Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.

8.5 SUMMARY
In this chapter we discussed the operation of continuous stirred-tank reactors. We
covered the following topics:
1. The underlying assumptions of the CSTR model and when they are satisfied
in practice.
2. The design equations, the energy balance equation, and the auxiliary relations
for species concentrations.
3. The reaction operating curves and species operating curves.
4. Design and operation of isothermal CSTRs with single reactions.
5. Determination of the reaction rate expression and its parameters.
6. Operation and optimization of a cascade of CSTRs.
7. Design and operation of isothermal CSTRs with multiple reactions.
8. A procedure to estimate the range of the HTN.
9. Design and operation of nonisothermal CSTRs with multiple reactions.
PROBLEMS*
8.1
1
The gas-phase chemical reaction
2A ! R
takes place in a CSTR. The rate expression is r ¼ 0.05C
A
2
mol/Ls
21
.
A stream consisting of 50% A–50% inert (by mole) is fed at a rate of
*
Subscript 1 indicates simple problems that require application of equations provided in the text.
Subscript 2 indicates problems whose solutions require some more in-depth analysis and modifi-
cations of given equations. Subscript 3 indicates problems whose solutions require more comprehen-
sive analysis and involve application of several concepts. Subscript 4 indicates problems that require
the use of a mathematical software or the writing of a computer code to obtain numerical solutions.
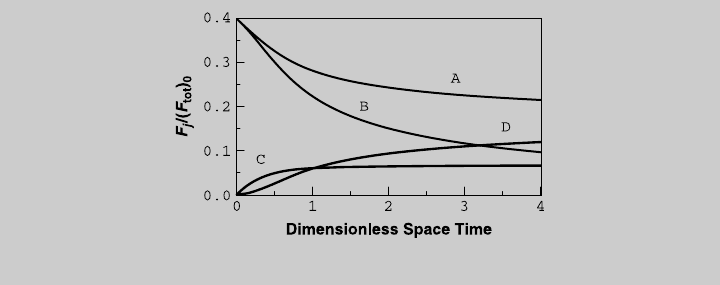
Figure E8.12.9 Species curves—nonisothermal operation.
370 CONTINUOUS STIRRED-TANK REACTOR
180 L/min into a 1-m
3
CSTR. Derive and plot the reaction and species
operating curves. If C
A
in
¼ 0.3 mol/L, what is the conversion of A?
What is the production rate of product R?
8.2
2
The gas-phase chemical reaction
A þ 2B ! 3C
takes place in a CSTR. A gas stream at 2 atm and 6778K contains reactant
A, and an inert at a proportion of 1/6 A and 5 /6 inert is fed at a rate of 2 L/
min. A second gas stream of reactant B at 1.95 atm and 3308K is fed at a
rate of 0.5 L/min into the reactor. The volume of the reactor is 0.75 L,
and it is kept at 4408K and 1.3 atm. The rate expression is r ¼ kC
A
C
B
.
The partial pressure of reactant A in the reactor and in the exit stream is
0.029 atm.
a. Derive and plot the reaction and species operating curves.
b. Determine the conversion of reactants A and B.
c. Determine the reaction rate constant of the reaction.
8.3
1
The liquid-phase reaction
A þ B ! R
takes place in a CSTR. An aqueous solution containing reactants A and B
(C
A
in
¼ 100 mmol/L and C
B
in
¼ 200 mmol/L) is fed at a rate of 400 L/min
to the reactor. The rate expression of the reaction is r ¼ 200C
A
C
B
mol/L
min
21
.
a. Derive and plot the reaction and species operating curves.
b. Determine the reactor volume needed for 90% conversion of A.
8.4
2
In the presence of an enzyme of fixed concentration, reactant A in aqueous
solution decomposes according to the reaction
A ! R þ P
The following data were collected on an isothermal CSTR:
C
A
(mol=L) 1234 5 6 810
r (mol=Lmin)12344:74:95 5
We plan to carry out this reaction in a large-scale CSTR at the same fixed
enzyme concentration. Use a graphical method (not requiring the derivation
of the rate expression) to derive and plot the reaction and species operating
curves for C
A
in
¼10 mol=L.
a. Determine and plot the reaction and species operating curves.
b. Find the volumetric flow rate of the stream that we can feed to 250 L
CSTR if we want to obtain 80% conversion.
PROBLEMS 371

8.5
2
The liquid-phase chemical reaction
A þ B ! R þ S
is investigated on a CSTR. A feed stream containing reactants A and B is
fed to a 1-L CSTR, and the following data are obtained:
Feed Composition (mol/L)
Flow Rate
(L/min)
Output
(mmol/L)
C
A
in
¼ C
B
in
¼ 100 v ¼ 1 C
A
out
¼ 50
C
A
in
¼ C
B
in
¼ 200 v ¼ 9 C
A
out
¼ 150
C
A
in
¼ 200; C
B
in
¼ 100 v ¼ 3 C
A
out
¼ 150
Determine the rate expression and the reaction rate constant.
8.6
2
The gas-phase decomposition reaction
A ! B þ 2C
is being studied in a CSTR. Reactant A at about 3 atm and 308C(C
A
in
¼
0.120 mol/L) is fed into a 1-L CSTR at various flow rates, and its exit con-
centration is measured for each flow rate.
a. Derive a rate expression of the reaction.
b. Derive and plot the reaction and species operating curves for a CSTR.
c. A stream of reactant A (C
A
in
¼ 0:320 mol=L) is fed into a 560-L CSTR.
Determine volumetric feed flow rate if we want to achieve 50%
conversion.
Data:
v
in
ðL=minÞ 0:06 0:48 1:58:1
C
A
out
ðmmol=LÞ 30 60 80 105
8.7
2
The gas-phase reaction
A ! 2B þ 2C þ D
is being investigated in a CSTR. A stream of a high-molecular-weight reac-
tant A is fed continuously to the reactor. By changing the feed flow rate,
different extents of cracking are obtained as follows:
F
A
in
(mmol=h) 300 1000 3000 5000
C
A
out
(mmol=L) 16 30 50 60
The volume of the reactor is 0.1 L, and, at the temperature of the reactor, the
feed concentration is C
A
in
¼ 0:1 mol=L. Determine the rate expression that
fits the experimental data.
8.8
2
The first-order, liquid-phase reaction
A ! R þ P
372 CONTINUOUS STIRRED-TANK REACTOR

is being studied in a CSTR. A liquid stream is fed into a CSTR, where reac-
tant A decomposes according to the first-order reaction. The reactor is oper-
ated at different temperatures, and the feed flow rate is adjusted to keep the
composition of the exit stream constant. Assuming constant feed concen-
tration and constant density, determine the activation energy of the reaction
from the following experimental results:
T ð8 CÞ 19 27 31 37
v
in
ðarbitrary unitsÞ 1235
8.9
2
The second-order, gas-phase reaction
A ! B þ C
is carried out in a cascade of two isothermal CSTRs. Reactant A is fed at a
rate of 100 mol/h into the first reactor, whose volume is 10 L. The molar
flow rate of reactant A at the exit of the first reactor is 60 mol/h, and at
the exit of the second reactor is 20 mol/h. The temperature in both reactors
is 1508C, and the pressure is 2 atm. Determine the volume of Reactor 2 by:
a. Taking the inlet stream into the cascade as the reference stream.
b. Taking the inlet stream into Reactor 2 as the reference stream.
8.10
2
A biological reagent A decomposes by the liquid-phase reaction
A ! R þ P
The rate expression is
r ¼
C
A
0:2 þ C
A
mol=L min
We wish to treat 10 L/min of a waste liquid stream containing A
(C
A
in
¼ 1 mol=L), and we want to achieve 99% conversion of A. Two
equal-size tanks (CSTR) are available. What is the best arrangement for
the two tanks. Determine the size of the two units needed.
8.11
4
The first-order gas-phase reaction
A ! B þ C
takes place in a CSTR. A stream consisting of 90% of reactant A and 10% I
(% mole) is fed into a 200-L reactor at a rate of 20 L/s. The feed is at 731 K
and 3 atm.
a. Derive and plot the reaction and species curves for isothermal operation.
b. Determine the conversion of reactant A when the reactor is operated
isothermally.
c. Determine the heating rate in part (b).
d. Determine the isothermal HTN.
e. Derive and plot the reaction, temperature, and species curves for adia-
batic operation.
PROBLEMS 373
f. Determine the conversion of reactant A when the reactor is operated
adiabatically.
g. Determine the reactor temperature in part (f).
h. Repeat parts (f) and (g) for nonisothermal operation with HTN half of
the isothermal HTN.
Data: At 731 K, k ¼ 0.2 s
21
, E
a
¼ 12,000 cal=mol
DH
R
¼10,000 cal= mol extent
^
c
p
A
¼ 25 cal=mol K
^
c
p
B
¼ 15 cal=mol K
^
c
p
C
¼ 18 cal=mol K
^
c
p
I
¼ 9 cal=mol K
8.12
4
The elementary liquid-phase reactions
A þ B ! C
C þ B ! D
take place in a cascade of two 100-L CSTRs. A solution
(C
A
in
¼ 2 mol=L and C
B
in
¼ 2 mol=L) at 808C is fed into the first reactor
at a rate of 200 L/min.
a. Derive and plot the reaction and species curves for isothermal operation.
b. Determine the conversion of reactant A and the production rate of pro-
duct C at the exit of the second reactor when both reactors are operated
isothermally.
c. Determine the heating rate of each reactor in part (b).
d. Determine the isothermal HTN of each reactor.
e. Derive and plot the reaction and species curves for adiabatic operation.
f. Determine the conversion of reactant A and the production rate of pro-
duct C at the exit of the second reactor when both reactors are operated
adiabatically.
g. Determine the temperature of each reactor in part (f).
h. Repeat parts (f) and (g) for nonisothermal operation with HTN half of
the isothermal HTN.
Data:At808C, k
1
¼ 0:1L=mol min
1
, k
2
¼ 0:2L=mol min
1
E
a
1
¼ 12,000 cal=mol E
a
2
¼ 16,000 cal=mol
DH
R
1
¼15,000 cal=mol extent DH
R
2
¼10,000 cal=mol extent
Density of the solution ¼ 900 g/L. Heat capacity ¼ 0.8 cal/g8C.
8.13
4
Methane is chlorinated in a gas phase at 4008C to produce mono-, di-, tri-,
and tetrachloromethane. The desired products are CH
2
Cl
2
and CCl
4
. The
374 CONTINUOUS STIRRED-TANK REACTOR
following reactions take place:
CH
4
þ Cl
2
! CH
3
Cl þ HCl
CH
3
Cl þ Cl
2
! CH
2
Cl
2
þ HCl
CH
2
Cl
2
þ Cl
2
! CHCl
3
þ HCl
CHCl
3
þ Cl
2
! CCl
4
þ HCl
The reactions are elementary and the rate constants at 4008Care
k
1
¼ 30 L=mol min k
2
=k
1
¼ 3 k
3
=k
1
¼ 1:5 k
4
=k
1
¼ 0:375
A gas stream containing CH
4
and Cl
2
in the proportion of 1 : 1.2 is fed at
1.2 atm into a CSTR operated at 4008C. The feed rate is 1 mol/min.
a. Derive and plot the reaction and species operating curves.
b. What is the reactor volume needed to maximize the production of
CH
2
Cl
2
?
c. What are the conversions of CH
4
and Cl
2
?
d. What are the production rates of CH
3
Cl, CH
2
Cl
2
, CHCl
3
, and CCl
4
in (b)?
8.14
4
Methanol is produced by the gas-phase reaction, where the following reac-
tions take place:
CO þ 2H
2
! CH
3
OH
CO þ 3H
2
! CH
4
þ H
2
O
Both reactions are second order (each is first order with respect to each reac-
tant) and k
2
/k
1
¼ 1.2. A synthesis gas stream is fed into a CSTR operated
at 4508C and 5 atm. Derive and plot the reaction and species operating
curves for:
a. A feed consisting of 1/3 CO and 2/3H
2
(mole basis).
b. A feed consisting of 50% CO and 50% H
2
(mole basis).
8.15
4
Below is a simplified kinetic model of the cracking of propane to produce
ethylene.
C
3
H
8
! C
2
H
4
þCH
4
r
1
¼k
1
C
C
3
H
8
C
3
H
8
C
3
H
6
þH
2
r
2
¼k
2
C
C
3
H
8
; r
2
¼(k
2
=K
2
)C
C
3
H
6
C
H
2
C
3
H
8
þC
2
H
4
! C
2
H
6
þC
3
H
6
r
3
¼k
3
C
C
3
H
8
C
C
2
H
4
2C
3
H
6
! 3C
2
H
4
r
4
¼k
4
C
C
3
H
6
PROBLEMS 375
C
3
H
6
C
2
H
2
þCH
4
r
5
¼k
5
C
C
3
H
8
; r
5
(k
5
=K
5
)C
C
2
H
2
C
CH
4
C
2
H
4
þC
2
H
2
! C
4
H
6
r
6
¼k
6
C
C
2
H
4
C
C
2
H
2
At 8008C, the values of the rate constants are k
1
¼ 2.341 s
21
, k
2
¼ 2.12
s
21
, K
2
¼ 1000, k
3
¼ 23.63 m
3
/kmol s, k
4
¼ 0.816 s
21
, k
5
¼ 0.305 s
21
,
K
5
¼ 2000, and k
6
¼ 4.06 10
3
m
3
/kmol s. You are asked to design a
CSTR for the cracking of propane to be operated at 2 atm. Plot the reaction
and species operating curves at 8008C.
376 CONTINUOUS STIRRED-TANK REACTOR

9
OTHER REACTOR CONFIGURATIONS
In this chapter, the analysis of chemical reactors is expanded to additional reactor
configurations that are commonly used to improve the yield and selectivity of the
desirable products. In Section 9.1, we analyze semibatch reactors. Section 9.2
covers the operation of plug-flow reactors with continuous injection along their
length. In Section 9.3, we examine the operation of one-stage distillation reactors,
and Section 9.4 covers the operation of recycle reactors. In each section, we first
derive the design equations, convert them to dimensionless forms, and then
derive the auxiliary relations to express the species concentrations and the energy
balance equation.
9.1 SEMIBATCH REACTORS
A semibatch reactor is a batch reactor into which one or more reactants are added
continuously during the operation and no material is withdrawn. Semibatch reactors
are used when it is desirable to maintain a low concentration of one reactant (the
injected reactant). By adding this reactant slowly, as it is being consumed, its con-
centration is low throughout the operation. In some cases an inert species is injected
to the reactor in order to supply (or remove) heat. For convenience, we divide semi-
batch reactors into two categories: reactors with liquid-phase reactions where the
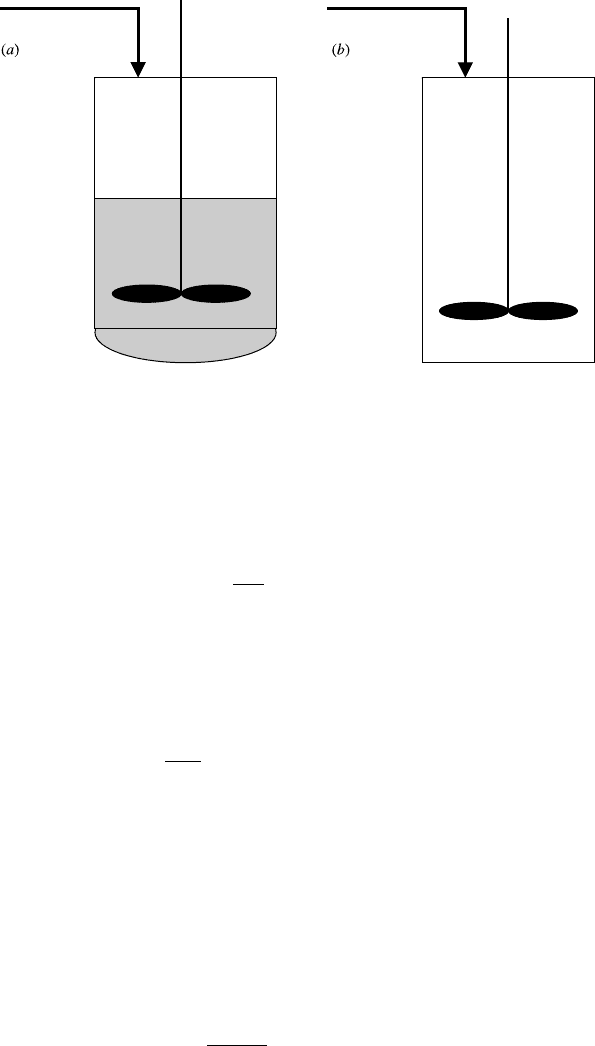
volume of the reacting fluid changes, shown schematically in the Figure 9.1a,
and reactors with gas-phase reactions where the volume does not change, shown
schematically in the Figure 9.1b.
Principles of Chemical Reactor Analysis and Design, Second Edition. By Uzi Mann
Copyright # 2009 John Wiley & Sons, Inc.
377
To derive the design equation of a semibatch reactor, we write a species balance
for any species, say species j, that is not fed continuously into the reactor. Its molar
balance equation is
dN
j
dt
¼ (r
j
)V
R
(t)(9:1:1)
where V
R
(t) is the volume of the reactor at time t. We follow the procedure used in
Chapter 4 to derive the design equation of an ideal batch reactor for the mth-inde-
pendent reaction:
dX
m
dt
¼ r
m
þ
X
n
D
k¼1
a
km
r
k
!
V
R
(t)(9:1:2)
where X
m
(t) is the extent of the mth-independent reaction in operating time t. Note
that Eq. 9.1.2 is identical to the design equation of an ideal batch reactor (Eq. 4.3.8).
The only differences between batch and semibatch operations are the way the reac-
tor volume and the species concentrations vary during the operation.
Consider first a semibatch reactor with liquid-phase reactions. To derive a
relation for the change in the volume of the reactor, we write an overall mass bal-
ance over the reactor:
d(rV
R
)
dt
¼ r
inj
(t)v
inj
(t)(9:1:3)
where v
inj
(t) is the volumetric flow rate of the injected stream, and r
inj
(t) and r are
the densities of the injected stream and the reacting liquid, respectively. For most
Figure 9.1 Semibatch reactors: (a) liquid phase (variable volume) and (b) gas phase
(constant volume).
378 OTHER REACTOR CONFIGURATIONS

liquid-phase reactions, the density of the feed is assumed to be the same as that of
reacting liquid; hence, r(t) ¼ r
inj
(t) ¼ r, and Eq. 9.1.3 reduces to
dV
R
dt
¼ v
inj
(t)(9:1:4)
Since the volumetric injection flow rate v
inj
(t) may vary with time, we have to solve
Eq. 9.1.4 simultaneously with Eq. 9.1.2.
The reactor volume at time t is
V
R
(t) ¼ V
R
(0) þ
ð
t
0
v
inj
(x) dx (9:1:5)
Substituting Eq. 9.1.5 into Eq. 9.1.2, the design equation of a semibatch reactor is
dX
m
dt
¼ r
m
þ
X
n
D
k¼1
a
km
r
k
!
V
R
(0) þ
ð
t
0
v
inj
(x) dx
2
4
3
5
(9:1:6)
To reduce the design equation to dimensionless form, we have to select a refer-
ence state and define dimensionless extents and dimensionless time. The reference
state should apply to all operations, including those with an initially empty reactor,
and should enable us to compare the operation of a semibatch reactor to that of a
batch reactor. Therefore, we select the molar content of the reference state,
(N
tot
)
0
, as the total moles of species added to the reactor. The dimensionless
extent is defined by
Z
m
(t) ;
Extent of the mth-independent reaction at time t
Total number of moles added to the reactor
¼
X
m
(t)
(N
tot
)
0
(9:1:7)
The total number of moles added to a semibatch reactor during the entire
operation is
(N
tot
)
0
; N
tot
(0) þ (N
tot
)
inj
¼ N
tot
(0) þ
ð
t
op
0
v
inj
(x)(C
0
)
inj
dx (9:1:8)
where N
tot
(0) is the total number of moles initially charged to the reactor, (N
tot
)
inj
is
the total moles injected, (C
0
)
inj
is the total concentration of the injected stream, and
t
op
is the total operating time.
The reference volume, V
R
0
, is defined as the total volume of reacting liquid
added to the reactor during the entire operation:
V
R
0
; V
R
(0) þ V
inj
¼ V
R
(0) þ
ð
t
op
0
v
inj
(x) dx (9:1:9)
9.1 SEMIBATCH REACTORS 379
