Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.


For the chemical formula used, the heat of reaction is
D H
R
(320 K) ¼
20 kcal
mol B
2 mol B
mol extent
¼40
kcal
mol extent
We select the inlet stream to the first reactor as the reference stream; hence,
y
A
0
¼ 1, y
B
0
¼ 0, T
0
¼ 3208K, y
A
0
¼ 1, y
B
0
¼ 0, T
0
¼ 3208K, and Z
1
in
¼ 0:
The molar flow rate of the reference stream is
(F
tot
)
0
¼ v
0
C
0
¼ 160 mol=min (a)
Using Eq. 5.2.60, the specific molar heat capacity of the reference stream is
^
c
p
0
¼
r
C
0
c
p
¼ 1250
cal
mol K
(b)
Using Eq. 5.2.62, for liquid-phase reactions and assuming constant heat
capacity, CF(Z
m
, u) ¼ 1.0. The dimensionless heat of reaction is
DHR ¼
D H
R
(T
0
)
T
0
^
c
p
0
¼0:10 (c)
The dimensionless activation energy is
g ¼
E
a
RT
0
¼ 14:15 (d)
Using Eq. 8.1.1, the design equation for each reactor is
Z
out
Z
in
¼ r
out
t
t
cr
C
0
(e)
Using Eq. 8.1.9 to express the species concentrations, the reaction rate is
r
out
¼ k(T
0
)C
0
(1 Z
out
)e
g
(u1)=u
(f)
We define the characteristic reaction time as
t
cr
¼
1
k(T
0
)
¼ 2:5 min (g)
Substituting (f) and (g) into (e), the design equation for the first reactor is
Z
1
out
(1 Z
1
out
)t
1
e
g
(u
1
1)=u
1
¼ 0 (h)
and the design equation for the second reactor is
Z
2
out
Z
1
out
(1 Z
2
out
)t
2
e
g
(u
2
1)=u
2
¼ 0 (i)
Using Eq. 8.1.14, the energy balance equation for the first reactor is
HTN
1
t
1
(u
F
u
1
) ¼ DHR(Z
1
out
0) þ CF(Z, u)
1
(u
1
1) ( j)
360 CONTINUOUS STIRRED-TANK REACTOR
The energy balance equation for the second reactor is
HTN
2
t
2
(u
F
u
2
) ¼ DHR(Z
2
out
Z
1
out
) þ CF(Z, u)
2
(u
2
u
1
) (k)
Once we solve the design equations, we can determine the species curves, using
Eq. 2.7.8:
F
A
out
(F
tot
)
0
¼ 1 Z
out
(l)
F
B
out
(F
tot
)
0
¼ 2Z
out
(m)
a. For isothermal operation, u
1
¼ u
2
¼ 1, we solve (h) and (i) and obtain the
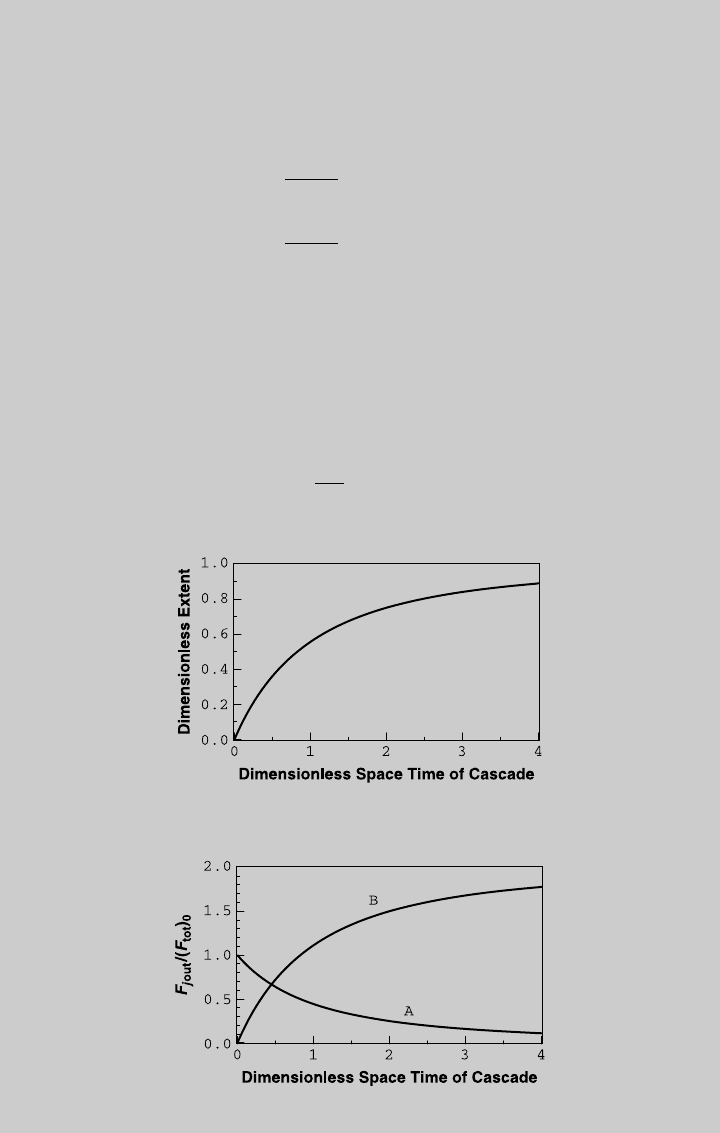
reaction operating curve of the cascade, shown in Figure E8.11.1. We then
use (l) and (m) to obtain the species curves, shown in Figure E8.11.2.
From the curve of reactant A, 80% conversion [F
A
=(F
tot
)
0
¼ 0:2] is reached
at t ¼ 2.48. At that cascade space time, Z
1
¼ 0.554, and Z
2
¼ 0.80. Using
Eq. 8.1.3 and (g), the flow rate of the reference stream is
v
0
¼
V
R
tt
cr
¼ 32:4L=min
Figure E8.11.1 Reaction curve for the cascade—isothermal operation.
Figure E8.11.2 Species curves for the cascade—isothermal operation.
8.4 NONISOTHERMAL OPERATIONS 361

b. Combining Eqs. 8.1.14 and 8.1.16, the heating load of the first reactor is
_
Q
1
(F
tot
)
0
T
0
^
c
p
0
¼ DHR(Z
1
out
0) (l)
and, the heating load of the second reactor is
_
Q
2
(F
tot
)
0
T
0
^
c
p
0
¼ DHR(Z
2
out
Z
1
out
) (m)
Using (c), the heating loads of the reactors are
_
Q
1
¼ (F
tot
)
0
D H
R
(T
0
)Z
out
1
¼2871 kcal=min
_
Q
2
¼ (F
tot
)
0
D H
R
(T
0
)(Z
out
2
Z
out
1
) ¼1274 kcal=min
The negative sign indicates that heat is being removed from the reactor.
c. To determine the isothermal HTN of each reactor, we use Eq. 8.1.20. For the
first reactor,
HTN
iso
¼
1
(u
F
1
1)t
1
DHR(Z
1
out
0) (n)
and for the second reactor
HTN
iso
¼
1
(u
F
2
1)t
2
DHR(Z
2
out
Z
1
out
) (o)
For t
1
¼ t
2
¼ 1.235, we solve (n) and (o) and obtain HTN
1
¼ 0.714, and
HTN
2
¼ 0.319.
d. For liquid-phase reaction, assuming constant heat capacity, CF (z, u) ¼ 1.
For adiabatic operations, (HTN ¼ 0), the energy balance for the first reactor,
( j), becomes
DHR(Z
1
out
0) þ (1)(u
1
1) ¼ 0(p)
The energy balance equation of the second reactor, (k), becomes
DHR(Z
2
out
Z
1
out
) þ (1)(u
2
u
1
) ¼ 0 (q)
We solve (h) and (i) simultaneously with (p) and (q) for different values of t.
Figure E8.11.3 shows the reaction curve for the cascade, and Figure E8.11.4
shows the temperature curves. We then use (l) and (m) to obtain the species
curves for the cascade, shown in Figure E8.11.5. From the curve of reactant
A at the exit of the second reactor, 80% conversion [F
A
=(F
tot
)
0
¼ 0:2] is
reached at t ¼ 1.04. At that cascade space time, Z
1
¼ 0.507, u
1
¼ 1.0507,
362 CONTINUOUS STIRRED-TANK REACTOR
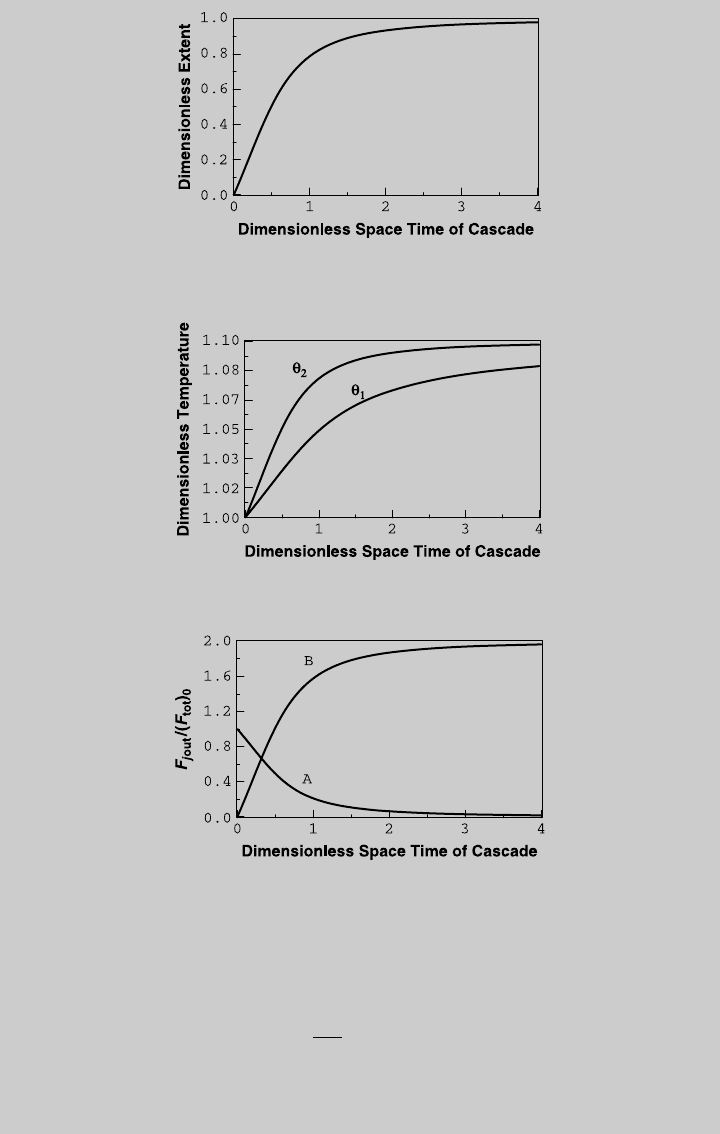
and Z
2
¼ 0.80, u
2
¼ 1.0802. Using Eq. 8.1.3 and (g), the flow rate of the
reference stream is
v
0
¼
V
R
tt
cr
¼ 76:9L= min
The temperature of the first reactor is 336.2 K, and of the second reactor is
345.7 K.
Figure E8.11.3 Reaction curve for the cascade—adiabatic operation.
Figure E8.11.4 The temperature curves—adiabatic operation.
Figure E8.11.5 Species curves for the cascade—adiabatic operation.
8.4 NONISOTHERMAL OPERATIONS 363

Example 8.12 The gas-phase chemical reactions
Reaction 1: A þ B ! C
Reaction 2: C þ B ! D
are carried out in a CSTR. A gas stream (at 1508C and 2 atm) consisting of 40%
reactant A, 40% reactant B, and 20% I (inert) is fed into a reactor at a rate of
0.4 m
3
/min. The reactor is cooled by condensing steam in the shell side at
T
F
¼ 1308C.
a. Derive the design equations and plot the reaction and species operating
curves for isothermal operation.
b. Determine the volume of isothermal reactor that provides the highest pro-
duction rate of product C.
c. Determine the heating rate in (b).
d. Determine the HTN for isothermal operation.
e. Derive the design equations and plot the reaction and species operating
curves for adiabatic operation.
f. Determine the volume of adiabatic reactor that provides the highest pro-
duction rate of product C. What is the operating temperature?
g. Determine the volume of the reactor that provides the highest production rate
of product C if the HTN is half of the isothermal HTN. What is the operating
temperature?
The rate expressions are: r
1
¼ k
1
C
A
C
B
, r
2
¼ k
2
C
B
C
C
Data: At 1508 C k
1
¼0:2L=mol s
1
, k
2
¼0:4L=mol s
1
DH
R
1
¼6000 cal=mol extent DH
R
2
¼4000 cal=mol extent
E
a
1
¼4900 cal=mol E
a
2
¼7000 cal=mol
^
c
p
A
¼16 cal=mol K
^
c
p
B
¼8 cal=mol K
^
c
p
C
¼20 cal=mol K
^
c
p
D
¼26 cal=mol K
^
c
p
I
¼10 cal=mol K
Solution Since each reaction has a species that does not appear in the other
reaction, the two reactions are independent, and there is no dependent reaction.
The stoichiometric coefficients are
s
A
1
¼1 s
B
1
¼1 s
C
1
¼ 1 s
D
1
¼ 0 D
1
¼1
s
A
2
¼ 0 s
B
2
¼1 s
C
2
¼1 s
D
2
¼ 1 D
2
¼1
We select the inlet stream as the reference stream; hence, Z
1
in
¼ Z
2
in
¼ 0, and
y
A
0
¼ 0:40, y
B
0
¼ 0:40, y
I
0
¼ 0:20, and y
C
0
¼ y
D
0
¼ 0. The reference
364 CONTINUOUS STIRRED-TANK REACTOR

concentration and flow rate are
C
0
¼
P
RT
0
¼ 5:76 10
2
mol=L
(F
tot
)
0
¼ v
0
C
0
¼ 23:05 mol= min
Using Eq. 5.2.58, the specific heat capacity of the reference stream is
^
c
p
0
¼
X
J
j
y
j
0
^
c
p
j
(1) ¼ y
A
0
^
c
p
A
(T
0
) þ y
B
0
^
c
p
B
(T
0
) þ y
I
0
^
c
p
I
(T
0
) ¼ 11:6 cal=mol K
The dimensionless activation energies of the two reactions are
g
1
¼
E
a
1
RT
0
¼ 5:83 g
2
¼
E
a
2
RT
0
¼ 8:33
The dimensionless heat of reactions are
DHR
1
¼
D H
R
1
(T
0
)
T
0
^
c
p
0
¼1:222 DHR
2
¼
D H
R
2
(T
0
)
T
0
^
c
p
0
¼0:815
Using Eq. 5.2.61, the correction factor of the heat capacity of the reacting fluid is
CF(Z
m
, u) ¼ 1 þ
1
^
c
p
0
X
J
j
^
c
p
j
(u)
X
n
I
m
(s
j
)
m
Z
m
¼ 1 þ
1
^
c
p
0
[
^
c
p
A
( Z
1
) þ
^
c
p
B
( Z
1
Z
2
) þ
^
c
p
C
(Z
1
Z
2
) þ
^
c
p
D
Z
2
]
¼
11:6 4Z
1
2Z
2
11:6
We write Eq. 8.1.1 for each independent reaction:
Z
1
out
¼ r
1
out
t
t
cr
C
0
(a)
Z
2
out
¼ r
2
out
t
t
cr
C
0
(b)
Using Eq. 8.1.13 to express the series concentrations, the rates of the two reac-
tions are
r
1
out
¼ k
1
(T
0
)C
0
2
( y
A
0
Z
1
out
)( y
B
0
Z
1
out
Z
2
out
)
[(1 Z
1
out
Z
2
out
)u]
2
e
g
1
(u1)=u
(c)
r
2
out
¼ k
2
(T
0
)C
0
2
(Z
1
out
Z
2
out
)( y
B
0
Z
1
out
Z
2
out
)
[(1 Z
1
out
Z
2
out
)u]
2
e
g
2
(u1)=u
(d)
8.4 NONISOTHERMAL OPERATIONS 365

We select Reaction 1 as the leading reaction, and the characteristic reaction
time is
t
cr
¼
1
k
1
(T
0
)C
0
¼ 86:85 ¼ 1:45 min (e)
Substituting (c), (d), and (e) into (a) and (b), the two design equations become
Z
1
out
( y
A
0
Z
1
out
)( y
B
0
Z
1
out
Z
2
out
)
[(1 Z
1
out
Z
2
out
)u]
2
te
g
1
(u1)=u
¼ 0(f)
Z
2
out
k
2
(T
0
)
k
1
(T
0
)
(Z
1
out
Z
2
out
)( y
B
0
Z
1
out
Z
2
out
)
[(1 Z
1
out
Z
2
out
)u]
2
te
g
2
(u1)=u
¼ 0 (g)
Substituting CF(Z
m
,u) into Eq. 8.1.14, the energy balance equation is
HTNt(u
F
u) DHR
1
Z
1
out
DHR
2
Z
2
out
11:6 4Z
1
out
2Z
2
out
11:6
(u 1) ¼ 0
(h)
We have to solve (f), (g), and (h) simultaneously for different values of t. Once
we have the solution, we use Eq. 2.7.8 to obtain the species curves:
F
A
out
(F
tot
)
0
¼ y
A
0
Z
1
out
(i)
F
B
out
(F
tot
)
0
¼ y
B
0
Z
1
out
Z
2
out
(j)
F
C
out
(F
tot
)
0
¼ y
C
0
þ Z
1
out
Z
2
out
(k)
F
D
out
(F
tot
)
0
¼ y
D
0
þ Z
2
out
(l)
a. For isothermal operation (u ¼ 1), the two design equations become
Z
1
out
(0:4 Z
1
out
)(0:4 Z
1
out
Z
2
out
)
(1 Z
1
out
Z
2
out
)
2
t ¼ 0 (m)
Z
2
out
(0 :5)
(Z
1
out
Z
2
out
)(0:4 Z
1
out
Z
2
out
)
(1 Z
1
out
Z
2
out
)
2
t ¼ 0 (n)
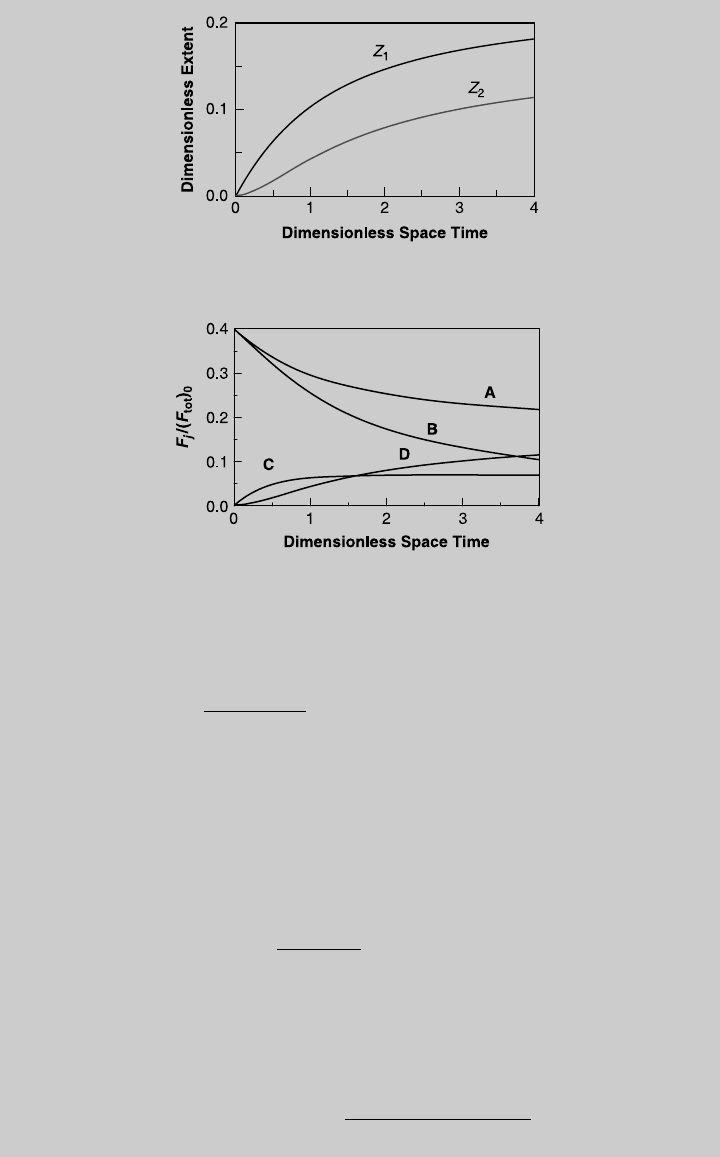
We solve (m) and (n) numerically for different values of t. Figure E8.12.1 shows
the rea ction operating curv es. Using (i) through (l), we obtain the species oper-
ating curv es, sho wn in Figure E8.12.2. From the curve of product C, the highest
F
C
=(F
tot
)
0
¼ 0:0686 and it is rea ched at t ¼ 2.84. F or that pace time,
Z
1
¼ 0.1664 and Z
2
¼ 0.0978. Using (e) and Eq. 8.1.3, the rea ctor volume is
V
R
¼ v
0
t
cr
t ¼ 1647 L
The production ra te of product C is 1.58 mol/min.
366 CONTINUOUS STIRRED-TANK REACTOR
b. For isothermal operation u ¼ 1, and combining Eq. 8.1.14 and Eq. 8.1.17,
_
Q
T
0
(F
tot
)
0
^
c
p
0
¼ DHR
1
Z
1
out
þ DHR
2
Z
2
out
(o)
which reduces to
_
Q ¼ (F
tot
)
0
bD H
R
1
(T
0
)Z
1
out
þ D H
R
2
(T
0
)Z
2
out
c¼32:02 kcal=min
The negative sign indicates that heat is removed from the reactor.
c. Using Eq. 8.1.20, the isothermal HTN is
HTN
iso
(t) ¼
1
(u
F
1) t
(DHR
1
Z
1
out
þ DHR
2
Z
2
out
)(p)
Figure E8.12.3 shows the HTN curve as a function of the reactor volume
(space time). For the given reactor, t ¼ 2.84, HTN ¼ 2.09.
d. For adiabatic operation, HTN ¼ 0, and (h) reduces to
DHR
1
Z
1
out
þ DHR
2
Z
2
out
þ
11:6 4Z
1
out
2Z
2
out
11:6
(u 1) ¼ 0 (q)
Figure E8.12.1 Reaction operating curves—isothermal operation.
Figure E8.12.2 Species curves—isothermal operation.
8.4 NONISOTHERMAL OPERATIONS 367
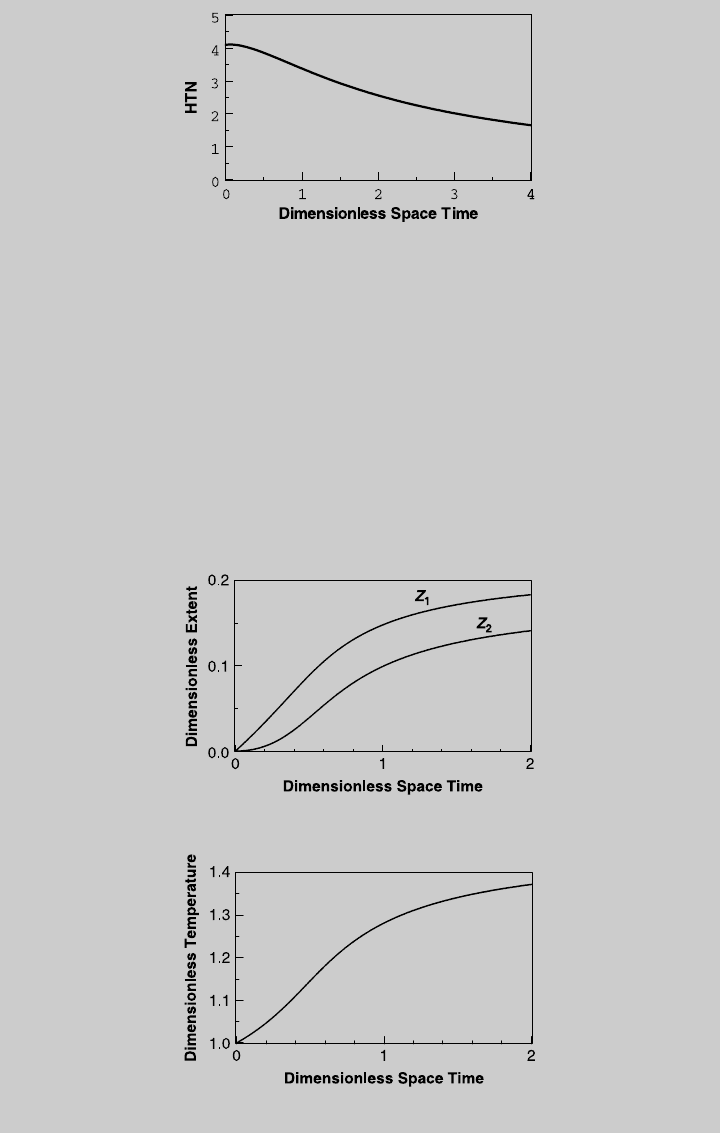
We solve (f), (g), and (q) numerically for different values of t. Figure E8.12.4
shows the reaction curves, and Figure E8.12.5 shows the temperature curve.
Using (i) through (l), we obtain the species operating curves, shown in
Figure E8.12.6. From the curve of product C, the highest F
C
=(F
tot
)
0
is
0.0516, and it is reached at t ¼ 0.66. At that space time, Z
1
¼ 0.1142,
Z
2
¼ 0.0626, and u ¼ 1.20. Using (e) and Eq. 8.1.3, the reactor volume is
V
R
¼ v
0
t
cr
t ¼ 382:8L
and the reactor temperature is T ¼ (1.2)(423) ¼ 507.8 K. The production rate
of product C is 1.19 mol/min.
Figure E8.12.3 Isothermal HTN curve.
Figure E8.12.4 Reaction operating curves—adiabatic operation.
Figure E8.12.5 Temperature curve—adiabatic operation.
368 CONTINUOUS STIRRED-TANK REACTOR
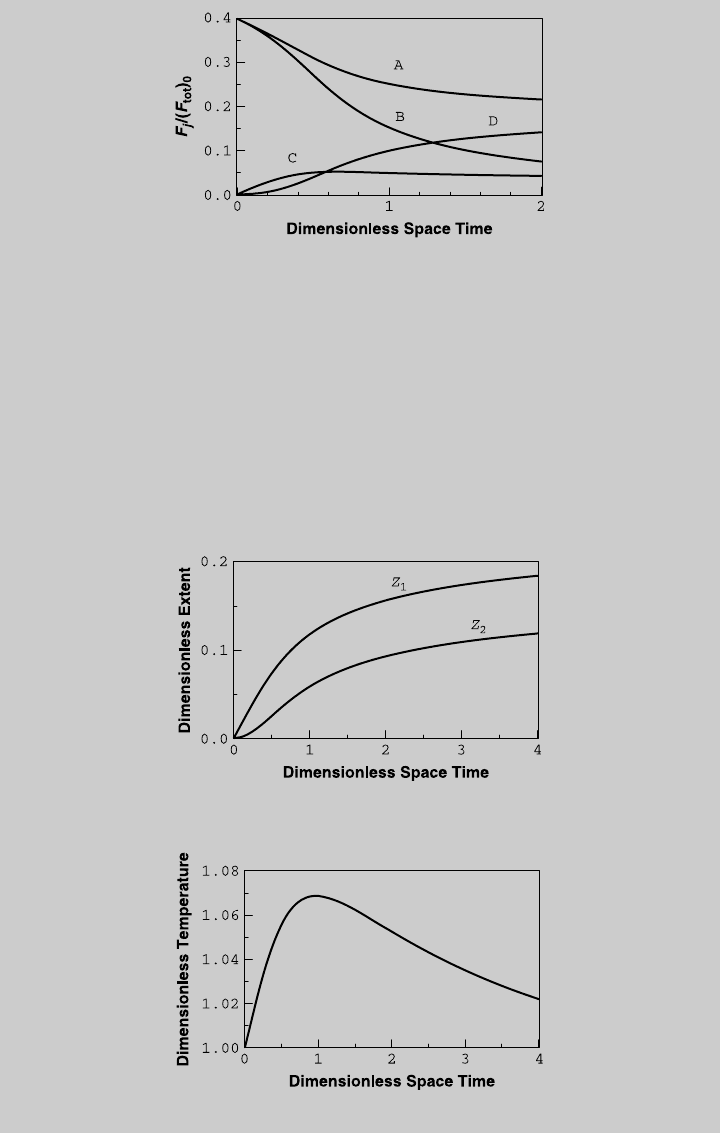
e. For nonisothermal operation with HTN ¼ 1.09, we solve (f), (g), and (h)
numerically for different values of t. Figure E8.12.7 shows the reaction
curves and Figure E8.12.8 shows the temperature curve. Using (i) through
(l), we obtain the species operating curves, shown in Figure E8.12.9. The
F
C
curve does not have a maximum. If we use the same feed rate as in
isothermal operation (t ¼ 2.84), Z
1
¼ 0.1722, Z
2
¼ 0.1073, u ¼ 1.0375,
and F
C
/(F
tot
)
0
¼ 0.0649. The reactor temperature is 439 K and the pro-
duction rate of product C is 1.495 mol/min.
Figure E8.12.6 Species operating curves—adiabatic operation.
Figure E8.12.7 Reaction curves—nonisothermal operation.
Figure E8.12.8 Temperature curve—nonisothermal operation.
8.4 NONISOTHERMAL OPERATIONS 369
