Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.

Alloys 103
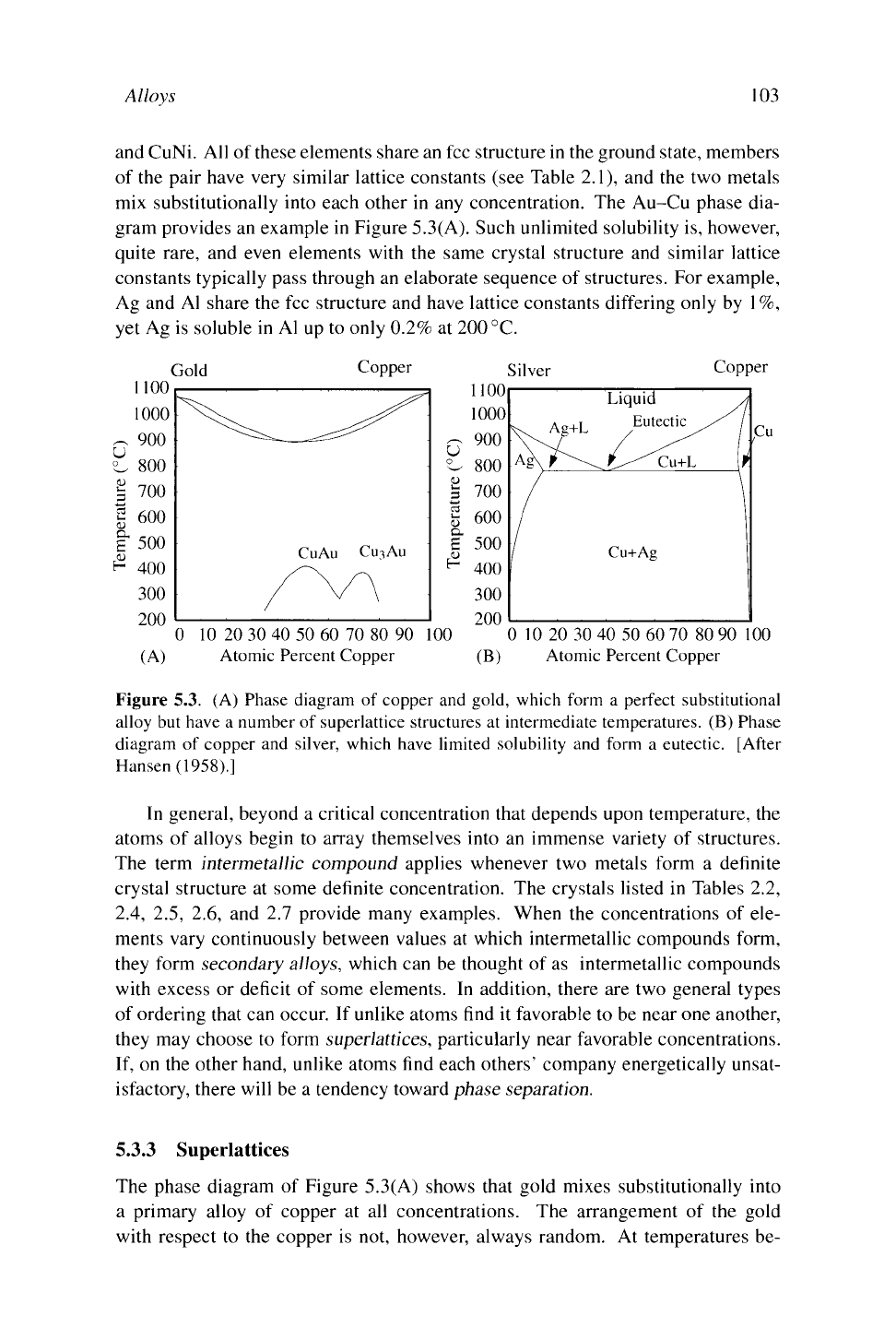
and CuNi. All of these elements share an fee structure in the ground state, members
of the pair have very similar lattice constants (see Table 2.1), and the two metals
mix substitutionally into each other in any concentration. The Au-Cu phase dia-
gram provides an example in Figure 5.3(A). Such unlimited solubility is, however,
quite rare, and even elements with the same crystal structure and similar lattice
constants typically pass through an elaborate sequence of structures. For example,
Ag and Al share the fee structure and have lattice constants differing only by 1%,
yet Ag is soluble in Al up to only 0.2% at 200 °C.
Gold
Copper
Silver
Copper
(A)
10 20 30 40 50 60 70 80 90
Atomic Percent Copper (B)
0 20 30 40 50 60 70 80 90 100
Atomic Percent Copper
Figure 5.3. (A) Phase diagram of copper and gold, which form a perfect substitutional
alloy but have a number of superlattice structures at intermediate temperatures. (B) Phase
diagram of copper and silver, which have limited solubility and form a eutectic. [After
Hansen (1958).]
In general, beyond a critical concentration that depends upon temperature, the
atoms of alloys begin to array themselves into an immense variety of structures.
The term intermetallic compound applies whenever two metals form a definite
crystal structure at some definite concentration. The crystals listed in Tables 2.2,
2.4, 2.5, 2.6, and 2.7 provide many examples. When the concentrations of ele-
ments vary continuously between values at which intermetallic compounds form,
they form secondary alloys, which can be thought of as intermetallic compounds
with excess or deficit of some elements. In addition, there are two general types
of ordering that can occur. If unlike atoms find it favorable to be near one another,
they may choose to form superlattices, particularly near favorable concentrations.
If, on the other hand, unlike atoms find each others' company energetically unsat-
isfactory, there will be a tendency toward phase separation.
5.3.3 Superlattices
The phase diagram of Figure 5.3(A) shows that gold mixes substitutionally into
a primary alloy of copper at all concentrations. The arrangement of the gold
with respect to the copper is not, however, always random. At temperatures be-

104
Chapter 5. Beyond Crystals
low 400 °C, X-ray diffraction off copper-gold mixtures, with three parts copper
to one part gold, shows a new set of diffraction peaks not present above 400 °C.
Furthermore, the strength of these peaks depends upon the way the mixture is pre-
pared. Bragg and Williams (1934) report that lowering of temperature from above
400 °C to 270 °C in a few seconds gives peaks no different from those present
above 400 °C—the mixture has been quenched. By cooling the solid solution in-
stead over a period of several days—annealing it—new peaks form. Following the
methods of Chapter 3, the locations of the new peaks can be used to show that the
copper and gold have formed a crystalline compound in which gold and copper are
arrayed as shown in Figure 5.4, called a
superlattice.
Many other combinations of
metals produce similar superlattices, usually when mixed in ratios of 1:1 or 3:1.
Figure 5.4. (A)
A
3:1
mixture of copper and gold has the equilibrium superlattice structure
depicted here below a temperature of
400
°C.
(B) Superlattice of copper and gold in equal
mixtures.The lattice constant c is 7% smaller than a.
The structures of superlattices are no less varied than the structures of inter-
metallic compounds, and all the crystal structures of Chapter 2 are possible. There
is no distinction in principle between intermetallic compounds and superlattices,
except that the latter lose their order at a definite transition temperature well below
the melting point of the crystal. Among elements mixed in 3:1 ratios, the structure
displayed in Figure 5.4(A) is common, while for elements mixed in proportion
of 1:1, the CsCl structure (Figure 2.8) frequently occurs, as in the cases of Cu-
Zn (/3-brass), CuBe, CuPd, AgMg, AgZn, AgCd, AuNi, NiAl, and FeCo. Phase
transformations in superlattices are studied in more detail in Section
24.5.1.
Many other structures are, however, possible. Mixed in a ratio of 1:1, copper
and gold form a superlattice involving alternating planes of copper and gold atoms,
as shown in Figure 5.4(B). When the atoms arrange themselves in this way, the
whole lattice contracts by 7% along the c axis, so the symmetry of the crystal
changes from cubic to tetragonal. Such macroscopic changes in the shape of the
crystal are another frequent feature of superlattices.
5.3.4 Phase Separation
Iron carbide, Fe3C, is a stable compound. Suppose that only 3 atomic percent of
carbon is mixed in with iron. The 3 carbon atoms out of 100 can combine with 9
Alloys
105
iron atoms to form 12 atoms' worth of Fe3C, leaving 88% iron. This process re-
quires the physical separation of
the
proper mixture of carbon and iron from the rest
of the soup, a rearrangement that cannot always occur quickly or spontaneously.
Because phase separation is based upon principles of thermodynamics much
more general than their application
in
alloy systems,
a
slightly abstract view
is
appropriate.
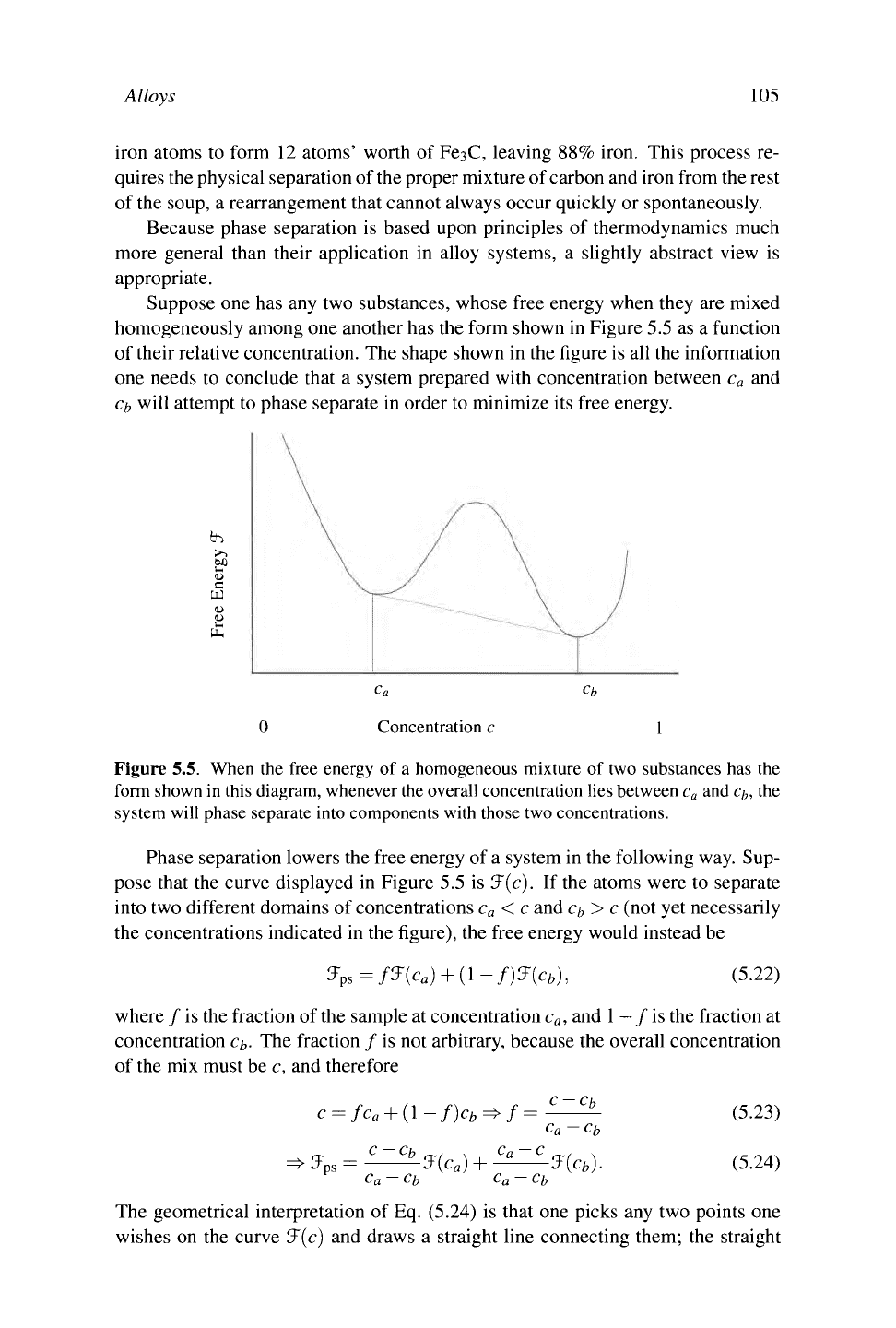
Suppose one has any two substances, whose free energy when they are mixed
homogeneously among one another has the form shown in Figure 5.5 as a function
of their relative concentration. The shape shown in the figure is all the information
one needs to conclude that
a
system prepared with concentration between c
a
and
c/,
will attempt to phase separate in order to minimize its free energy.
>>
EP
<u
c
ω
<u
ω
Ca
C
h
0 Concentration c
1
Figure 5.5. When the free energy of a homogeneous mixture of two substances has the
form shown in this diagram, whenever
the
overall concentration lies between c
a
and c/„ the
system will phase separate into components with those two concentrations.
Phase separation lowers the free energy of a system in the following way. Sup-
pose that the curve displayed in Figure 5.5 is iF(c). If the atoms were to separate
into two different domains of concentrations c
a
< c and
Q,
>
c (not yet necessarily
the concentrations indicated in the figure), the free energy would instead be
3
r
ps
= f3
r
(c
a
) + (l-f)?(c
b
), (5.22)
where
/
is the fraction of the sample at concentration c
a
, and
1 —
/
is the fraction at
concentration
Q,.
The fraction
/
is not arbitrary, because the overall concentration
of the mix must be c, and therefore
c =
fc
a
+
(l-f)c
b
^f=^^-
(5.23)
Ca-Cb
=*
?ps
=
^^Hc
a
)
+
^-2(c
b
).
(5.24)
The geometrical interpretation of Eq. (5.24) is that one picks any two points one
wishes on the curve 3(c) and draws
a
straight line connecting them; the straight

106
Chapter 5. Beyond Crystals
line describes free energy of phase separation between those two points. The points
c
a
and
Cb
in Figure 5.5 have been chosen so that this construction resulted in the
lowest possible free energy.
A typical phase diagram is largely occupied with regions of phase separation.
Consider the diagram in Figure
5.3(B)
for copper and silver. Regions Ag and Cu
are primary phases of fee metal with substitutional impurities. The region marked
"Liquid" is also a homogeneous mixture. Everywhere else, the two metals phase
separate. The solid lines then indicate the concentrations c
a
and
Q,
as a function of
temperature. For example, at 700
°C,
separation occurs between the two concen-
trations that have been marked. In the region denoted "Ag+liquid" a primary alloy
of silver coexists with a liquid containing a greater percentage of copper. The line
describing the composition of the liquid is called the liquidus, while the line de-
termining the composition of the solid is the solidus. The point marked "Eutectic"
has technological significance, since it provides the lowest possible temperature at
which the two metals mix in the molten state. As soon as the two metals are cooled
below the eutectic temperature, however, they begin to phase separate, so if the
goal is a homogeneous material there is a race against time.
Binary phase diagrams have a number of characteristic shapes that appear
bizarre at first glance, but which have a rather natural explanation in terms of sim-
ple assumptions concerning the free energies of solid and liquid phases. The idea
is best illustrated geometrically and is shown in Figure 5.6.
5.3.5 Nonequilibrium Structures in Alloys
A material composed of large numbers of small crystalline regions of different
orientations is said to be built out of
grains,
and the interfaces between them are
grain boundanes. These boundaries may appear on scales ranging from tens of
nanometers to meters. When the crystallites are at the small end of the scale, one
calls the material microcrystalline. Frequently in metals, the crystalline regions are
on the micron scale, and the materials are called polycrystalline. In sea ice, grains
may grow to scales of
meters.
The orientations of adjoining crystalline regions are
fairly random, and if one takes a two-dimensional slice through such a solid, the
grain boundaries form a characteristic network, with the grain boundaries meeting
in vertices, as shown in Figure 5.7. The manner in which grains grow is the subject
of Problem 6.
A type of grain boundary that occurs in substitutional alloys is the antiphase
boundary, which is a grain boundary where the orientations of the crystals on the
two sides are the same, but there is a shift of phase in the lattice as one crosses
the boundary. Antiphase boundaries can form snaking labyrinthine structures of
great complexity [Figure 5.8(A)]. As the concentration of one element in another
increases, one tends to get crystals dominated by one element embedded in crystals
dominated by the other element. For example, one can have small crystals of N13AI
sitting in a background of nearly pure Al. The forms that these intermixed crystals
can take depend upon the dynamical processes by which they form. In the simplest
Alloys 107
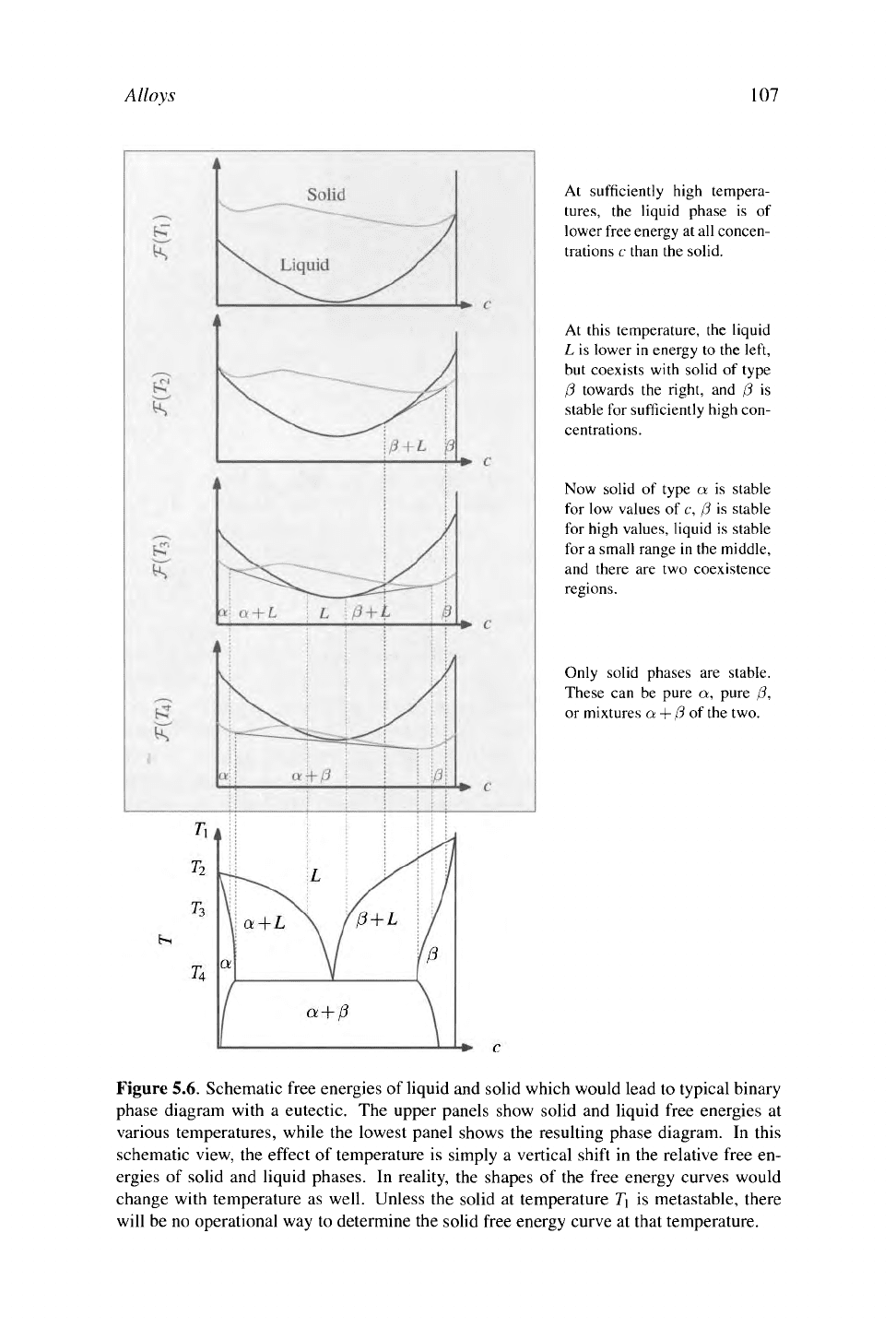
At sufficiently high tempera-
tures,
the liquid phase is of
lower free energy at all concen-
trations c than the solid.
At this temperature, the liquid
L is lower in energy to the left,
but coexists with solid of type
0 towards the right, and 0 is
stable for sufficiently high con-
centrations.
Now solid of type a is stable
for low values of c, 0 is stable
for high values, liquid is stable
for a small range in the middle,
and there are two coexistence
regions.
Only solid phases are stable.
These can be pure a, pure 0,
or mixtures a + 0 of the two.
Figure 5.6. Schematic free energies of liquid and solid which would lead to typical binary
phase diagram with a eutectic. The upper panels show solid and liquid free energies at
various temperatures, while the lowest panel shows the resulting phase diagram. In this
schematic view, the effect of temperature is simply a vertical shift in the relative free en-
ergies of solid and liquid phases. In reality, the shapes of the free energy curves would
change with temperature as well. Unless the solid at temperature T\ is metastable, there
will be no operational way to determine the solid free energy curve at that temperature.

108 Chapter 5. Beyond Crystals
Figure 5.7. Transmission electron micrograph of the grain structure of alumina. Grains
are approximately
1
μπι across. [Source: B. Hockey, National Institutes of Science and
Technology.]
case,
one has spheres of one type sitting in a background of the other type. The
spheres can be replaced by rods or plates, or can be sufficiently dense that they form
an interconnected network. The structures can include treelike dendritic shapes
[Figure 5.8(B)], arrays of parallel fingers, and bands or stripes.
5.3.6 Dynamics of Phase Separation
Any alloys heated sufficiently form a homogeneous liquid mixture. Upon cooling,
the mixture will typically remain homogeneous for a time, even if a phase sepa-
rated state is ultimately of lower free energy. In some cases, the initial process
of breaking apart into spatially separated regions of different phases happens eas-
ily, because the homogeneous state is unstable, and the result is called spinodal
decomposition. It can also happen that the cooled homogeneous state is stable
against small fluctuations, and only large rare fluctuations can disturb the situation.
The appearance of new domains by such fluctuations is called nucleation, and is
reviewed by Wu (1997) and Kelton (1991). As shown in Figure 5.8, phase sepa-
ration during the cooling of solid solutions can produce exceedingly complicated
spatial patterns. The patterns have an intrinsic aesthetic appeal, although from a
technological point of view they are usually undesirable, and one reason to under-
stand the physics underlying them is to prevent them from occurring.
The basic equation underlying the dynamics of phase separation is simply the
law of diffusion, presented in Section 5.2.2. The change in concentration with time
following from Eq. (5.1) is
β The concentration c is often taken to be a linear func-
Z__ _ 'Y)\7^r tion of actual atomic concentrations; for example, when Γ5 25Ì
ßf phase separating between c
a
and
Q,,
one might take c
—>
\ · /
(c-C
a
)/(c
b
-C
a
).
The diffusion equation, (5.25), appears innocent, but, when coupled with ap-
propriate boundary conditions, it is capable of producing the sort of complexity that
appears in Figure 5.8. As an illustration of how it functions, consider the problem
of a spherical droplet of
carbide,
carbon concentration c
a
, growing in a background

Allovs 109
Figure 5.8. (A) Antiphase boundaries in FÎ76AI23. The boundaries mark the dividing
line between two chemically identical regions, but where the placement of atoms is out
of synchrony. [Source: Allen and Cahn (1979), p. 1093.] (B) Dendrite formed during
solidification of stainless steel (F^oNiisCris). (Courtesy of L. A. Boatner, J. Gardner, and
D.
Corrigan, Oak Ridge National Laboratory. )
mixture of iron and carbon whose overall carbon concentration is c^ > c
a
. Car-
bon atoms flow toward the droplet, because phase separation is thermodynamically
favorable. The simplest calculations employ the quasi-static approximation. This
approximation requires the time it takes for carbon atoms actually to join the drop
and make it grow to be long compared to the time for the concentration of carbon
outside the drop to converge to a steady state. In this case, the time variation of
c(r, t) can be neglected, and Eq. (5.25) becomes Laplace's equation
V c[r) = 0 Place the origin of a system of polar coordi- (5.26)
nates at the center of the droplet.
with boundary conditions
c(R) = c
a
and lim c(r) — c^. (5.27)
The unique solution of Eq. (5.26) satisfying (5.27) is
p
c{r) = c
0O
+ -(c
a
-c
oo
) (5.28)
r
and from Eq. (5.1) the flux of material into the drop is
ATTR
2
T>——
-. Take the gradient of Eq. (5.28) at« = r, and (5-29)
R multiply by the surface area of the drop.
Suppose that for each unit of concentration entering the drop, the volume of the
drop changes by v. Then the drop changes in size according to
j
t
~R
3
=v4irR'D(c
00
-c
a
)
(5.30)
• VD,
=>R=—{coc-c
a
) (5.31)
K
RoiJ2vT>(c
00
-c
a
)t. (5.32)

110 Chapter 5. Beyond Crystals
The radius
of
the drop grows
as the
square root
of
time.
This calculation oversim-
plifies even
the
problem
of
growing drops.
It
neglects
the
fact that
the
surface
of
each drop exerts
a
pressure
on the
material inside, like
the
skin
of a
balloon, that
causes very small drops
to
shrink rather than grow.
It
neglects
the
fact that after
many drops have grown,
the
background concentration
CQO
of
carbon must dimin-
ish. Taking these effects into account, Lifshitz
and
Slyozov (1961) showed that
the
average size
of
drops increases
as the
cube root
of
time,
not the
square root.
The reason that
Eq.
(5.25)
is
able
to
exhibit complicated behavior
is
that
the
shape
of the
region where
the
equation applies
is
being made
to
depend upon
the
concentration field;
the
coupled motion
of the
boundaries
and
concentration field
is
a
complicated nonlinear problem. Further discussion
of
such pattern-forming
problems
is
provided
by
Langer (1980).
5.4 Simulations
As
the
power
of
computation increases,
it is
gradually becoming possible
to
imag-
ine computing
the
evolution
of
structures such
as
shown
in
Figure
5.8,
either from
partial differential equations
or by
starting down
at the
atomic level.
Two
impor-
tant methods
for
atomic scale calculation
are
Monte
Carlo
and
molecular dynam-
ics.
They
are
very similar
in
conception. Each method treats atoms
as
classical
interacting particles,
and
each watches them evolve
in a
fashion meant
to
mimic
the actual evolution they might display
in
nature.
The
methods differ only
in the
detailed manner that they employ
to
move atoms about.
5.4.1 Monte Carlo
The Monte Carlo method shuffles atoms about randomly, like
the
cards
in the
casi-
nos from which
it
takes
its
name.
The
idea behind
the
method
is
that
if a
solid
is
at some temperature
T,
then
the
probability
of
its atoms adopting
a
position
so
that
their energy
is E is
exp[—/?£], where
ß = 1/kßT. As a
corollary,
it
follows that
if
one
has any two
states
of
the system whose energy differs
by SE,
then
the
relative
probability that these
two
states
be
occupied
is
exp[—ßSE}.
So suppose one has
a
collection of N atoms
at
7\
. .
.
?N,
and an
energy function
E{7\
... ?/v).
What exactly this energy functional might
be is
discussed further
in
Section
11.8. In brief, the
Monte Carlo method chooses
an
atom randomly from
this collection,
and
randomly moves
it a
small distance.
If the
result
of
this small
move
is to
lower
the
energy
of the
system,
the
move
is
always accepted.
If the
move raises
the
energy
of
the system
by an
amount SE, then
the
move
is
accepted
with probability exp[—ßSE}.
Suppose
one
wants
to
compute
the
average value
in
thermal equilibrium
of
any
function
g(r\ . . . r#) of
particle positions.
The
function
g
could
be the
energy,
or
anything else.
The
Monte Carlo method proceeds
as
follows:

Simulations 111
1.
Begin with a collection of particles whose locations are known and whose
energy E(r\ ... r#) = £1
nas
been computed. Compute g(r\ . . . r#) and
store the result in a variable G.
2.
Choose one of these particles at random. Call the particle chosen particle /.
3.
Create a random displacement vector. One way to do this is to choose three
random numbers p\ . . . pi lying in [0, 1] and to form the vector
Ξ =
2a{
Pi
- 1/2, p
2
- 1/2, p
3
- 1/2), (5.33)
with a setting the length scale. A natural choice of a is a typical interparticle
spacing, although final results should not depend upon the choice of a.
4.
Compute the energy difference,
δε = ε(?
ι
...η+Ε...Γ
Ν
)-ε
ι
. (5.34)
When particles interact only with near neighbors, it will always be possible to
compute this change in energy more efficiently than by computing the energy
from scratch for all particles in the system.
5.
Check whether δΖ is positive or negative. If
<5£
is negative, replace r/ by r/ + Ξ
and return to step 1. In either case, add the new value of
g(T\
. . .
?N)
to G.
6. However, if
δΕ
is positive decide randomly whether to allow particle i to move
or not. Pick a new random number p E [0,
1],
and compare p to exp[—βδ£]. If
p is greater than this Boltzmann factor, then leave ?/ where it is and return to
step 1. However, if p is less, set ?/ to η + Ξ before returning to step 1, despite
the fact that this move raises the energy of the system. Once again, add the
new value of g(r\ . . . r#) to G.
At the very end, after M steps of the process, an estimate of the thermal average of
g(r\ . . .
7N)
is given by taking the variable G that accumulated the sum of g and
computing
At low temperatures, almost the only moves accepted are those which lower the
energy of the system. At very high temperatures, almost every move is accepted.
The probability of accepting a move with positive δ£ has been arranged in just
such a way that the probability of occupying states differing by δΕ is exp[—βδ£].
This fact is easiest to understand by considering a system with just one particle,
and which can occupy just two states, but is true generally.

112
Chapter 5. Beyond Crystals
5.4.2 Molecular Dynamics
Molecular dynamics makes use of Newton's laws and random forces, rather than
random hops, in order to emulate thermal equilibrium. One form of molecular
dynamics operates by computing
Fi = -^r, (5-36)
on
the force on every particle due to every other particle, and then has the particles
move according to
d
2
n
fill
— f, mi is the mass of particle /. (5.37)
dt
2
Practitioners usually want to use the largest number of particles that can possibly
fit on their computers, and they also want to obtain reasonable accuracy while
minimizing intermediate storage. For this reason, an algorithm due to Verlet (1967)
is widely employed; a variety of other algorithms is described by Rapaport (1995).
Pick a time step dt that is much smaller than any time scale on which forces would
cause particles to move appreciably. Let the position of particle / after n steps of
the algorithm be rf. In order to find positions at the n + l'st step, compute
r?
+1
=2rf-r?-' + ^/
2
(5.38)
mi
with
Pp = P
l
{7{r
2
...r
N
) (5.39)
This sequence of computations requires only the storage needed to hold rf and
rf~ ', but makes errors only at order (dt)
4
after each time step, as shown in Problem
2.
Nothing more than Newton's laws is required in order to carry out computer
simulations of a physical system at temperature T. However, it is not easy to know
what the temperature of the system will be before the simulation begins, because
while the total energy £ is easily specified in initial conditions, the temperature
would have to be found later—for example, from the root mean square velocity of
particles. The fluctuation dissipation theorem may be used to modify the method
so as to specify temperature directly. According to this theorem, interacting parti-
cles head toward equilibrium at temperature T when two terms are added to their
equation of motion, one a damping term, and the other a random force, giving the
Langevin equation
p The damping constant b is somewhat arbitrary. The
-jt _ i_ _ iy-γ
_i_?/f\
smaller it is, the more closely particles follow Hamil- /-c ΛΓ\\
'
m
' >
V
/ tonian mechanics, while the larger it is, the more ^ ' '
quickly they come into thermal equilibrium. De-
tails of a particular physical problem are needed to
... fixé.
with
