Marder M.P. Condensed Matter Physics
Подождите немного. Документ загружается.


Liquids 113
The brackets refer either to a time average, or to
an
_
(5.41 )
average over statistical realizations of the system.
See Landau and Lifshitz
(
1980) p. 362 or Problem
10
A practical implementation of this idea replaces F" in Eq. (5.38) with G" where
tyn_jf-l] i
Ö" = Ff-bmi^—
1
—
i
- + Êi^6bmik
B
T/dt; (5.42)
Ξ/ is a vector whose components lie randomly between —1 and 1, and it can be
computed from
Ξ = 2(ρι — 1/2 ΏΊ — 1/2. P3 — 1/2) Each p
a
is a random number between 0 and 1. (5.43)
Because molecular dynamics keeps track of both positions and momenta, it is
computationally more costly than Monte Carlo, and Monte Carlo is preferable if
one only wants to find thermodynamic averages for a system in equilibrium, be-
cause it runs faster. However, molecular dynamics paints a more realistic picture of
the dynamical fashion in which a system approaches equilibrium. Representative
results from molecular dynamics calculations appear in Figures 5.9 and 5.14.
5.5 Liquids
5.5.1 Order Parameters and Long- and Short-Range Order
Every element melts at some temperature, at which point crystalline order disap-
pears.
The presence and absence of order is captured by an order
parameter,
which
is a function designed to (a) vanish when the desired form of order is absent, and (b)
rise up from zero as soon as it is present. Order parameters are often single num-
bers,
although they may also be tensors. The sharp Bragg peaks that characterize
scattering from crystalline lattices can be used as crystalline order parameters.
Formally, to create an order parameter 0^ distinguishing between crystal and
liquid, choose any reciprocal lattice vector K ψ 0 of the crystal, look back to the
correlation functions defined in Section 3.5 and define
V
0 - = —n
2
(q = K,0). Where n
2
{q, t) was defined in Eq. (3.54). (5.44)
For a crystal where N
2
terms contribute to the sum in Eq. (3.54), this quantity
should be of order unity. In a liquid where crystalline order has been lost, it will
instead be of order
1
/N.
Radial Correlation Functions.
Comparisons of solids and liquids are often made by additionally averaging the
correlation function
«2(^1,
^2;
0) defined in Eq. (3.49) over all angular orientations
(uo)m)=™
BTSaßm
mi

114
Chapter 5. Beyond Crystals
of the sample and making the result dimensionless by dividing through by the
square of the density n = N/V. The result is the
radial correlation
function
g{r)
(n
2
(r\,r
2
;0))e δ(7
χ
-7
2
)
Here r = \7\
—
τ\\. The angular braces mean
one must average over all angular orientations
of the sample. Subtracting off
the
second term
is conventional.
(5.45)
Liquids and glasses are isotropie by nature, while polycrystalline and powdered
crystalline samples have been made isotropie as described in Section 3.3.3. For
scattering off such materials the structure factor in Eq. (3.52) can be rewritten as
S(q) = \+n
J
drg[
re
iq-r
=
,+„/
dr(g(r)-\)é^
r
+ n
Using Eq. (5.45) and the static (5.46)
structure factor of
Eqs.
(3.55) and
(3.54).
dr e'Q'
r
Since g(r) ~^
1
for large r the (5.47)
integral only converges well after
subtracting 1.
\+n / dre^
7
{g{r)-\).
The last term on the right hand
side of (5.47) is a delta function
that only rises above zero when
one is staring directly into the
scattering beam, and which one
therefore can drop.
(5.48)
An integral of the area under the first peak,
I
first peak
dr 4πΓ g(r), (5.49)
gives the average number of nearest neighbors of each atom, known as the coor-
dination number. This quantity is slightly ambiguous to the extent that the precise
ending point of the first peak is ambiguous.
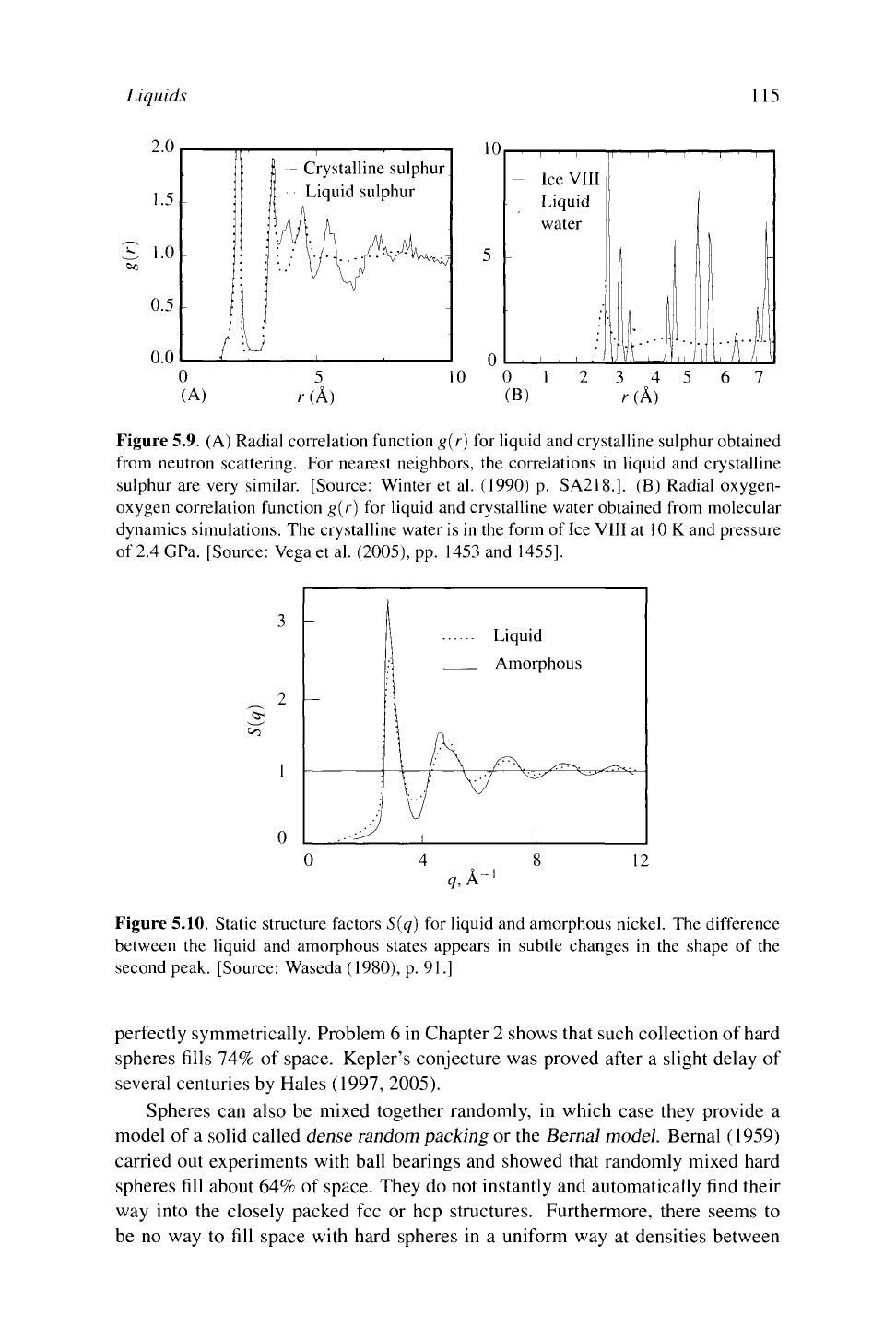
Figure 5.9 provides typical examples of correlation functions for crystals and
liquids taken from experiment and from computer simulation. The defining prop-
erty of crystals is long-range order (Section 3.5.1), yet the long-range order found
for crystals in these examples is not so very long: only around 10 Â. Nevertheless
order over these distances is easily sufficient to distinguish the liquids and crystals
from each other.
Figure 5.10 displays the static structure factors S(q) for liquid and amorphous
nickel. The radial correlation function g(r) can be obtained by inverting the Fourier
transform in Eq. (5.48). Differences between static structure functions of liquids
and amorphous solids are quite subtle.
5.5.2 Packing Spheres
One of the oldest questions in condensed matter physics goes back to a conjecture
of Kepler that the most efficient way to fill space with spheres is to stack them in
an fee or hep lattice (Figure 2.2 (D)). If one examines any given sphere in the close
packed state it has 12 neighbors, but these neighbors are not distributed about it
Liquids
115
Figure 5.9. (A) Radial correlation function g(r) for liquid and crystalline sulphur obtained
from neutron scattering. For nearest neighbors, the correlations in liquid and crystalline
sulphur are very similar. [Source: Winter et al. (1990) p. SA218.]. (B) Radial oxygen-
oxygen correlation function g(r) for liquid and crystalline water obtained from molecular
dynamics simulations. The crystalline water is in the form of Ice VIII at 10 K and pressure
of 2.4 GPa. [Source: Vega et al. (2005), pp. 1453 and 1455].
5ί
Figure 5.10. Static structure factors S(q) for liquid and amorphous nickel. The difference
between the liquid and amorphous states appears in subtle changes in the shape of the
second peak. [Source: Waseda (1980), p. 91.]
perfectly symmetrically. Problem 6 in Chapter 2 shows that such collection of hard
spheres fills 74% of space. Kepler's conjecture was proved after a slight delay of
several centuries by Hales (1997, 2005).
Spheres can also be mixed together randomly, in which case they provide a
model of a solid called dense random packing or the Bernal model. Bernal ( 1959)
carried out experiments with ball bearings and showed that randomly mixed hard
spheres fill about 64% of space. They do not instantly and automatically find their
way into the closely packed fee or hep structures. Furthermore, there seems to
be no way to fill space with hard spheres in a uniform way at densities between
116 Chapter 5. Beyond Crystals
74%
and 64%. Ensembles of hard spheres provide attractive settings for carrying
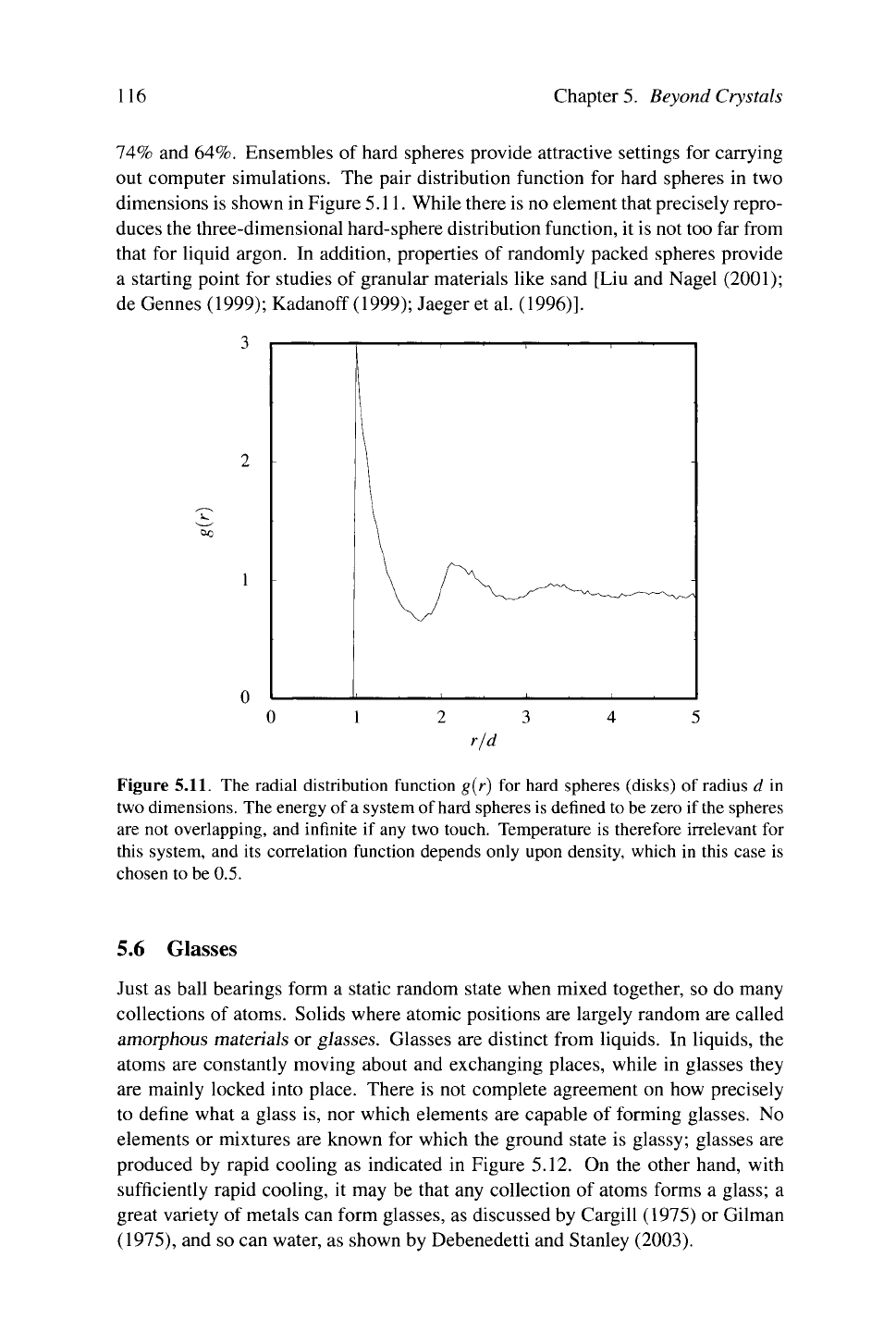
out computer simulations. The pair distribution function for hard spheres in two
dimensions is shown in Figure
5.11.
While there is no element that precisely repro-
duces the three-dimensional hard-sphere distribution function, it is not too far from
that for liquid argon. In addition, properties of randomly packed spheres provide
a starting point for studies of granular materials like sand [Liu and Nagel (2001);
de Gennes (1999); Kadanoff (1999); Jaeger et al. (1996)].
3
2
"öo
1
0
0 12 3 4 5
r/d
Figure 5.11. The radial distribution function g(r) for hard spheres (disks) of radius d in
two dimensions. The energy of
a
system of
hard
spheres is defined
to
be zero if
the
spheres
are not overlapping, and infinite if any two touch. Temperature is therefore irrelevant for
this system, and its correlation function depends only upon density, which in this case is
chosen to be 0.5.
5.6 Glasses
Just as ball bearings form a static random state when mixed together, so do many
collections of atoms. Solids where atomic positions are largely random are called
amorphous materials or glasses. Glasses are distinct from liquids. In liquids, the
atoms are constantly moving about and exchanging places, while in glasses they
are mainly locked into place. There is not complete agreement on how precisely
to define what a glass is, nor which elements are capable of forming glasses. No
elements or mixtures are known for which the ground state is glassy; glasses are
produced by rapid cooling as indicated in Figure 5.12. On the other hand, with
sufficiently rapid cooling, it may be that any collection of atoms forms a glass; a
great variety of metals can form glasses, as discussed by Cargill (1975) or Gilman
(1975),
and so can water, as shown by Debenedetti and Stanley (2003).
Glasses 117
To make a glass, start from a liquid and lower the temperature quickly. Below
the melting temperature
7M,
thermodynamic equilibrium requires atoms to arrange
themselves into a crystal. For fast enough cooling rates, which for window glass
are around 10 K s
_1
, and for nickel are 10
7
K s
_1
, glass forms instead. Density of
the liquid increases slowly, but viscosity η increases dramatically, up to a value of
around 10
12
Pa s, obeying the empirical formula, known as the Vogel-Fulcher law
η (X exp [C/(T - T
0
)} . C is a constant (5.50)
There is now a variety of theories, discussed by Angeli (1988), which can calculate
a divergence similar in shape to that of Eq. (5.50). Gradually, the material changes
its mechanical nature from a viscous liquid to a (frequently) brittle elastic solid.
The first peak in the correlation function g(r) narrows, indicating an increase in
short-range order, and the second peak then splits, as shown for amorphous nickel
in Figure 5.10.
(B) Composition (C) Composition
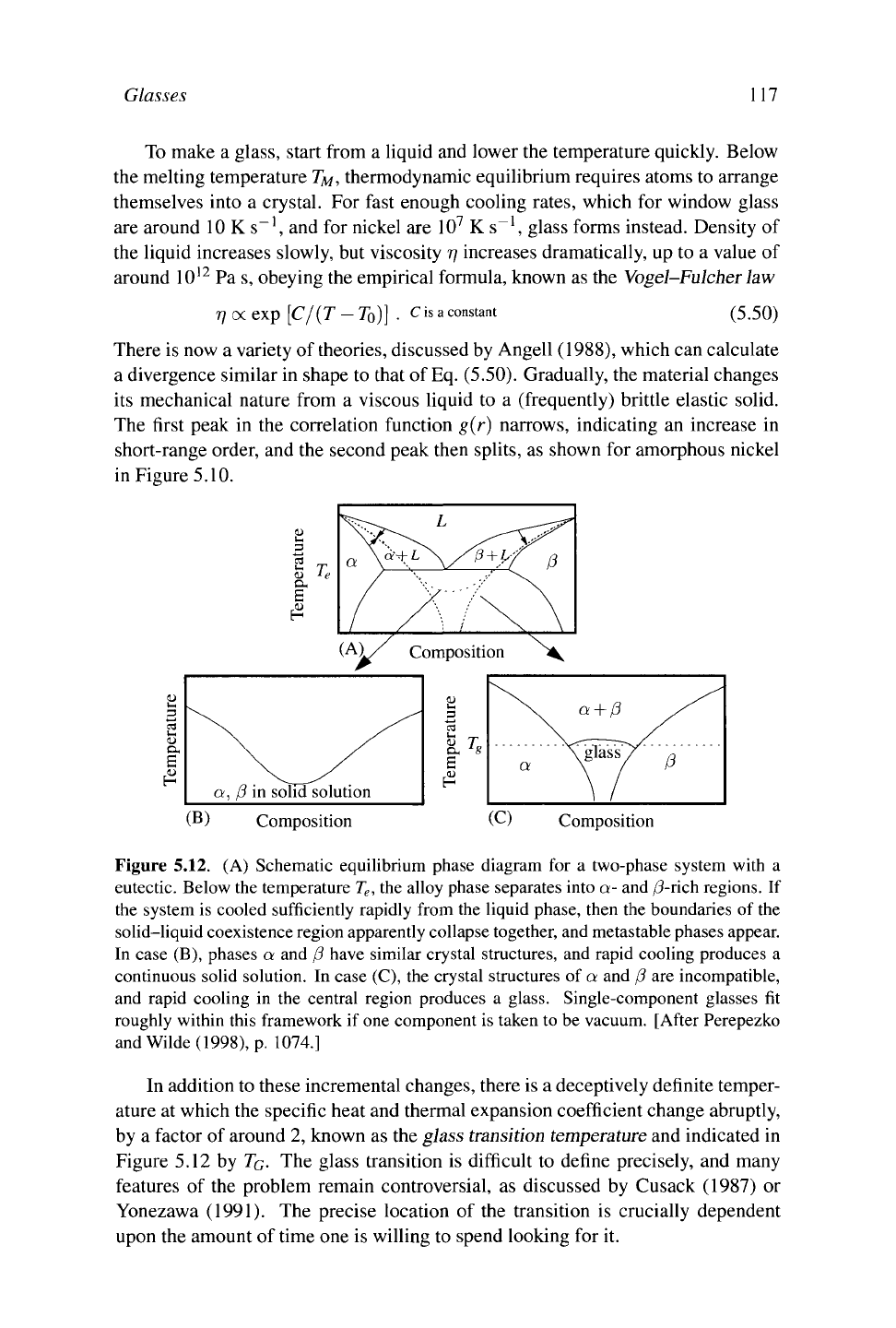
Figure 5.12. (A) Schematic equilibrium phase diagram for a two-phase system with a
eutectic. Below the temperature
T
e
,
the alloy phase separates into a- and /3-rich regions. If
the system is cooled sufficiently rapidly from the liquid phase, then the boundaries of the
solid-liquid coexistence region apparently collapse together, and metastable phases appear.
In case (B), phases a and β have similar crystal structures, and rapid cooling produces a
continuous solid solution. In case (C), the crystal structures of a and β are incompatible,
and rapid cooling in the central region produces a glass. Single-component glasses fit
roughly within this framework if
one
component is taken to be vacuum. [After Perepezko
and Wilde (1998), p. 1074.]
In addition to these incremental changes, there is a deceptively definite temper-
ature at which the specific heat and thermal expansion coefficient change abruptly,
by a factor of around 2, known as the glass transition
temperature
and indicated in
Figure 5.12 by
TQ.
The glass transition is difficult to define precisely, and many
features of the problem remain controversial, as discussed by Cusack (1987) or
Yonezawa (1991). The precise location of the transition is crucially dependent
upon the amount of time one is willing to spend looking for it.
118 Chapter 5. Beyond Crystals
X
E
o
I
o
6
5
4
3
0.
4r l
180
200 220
Temperature T (K)
240
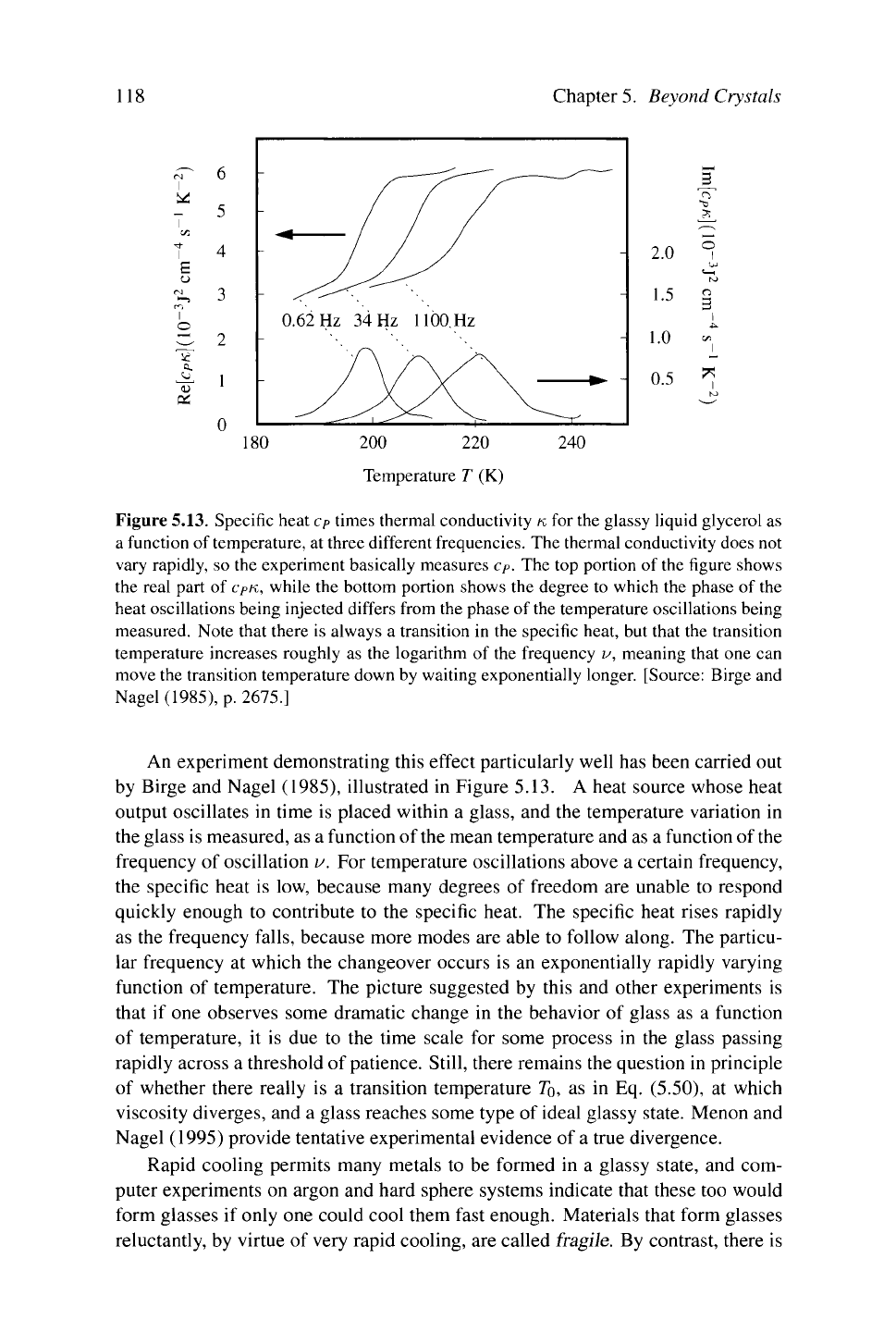
Figure 5.13. Specific heat cp times thermal conductivity κ for the glassy liquid glycerol as
a function of temperature, at three different frequencies. The thermal conductivity does not
vary rapidly, so the experiment basically measures c
P
. The top portion of the figure shows
the real part of
cpK,
while the bottom portion shows the degree to which the phase of the
heat oscillations being injected differs from the phase of the temperature oscillations being
measured. Note that there is always a transition in the specific heat, but that the transition
temperature increases roughly as the logarithm of the frequency v, meaning that one can
move the transition temperature down by waiting exponentially longer. [Source: Birge and
Nagel (1985), p. 2675.]
An experiment demonstrating this effect particularly well has been carried out
by Birge and Nagel (1985), illustrated in Figure 5.13. A heat source whose heat
output oscillates in time is placed within a glass, and the temperature variation in
the glass is measured, as a function of the mean temperature and as a function of the
frequency of oscillation v. For temperature oscillations above a certain frequency,
the specific heat is low, because many degrees of freedom are unable to respond
quickly enough to contribute to the specific heat. The specific heat rises rapidly
as the frequency falls, because more modes are able to follow along. The particu-
lar frequency at which the changeover occurs is an exponentially rapidly varying
function of temperature. The picture suggested by this and other experiments is
that if one observes some dramatic change in the behavior of glass as a function
of temperature, it is due to the time scale for some process in the glass passing
rapidly across a threshold of patience. Still, there remains the question in principle
of whether there really is a transition temperature
TQ,
as in Eq. (5.50), at which
viscosity diverges, and a glass reaches some type of ideal glassy state. Menon and
Nagel (1995) provide tentative experimental evidence of a true divergence.
Rapid cooling permits many metals to be formed in a glassy state, and com-
puter experiments on argon and hard sphere systems indicate that these too would
form glasses if only one could cool them fast enough. Materials that form glasses
reluctantly, by virtue of very rapid cooling, are called fragile. By contrast, there is

Glasses
119
a tendency among certain materials to form glasses especially readily. These are
strong or network glasses.
The prototypical network glass is S1O2, which is at the heart of most commer-
cial glasses. The basic structure is that of a silicon atom, which wants to bond
with four neighbors, surrounded by four oxygens, each of which wants to bond
with two neighbors. One model of the glass structure is the continuous random
network, shown in Figure 5.14. It is crucial that the angles between all the bonds
be somewhat variable, or else a crystalline structure must resuk instead.
Figure 5.14. The continuous random network was proposed as a model of S1O2 by
Zachariasen (1932). Each silicon tends to have four neighboring oxygens and each oxygen
tends to have two neighboring silicons. The picture was produced by a molecular dynamics
simulation where silicon and oxygen were raised to a high temperature and then rapidly
cooled and allowed to settle (stereo pair).
A rough argument of Phillips (1982b) gives an idea why mixtures where the
the average coordination number z lies between two and three tend to form glasses.
If one views the solid as a mechanical system where atoms bond to neighbors,
then it tends to form a glass when the number of degrees of freedom in the system
just equals the number of mechanical constraints. If there were more degrees of
freedom than needed to optimize the constraints, then the network would be me-
chanically unstable and would flop around, as for polymers (Section 5.8). If there
were fewer, the local structure of the network would have a deep energy minimum
at some particular configuration, and crystals would be favored. More specifically:
When the average coordination number provides z neighbors per atom in a system
of N atoms, the system has a total of Nz/2 bonds and must try to choose an opti-
mum length for each of them, providing Nz/2 constraints. Angles between bonds
provide more constraints. An atom with two bonds must try to optimize the angle
between them. An atom with three bonds must try to optimize the three angles
between the three pairs. For these two cases, the number of constraints to try to
satisfy is given by N(2z
—
3). Setting the total number of degrees of freedom, 3N,

120 Chapter 5. Beyond Crystals
equal to the total number of constraints,
Nz
3Ν = Ν(2ζ-3) + γ, (5.51)
it follows that
z = 2.4. (5.52)
Thus according to this argument, systems form glasses roughly when the average
number of neighbors per atom lies between 2 and 3. This argument is in accord
with the picture of the continuous random network in Figure 5.14 and is consistent
with observations in silicon oxides, boron oxides (B2O3), and the chalcogenide
glasses (AS2S3 and As3Se, for example). It does not fit amorphous silicon, how-
ever, which has fourfold coordination. In attempting more detailed accounts of
structure, the most frequently studied case is S1O2. While the general picture
shown in Figure 5.14 is correct, attempts to calculate the distribution of bond an-
gles and compare predictions with experiment have not yet been conclusive.
5.7 Liquid Crystals
CH
3
O -N=N O CH
3
O
T
^ ►
20
Â
Figure
5.15.
Picture of the organic molecule p-azoxyanisole
(PAA),
which forms
a
nematic
liquid crystal between 116°C and
135
°C.
It can roughly be regarded as a rigid rod of
length 20 Â and width
5
Â.
Intermediate in order between liquids and crystals are the liquid
crystals.
Their
mechanical properties are those of a liquid, yet certain types of order, particularly
in orientation, persist over large distances. The main structural element is a rodlike
molecule as shown in Figure 5.15, often made by two linked aromatic rings with
various flexible chains hanging off the ends.
5.7.1 Nematics, Cholesterics, and Smectics
Nematics. A nematic liquid crystal consists of a series of rods whose centers
are arrayed randomly, as are molecules in a liquid. Just as in a liquid, there is no
long-range positional order. However, there is long-range orientational
order,
as

Liquid Crystals 121
shown in Figure 5.16. As a consequence, the refractive index of the liquid varies
by around 20% in different directions and must be regarded as a tensor. This tensor
has complete rotational symmetry about the axis n and has mirror symmetry about
the plane normal to n as well as the planes containing it; its point group is D^/, in
Schönflies notation, or oo/mmm in international notation.
Figure 5.16. The molecules of
a
nematic liquid crystal have long-range orientational order,
but only short-range positional order.
Cholesterics. A variant of the nematic liquid crystal is the cholesteric (see Figure
5.17). Now the director n rotates slowly along an axis that is perpendicular to it,
described by
n
x
= 0 (5.53a)
n
y
= cos qox (5.53b)
n
z
= sin qox. (5.53c)
The wavelength of the twist λ = Ιπ/qo is on the order of thousands of angstroms
and is therefore much larger than the lengths of
the
molecules. This length can vary
rapidly as a function of temperature. The twist breaks the mirror symmetries about
the planes containing the x axis, and this loss of symmetry is directly connected
to the fact that cholesterics are produced by chimi molecules; these molecules are
rodlike, but they twist slightly as one moves up the rod.
Smectìcs. The final major class of liquid crystals is the smectic. Now, there is not
only long-range orientational order, but long-range positional order in one direction
as well. In smectìcs A, the rodlike molecules arrange themselves in layers with a
well-defined spacing, although within each layer the structure is liquid-like. The
layers are perpendicular to the director n. In smectìcs C, the director is no longer
perpendicular to the layers, and it may or may not rotate as one moves along the x
axis,
depending upon whether or not the liquid crystal is made of chiral molecules.

122
Chapter 5. Beyond Crystals
Figure 5.17. The director
h
of
a
cholestenc liquid crystal rotates as one moves along the x
axis.
Finally, in smectics B, molecules are arranged in a crystalline fashion within the
layers, and only the fact that the layers slide about with respect to one another
distinguishes the structure from a perfect crystal. These three phases are shown in
Figure 5.18.
Figure 5.18. The three main smectic phases, A, B, and C display one-dimensional long-
range positional order.
5.7.2 Liquid Crystal Order Parameter
One way to define an order parameter that will pick out nematic order is to consider
the probability n\ that a particle is at
?\
pointing along θ\ and define
Ό= ί ά
3
ηαθι
ηι
(7ι, 0i)i(3 cos
2
0i - 1). (5.54)
