Mann U. Principles of Chemical Reactor Analysis and Design: New Tools for Industrial Chemical Reactor Operations
Подождите немного. Документ загружается.

time, N
V
(t)=(N
tot
)
0
¼ 0:151 and N
W
(t)=(N
tot
)
0
¼ 0:388, which correspond
to 120.8 and 302.4 mol, respectively. Hence, the operating time of semibatch
is about 8 times longer than that of a batch operation, but more than twice the
amount of product V, and a smaller amount of undesirable product W are
generated.
d. The heating load curve is calculated by Eq. 9.1.50, and is shown in
Figure E9.1.5. For 90% conversion, t
op
¼ 4.4, and Q=[(N
tot
)
0
^
c
p
0
T
0
] ¼
Figure E9.1.3 Comparison of V production—isothermal operation.
Figure E9.1.4 Comparison of W production—isothermal operation.
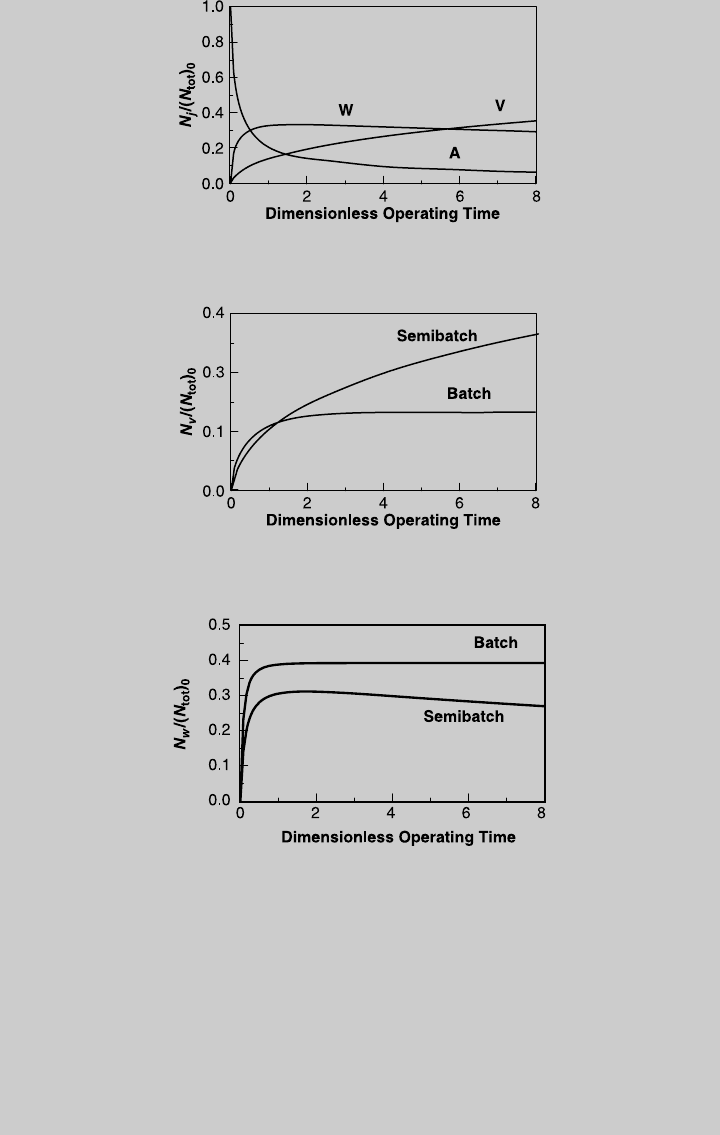
Figure E9.1.2 Species operating curves—semibatch; isothermal operation.
390 OTHER REACTOR CONFIGURATIONS
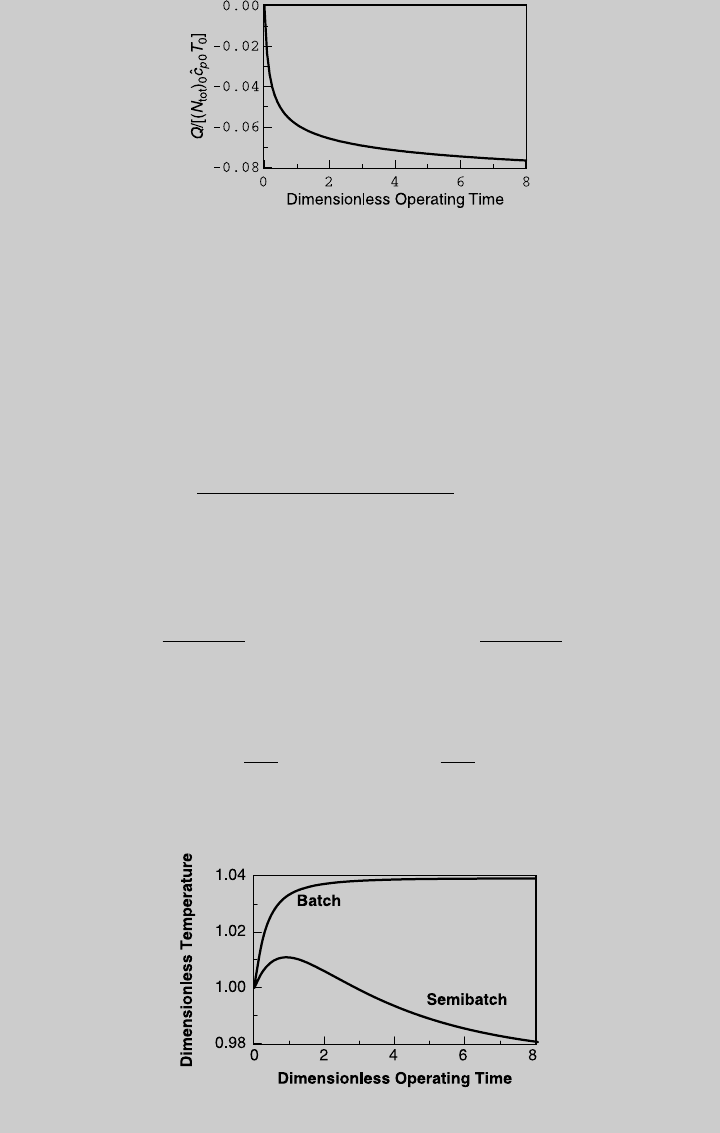
20.0723, which corresponds to 24817.3 kcal. The instantaneous isothermal
HTN is calculated by Eq. 9.1.51.
e. For adiabatic semibatch operation, we have to solve design equations (g) and
(h) simultaneously with the energy balance equation. Using Eq. 9.1.35, the
specific molar heat capacity of the reference state is
^
c
p0
¼
(200 L)(1000 g=L)(1 cal=gK)
800 mol
¼ 250 cal=mol K
The reference temperature is T
0
¼ 333 K, and the dimensionless heats of
reactions are
DHR
1
¼
DH
R
1
(T
0
)
T
0
^
c
p
0
¼0:0961 DHR
2
¼
DH
R
2
(T
0
)
T
0
^
c
p
0
¼0:1441
The dimensionless activation energies of the two chemical reactions are
g
1
¼
E
a
1
RT
0
¼ 13:6 g
2
¼
E
a
2
RT
0
¼ 18:13
Figure E9.1.5 Heat-transfer curve—semibatch; isothermal operation.
Figure E9.1.6 Temperature curve—adiabatic operation.
9.1 SEMIBATCH REACTORS 391
For adiabatic operation, HTN ¼ 0. For uniform injection rate, using
Eq. 9.1.39, the correction factor of the heat capacity is
CF(Z
m
, u) ¼
t
t
op
(r)
Since T
inj
¼ T
0
(u
inj
¼ 1), the energy balance equation (Eq. 9.1.38) reduces to
du
dt
¼
t
op
t
1
t
op
V
inj
V
R
0
(1 u) DHR
1
dZ
1
dt
DHR
2
dZ
2
dt
(s)
We solve (s) simultaneously with (g) and (h) for different values of operating
times, t
op
, subject to the initial conditions that at t ¼ 0, Z
1
¼ Z
2
¼ 0, and
u ¼ 1, and we determine Z
1
, Z
2
, and u for each t
op
. Once we obtain the reac-
tion operating curves (Z
1
and Z
2
versus t
op
), we use Eq. 9.1.15 to obtain the
species operating curves. Figure E9.1.7 shows the curve for product V and
compares it to that of an adiabatic batch reactor. Figure E9.1.8 shows the
curve for product W and compares it to that of an adiabatic batch reactor.
Figure E9.1.6 shows the temperature curve and compares it to that of a
Figure E9.1.7 Comparison of V production—adiabatic operation.
Figure E9.1.8 Comparison of W production—adiabatic operation.
392 OTHER REACTOR CONFIGURATIONS

batch reactor. Note that since the activation energy of the second reaction is
higher than that of the first, and both reactions are exothermic, isothermal
operation is preferable.
For a batch reactor (constant volume), the design equations (Eq. 7.1.1) are
dZ
1
dt
¼ r
1
t
cr
C
0
(t)
dZ
2
dt
¼ r
2
t
cr
C
0
(u)
Using Eq. 6.1.5 to express the species concentrations, the reaction rates are
r
1
¼ k
1
(T
0
)C
0
(1 Z
1
2Z
2
)e
g
1
(u1)=u
(v)
r
2
¼ k
2
(T
0
)C
2
0
(1 Z
1
2Z
2
)
2
e
g
2
(u1)=u
(w)
Substituting (v), (w), and (f) into (t) and (u), the design equations become
dZ
1
dt
¼ (1 Z
1
2Z
2
)e
g
1
(u1)=u
(x)
dZ
2
dt
¼
k
2
(T
0
)C
0
k
1
(T
0
)
(1 Z
1
2Z
2
)
2
e
g
2
(u1)=u
(y)
For isothermal operation, u ¼ 1, and (x) and (y) are solved simultaneously.
For adiabatic operation, the energy balance equation is
du
dt
¼DHR
1
dZ
1
dt
DHR
2
dZ
2
dt
(z)
We solve (x), (y), and (z) for different values of operating times, t
op
, subject
to the initial conditions that t ¼ 0, Z
1
¼ Z
2
¼ 0, and u ¼ 1. The species
curves of the product V, and the undesirable product W, are compared in
the figures above.
Example 9.2 Valuable product V is produced in a semibatch reactor where the
following simultaneous, liquid-phase chemical reactions take place.
Reaction 1: A þ B ! V
Reaction 2: 2A ! W
The rate expressions are r
1
¼ k
1
C
A
C
B
, and r
2
¼ k
2
C
A
2
. The reactor is initially
charged with a 200-L solution of reactant B with a concentration of C
B
(0) ¼ 3
mol/L, and 200-L solution of reactant A (C
A
¼ 3 mol/L) is fed continuously
9.1 SEMIBATCH REACTORS 393

into the reactor at a constant rate. Both the injected stream temperature and the
initial reactor temperature are 608C.
a. Show qualitatively the advantage of semi-batch operation over batch
operation.
b. Derive the design equations, and plot the reaction and species curves for
isothermal semi-batch operation. Compare the product curves to those of
isothermal batch operation. What is the operating time for each mode of
operation for 80% conversion of reactant A? What is the amount of product
V and W generated in each mode?
c. Derive the design equations, and plot the reaction and species curves for
adiabatic semi-batch operation. Compare the product curves to those of
adiabatic batch operation with T(0) ¼ 608C, and T
inj
¼ 508C.
d. Derive the design and energy balance equations for batch operation.
Data:At608C, k
1
¼ 0:02 L=mol min
1
, k
2
¼ 0:04 L=mol min
1
DH
R
1
¼9000 cal=mol DH
R
2
¼13,000 cal=mol
E
a
1
¼ 12,000 cal=mol E
a
2
¼ 20,000 cal=mol
The heat capacity of the solution is
c
p
¼ 0:9 cal g
1
K
1
, and its density is
0.85 kg/L.
Solution
a. To identify the preferable reactor operation mode, we write the ratio of the
formation rates of the desired and undesired products:
r
V
r
W
¼
k
1
(T)C
A
C
B
k
2
(T)C
2
A
¼
k
1
k
2
C
B
C
A
exp
E
a
1
E
a
2
RT
Hence, we would like to maintain high concentration of reactant B and low
concentration of reactant A. This is achieved by charging the reactor with
reactant B and then injecting reactant A slowly. Also, since E
a
1
, E
a
2
,itis
preferable to operate the reactor at a lower temperature.
b. The stoichiometric coefficients of the chemical reactions are
s
A
1
¼1 s
B
1
¼1 s
V
1
¼ 1 s
W
1
¼ 0 D
1
¼1
s
A
2
¼2 s
B
2
¼ 0 s
V
2
¼ 0 s
W
2
¼ 1 D
2
¼1
Since each reaction has a species that does not appear in the other, the two
reactions are independent, and there is no dependent reaction. We select
the total amount introduced into the reactor during the operation as the
394 OTHER REACTOR CONFIGURATIONS

reference state. Hence, V
R
(0) ¼ 200 L, and, for constant injection rate,
(V
inj
)
0
¼ v
0
t
op
¼ 200 L, and
V
R
0
¼ V
R
(0) þ (V
inj
)
0
¼ 200 þ 200 ¼ 400 L
Since only reactant B is charged initially, N
tot
(0) ¼ N
B
(0) ¼ 600 mol,
N
A
(0) ¼ 0, N
V
(0) ¼ 0, and N
W
(0) ¼ 0. Also, since only reactant A is
injected, (N
tot
)
inj
¼ (C
A
)
inj
(V
inj
)
0
¼ 600 mol, and the total molar content of
the reference state is
(N
tot
)
0
¼ N
tot
(0) þ (N
inj
)
0
¼ 1200 mol
The reference concentration is
C
0
¼
(N
tot
)
0
V
R
0
¼
1200 mol
400 L
¼ 3 mol=L
We write Eq. 9.1.20 for each independent reaction:
dZ
1
dt
¼ r
1
1
2
1 þ
t
t
op
t
cr
C
0
0 t t
op
(a)
dZ
2
dt
¼ r
2
1
2
1 þ
t
t
op
t
cr
C
0
0 t t
op
(b)
Using Eq. 9.1.21, the concentrations of the reactants in the reactor are
C
A
(t) ¼ C
0
2t
op
t
op
þ t
(0:5)
t
t
op
þ (1)Z
1
(t) þ(2)Z
2
(t)
(c)
C
B
(t) ¼ C
0
2t
op
t
op
þ t
[(0:5) þ (1)Z
1
(t)] (d)
The rates of the two reactions are
r
1
¼ k
1
(T
0
)C
2
0
2t
op
t
op
þ t
2
(0:5)
t
t
op
Z
1
2Z
2
(0:5 Z
1
)e
g
1
(u1)=u
(e)
r
2
¼ k
2
(T
0
)C
2
0
2t
op
t
op
þ t
2
(0:5)
t
t
op
Z
1
2Z
2
2
e
g
2
(u1)=u
(f)
We select Reaction 1 as the leading reaction; hence, the characteristic reaction
time is
t
cr
¼
1
k
1
(T
0
)C
0
¼ 16:67 min (g)
9.1 SEMIBATCH REACTORS 395

Substituting (e), (f), and (g) into (a) and (b), the design equations reduce to
dZ
1
dt
¼
2t
op
t
op
þt
(0:5)
t
t
op
Z
1
2Z
2
(0:5Z
1
)e
g
1
(u1)=u
0 t t
op
(h)
dZ
2
dt
¼
k
2
(T
0
)
k
1
(T
0
)
2t
op
t
op
þt
(0:5)
t
t
op
Z
1
2Z
2
2
e
g
2
(u1)=u
0 t t
op
(i)
For isothermal operation, u ¼ 1, and the design equations reduce to
dZ
1
dt
¼
2t
op
t
op
þt
(0:5)
t
t
op
Z
1
2Z
2
(0:5Z
1
)0t t
op
(j)
dZ
2
dt
¼
k
2
(T
0
)
k
1
(T
0
)
2t
op
t
op
þt
(0:5)
t
t
op
Z
1
2Z
2
2
0 t t
op
(k)
We solve ( j) and (k) for different values of operating times, t
op
, subject to the
initial conditions that at t ¼ 0, Z
1
¼ Z
2
¼ 0, and then determine the final
extents for each t
op
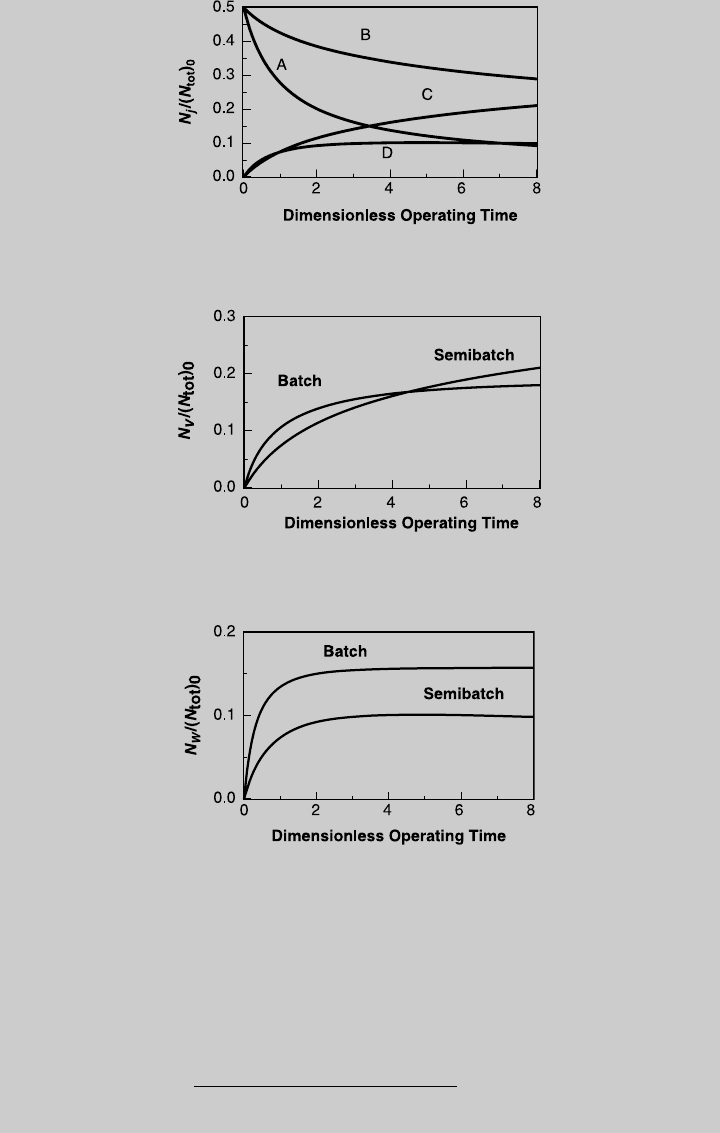
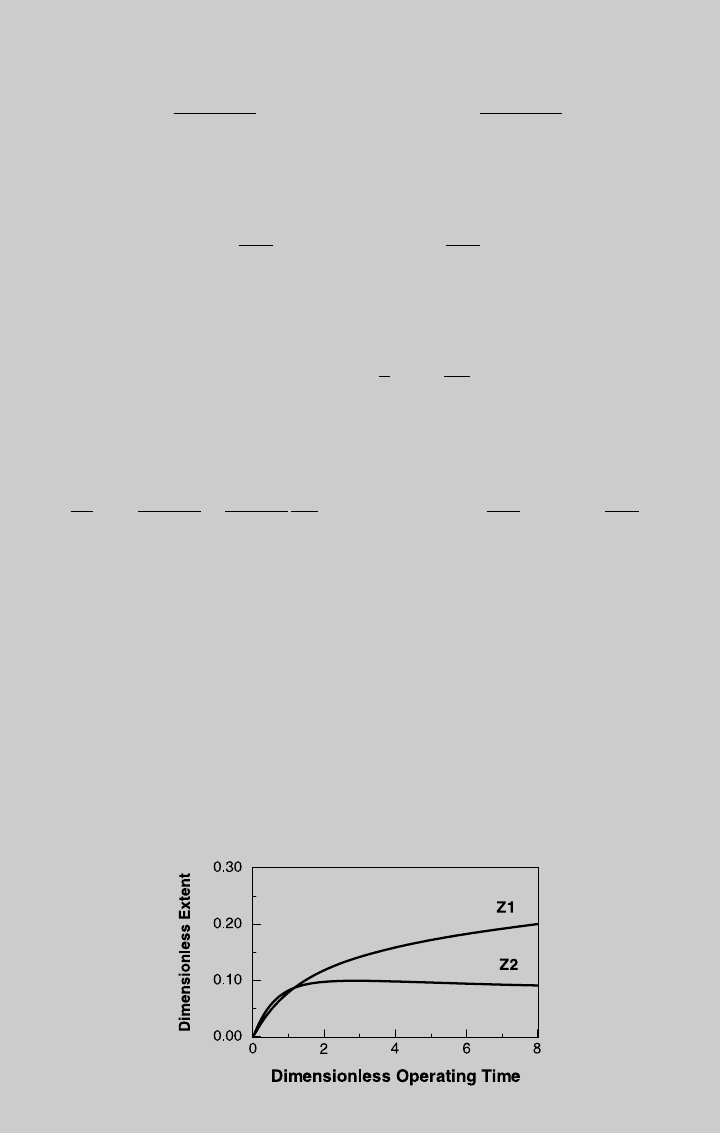
. Figure E9.2.1 shows the reaction curves for isothermal
semibatch operation. Using Eq. 9.1.15, we determine the species curves,
shown in Figure E9.2.2. The curves of products V and W are compared to
those of isothermal batch operation (derived below) in Figures E9.2.3 and
E9.2.4, respectively.
For semibatch operation, 80% conversion of reactant A (N
A
/
(N
tot
)
0
¼ 0.1), is reached at t
op
¼ 6.95, which corresponds to 116 min. At
that operating time, Z
1
¼ 0.180, Z
2
¼ 0.08, N
V
/(N
tot
)
0
¼ 0.20, and N
W
/
(N
tot
)
0
¼ 0.10. Hence, 240 mol of product V and 120 mol of product W
were produced. For batch operation, 80% conversion is reached at
t
op
¼ 1.26, which corresponds to 21 min. At that operating time, N
V
/
(N
tot
)
0
¼ 0.118, and N
W
/(N
tot
)
0
¼ 0.141. Hence, 141.6 mole of product V
and 169.2 mol of product W were produced.
Figure E9.2.1 Reaction operating curves; isothermal semibatch operation.
396 OTHER REACTOR CONFIGURATIONS
c. For nonisothermal semibatch operations, we have to solve design equations
(h) and (i) simultaneously with the energy balance equation. Using
Eq. 9.1.35,
^
c
p0
¼
(400 L)(850 g=L)(0 :9 cal=gK)
(1200 mol)
¼ 255 cal=mol K
Figure E9.2.2 Species operating curves; isothermal semibatch operation.
Figure E9.2.3 Comparison with batch reactor—product V (isothermal operation).
Figure E9.2.4 Comparison with batch reactor—product W (isothermal operation).
9.1 SEMIBATCH REACTORS 397
The reference temperature is T
0
¼ 333 K, and the dimensionless heats of
reactions are
DHR
1
¼
DH
R
1
(T
0
)
T
0
^
c
p
0
¼0:106 DHR
2
¼
DH
R
2
(T
0
)
T
0
^
c
p
0
¼0:153
The dimensionless activation energies of the two reactions are
g
1
¼
E
a
1
RT
0
¼ 18:14 g
2
¼
E
a
2
RT
0
¼ 30:21
For adiabatic operation, HTN ¼ 0. Using Eq. 9.1.39,
CF(Z
m
, u) ¼
1
2
1 þ
t
t
op
(l)
The energy balance equation (Eq. 9.1.37) reduces to
du
dt
¼
2t
op
t
op
þ t
(N
tot
)
inj
(N
tot
)
0
1
t
op
(u
inj
u) þ DHR
1
dZ
1
dt
þ DHR
2
dZ
2
dt
(m)
In this case, u
inj
¼ 0.97. We solve (m) simultaneously with (h) and (i) for
different values of t
op
, subject to the initial conditions that at t ¼ 0,
Z
1
¼ Z
2
¼ 0, u ¼ 1, and then determine the extents and u at each t
op
.
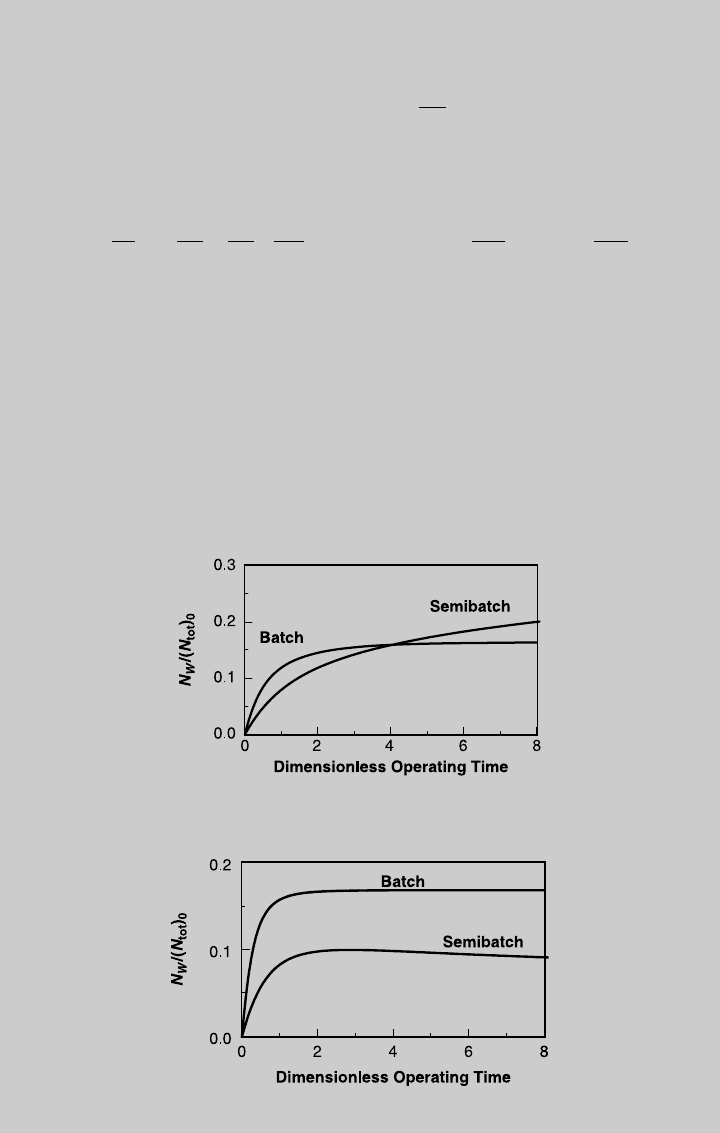
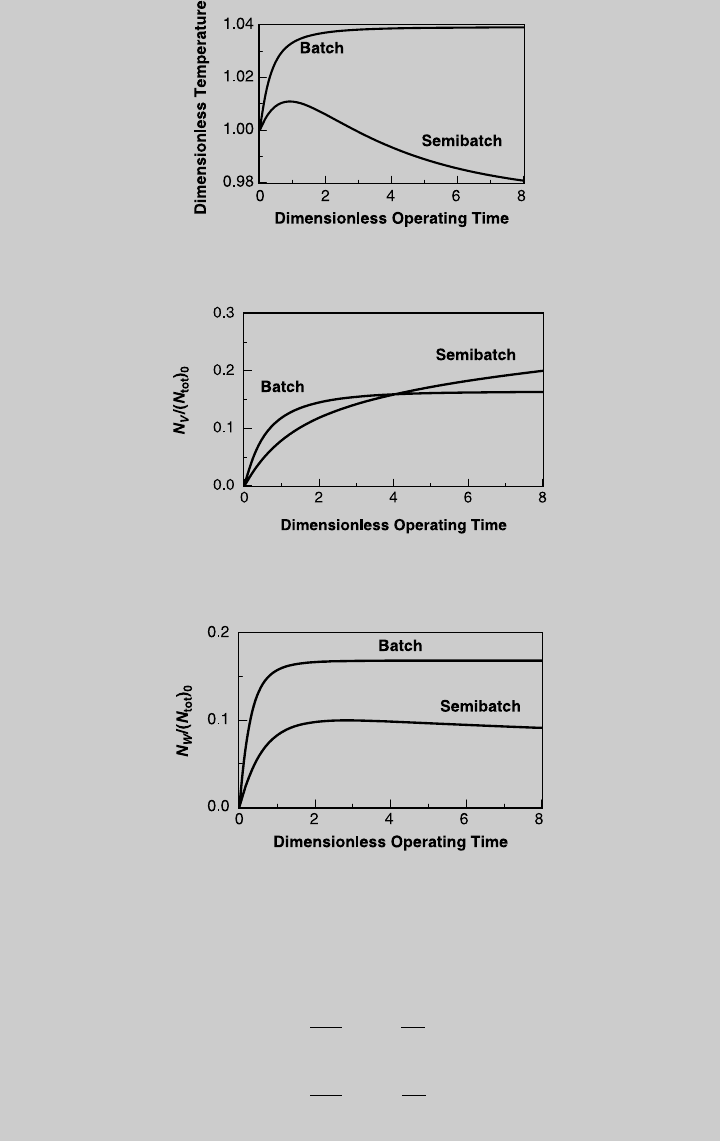
Figure E9.2.5 shows the reaction operating curves and Figure E9.2.6
shows the temperature curve. Using Eq. 9.1.15 we determine the
species curves. The curves of products V and W are shown in Figures
E9.2.7 and E9.2.8, respectively, and are compared to those of adiabatic
batch operation.
Figure E9.2.5 Reaction curves—semibatch; adiabatic operation.
398 OTHER REACTOR CONFIGURATIONS
d. For a constant-volume batch reactor, the design equations (Eq. 6.1.1) are
dZ
1
dt
¼ r
1
t
cr
C
0
(n)
dZ
2
dt
¼ r
2
t
cr
C
0
(o)
Figure E9.2.6 Temperature curve; adiabatic operation.
Figure E9.2.8 Comparison with batch reactor—W production; adiabatic operation.
Figure E9.2.7 Comparison with batch reactor—V production; adiabatic operation.
9.1 SEMIBATCH REACTORS 399
