating curves—feed of species D.
358 CONTINUOUS STIRRED-TANK REACTOR

3. Determine the dimensionless activation energies, g
i
’s, of all chemical
reactions.
4. Determine the dimensionless heat of reactions, DHR
m
’s, of the independent
reactions.
5. Determine the correction factor of the heat capacity of the reacting fluid, CF
(Z
m
, u).
6. Specify the dimensionless heat-transfer number, HTN (using Eq. 8.1.22).
7. Determine (or specify) the inlet temperature, u
in
.
8. Determine (or specify) the temperature of the heating/cooling fluid, u
F
.
9. Solve the energy balance equation simultaneously with the design equations
to obtain Z
m
’s and u
out
as functions of the dimensionless space time, t.
The design formulation of nonisothermal CSTRs consists of (n
I
þ 1) simul-
taneous, nonlinear algebraic equations. We have to solve them for different
values of dimensionless space time, t. Below, we illustrate how to design noni-
sothermal CSTRs.
Example 8.11 The first-order chemical reaction
A ! 2B
takes place in an aqueous solution. A solution of reactant A (C
A
¼ 0:8 mol=L) is
fed at a rate of 200 L/min into a cascade of two equal-size 100-L CSTRs con-
nected in series. The feed temperature is 478C. Based on the data below, for the
indicated operations, determine the conversion of reactant A and the outlet temp-
erature of each reactor:
a. Derive the reaction and species operating curves of each reactor for isother-
mal operation. Determine the feed flow rate if we would like to achieve 80%
conversion.
b. Determine the heating load of each reactor in (a).
c. Determine the isothermal HTN of each reactor.
d. Derive the reaction and species operating curves of each reactor for adiabatic
operation. Determine the feed flow rate if we would like to achieve 80% con-
version.
Data:At478C, k
1
¼ 0: 4 min
1
, DH
R
(T
0
) ¼20 kcal=mol B
E
a
¼ 9000 cal=mol r ¼ 1:0kg=L
c
p
¼ 1:0 kcal=kg K
The temperature of the cooling fluid is 278C.
Solution The stoichiometric coefficients of the reaction are
s
A
¼1 s
B
¼ 2 D ¼ 1
8.4 NONISOTHERMAL OPERATIONS 359
